Lumbar Intradural Extramedullary Isolated Hemagioblastoma: A Rare Entity and Review of Literature
Article Information
Muhammad Mohsin Khan1*, Kazim Mohammed2, Javeed Iqbal2, Surya Batas1, Khaled Al-sawalmeh3, Issam Al Bozom3, Ali Raza4
1Resident neurosurgery, Hamad Medical Corporation, Doha, Qatar
2Specialist neurosurgery, Hamad Medical Corporation, Doha, Qatar
3Department of pathology, Hamad Medical Corporation, Doha, Qatar
4Senior consultant neurosurgery, Hamad Medical Corporation, Doha, Qatar
*Corresponding Author: Muhammad Mohsin Khan, Resident neurosurgery, Hamad Medical Corporation, Doha, Qatar
Received: 03 May 2020; Accepted: 16 May 2020; Published: 04 June 2020
Citation:
Muhammad Mohsin Khan, Kazim Mohammed, Javeed Iqbal, Surya Batas, Khaled Al-sawalmeh, Issam Al Bozom, Ali Raza. Lumbar Intradural Extramedullary Isolated Hemagioblastoma: A Rare Entity and Review of Literature. Journal of Spine Research and Surgery 2 (2020): 077-084.
View / Download Pdf Share at FacebookAbstract
The terminology “Intradural Extramedullary” is often associated with Nerve sheath tumors (schwannoma, Neurofibroma) and Meningioma, while dermoid cyst, epidermoid, neurenteric cyst are other benign entities. Hemangioblastomas are rare, accounting for 1-5% of all spinal cord tumours.1 75% of the Spinal hemangiblastoma are Intramedullary, and another 10-15% have combined intramedullary and extramedullary-intradural component, while isolated extramedullary hemangioblastoma are rare1. Our case had an isolated intradural extramedullary hemangioblastoma without any associated manifestations of VHL syndrome. Surgery is a preferred option where total resection is possible, where as SRS is also an option in case of residual or incomplete resection.
Keywords
Intradural Extrameduallary, Hemangiblastoma, VHL syndrome, MRI
Intradural Extrameduallary articles Intradural Extrameduallary Research articles Intradural Extrameduallary review articles Intradural Extrameduallary PubMed articles Intradural Extrameduallary PubMed Central articles Intradural Extrameduallary 2023 articles Intradural Extrameduallary 2024 articles Intradural Extrameduallary Scopus articles Intradural Extrameduallary impact factor journals Intradural Extrameduallary Scopus journals Intradural Extrameduallary PubMed journals Intradural Extrameduallary medical journals Intradural Extrameduallary free journals Intradural Extrameduallary best journals Intradural Extrameduallary top journals Intradural Extrameduallary free medical journals Intradural Extrameduallary famous journals Intradural Extrameduallary Google Scholar indexed journals Hemangiblastoma articles Hemangiblastoma Research articles Hemangiblastoma review articles Hemangiblastoma PubMed articles Hemangiblastoma PubMed Central articles Hemangiblastoma 2023 articles Hemangiblastoma 2024 articles Hemangiblastoma Scopus articles Hemangiblastoma impact factor journals Hemangiblastoma Scopus journals Hemangiblastoma PubMed journals Hemangiblastoma medical journals Hemangiblastoma free journals Hemangiblastoma best journals Hemangiblastoma top journals Hemangiblastoma free medical journals Hemangiblastoma famous journals Hemangiblastoma Google Scholar indexed journals VHL syndrome articles VHL syndrome Research articles VHL syndrome review articles VHL syndrome PubMed articles VHL syndrome PubMed Central articles VHL syndrome 2023 articles VHL syndrome 2024 articles VHL syndrome Scopus articles VHL syndrome impact factor journals VHL syndrome Scopus journals VHL syndrome PubMed journals VHL syndrome medical journals VHL syndrome free journals VHL syndrome best journals VHL syndrome top journals VHL syndrome free medical journals VHL syndrome famous journals VHL syndrome Google Scholar indexed journals MRI articles MRI Research articles MRI review articles MRI PubMed articles MRI PubMed Central articles MRI 2023 articles MRI 2024 articles MRI Scopus articles MRI impact factor journals MRI Scopus journals MRI PubMed journals MRI medical journals MRI free journals MRI best journals MRI top journals MRI free medical journals MRI famous journals MRI Google Scholar indexed journals Lumbar Intradural articles Lumbar Intradural Research articles Lumbar Intradural review articles Lumbar Intradural PubMed articles Lumbar Intradural PubMed Central articles Lumbar Intradural 2023 articles Lumbar Intradural 2024 articles Lumbar Intradural Scopus articles Lumbar Intradural impact factor journals Lumbar Intradural Scopus journals Lumbar Intradural PubMed journals Lumbar Intradural medical journals Lumbar Intradural free journals Lumbar Intradural best journals Lumbar Intradural top journals Lumbar Intradural free medical journals Lumbar Intradural famous journals Lumbar Intradural Google Scholar indexed journals spinal cord articles spinal cord Research articles spinal cord review articles spinal cord PubMed articles spinal cord PubMed Central articles spinal cord 2023 articles spinal cord 2024 articles spinal cord Scopus articles spinal cord impact factor journals spinal cord Scopus journals spinal cord PubMed journals spinal cord medical journals spinal cord free journals spinal cord best journals spinal cord top journals spinal cord free medical journals spinal cord famous journals spinal cord Google Scholar indexed journals spinal canal articles spinal canal Research articles spinal canal review articles spinal canal PubMed articles spinal canal PubMed Central articles spinal canal 2023 articles spinal canal 2024 articles spinal canal Scopus articles spinal canal impact factor journals spinal canal Scopus journals spinal canal PubMed journals spinal canal medical journals spinal canal free journals spinal canal best journals spinal canal top journals spinal canal free medical journals spinal canal famous journals spinal canal Google Scholar indexed journals L4 vertebral articles L4 vertebral Research articles L4 vertebral review articles L4 vertebral PubMed articles L4 vertebral PubMed Central articles L4 vertebral 2023 articles L4 vertebral 2024 articles L4 vertebral Scopus articles L4 vertebral impact factor journals L4 vertebral Scopus journals L4 vertebral PubMed journals L4 vertebral medical journals L4 vertebral free journals L4 vertebral best journals L4 vertebral top journals L4 vertebral free medical journals L4 vertebral famous journals L4 vertebral Google Scholar indexed journals anterior articles anterior Research articles anterior review articles anterior PubMed articles anterior PubMed Central articles anterior 2023 articles anterior 2024 articles anterior Scopus articles anterior impact factor journals anterior Scopus journals anterior PubMed journals anterior medical journals anterior free journals anterior best journals anterior top journals anterior free medical journals anterior famous journals anterior Google Scholar indexed journals Nerve root articles Nerve root Research articles Nerve root review articles Nerve root PubMed articles Nerve root PubMed Central articles Nerve root 2023 articles Nerve root 2024 articles Nerve root Scopus articles Nerve root impact factor journals Nerve root Scopus journals Nerve root PubMed journals Nerve root medical journals Nerve root free journals Nerve root best journals Nerve root top journals Nerve root free medical journals Nerve root famous journals Nerve root Google Scholar indexed journals
Article Details
1. Case Report
33 years old female, no chronic medical condition, presented to emergency department with the complaints of low back pain radiating to right lower limb for the past 3months with exacerbation of symptoms since 2 weeks, necessitating repeated visits to ED. She also complained of urinary retention and weak urine stream for one day prior to presentation.
1.1 Examination on admission
Patient seemed to be in distress because of pain.Power in both limbs was grossly 4+/5, numbness both LL, saddle anesthesia, lax anal tone. Deep tendon reflexes seemed to be normal. An urgent MRI revealed anIntradural mass at L3-L4 vertebral level with intense enhancementlabelled by radiologist as meningioma.
1.2 Operation
The tumor was exposed byL3 total and L4 partial Laminectomy.The tumor waswell circumscribed, highly
Vascular. Multiple blood vessel were covering the tumor.
Complete excision of intradural extramedullary lesion was achieved by microdissection.The patients recovery was uncomplicated. Post operatively she received active Physio and rehabilitation.Patient did not have any neuro deficit at discharge.
1.3 Follow up
Followed in OPD at 4 months. No neurological deficits. Screening MRI of brain and whole spine showed no evidence of any associated lesion. Other screening for VHL came out to be negative.
PRE-OP MRI :
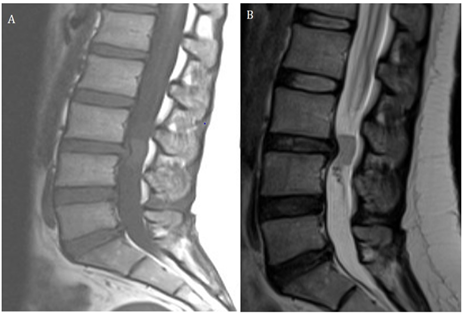
Figure 1: A and B. A well-defined intradural mass is seen at L3-L4 disk level. It appears isointense to the spinal cord on T1WI and T2WI.
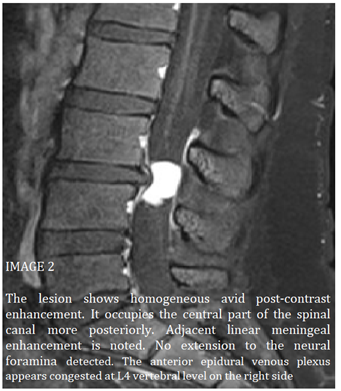
Figure 2: The lesion shows homogeneous avid post-contrast enhancement. It occupies the central part of the spinal canal more posteriorly. Adjacent linear meningeal enhancement is noted. No extension to the neural foramina detected. The anterior epidural venous plexus appears congested at L4 vertebral level on the right side.
POST-OP MRI:
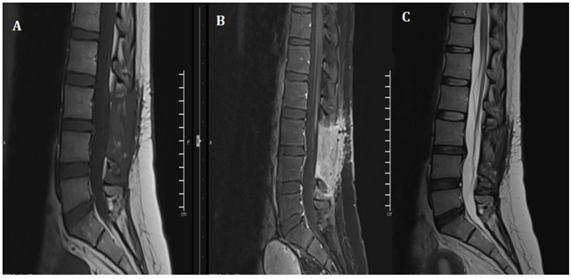
Figure 3: POST –OP follow up MRI shows total resection of the Lesion.
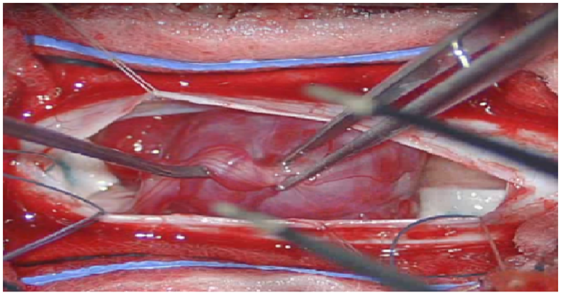
Figure 4: Intra-operative images Showing a solitary well capsulated Red colored Lesion with surrounding vessels and attached to Nerve root.
1.4 Pathology slides
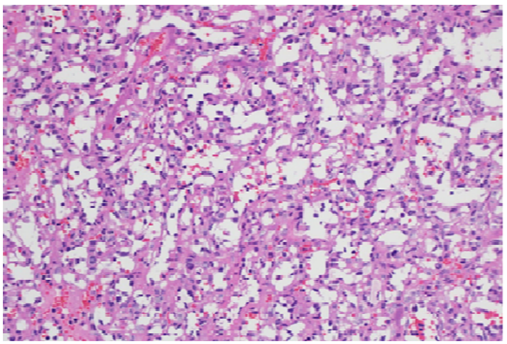
Figure 5: Light microscopic view showing richly vascular tumor (H&E X200).
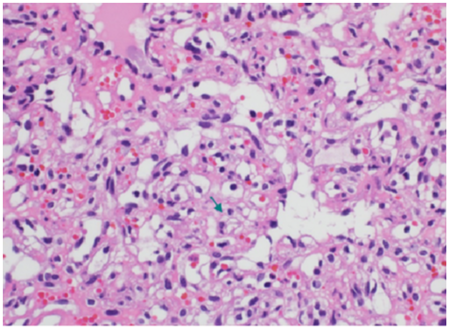
Figure 6: High power view showing interstitial tumor cells with abundant vacuolated cytoplasm (green arrow) with occasional hyperchromatic nuclei (H&E X00).
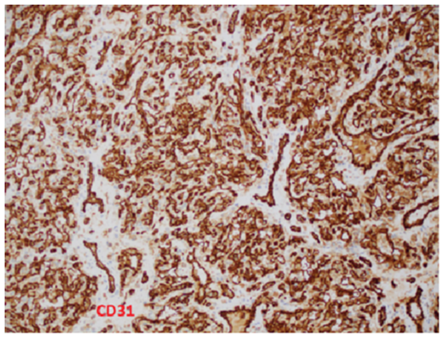
Figure 7: Immunohistochemical stain for CD31 highlights the lobulated capillaries and the rich vascular network (Immunohistochemistry X100).
1.4.1 Pathological examination: Sections show a well-circumscribed lobular proliferation of closely packed variable sized capillaries (Figure 1). The capillaries are lined by bland endothelium. The interstitium between the capillaries show stromal cell proliferation with vacuolated cytoplasm and occasional hyperchromatic nuclei (Figure 2). By immunohistochemistry, the interstitial cells are focally positive with CD68 while negative with CD31, CD34, Inhibin, EMA, GFAP and S-100. The rich capillary network ishighlighted with CD34 andCD31 (Figure 3).
2. Discussion
Hemangioblastoma are benign neoplasm of the CNS and they are basically classified as meningeal lesions of uncertain origin [1, 2]. Hemangioblastomas are rare, accounting for 1-5% of all spinal cord tumours.K1 75% of the Spinal hemangioblastoma are Intramedullary, and another 10-15% have combined intramedullary and extramedullary-intradural component, while isolated Extramedullary hemangioblastoma are rareK1 and associated with Pia mater. They most frequently are localized at the level of the cervical and thoracic vertebrae [3]. On the axial plane, the epicenters of hemangioblastomas are most commonly found in the posterior aspect of the spinal cord, in the region of the dorsal root entry zone [3, 4]. Chang Hyun Park [5], in a single center study from 2003-2012, have operated on 16 cases of Spinal Hemangioblastoma out of which 12 were solitary sporadic and 4 fulfilled the criteria of VHL, and all of these were intra-medullary. Out of the 12 solitary, 7 were cervical, 4 were thoracic and one at L1/2 level. The 4 patients with VHL had multiple and all were in cervical or thoraxic region with one intramedullary at L1. This study again shows the occurrence of solitary extramedullary Lesion is a rare entity [5]. Von Hippel-Lindau syndrome is autosomal dominant disorder with incomplete penetrance that has variable manifestations, linked to loss of function and mutation in the tumor suppressor gene on Chromosome no 3. Intramedullary Hemangioblastoma are seen in 10-30 % of patients with VHL syndrome. VHL syndrome is usually manifesting itself as central nervous system hemangioblastomas, renal cysts, and renal cell carcinomas. Other lesions include retinal angiomas, pheochromocytomas, pancreatic cysts, and epididymal cystadenomas. Tumours that are rich in blood vessels grow in the neural tissue in these tumours are called hemangioblastomas. cerebellum, cervical spine is the most common location of these tumours .spinal hemangioblastoma patients tend to present with neurological symptoms And signs at a younger age, and experience multiple small lesions .these tumours are only diagnosed when they are associated with compression effect and loss of function due to mass effect on the surrounding nervous tissue. Radical advancement of hemangioblastomas from solid to cystic component. Patients usually appear with symptoms due to mass effect, most often from an enlarging peritumoral cyst. An awareness of this advancement can assist in clinical decisions concerning follow-up intervals and the timing of surgical intervention. Our review of literature shows that a haemangioblastoma is actually rare entity and it does not occur commonly in the spinal cord. According to the literature the most common location is the posterior cranial fossa [6] and the second most common location comes out to be the spinal cord followed by spinal nerves, brain stem, thalamus and pituitary stalk [7, 8]. Infact spinal cord lesions account for very small percentage of the primary tumors overall, furthermore most of the patients that come with this lesion have actually and underline pretext to it that is VHL syndrome [9]. The actual incidence of sporadic lesion is unknown, one study [10] showed 20% of the cases have sporadic lesions and in our case we also think that this is a sporadic and isolated lesion as the patient does not have any other manifestations of Von Hippel Lindau syndrome, clinically or radiologically. Literature shows that sporadic lesions present much later in life and have less neurological symptoms as compared to the other counterpart with VHL syndromepatients. Also these patients are at much higher risk of recurrence, These patients usually require a lifelong surveillance and serial imaging to rule out other lesions [11]. In patients with sporadic and isolated lesions of hemangioblastoma, only 4% of the patients have detectable mutations in the respective VHL gene, however, new literature shows that these mutations are only specific to hemangioblastoma and have lower penetrance [12]. Because of the slow growing nature of the tumor mostly patients are asymptomatic, gets symptomatic because of local compression of neurological tissue from the mass lesion, mostly associated with the Back Pain, which later on may involve sensory loss or motor loss. VHL syndrome is a genetic disorder that is autosomal dominant in nature, and it is caused by mutations of the VHL tumour suppressor gene which is a germline mutation present on little arm of the third chromosome (3p25-26). The prevalence is 1:40000 to 1:50000. VHL syndrome is usually manifesting itself as central nervous system hemangioblastomas, renal cysts, and renal cell carcinomas. Other lesions include retinal angiomas, pheochromocytomas, pancreatic cysts, and epididymal cystadenomas. Tumours that are rich in blood vessels grow in the neural tissue in these tumours are called hemangioblastomas. cerebellum, cervical spine is the most common location of these tumours .spinal hemangioblastoma patients tend to present with neurological symptoms And signs at a younger age, and experience multiple small lesions .these tumours are only diagnosed when they are associated with compression effect and loss of function due to mass effect on the surrounding nervous tissue. Radical advancement of hemangioblastomas from solid to cystic component. Patients usually appear with symptoms due to mass effect, most often from an enlarging peritumoral cyst. An awareness of this advancement can assist in clinical decisions concerning follow-up intervals and the timing of surgical intervention. Tumour resection is the mainstay of management and can be curative for patients with solitary lesions, but constant follow-up is essential due to the high frequency of mutations in these patients with these tumours and a possible risk of a new tumour. Most of the literature we encountered patients had follow up for 1 to 2 years continuously. In a study done by Sven and Frank [14], and literature review follow-up after treatment for sporadic CNS hemangioblastoma were not found in the current literature. Based upon their experience they suggested to perform a MRI scan of the involved neuronal axis at 6 and at 12-24 months in the postoperative period. These follow-up modalities are to be advanced or extended based upon clinical indication. After intervention for a VHL associated lesion, there are detailed follow-up schedules available. The literature suggests performing a yearly MRI driven craniospinal control as well as an annual ophthalmoscopy, a yearly abdominal ultrasound with triennial computed tomography (CT) imaging, a yearly audiometry and pheochromocytoma investigation by urine analysis (metanephrine – VMA) Based upon clinical indication these follow-up modalities should be advanced or extended.
3. Conclusion
Tumour resection is the mainstay of management and can be curative for patients with solitary lesions, but constant follow-up is essential due to the high frequency of mutations in these patients with these tumours and a possible risk of a new tumour. Most of the literature we encountered patients had follow up for 1 to 2 years continuously. Once considered a rare entity the actual incidence rate of isolated intradural extramedullary Hemangioblastoma still remains unknown Keeping a higher index of suspicion in mind, a thorough review of MRI can lead to higher index of diagnosis pre-operatively. Total tumor resection appears to be one of the best options of treatment.
References
- Cushing HB, Bailey P. Tumors arising from blood vessels of the brain: angiomatous malformations and hemangioblastomas. Charles C. Thomas Springfield, IL 105 (1928): 212.
- Rubinstein LJ. Tumors of the central nervous system. Armed Forces Institute of Pathology; sold by American Registry of Pathology (1972).
- Lonser RR, Oldfield EH. Spinal cord hemangioblastomas. Neurosurgery Clinics 17 (2006): 37-44.
- Mandigo CE, Ogden AT, Angevine PD, et al. Operative management of spinal hemangioblastoma. Neurosurgery 65 (2009): 1166-1177.
- Park CH, Lee CH, Hyun SJ, et al. Surgical outcome of spinal cord hemangioblastomas. Journal of Korean Neurosurgical Society 52 (2012): 221.
- Neumann HP, Eggert HR, Weigel K, et al. Hemangioblastomas of the central nervous system: a 10-year study with special reference to von Hippel-Lindau syndrome. Journal of neurosurgery 70 (1989): 24-30.
- Gläsker S, Berlis A, Pagenstecher A, et al. Characterization of hemangioblastomas of spinal nerves. Neurosurgery 56 (2005): 503-509.
- Kanno H, Yamamoto I, Nishikawa R, et al. Spinal cord hemangioblastomas in von Hippel–Lindau disease. Spinal cord 47 (2009): 447-452.
- Kinney TD, Fitzgerald PJ. Lindauvon Hippel disease with hemangioblastoma of the spinal cord and syringomyelia. Archives of pathology 43 (1947): 439.
- Takai K, Taniguchi M, Takahashi H, et al. Comparative analysis of spinal hemangioblastomas in sporadic disease and Von Hippel-Lindau syndrome. Neurologia medico-chirurgica 50 (2010): 560-567.
- Conway JE, Chou D, Clatterbuck RE, et al. Hemangioblastomas of the central nervous system in von Hippel-Lindau syndrome and sporadic disease. Neurosurgery 48 (2001): 55-63.
- Woodward ER, Wall K, Forsyth J, et al. VHL mutation analysis in patients with isolated central nervous system haemangioblastoma. Brain 130 (2007): 836-842.
- Murota T, Symon L. Surgical management of hemangioblastoma of the spinal cord: a report of 18 cases. Neurosurgery 25 (1989): 699-708.
- Bamps S, Van Calenbergh F, De Vleeschouwer S, et al. What the neurosurgeon should know about hemangioblastoma, both sporadic and in Von Hippel-Lindau disease: A literature review. Surgical neurology international 4 (2013).
