A New Office Based Protocol to Reverse AGA Miniaturization at Early and Advanced Stages
Article Information
Piccolo D1*, Tesauro P2, Conforti C3, Crisman G1, Tramontozzi S1, Bovani B4, Melfa F5, Gennai A6
1Novea Skin Center Avezzano, private practice, Avezzano, L’Aquila, Italy
2Plastic Surgeon, PoliambulaTorio Gioia, private practice, Milano, Italy
3Dermatology Clinic, Maggiore Hospital of Trieste, Trieste, Italy
4Surgery and Aesthetic Plastic Surgeon, private practice, Perugia, Italy and Republic of San Marino
5Mediaging Clinic Center, private practice, Palermo, Italy
6Plastic Surgeon, Studio Gennai, private practice, Bologna, Italy
All authors contributed equally to this work
*Corresponding Author: Domenico Piccolo, Dermatologist, Novea Skin Center Avezzano, Avezzano, L’Aquila, Italy.
Received: 30 July 2023; Accepted: 07 August 2023; Published: 25 August 2023
Citation: Piccolo D, Tesauro P, Conforti C, Crisman G, Tramontozzi S, Bovani B, Melfa F, Gennai A. A New Office Based Protocol to Reverse AGA Miniaturization at Early and Advanced Stages. Journal of Surgery and Research. 6 (2023): 295-302.
View / Download Pdf Share at FacebookAbstract
Introduction: Several types of hair loss diseases have been identified and classified over the past decades, and Androgenetic Alopecia (AGA) represents the most common condition among these. It can affect both sexes with an aesthetic and psychological impact that goes from moderate to severe. Several medical treatments, topical and systemic, have been proposed for it, with different degrees of response.
Objective: To analyze the efficacy and outcome of an innovative therapeutic protocol, which combined the effect of the autologous regenerative treatment performed by medical device SEFFIHAIR™ and six sessions of TRICHOBIOLIGHT®, a combination of mesotherapy directly performed on the scalp with a subsequent photobiostimulation session with red LED light (630nm).
Methods: Forty patients (12 women and 28 men, mean age 36,4 year-old) with clinical and trichoscopic diagnosis of AGA have been enrolled during our private practice and have all been treated with the combination of SEFFIHAIR™ and TRICHOBIOLIGHT®.
Results: All patients responded to the treatment with an impressively excellent outcome reached in the 82,5% of patients (33/40) and with good results for the other 7 patients (17,5%).
Conclusions: The combination of SEFFIHAIR™ and TRICHOBIOLIGHT® has proved to be a very promising protocol for reversing the miniaturization process of follicles induced by AGA, in the absence of adverse effects. This synergy of actions has determined an acceleration of hair growth and thickening of the hair shaft, thus resulting in a consistent aesthetic result and a substantial psychological improvement.
Keywords
TRICHOBIOLIGHT®, Androgenetic alopecia, Photobiostimulation, Mesotherapy, SEFFIHAIR™, Autologous regenerative treatment
TRICHOBIOLIGHT? articles; Androgenetic alopecia articles; Photobiostimulation articles; Mesotherapy articles; SEFFIHAIR? articles; Autologous regenerative treatment articles
TRICHOBIOLIGHT® articles TRICHOBIOLIGHT® Research articles TRICHOBIOLIGHT® review articles TRICHOBIOLIGHT® PubMed articles TRICHOBIOLIGHT® PubMed Central articles TRICHOBIOLIGHT® 2023 articles TRICHOBIOLIGHT® 2024 articles TRICHOBIOLIGHT® Scopus articles TRICHOBIOLIGHT® impact factor journals TRICHOBIOLIGHT® Scopus journals TRICHOBIOLIGHT® PubMed journals TRICHOBIOLIGHT® medical journals TRICHOBIOLIGHT® free journals TRICHOBIOLIGHT® best journals TRICHOBIOLIGHT® top journals TRICHOBIOLIGHT® free medical journals TRICHOBIOLIGHT® famous journals TRICHOBIOLIGHT® Google Scholar indexed journals Androgenetic alopecia articles Androgenetic alopecia Research articles Androgenetic alopecia review articles Androgenetic alopecia PubMed articles Androgenetic alopecia PubMed Central articles Androgenetic alopecia 2023 articles Androgenetic alopecia 2024 articles Androgenetic alopecia Scopus articles Androgenetic alopecia impact factor journals Androgenetic alopecia Scopus journals Androgenetic alopecia PubMed journals Androgenetic alopecia medical journals Androgenetic alopecia free journals Androgenetic alopecia best journals Androgenetic alopecia top journals Androgenetic alopecia free medical journals Androgenetic alopecia famous journals Androgenetic alopecia Google Scholar indexed journals Photobiostimulation articles Photobiostimulation Research articles Photobiostimulation review articles Photobiostimulation PubMed articles Photobiostimulation PubMed Central articles Photobiostimulation 2023 articles Photobiostimulation 2024 articles Photobiostimulation Scopus articles Photobiostimulation impact factor journals Photobiostimulation Scopus journals Photobiostimulation PubMed journals Photobiostimulation medical journals Photobiostimulation free journals Photobiostimulation best journals Photobiostimulation top journals Photobiostimulation free medical journals Photobiostimulation famous journals Photobiostimulation Google Scholar indexed journals Mesotherapy articles Mesotherapy Research articles Mesotherapy review articles Mesotherapy PubMed articles Mesotherapy PubMed Central articles Mesotherapy 2023 articles Mesotherapy 2024 articles Mesotherapy Scopus articles Mesotherapy impact factor journals Mesotherapy Scopus journals Mesotherapy PubMed journals Mesotherapy medical journals Mesotherapy free journals Mesotherapy best journals Mesotherapy top journals Mesotherapy free medical journals Mesotherapy famous journals Mesotherapy Google Scholar indexed journals SEFFIHAIR™ articles SEFFIHAIR™ Research articles SEFFIHAIR™ review articles SEFFIHAIR™ PubMed articles SEFFIHAIR™ PubMed Central articles SEFFIHAIR™ 2023 articles SEFFIHAIR™ 2024 articles SEFFIHAIR™ Scopus articles SEFFIHAIR™ impact factor journals SEFFIHAIR™ Scopus journals SEFFIHAIR™ PubMed journals SEFFIHAIR™ medical journals SEFFIHAIR™ free journals SEFFIHAIR™ best journals SEFFIHAIR™ top journals SEFFIHAIR™ free medical journals SEFFIHAIR™ famous journals SEFFIHAIR™ Google Scholar indexed journals Autologous regenerative treatment articles Autologous regenerative treatment Research articles Autologous regenerative treatment review articles Autologous regenerative treatment PubMed articles Autologous regenerative treatment PubMed Central articles Autologous regenerative treatment 2023 articles Autologous regenerative treatment 2024 articles Autologous regenerative treatment Scopus articles Autologous regenerative treatment impact factor journals Autologous regenerative treatment Scopus journals Autologous regenerative treatment PubMed journals Autologous regenerative treatment medical journals Autologous regenerative treatment free journals Autologous regenerative treatment best journals Autologous regenerative treatment top journals Autologous regenerative treatment free medical journals Autologous regenerative treatment famous journals Autologous regenerative treatment Google Scholar indexed journals psychological improvement articles psychological improvement Research articles psychological improvement review articles psychological improvement PubMed articles psychological improvement PubMed Central articles psychological improvement 2023 articles psychological improvement 2024 articles psychological improvement Scopus articles psychological improvement impact factor journals psychological improvement Scopus journals psychological improvement PubMed journals psychological improvement medical journals psychological improvement free journals psychological improvement best journals psychological improvement top journals psychological improvement free medical journals psychological improvement famous journals psychological improvement Google Scholar indexed journals trichoscopic diagnosis articles trichoscopic diagnosis Research articles trichoscopic diagnosis review articles trichoscopic diagnosis PubMed articles trichoscopic diagnosis PubMed Central articles trichoscopic diagnosis 2023 articles trichoscopic diagnosis 2024 articles trichoscopic diagnosis Scopus articles trichoscopic diagnosis impact factor journals trichoscopic diagnosis Scopus journals trichoscopic diagnosis PubMed journals trichoscopic diagnosis medical journals trichoscopic diagnosis free journals trichoscopic diagnosis best journals trichoscopic diagnosis top journals trichoscopic diagnosis free medical journals trichoscopic diagnosis famous journals trichoscopic diagnosis Google Scholar indexed journals photobiostimulation articles photobiostimulation Research articles photobiostimulation review articles photobiostimulation PubMed articles photobiostimulation PubMed Central articles photobiostimulation 2023 articles photobiostimulation 2024 articles photobiostimulation Scopus articles photobiostimulation impact factor journals photobiostimulation Scopus journals photobiostimulation PubMed journals photobiostimulation medical journals photobiostimulation free journals photobiostimulation best journals photobiostimulation top journals photobiostimulation free medical journals photobiostimulation famous journals photobiostimulation Google Scholar indexed journals
Article Details
Introduction
Hair have taken over, in today’s society, an identifying function and their loss can cause, right from its first manifestations, serious discomfort for individual of all ages that are afflicted with it, generating a deterioration in the quality of life, with direct consequences on the overall well-being. Hair loss can be ascribed to numerous causes depending on which one observes transient alopecia, as in Acute Telogen or some forms of Alopecia Areata, or permanent alopecia as occurs in various autoimmune diseases (lichen, lupus) or iatrogenic lesions. In most cases, however, hair loss recognizes a genetic origin, correlated with androgen hormone production (AGA), which is often worsened by numerous engraving condition such as digestive disorders, nervous breakdown, physical or mental discomfort. AGA and its precursors such as Miniaturization of Anagen phase (MA) represent the most common cause of alopecia, affecting 70% of men, who may show the first signs of the disease from late adolescence, and 40% of women, generally affected by the third or fourth decade onwards [1]. Men typically show a recession of the hairline at the temples and hair loss at the vertex, while women normally show a widespread thinning over the whole upper part of the scalp [1-5]. The most frequentdermatoscopicfeatures associated with AGA are a miniaturization of the hair shafts, a reduction in the number of hairs per pilosebaceous unit and numerous yellow dots. The latter represent, in this pathology, follicles "stuck" in kenogen phase, which are unable to express a new stage of growth. This exam allows detection of the earliest signs of AGA, which become increasingly evident as the years go by and the disease worsen. Patients almost invariably report a loss of mass and a progressive increasing of transparency [6,7]. AGA recently described as an autosomal dominant polygenic inherited disease, is caused by the action of androgens on the epithelial cells of hair follicles of genetically predisposed individuals. In these subjects, DHT causes, in androgen-dependent areas, a shortening of the anagen phase and an early entry of the follicle into telogen [1-5].
This acceleration of the follicular cycle generates a functional incapacity and subsequent miniaturization of hair perceived by patients as a “loss of hair” [2-6]. Several therapeutic approaches to AGA have been tried in the last decades. Drugs or cosmeceuticals have been prescribed to patients and administered either by topical or systemic routes, as well as physical therapies of various types and all the above individually or in combination. Variable success rates are reported in the literature, and the outcome depends mainly on the earliness of therapy and disease progression. [1-5]. In the last few years, new studies proved the efficacy of therapies based on the autologous grafting of adult mesenchymal stem cells to accelerate the healing and regenerative processes of the skin and mesenchymal tissues [8-16].
The adipose-derived stem cells (ADSCs) are, of the above, the most widely used by clinician, due to the simplicity of their harvesting. ADSCs are pluripotent adult progenitor cells derived from embryonic connective tissue. It can differentiate towards adipogenic, osteogenic, condrogenic, myogenic cells but also towards not mesodermic cell lines (neuronal, endothelial, epithelial cells, etc.). According to the literature, some authors proposed the possibility to restore the hair cycle in male and female pattern baldness by stimulating the niche with autologous fat enriched with SVF. Their studies have shown that ADSCs promote bulb function by stimulating hair follicle stem cells (HFSCs). [11-22]. Taking into consideration all the above therapeutic strategies, and the strengths and weaknesses of each of them, we decided to test a new type of outpatient protocol for the treatment of AGA. We structured a therapeutic process based on three innovative methods placed in synergy with each other, each capable of enhancing the action of the previous one. The first step of our protocol is based on the use of an ADSCs extract enriched with vasculo-stromal cells (SVF). For adipose tissue harvesting, we used a specific medical device, SEFFIHAIR™, Manufactured by SEFFILINE S.r.l. Bologna - Italy. The word SEFFI stands for Superficial Enhanced Fluid Fat Injection [13,14].
For the second step, we chose to subject patients to a sequence of Mesotherapy and Photobiostimulation. Mesotherapy, "therapy in the middle layer of the skin," is based on multiple superficial dermal microinjections and has shown significant potential in the treatment of AGA. In this protocol, we included a selection of natural substances in the therapeutic cocktail, excluding pharmacological active ingredients and choosing the one-month interval as the right compromise between efficacy and compliance [23-27]. Photobiostimulation with red LED light at 630 nm proved to be very effective in activating mitochondria and the ATP cycle, thus leading to increased receptivity of the cells to nearby substances (in this case those conveyed by mesotherapy) and would promote the development of mesenchymal stem cells transplanted around the hair follicles [22,28]. This second step represents the previously structured and registered protocol named TRICHOBIOLIGHT®, consistent results of which have already been published [28]. This combined protocol, SEFFIHAIR™ and TRICHOBIOLIGHT®, was implemented with the aim of verifying whether the results previously obtained only with the latter could be further improved. In this retrospective study, we present the results obtained in almost two years of experimentation.
Methods
A sample of 40 Caucasians patients (12 F: 28 M, with an average age of 36,4 year-old (range 16-67)) with clinical diagnosis of AGA (with clinically diagnosed mild to moderate AGA (Grades II-V), according to Norwood-Hamilton classification), have been enrolled in our private practice at our four “Novea Skin Center” located in L’Aquila, Avezzano, Pescara, Catania and other Italian associated centers) from June 2020 to August 2022. All patients signed informed consent. The inclusion criteria in the protocol were the clinical and trichoscopic diagnosis of AGA and the suspension for at least 6 months of other trichological therapies, while the diagnosis of other scalp pathologies and/or the unavailability of a complete adherence to the treatment protocol represented the exclusion criteria. General patient information was collected, including age at AGA’s onset, clinical history of the disease, and previously prescribed therapies. Then, a global scalp photography, pull test, trichogram and trichoscopy have been performed to all patients. Trichoscopy helped to visualize the hair shaft, scalp epidermis, and follicular openings with higher magnification and accuracy. Trichoscopic parameters such as variation in hair shaft diameter or differences in number of hair shafts per follicular unit between the disease-affected areas and the comparison occipital region can be used to identify the disease severity and assess the therapeutic response [7-9]. The first step of our protocol. consisted with the autologous regenerative treatment with SVF and ADSc with SEFFIHAIR™ medical device. Once the sampling area has been established (in most cases the abdomen or hips), the procedure involves the preparation of the surgical field, followed by local anesthesia of the entry point and the small incision on the edge of the selected area. Subsequently, with the same instrument, both the infiltration of the anesthetic solution and, after a 10-minute break needed for vasoconstriction, the aspiration of the adipose tissue is performed. This instrument consists of a syringe with an automatic plunger lock onto which a 2 mm.Ø cannula with 1mm.Ø micro-holes is screwed, inserted inside a special guide (Figure 1). The instrument ensures that the tunneling is performed in the superficial subcutaneous tissue, next to the dermis, thus harvesting the ideal tissue richest in SVF and ADSC, able to offer the best quantity and quality of cells. Once collected, the already micro-fragmented adipose tissue is gently washed and then emulsified (A-B), thanks to 20 passages between two syringes joined by a connector. The obtained product is then transferred into test tubes and centrifuged for 10 minutes at 3500 rpm. At the end of the centrifugation the infranatant (in the lower part of the tubes) is separated from the tissue (deposited on the top) and can be easily aspirated from the bottom of the tubes using a long needle; it is now ready to be easily and painlessly injected into the dermis of the scalp, using a multi injector 7 needles, 32G, 3 mm length. One month after a single session of SEFFIHAIR™ begins the second phase of our protocol which consists of a therapeutic combined approach of mesotherapy and photobiostimulation called TRICHOBIOLIGHT® (registered trademark at the EUIPO -ID No. 2020/098 - already available online at http://www.euipo.europa. eu/). Mesotherapy is performed with a 1.5-mL syringe equipped with a 30G micro-needle, 4mm in length. 3 cc of a hyaluronic acid-based solution is used, which contains polydeoxyribonucleotides (5.625mg/3mL), 7 vitamins, GF fibroblasts + polypeptides + NaDNA, 16 amino acids, 8 minerals, 2 Ginkgo Biloba extracts, linoleic acid, oleic acid, quercetin, kaempferol, chlorogenic acid, gallic acid and water (Table 1). The mesotherapy session is immediately followed by a 15-minute photobiostimulation session with a YOUNG-AGAIN® E-Light technology scalp conformal lamp (Espansione Group®, Italy) (630 nm LED light). Patients underwent 6 sessions of TRICHOBIOLIGHT, once monthly. Follow-up examination have been performed every four months and one or two maintenance sessions/year have been prescribed depending on the gravity of the disease and the obtained results. In addition, we asked each patient to fill in an end-of-treatment satisfaction questionnaire.
|
"Hyaluronic Acid Plydeoxyribonucleotides /5,625mg/3mL)" |
||
|
Vit E |
Fibroblasts GF + Polipeptides + NaDNA |
Water |
|
Arginine Lysine |
Silica |
|
|
Quercetin |
||
|
Bliobalide |
||
|
Chlorogenic Acid |
Lynoleum Acid |
|
Table 1: The cocktail of active ingredients conveyed by mesotherapy directly on the scalp during the six TRICHOBIOLIGHT® sessions.
Results
All patients responded to the treatment and an impressive and excellent outcome has been reached in 82.5% of patients (33/40) (Figures 2-6), and a good result was observed in the remaining 7 patients (17,5%) (Figure 7). The data collected proved the excellent efficacy of our protocol in the treatment of baldness, showing an early clinical response after only four months of therapy, with results that were maintained over the following 12 to 18 months. Results have been confirmed by trichogram and trichoscopy and the increase of hair shaft diameter has been measured at the follow up visit, as well as by the clinical image/patient self-perception at the mirror. A 20% increase of hair shaft diameter has been observed (Figure 1). Common side effects included mild to moderate pain after the mesotherapy session, itchy or burning sensation self-resolving within 24-hour. At the end-of-treatment satisfaction questionnaire, 97,5% (39/40) of patients reported a high satisfaction with the treatment, referring to the better perception of their image for a visible increase in thickness and shine of the hair, associated with a decrease in hair loss.
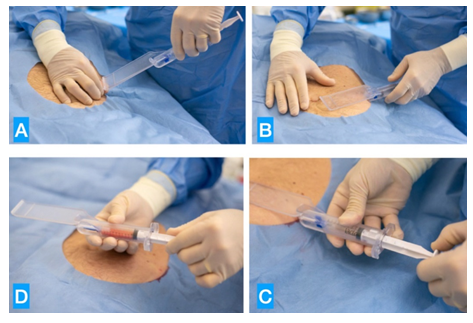
Figure 1: The aspiration of the adipose tissue is performed with a syringe with an automatic plunger lock onto which a 2 mm.Ø cannula with 1 mm.Ø micro-holes is screwed, inserted inside a special guide (Figure 1A). The instrument ensures that the tunneling is performed in the superficial subcutaneous tissue (Figure 1B), next to the dermis, thus harvesting the ideal tissue richest in SVF and ADSC (Figure 1C), able to offer the best quantity and quality of cells and to repeat the procedure if needed ((Figure 1D).
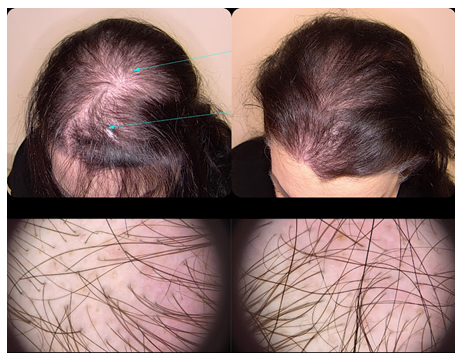
Figure 2: Trichoscopy as a diagnostic and monitoring tool in hair loss diseases. Left: Clinical and trichoscopic findings before the SEFFIHAIR™ and TRICHOBIOLGIHT® treatment. Right: Clinical and tirchoscopic findings after one session of SEFFIHAIR™ and six sessions of TRICHOBIOLGIHT®. A consistent thickening of hairs (variation in hair shaft diameter is >35%) is observed, while there is no change in the number of follicular units with single and multiple hairs and perifollicular halo.
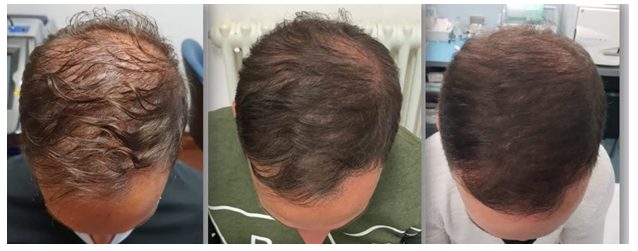
Figure 3: Excellent improvement after one session of SEFFIHAIR™ and 6 TRICHOBIOLIGHT® sessions, with a consistent thickening of the patient’s hair in a 46-year old man.
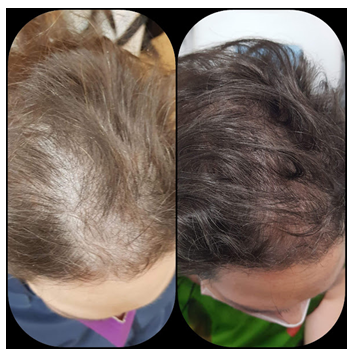
Figure 4: Excellent improvement after one session of SEFFIHAIR™ and 6 TRICHOBIOLIGHT® sessions, with a consistent thickening of the patient’s hair in a 38-year-old man.
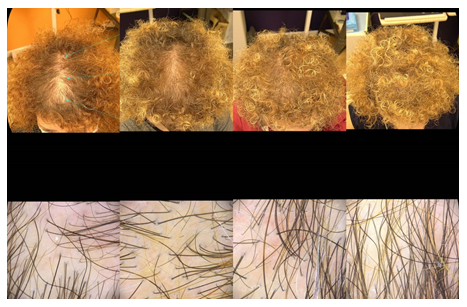
Figure 5: Trichoscopy as a diagnostic and monitoring tool in hair loss diseases. From Left to Right: Clinical and trichoscopic findings before the SEFFIHAIR™ and TRICHOBIOLGIHT® treatment (T0) (Second images): Clinical and trichoscopic findings after one session of SEFFIHAIR™ and two sessions of TRICHOBIOLGIHT® (T2). A consistent thickening of hairs (variation in hair shaft diameter is >35%) is observed, while there is no change in the number of follicular units with single and multiple hairs and perifollicular halo. (Third Images): Clinical and trichoscopic findings after one session of SEFFIHAIR™ and four TRICHOBIOLGIHT® treatment (T4). (Fourth Images): Clinical and trichoscopic findings after one session of SEFFIHAIR™ and sixTRICHOBIOLGIHT® treatment, one months of follow up at the end of the treatment (T7) in a 55-year-old woman.
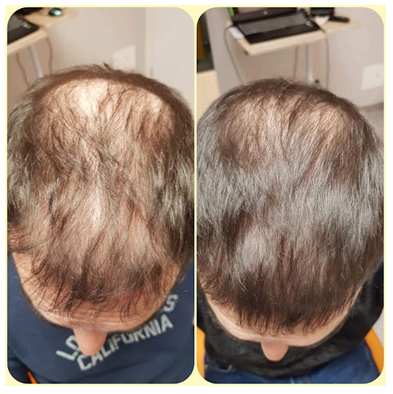
Figure 6: Excellent outcome after one session of SEFFIHAIR™ and 6 TRICHOBIOLIGHT® sessions in a 35-year.old man.
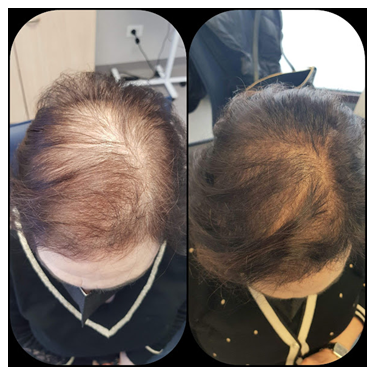
Figure 7: Good outcome after one session of SEFFIHAIR™ and 4 TRICHOBIOLIGHT® sessions in a 49-year old woman.
Discussion
Among the past decades, more and more studies have been published in the aim to find out effective therapy for AGA, especially because baldness has a significant psychological impact on the quality of life of affected patients. The most common therapies offered to patients are natural therapies, which include a vast array of stimulating lotions and food supplements containing natural substances with anti-androgenic and antioxidant effects, and pharmacological treatments. It must be said that "natural" therapies based on herbal treatments such as Saw palmetto (Serenoa Repens), Rosemary (Rosmarinus officinalis), Licorice (Glycyrrhiza glabra), grape seeds (Vitis vinifera), pumpkin seeds (Curcurbita pepo), green tea (Camellia sinensis) etc. It should be mentioned that "natural" therapies based on herbal treatments such as Saw palmetto (Serenoa Repens), Rosemary (Rosmarinus officinalis), Licorice (Glycyrrhiza glabra), grape seed (Vitis vinifera), pumpkin seed (Curcurbita pepo), green tea (Camellia sinensis) etc. however popular they are, often generate questionable results [1]. To date, pharmacological treatments for the medical treatment of AGA are based on FDA-approved formulations of minoxidil and finasteride, and dutasteride the most popular of the off-label protocols in use today. These substances can be administered both topically and systemically and due to their proven action often offer significant and stable results over time. However, there are several side effects associated with the use of these drugs (burning sensation, allergic contact dermatitis, hypertrichosis in adjacent areas), and even more significant has become the patients’ resistance to their use related to the possible onset of side effects on the sexual sphere (decreased libido and ejaculation disorders) [9-12]. The above-described treatments are delegate to the patient’s compliance, which means that results are strictly related to the respect of the therapeutic scheme and how the prescribed products are used/applied. In addition, it is also worth noting that, in addition to or as a substitute for therapies, there is an exponential increase in hair transplant surgeries procedures (FUE or FUS), which today offer an excellent possibility to successfully correct baldness. Without going into the details of this method the limits of the surgical approach are inextricably linked to the capacity of the donor area and the aggressiveness of the baldness [7-9].
When the operation is performed on a still evolving form of AGA, the real success of this approach should be understood as the durability of the result without resorting to early integrations. It follows once again the need to find a therapeutic balance for these patients before the operation. This stability, if achieved, represents the best requirement for a favorable long-term evolution of a hair transplantation [8-10].
After a systematic review of the literature and based on our clinical experience, we decided to develop this three-steps therapeutic protocol, responding to these essential characteristics:
- demonstrable efficacy on AGA, (but also, as we will report in future works, on other pathologies causing alopecia);
- simplicity of execution and short administration times, to meet the needs of both the doctor and the patient;
- low costs to allow a wider accessibility to the treatment;
- use of techniques and active ingredients of proven efficacy, to guarantee a satisfactory result.
Moreover, this combination of therapies, an alternative to conventional options, allows the physician to follow the patient, step by step, from the initial phase to the first therapeutic achievements. The absence of drugs promotes positive compliance and creates the right conditions for a very good therapeutic adherence further facilitated by the beneficial results obtained, motivating patients to keep their monthly appointments on time.
We are aware in our clinical research activities that these protocols act on molecular mechanisms about which we still know relatively little. Recent work has shown that the hair follicle and dermal white adipose tissue (dWAT) constitute two compartments that, since late embryogenesis, are physiologically coupled. This epithelium/mesenchyme interaction is based on well-defined molecular pathways. In AGA this delicate imbalance is altered, and the miniaturized follicles are then characterized by a decrease in the number of cells within the dermal papilla (DP). This condition does not correspond to a reduction in the number of hair follicle stem cells (HFSC) located in the mesenchyme at the base of the HF. So, two possible causes of the miniaturization associated with the pathology have been proposed: an intrinsic defect of the HFSCs located in the bulge region and an impaired (or weakened) signal from the DP to the HFSC bulge. The inoculation of stem cells in sufficient concentration aims to unlock both the above-mentioned mechanisms by trying to reverse the pathogenesis of hair loss by reactivating the dermal papilla cells or activating the HFSCs of the bulge. This action would be carried out through paracrine signals, in particular cytokines and growth factors such as platelet-derived growth factor (PDGF), vascular endothelial growth factor (VEGF), insulin-like growth factors (IGFBP-1 binding proteins and IGFBP-2), which once secreted stimulate hair follicle stem cells (HFSCs) and niche cells. The effectiveness of the proposed therapy is based on the ability of the implanted cells (ADSc and VSC) to induce the regeneration of the contiguous follicular structures, positively supported by the nutrients and energy provided by the mesotherapy and photobiostimulation sessions. The TRICHOBIOLIGHT® system had already proven to be extremely effective in AGA without additional treatments [28]. Mesotherapy, which constitutes the first phase, allows the direct delivery of the active ingredients into the scalp, at the exact depth to which effect is added the local release of cytokines and growth factors induced by the microtrauma induced by the sequential injections. The benefits of this method which offers the possibility of administering numerous substances at different times and concentrations are widely described in the literature [23-27].
Compared to other proposed therapies, the combination of SEFFIHAIR™ and TRICHOBIOLIGHT® protocol stands out for a series of advantages:
- the administration of more than one single active agent resulted to be safer than the administration of a single one, to avoid the risk of developing intolerance towards the active principle or side effects;
- the use of autologous regenerative treatment enhances the chances of success rate of the following stimulating steps;
- mesotherapy guarantees precise delivery on the affected area, unlike topical administrations of other active ingredients, which must be performed on the entire scalp with a whole series of more or less complex precautions for the patient;
- the adjunctive use of photobiostimulation by a red LED light at 630nm, which has already proven to be very effective in increasing cell receptivity to substances (in this case those conveyed by mesotherapy) present nearby;
- We decided to use the SEFFIHAIR™ kit both for its effectiveness and for the peculiar characteristics of this device, created with the aim of making the entire procedure safe, reproducible and simple both for the expert doctor and for the doctor without previous skills in liposuction, offering the latter a very rapid and effective learning curve.
- This method also allows the regenerative potential of the SVF and ADSC to be used, avoiding the problems due to the difficult and painful injection of micro-fragmented adipose tissue into the scalp.
Consequently, the combined SEFFIHAIR™ and TRICHOBIOLIGHT® protocol proves to be effective and safe precisely because it targets the structures still present in the patient's scalp, without trying to give birth to new hair follicles, and lead to a significant patient satisfaction because the obtained results is made by his own hair, giving an increased sense of self- esteem and confidence that other techniques cannot guarantee.
Conclusions
Hair loss can lead to moderate to severe psychological discomfort and AGA is the most common condition for which countless treatments are now offered to patients, not always effective. As a result of years of studies and research, our protocol, which proposes the combination of three well-known therapies such as autologous regenerative therapy, mesotherapy and photobiostimulation, has proven to be effective in all cases, treated regardless of their severity. This is because both SEFFIHAIR™ and TRICHOBIOLIGHT® act on existing structures that are still able to regenerate, if adequately stimulated, with a significant reduction in side effects. This combined SEFFIHAIR™ and TRICHOBIOLIGHT® protocol has resulted in consistent patient satisfaction, who are often surprised to have achieved undreamed of results in significantly less time than expected. This generates a recovery of self-esteem and confidence that, in our opinion, other techniques cannot guarantee. Among the advantages of this method, we would also like to highlight the strict scheduling in the application of the therapy, guaranteed by the fact that it is performed exclusively by the doctor for the entire duration of the treatment. The absence in the protocol of so-called "suspicious" drugs that a very high percentage of patients are wary, makes the initial approach immediately favorable. Added to this, the patient will have the perception of being carefully assisted by the practitioner followed throughout the entire course of treatment, thus resulting in a consistent psychological reassurance, a factor that can be decisive in this pathology that has major psychological repercussions. We have highlighted how this practice is based on solid scientific foundations. Although many questions concerning the understanding of the mechanisms underlying AGA still fall into a gray area, we believe that this original protocol can filled some therapeutic gaps still present today in the treatment of AGA too often focused on showing success rates and not paying enough attention to the very high rate of therapeutic dropout and subsequent relapse. We also are confident that it offers to stem cell researcher a strong clinical support, which is unquestionably needed today. This multimodal approach provides the physician a therapeutic protocol capable of treating AGA in a broad, complete and, at the same time, highly targeted way. In conclusion, we can therefore state that the combination of SEFFIHAIR™ and TRICHOBIOLIGHT® represents an innovative and effective protocol in the treatment of androgenetic alopecia, with constant and noteworthy results.
References
- Kabir Y, Goh C. Androgenetic alopecia: update on epidemiology, pathophysiology, and treatment. Journal of the Egyptian Women's Dermatologic Society 10 (2013): 107-116.
- Piraccini BM, Alessandrini A. “Androgenetic alopecia,” Giornale Italiano di Dermatologia e Venereologia, 149 (2014): 15-24.
- Kaliyadan F, Nambiar A, Vijayaraghavan S. Androgenetic alopecia: an update. Indian Journal of Dermatology, Venereology, and Leprology 79 (2013): 613-625.
- Zhang Y. Study of the differences on hair follicle between AGA and healthy. Second Military Medical University, Shanghai, China (2016).
- Ludwig E. Classification of the types of androgenetic alopecia (common baldness) occurring in the female sex. Br J Dermatol 97 (1977): 247-254.
- Inui S, Nakajima T, Itami S. Significance of dermoscopy in acute diffuse and total alopecia of the female scalp: review of twenty cases. Dermatology 217 (2008): 333-336.
- Seleit IA, El-Bakry OA, El-Sherif RM. Dermoscopy in hair disorders. Menoufia Med J 27 (2014): 762-765.
- Sharma R, Ranjan A. Follicular Unit Extraction (FUE) Hair Transplant: Curves Ahead. J Maxillofac Oral Surg 18 (2019): 509-517.
- Jimeenez-Acosta F, Ponce-Rodriguez I. Follicular Unit Extraction for Hair Transplantation: An update. Actas Dermosifiliogr 108 (2017): 532-537.
- Kerure AS, Patwardhan N. Complications in Hair Transplantation. J Cutan Aesthet Surg 11 (2018): 182-189.
- Lubart R, Eichler M, Lavi R, et al. Low-energy laser irradiation promotes cellular redox activity. Photomed Laser Surg 23 (2005): 3-9.
- Smith AN, Willis E, Chan VT, et al. Mesenchymal stem cells induce dermal fibroblast responses to injury. Exp Cell Res 316 (2010): 48-54.
- Rossi MAF, Ricci F, Vignoli F, et al. In vitro multilineage potential and immunomodulatory properties of adipose derived stromal/stem cells obtained from nanofat lipoaspirates. CellR4 4 (2016): e2212.
- Martina Rossi, Barbara Roda, Silvia Zia, et al. Characterization of the Tissue and Stromal Cell Components of Micro-Superficial Enhanced Fluid Fat Injection (Micro-SEFFI) for Facial Aging Treatment, Aesthetic Surgery Journal 40 (2020): 679-690.
- Ogura F, Wakao S, Kuroda Y, et al. Human adipose tissue possesses a unique population of pluripotent stem cells with nontumorigenic and low telomerase activities: potential implications in regenerative medicine. Stem Cells Dev 23 (2014): 717-728.
- Tesauro P, Trivisonno A, Gennai A, et al. Hair transplantation in cicatricial alopecia: The role of autologous fat transfer. International Journal of Regenerative Medicine 3 (2020): 11.
- Bernardini FP, Gennai A. Superficial enhanced fluid fat injection for volume restoration and skin regeneration of the periocular aesthetic unit. An improved fat grafting technique to enhance the beauty of the eye. JAMA Plastic Facial Surgery 18 (2016): 1.
- Gennai A, Tesauro P, Colli M et al. Infranatant Portion of Microfragmented Adipose Tissue: A Promising Source of SVF for the Management of Androgenic Alopecia. Int J Regenr Med 12 (2021): 1-7
- Gennai A, Bovani B, Colli M et al. Comparison of Harvesting and Processing Technique for Adipose Tissue Graft: Evaluation of Cell Viability. Int J Regen Med 4 (2021): 2-5.
- Gennai A, Bernardini FP, Baldessin M et al. Evaluation of The Viability and Phenotipe of Adipose Derived Cells Harvested Using Different Harvesting and Processing Procedures: A Pilot Study. J stem cells clin pract 2 (2022): 101.
- Antonio L, Bartoletti E, Gennai A. The role of regenerative medicine in androgenetic alopecia treatment: an uncontrolled clinical trial. Aesthetic Medicine 8 (2022): 457.
- El-Khalawany M, Rageh MA, Elnokrashy I et al. Efficacy of autologous stroll vascular fraction injection in the treatment of androgenic alopecia. Arch Dermatol Res
- Khorosandi K, Hosseinzadeh R, Abrahamse H, et al. Biological Responses of Stem Cells to Photobiomodulation Therapy. Curr Stem Cell Res Ther 15 (2020): 400-413.
- Moftah N, Moftah N, Abd-Elaziz G, et al. Mesotherapy using dutasteride-containing preparation in treatment of female pattern hair loss: Photographic, morphometric and ultrustructural evaluation. J Eur Acad Dermatol Venereol 27 (2013): 686-693.
- Sobhy N, Aly H, El Shafee A, et al. Evaluation of the effect of injection of dutasteride as mesotherapeutic tool in treatment of androgenetic alopecia in males. Our Dermatol Online 4 (2013): 40-45.
- Barbareschi M. The use of minoxidil in the treatment of male and female androgenetic alopecia: a story of more than 30 years. G Ital Dermatol Venereol 153 (2018): 102-106.
- Gajjar PC, Mehta HH, Barvaliya M, et al. Comparative Study between Mesotherapy and Topical 5% Minoxidil by Dermoscopic Evaluation for Androgenic Alopecia in Male: A Randomized Controlled Trial. Int J Trichology 11 (2019): 58-67.
- Abdallah MA, El-Zawahry KA, Besar H. Mesotherapy using dutasteride-containing solution in male pattern hair loss: A controlled pilot study. J Pan Arab Leag Dermatol 20 (2009): 137-145.
- Piccolo D, Crisman G, Conforti C et al. Trichobiolight: A new, effective protocol in the treatment of androgenetic alopecia and telone effluvium. Dermatol There 34 (2021): e14799.
