Virtual Surgical Planning and Pre-operative Simulation for Maxillofacial Reconstruction with 3D Printing Technology in Tumor-affected Facial Deformity
Article Information
Gyeong-Man Kim1,2*, Masreshaw Demelash Bayleyegn1, Song-Jung Kim2
1Center of Biomedical Engineering, Addis Ababa Institute of Technology, Addis Ababa University, Addis Ababa, Ethiopia
2Department of Radiology, Myungsung Medical College, Addis Ababa, Ethiopia
*Corresponding Author: Gyeong-Man Kim, Center of Biomedical Engineering, Addis Ababa Institute of Technology, Addis Ababa University, Addis Ababa, Ethiopia
Received: 21 June 2022; Accepted: 05 July 2022; Published: 12 July 2022
Citation: Gyeong-Man Kim, Masreshaw Demelash Bayleyegn, Song-Jung Kim. Virtual Surgical Planning and Pre-operative Simulation for Maxillofacial Reconstruction with 3D Printing Technology in Tumor-affected Facial Deformity. Journal of Radiology and Clinical Imaging 5 (2022): 23-29.
View / Download Pdf Share at FacebookAbstract
Background: The high anatomical complexity of maxillofacial defects caused by tumor can pose a formidable challenge for clinicians when designing an appropriate plan for surgical reconstruction. The intention of this work was to restore the complex anatomy with maximum possible facial functionality and aesthetics of the patient. Based on the medical images generated by Computed Tomography (CT) scan an optimal therapeutic planning for complex maxillofacial reconstruction was designed.
Method: Firstly, the volumetric data sets were carefully evaluated and deeply inspected for accurate diagnosis. Regarding 3D visualization of the CT scan images 3D virtual models for regions of interest were created using special software of 3D Slicer. Using the resulting 3D virtual models a well-defined virtual surgical planning was generated for multiple surgical procedures, including the osteotomies for bone defects, harvesting autogenous bone graft and creating a customized implant.
Results: The relevant patient-specific anatomical models for real surgery were translated into the 3D printed physical models, with which the surgeons can rehearse the surgery before coming into the operating room. Precisely defined multiple surgeries for complex maxillofacial reconstruction were proposed in this research that could be transferred to the real-time surgery.
Conclusions: The proposed surgical approach will be beneficial both for the surgeons and patient, including improvement in surgical precision and outcomes, reduction in operating time, as well as understanding surgical procedures and decision making etc. In addition, it is worth noting that such an appropriate methodology with free, open source software is highly adaptable in medical education, especially for lowresource countries.
Keywords
<p>3D Medical images; Computed Tomography (CT); Virtual Surgical Planning (VSP); 3D Printing; Pre-operative simulation; Virtual model; Physical model; Maxillofacial reconstruction</p>
3D Medical images articles; Computed Tomography (CT) articles; Virtual Surgical Planning (VSP) articles; 3D Printing articles; Pre-operative simulation articles; Virtual model articles; Physical model articles; Maxillofacial reconstruction articles
3D Medical images articles 3D Medical images Research articles 3D Medical images review articles 3D Medical images PubMed articles 3D Medical images PubMed Central articles 3D Medical images 2023 articles 3D Medical images 2024 articles 3D Medical images Scopus articles 3D Medical images impact factor journals 3D Medical images Scopus journals 3D Medical images PubMed journals 3D Medical images medical journals 3D Medical images free journals 3D Medical images best journals 3D Medical images top journals 3D Medical images free medical journals 3D Medical images famous journals 3D Medical images Google Scholar indexed journals Computed Tomography articles Computed Tomography Research articles Computed Tomography review articles Computed Tomography PubMed articles Computed Tomography PubMed Central articles Computed Tomography 2023 articles Computed Tomography 2024 articles Computed Tomography Scopus articles Computed Tomography impact factor journals Computed Tomography Scopus journals Computed Tomography PubMed journals Computed Tomography medical journals Computed Tomography free journals Computed Tomography best journals Computed Tomography top journals Computed Tomography free medical journals Computed Tomography famous journals Computed Tomography Google Scholar indexed journals Virtual Surgical Planning articles Virtual Surgical Planning Research articles Virtual Surgical Planning review articles Virtual Surgical Planning PubMed articles Virtual Surgical Planning PubMed Central articles Virtual Surgical Planning 2023 articles Virtual Surgical Planning 2024 articles Virtual Surgical Planning Scopus articles Virtual Surgical Planning impact factor journals Virtual Surgical Planning Scopus journals Virtual Surgical Planning PubMed journals Virtual Surgical Planning medical journals Virtual Surgical Planning free journals Virtual Surgical Planning best journals Virtual Surgical Planning top journals Virtual Surgical Planning free medical journals Virtual Surgical Planning famous journals Virtual Surgical Planning Google Scholar indexed journals 3D Printing articles 3D Printing Research articles 3D Printing review articles 3D Printing PubMed articles 3D Printing PubMed Central articles 3D Printing 2023 articles 3D Printing 2024 articles 3D Printing Scopus articles 3D Printing impact factor journals 3D Printing Scopus journals 3D Printing PubMed journals 3D Printing medical journals 3D Printing free journals 3D Printing best journals 3D Printing top journals 3D Printing free medical journals 3D Printing famous journals 3D Printing Google Scholar indexed journals Pre-operative simulation articles Pre-operative simulation Research articles Pre-operative simulation review articles Pre-operative simulation PubMed articles Pre-operative simulation PubMed Central articles Pre-operative simulation 2023 articles Pre-operative simulation 2024 articles Pre-operative simulation Scopus articles Pre-operative simulation impact factor journals Pre-operative simulation Scopus journals Pre-operative simulation PubMed journals Pre-operative simulation medical journals Pre-operative simulation free journals Pre-operative simulation best journals Pre-operative simulation top journals Pre-operative simulation free medical journals Pre-operative simulation famous journals Pre-operative simulation Google Scholar indexed journals Virtual model articles Virtual model Research articles Virtual model review articles Virtual model PubMed articles Virtual model PubMed Central articles Virtual model 2023 articles Virtual model 2024 articles Virtual model Scopus articles Virtual model impact factor journals Virtual model Scopus journals Virtual model PubMed journals Virtual model medical journals Virtual model free journals Virtual model best journals Virtual model top journals Virtual model free medical journals Virtual model famous journals Virtual model Google Scholar indexed journals Physical model articles Physical model Research articles Physical model review articles Physical model PubMed articles Physical model PubMed Central articles Physical model 2023 articles Physical model 2024 articles Physical model Scopus articles Physical model impact factor journals Physical model Scopus journals Physical model PubMed journals Physical model medical journals Physical model free journals Physical model best journals Physical model top journals Physical model free medical journals Physical model famous journals Physical model Google Scholar indexed journals Maxillofacial reconstruction articles Maxillofacial reconstruction Research articles Maxillofacial reconstruction review articles Maxillofacial reconstruction PubMed articles Maxillofacial reconstruction PubMed Central articles Maxillofacial reconstruction 2023 articles Maxillofacial reconstruction 2024 articles Maxillofacial reconstruction Scopus articles Maxillofacial reconstruction impact factor journals Maxillofacial reconstruction Scopus journals Maxillofacial reconstruction PubMed journals Maxillofacial reconstruction medical journals Maxillofacial reconstruction free journals Maxillofacial reconstruction best journals Maxillofacial reconstruction top journals Maxillofacial reconstruction free medical journals Maxillofacial reconstruction famous journals Maxillofacial reconstruction Google Scholar indexed journals Stereolithography articles Stereolithography Research articles Stereolithography review articles Stereolithography PubMed articles Stereolithography PubMed Central articles Stereolithography 2023 articles Stereolithography 2024 articles Stereolithography Scopus articles Stereolithography impact factor journals Stereolithography Scopus journals Stereolithography PubMed journals Stereolithography medical journals Stereolithography free journals Stereolithography best journals Stereolithography top journals Stereolithography free medical journals Stereolithography famous journals Stereolithography Google Scholar indexed journals Fused Deposition Modeling articles Fused Deposition Modeling Research articles Fused Deposition Modeling review articles Fused Deposition Modeling PubMed articles Fused Deposition Modeling PubMed Central articles Fused Deposition Modeling 2023 articles Fused Deposition Modeling 2024 articles Fused Deposition Modeling Scopus articles Fused Deposition Modeling impact factor journals Fused Deposition Modeling Scopus journals Fused Deposition Modeling PubMed journals Fused Deposition Modeling medical journals Fused Deposition Modeling free journals Fused Deposition Modeling best journals Fused Deposition Modeling top journals Fused Deposition Modeling free medical journals Fused Deposition Modeling famous journals Fused Deposition Modeling Google Scholar indexed journals
Article Details
Abbreviations:
3D: Three dimensional; ABS: Acrylonitrile Butadiene Styrene; PLA: Polylactic Acid; CAD: Computer-Aided Design; CAM: Computer-Aided Manufacturing; CT: Computed Tomography; DICOM: Digital Imaging And Communications in Medicine; FDM: Fused Deposition Modeling; IV: Intravenous; MRI: Magnetic Resonance Imaging; STL: Stereolithography or Standard Tessellation Language; ROI: Region of Interest; ZMC: Zygomaticomaxillary Complex; DVR: Direct Volume Rendering; VSP: Virtual Surgical Planning
1. Introduction
Extensive maxillofacial defects, which can be caused ether by secondary to trauma, ablative tumor resection, and refractory infection or by congenital/developmental deformities, lead to the abnormal contours of the face and functional disorders [1,2]. The face is vital for human identity, and plays a crucial role in the individual's social interaction. Thus, individuals with even minor facial deformities are at risk for experiencing social and psychological stress and thus for affecting their quality of life adversely [3,4].
Reconstruction of maxillofacial defects, especially the restoration of aesthetics and function, is always a difficult task, because the bone lesions that lead to deformities of the normal maxillofacial anatomy are complex in general [5,6]. There are several factors which are needed to be considered in the planning of these reconstructions, including the anatomic diversity of the bone defects and the complexity of the oral function based on mandibular movement, and normal facial appearance. All these aspects are important for maintaining quality of life.
The demand for performing accurate and low risk surgery, which is still a great challenge for the surgeons at the present time, has led to the discovery of computer-based surgical planning [7,8]. Nowadays computer-assisted surgery is becoming more and more important in the field of oral and maxillofacial surgery, because of the possibility of simulating almost every scenario imaginable with a high accuracy [9]. Currently, the great challenges are not only how best to 3D display the volumetric data sets generated by CT and MRI scan, but also how accurate to represent those data into the tangible, physical 3D objects [10]. 3D printing enables the graspable 3D objects to be created from volumetric images [11-13].
The purpose of this study was twofold. The first purpose was to develop a Virtual Surgical Plan (VSP) as well as to design the customized intraoperative surgical tools with greater accuracy. That helps doctors in taking right decisions even before performing the surgery. The second was to simulate the surgery assisted by 3D printed patient-specific anatomical models as templates with customized implant and autogenous bone graft for training residents as well as experienced surgeons prior to entering the operating room, by which it will be produced more predictable outcomes and reduced duration of surgery and surgical risks significantly.
We transfer the accurate VSP with 3D virtual models and pre-operative simulation with 3D physical models to the real-surgery that will perfectualize an entire surgical plan for maxillofacial reconstruction, which results in improved outcome meaningfully.
2. Methods
2.1 Patient
A 24 year-old-male presented with a gradually enlarging, refractory swelling on the left side of his face, leading to obvious facial asymmetry. The patient denied any pain or numbness 3 associated with this swelling on palpation. The teeth in the area of interest were nonmobile, non-painful. The histopathological analysis from incisional biopsy showed that this was a benign bone lesion, suggesting the provisional diagnosis of an ossifying fibroma, fibrous dyplasis and ameloblastoma. For accurate diagnosis and proper treatment plan of the lesion the maxillofacial computed tomography (CT) scan was performed to obtain valuable information regarding the size and extent as well as the localization of the pathological patient’s condition affected by tumor.
2.2 Medical image processing for 3d modelling and 3d printing
Patient underwent the preoperative maxillofacial and pelvis Computed Tomography (CT) scans (GE Brightspeed Edge 8, USA). For the exams, an image acquisition protocol was used 1 mm for increment between slices and 1 mm of thickness, zero degrees of gantry inclination of the orientation. The CT scan images of the maxillofacial region (257 images) and the pelvis region (585 images) were stored in Digital Imaging and Communications in Medicine (DICOM) file format, which is the international standard format for medical imaging.
To 3D visualize the CT imaging data as in DICOM file, the images were imported into 3D Slicer (version 4.10.1) software, which is a free, open-source software package for 3D visualization of medical images and image analysis. This software provides the advanced 3D visualization functionalities such as surface rendering as well direct volume rendering. The uploaded images were processed, edited and further segmented to perform the accurate 3D visualization of patient’s anatomical particularities on the basis of desired tissues and pathophysiology in the form of 3D rendered virtual models through volume rendering in 3D Slicer software.
In general, 3D virtual models generated in 3D Slicer software are surface models that are building up out of triangle meshes. 3D virtual models as saved in STL (STereoLithography or Standard Triangulation Language) file format were further edited and refined using Meshmixer® 3.5 version (Autodesk, Inc., USA) to generate the error-free 3D printable meshes, to remove all artifacts, and to minimize surface imperfections amendable to 3D printing. Meshmixer is also an open-source software for working with triangle meshes.
All the resulting 3D virtual models completed in Meshmixer were saved once again in STL file format and loaded into the Flashprint software (FlashForge Corporation, Zhejiang, China) to set up parameters such as resolution, temperature, wall thickness, support structure, etc. for 3D printing. At the final stage, the resulting STL files were exported to a Dreamer NX 3D Printer (FlashForge Corporation, China), Fused Deposition Modelling (FDM) ultimately to fabricate patient-specific, highly accurate 3D anatomical models as 3D physical models.
The materials used for 3D printing were Polylactic Acid (PLA), and Acrylonitrile Butadiene Styrene (ABS) filaments from Material 4print, Germany or Polymaker, China. Printing took 4 around 8 to 10 hours for a typical physical model, e.g., patient’s skull, however this was variable depending on the size of the model required.
3. Results
3.1 3D Visualization of CT scan images and creation of virtual 3D models
To 3D visualize the CT imaging data as in DICOM file, the images were firstly imported into 3D Slicer Software. Once uploaded, the images were processed, manipulated and edited to perform the accurate 3D visualization of patient’s anatomical particularities on the basis of desired tissues and pathophysiology in the form of 3D virtual models through volume rendering [14]. By properly choosing the Hounsfield ranges, different types of tissues were selected the face as soft tissue and the facial skeleton as hard tissue, by which in turn 3D virtual models could be created on the computer (Figure 1) [15,16]. As clearly seen Figure 1c, the lesion presented irregular osseous defects, including a partial rupture of zygomatic arch and also extensive resorption of zygoma and coronoid. In addition, an expansive mass of cortical plate on the ramus region in the mandible was identified in this 3D virtual model.
Based on the diagnosis with CT scan and histological analysis, it was decided to undertake multiple surgical procedures, including the management of damaged Zygomaticomaxillary Complex (ZMC) and a segmental mandibulectomy, i.e. a unilateral partial resection of mandible affected by tumor, and ultimately followed by maxillofacial reconstruction.
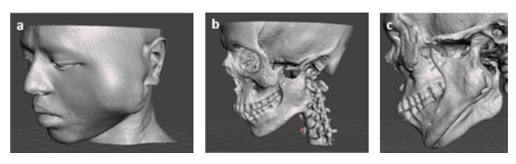
Figure 1: Virtual 3D models: a) skin model taken from lower Hounsfield unit, b) facial skeleton model taken from higher Hounsfield unit, and c) close inspection of the tumor area.
3.2 VSP with virtual 3D models on the computer screen
3.2.1 Procedure 1: Repair planning for Zygomaticomaxillary Complex (ZMC) defect
Among the multiple surgical procedures, at first we started to manage the damages in ZMC. Dedicated 3D virtual model of facial skeleton (Figure 1c) demonstrated that free-floating rest of the damaged zygomatic arch was retained barely to the temporal bone. After the careful evaluation of the residual maxillary bone, a zygomatic implant was designed virtually to restore the facial contour and to balance the zygomatic bone as cheekbone with the rest of the face. At first it was defined the resection line for osteotomy of bony defects on ZMC (Figure 2). The damaged areas with sufficient margin in the ZMC were resected parallel to the zygomatic arch, where eventually will be used the basement for placing the implant (Figure 3).
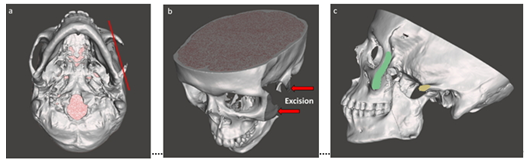
Figure 2: Preparation for osteotomy: a) defining the cutting line, b) excision of Zygomaticomaxillary Complex (ZMC) and c) prepared basement for placing the implant.
The resulting zygomatic Implant functions just like a real bone, which integrates with the structures of the face. As a consequence, it provides the patient with an exceptional anatomical fit and enhanced aesthetic results.
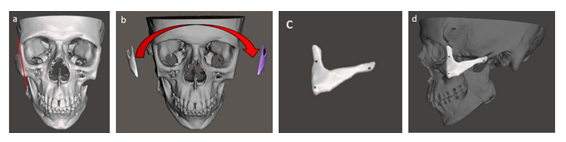
Figure 3: Creating a zygomatic implant: a) defining the cutting line on the healthy side from symmetric Zygomaticomaxillary Complex (ZMC) basement, b) mirroring an image on the defect side, c) creating implant and d) placing the implant on the prepared basement with three fixing holes.
3.2.2 Procedure 2: Repair planning for mandible
To remove the tumor-affected area with sufficient tumor-clear margin where possible in the ramus completely [18], the precise resection lines were acquired on the virtual 3D model (Figure 4a).
To assist surgeon to perform mandibulectomy easily, conveniently and accurately, a cutting guide was designed with the Boolean operations in the Meshmixer software, according to the well-defined resection lines (Figure 4b). As a consequence, the cutting guide exhibited the identical surface topology of the mandible that is a prerequisite for accurate positioning the cutting guide on the mandible.
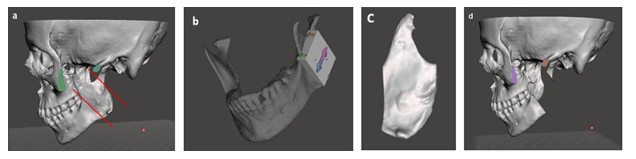
Figure 4: a) Virtual 3D model as a defect resection template, exhibiting the defined resection lines, b) mandibulectomy with cutting guide, c) the resected mandible and d) virtual 3D model of the resected both mandible and ZMC.
In addition, the surface of the cutting guide was marked with arrows, where the arrows on the upper surface indicate its exact position on the ramus to be placed – between both sides of coronoid process, whereas a double sided arrow indicates where the cutting to be carried out in the surgery. Once the cutting guide was adapted at an exact place onto the mandible and then secured with 2 cortical locking screws on the posterior border of the mandibular ramus, the segmental mandibulectomy was performed using a sagittal saw along the proximal and distal edges of the mandible cutting guide. The resected part from the mandible will be used as template for harvesting of graft segment from a donor site (Figure 4c). A careful evaluation on the 3D virtual models clearly indicated that the condyle remained intact. In general, accurate repositioning of the condylar head in the temporomandibular fossa is difficult to maintain the maxillomandibular relationship, because false condyle position and condylar disk displacement often induce temporomandibular joint dysfunction associated with malocclusion [19,20]. Therefore, it was decided that the intact mandibular condyle should be preserved in its initial position without further effort (Figure 4d).
3.2.3 Procedure 3: Preparation of autogenous bone graft
Bony reconstruction of the discontinuity in mandible can be achieved by different types of autologous bone grafts [21,22]. To decide which bone graft is best to restore bony continuity and facial contour, the settings at the defect site are crucial, e.g., the defect size, location, need for soft tissue and status of the blood vessels. In addition, the availability of donor sites, e.g., adequate amount of bone volume, is also important [23]. Through the evaluation to the size of resected bone, the defect size was relatively large in dimension with 3 × 4 × 1 cm3 (height, length and width respectively) but no additional tissue was needed. Therefore, for achieving sufficient anatomical bulk being replaced the continuity defect in mandible we decided to harvest the bone graft from patient’s right-sided pelvis (Figure 5) [24].
To precisely define the area for harvesting the bone graft on the ilium two surgical tools were patient-specifically designed which were fitted uniquely at the planned position with certain orientation (Figure 5a). The left one was an iliac crest cutting guide to harvest the bone graft. Such cutting guide included all necessary information, such as graft size, angulation, and osteotomies, according to the virtual plan. The other in the right side was a jig, which was used to assist the iliac crest cutting guide for locating at the exact position. Ultimately, the iliac crest bone graft was resected virtually as guided by the iliac crest resection guide, and then harvested, as shown in Figure 5b.
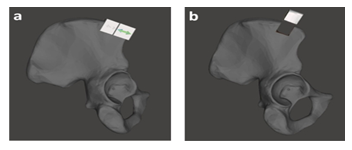
Figure 5: a) Iliac bone segmented from pelvis, a) Attaching the cutting guide with jig together at the iliac bone and b) removing the jig to fix the cutting guide only and harvesting bone for grafting.
Once the iliac crest bone graft was released, that should be shaped precisely to match with the discontinuity (Figure 6). If reassembled all the parts together in planed position with right angulation, by which it was achieved excellent virtual mandible symmetry as shown in Figure 7c.
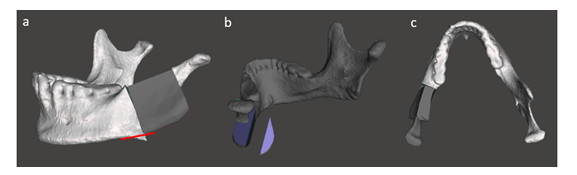
Figure 6: a) Refining the mandible, b) the harvested bone graft and c) adjusting the symmetric according to the right part of healthy side.
According to the stress states on the mandible during mastication the number, orientation and length of the osteosythesis plates were determined: the one was at the area where the tensile stress was dominated, on the other hand the other was at the location where the compression was mostly subjected (Figure 7a) [25,26]. For the best possible 3D adaptation of the osteosynthesis plates throughout the rest of mandible stump, the bone graft and the preserved condyle the osteosynthesis plates were pre-bent, whose curvature and contour were adjusted preoperatively on the virtual 3D mandible model and trimmed for a customized fit ready for the real surgery (Figure 7b).
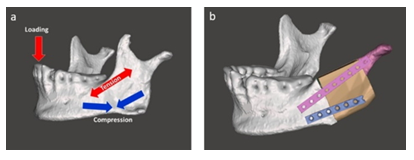
Figure 7: a) Stress states in the mandible and b) attaching the osteosynthesis plates.
Thus, the patient-specifically pre-bent osteosythesis plates exhibited an anatomical shape precisely fit to the maxillofacial contour without further bending or adjustment at the time of surgery, so that the surgical time will be reduced significantly.
3.3.4 Pre-surgical simulation with 3D printed physical models
Once VSP with 3D virtual models was accomplished, the relevant anatomical models of the regions of interest being involved in the real surgery were then printed out in 3D, under the following aspects; firstly to complement and improve the VSP in the feasibility aspect, secondly to simulate a real surgery in the accuracy and effectively aspect, lastly to prepare the surgical tools and implant in the technological aspect.
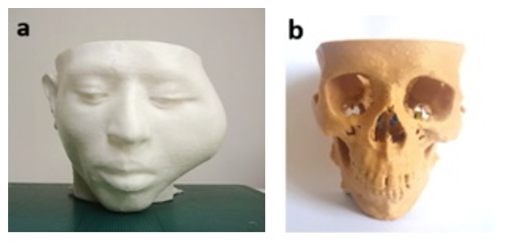
Figure 8: 3D printed physical models: a) for patient’s head and b) for skull.
With the 3D printed physical models (Figure 8), on anywhere without presenting the patient surgeons can simply and precisely recognize the lesion affected by tumor in a 3D environment, and also in any time use them to discuss within a surgical team. To simulate for osteotomies both mandible and ZMC the 3D printed anatomical models were used as surgical templates. Firstly, to simulate the management of defects on ZMC we fabricated the zygomatic implant by 3D printer (Figure 9a), hereby used the STL file created in Figure 4c. The screw hole locations on the zygomatic implant were used to position the implant at the appropriate location exactly (Figure 9b). Fixation was performed with three osteosynthesis screws through the existing holes on the prepared ZMC.
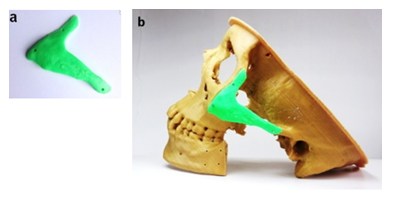
Figure 9: a) 3D printed zygomatic implant with three fixation holes and b) implant fixation on the osteotomized basement of ZMC.
As the next, to simulate mandibulectomy we produced a cutting guide and fixed it temporarily to the lateral side of mandible using two osteosynthesis screws through the existing holes, and then the defect area was resected using an oscillating saw, along the both sides of cutting guide as marked with arrows, as shown in Figure 10.

Figure 10: Attachment of cutting guide for surgical simulation of mandibulectomy.
For harvesting the autogenous bone graft from iliac crest, the iliac crest of the part pelvis was 3D printed in the real size as used a simulation template (Figure 11). The cutting guide combined with jig together was fixed onto the iliac crest and then removed the jig by keeping the cutting guide position at the defined position (Figure 11b), so that one can precisely resect the iliac crest to harvest the bone graft with exact volume without having to wait for the exact measurements.

Figure 11: Surgical simulation for harvesting bone graft from iliac bone: a) virtual 3D model, b) real-sized 3D printed physical model for exact positioning cutting guide on the iliac crest after removal of jig.
The harvested bone graft was refined and reshaped in vitro to adapt to the discontinuity in the mandible. The customized osteosynthesis plates were primarily adjusted to the predefined orientation using the contour line as designed in the virtual planning, and then fixed with 9 titanium screws to bridge between the residual mandible stump, iliac bone graft and condyle, for achieving the best possible restoration of the patient’s appearance and oral function (Figure 12).
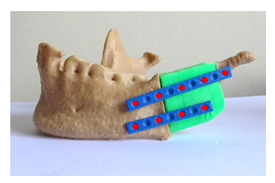
Figure 12: Surgical simulation of mandible reconstruction (red dots are marked for screws).
Accurately 3D printed anatomical model of the resected mandible and zygomatic bone were used as surgical templates to precisely replace the maxillofacial bone defects both with iliac crest bone graft and zygomatic implant by adapting the pre-bent osteosynthesis plates with specific length of screws (Figure 13).
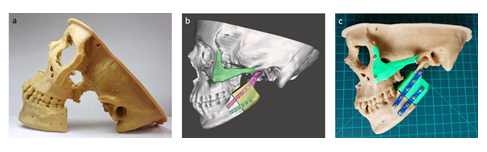
Figure 13: Maxillofacial reconstruction; a) real-sized 3D printed physical model of osteomized skull, b) 3D virtual model and c) reconstructed 3D physical model.
After adjusting and re-validating in a pre-surgical simulation with 3D physical models by doing repetitive practice, the entire surgical procedure will be perfectualized and fully understood by surgical teams as a training platform in a non-threatening environment before they practice on patients. Finally, the expected surgical outcome was presented with a hand-on 3D printed model (Figure 14), which can be shown to the patient for his decision making to accept this kind of surgical procedure.

Figure 14: 3D printed physical model for the expected outcome after surgery.
4. Discussion
Recent advances in computer graphics for medical imaging make interactive volume visualization both in virtual and physical modes possible. [27] Using a combination of 3D virtual modelling and VSP it is possible [28]
- to increase the accuracy of the surgical procedure significantly,
- to decrease the necessity for intraoperative trial and error drastically,
- to design the customized implant and surgical tools precisely,
- to improve the predictability of surgical outcomes effectively, and
- to reduce the overall duration of surgery efficiently.
Although the virtual 3D models can provide the surgeon with interactive 3D view of the detailed anatomy, they are strictly confined on the computer screen, so their effects on the preparation of a successful surgical plan are strictly limited. Through in vitro simulation with tactile 3D physical models, the surgeon can foresee intra- operative complications in both anatomical and technical aspects of the procedure, e.g., how to handle the patient’s anatomy for accessing the tumor and performing the osteotomy, which may result in improved intraoperative precision with the reduced operating time. In addition, surgical simulations involving the life-size 3D physical models allow any trainees, such as novices, residents and medical students to learn the surgery with the basic operative steps of specific procedures, enabling effective training in a safe environment to significantly improve their knowledge and skills [29-31].
Furthermore, in the complicated cases, where the patients undergoing new, uncommon or complex multiple surgeries, 3D physical models are inevitable and beneficial with, by which it increases surgeon’s self-confidence approaching difficult anatomies competently with new medical devices, which would be better accommodated the patient’s anatomy. To achieve the best possible facial contour in terms of esthetics and functional recovery, which are the ultimate goal of maxillofacial reconstruction and thus considered as a measure of evaluation for the clinical outcomes, the need for precise virtual surgical planning and pre-surgical simulation with the life-size 3D printed models cannot be over emphasized.
5. Conclusions
Virtual surgical planning with 3D virtual models can be used to manage almost every scenario that could arise during the real-time surgical procedure as well as to design the customized implant and intraoperative surgical tools with greater accuracy. In addition to that, pre-operative simulation with 3D physical models can lead to optimize the surgical intervention for complex maxillofacial defects that has the potential to improve outcomes in the shortest possible surgical times and the fewest number of surgical procedures. When resources are limited, these methods can be simulated with free and open source software, enhancing educational opportunities and increasing confidence of the operating teams.
Acknowledgements
Not applicable.
Funding
There was no funding source for the research reported.
Availability of data and materials
All data generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors’ contributions
GMK primarily conceived the study and drafted the manuscript. All authors contributed to revising of the manuscript and read and approved the final version.
Ethics approval and consent to participate
This study was reviewed by the Institutional Review Board (IRB) of Myungsung Medical College, Addis Ababa, Ethiopia and was approved after thorough deliberation.
Consent for publication
Not applicable.
Competing interests
The authors declare no conflict of interest.
References:
- Essig H, Rana M, Kokemueller H, et al. Pre-operative planning for mandibular reconstruction - a full digital planning workflow resulting in a patient specific reconstruction. Head Neck Oncol 3 (2011): 45-51.
- Maciej P, Banaszewski J, Pastusiak T, et al. Mandibular reconstruction with osteo-cutaneous free flaps in a patient after extensive surgery supported with 3d printed models. Integr Oncol 5 (2016): 5:3.
- Pomahac B, Pribaz J, Eriksson E, et al. Three patients with full facial transplantation. N Engl J Med 366 (2012): 715-722.
- Kumamaru KK, Sisk GC, Mitsouras D, et al. Vascular communications between donor and recipient tissues after successful full face transplantation. Am J Transplant 3 (2014): 711-719.
- Foster BL, Ramnitz MS, Gafni RI, et al. Rare Bone Diseases and Their Dental, Oral, and Craniofacial Manifestations. JDR Clinical Research Suplement 93 (2014): 6, 7S-19S.
- Marmulla R, Niederdellmann H. Computer-assisted bone segment navigation. J Cranio- Maxillofac Surg 26 (1998): 347-359.
- Pratt R, Deprest J, Vercauteren T, et al. Computer-assisted surgical planning and intraoperative guidance in fetal surgery: a systematic review. Prenatal Diagnosis 35 (2015): 1159-1166.
- Xia J, Horace HS, NabilSamman LP, et al. Computer-assisted three-dimensional surgical planning and simulation: 3D virtual osteotomy. Int J Oral Max Surg 29 (2000): 11-17.
- Sukegawa S, Kanno T, Furuki Y. Application of computer-assisted navigation systems in oral and maxillofacial surgery. Japanese Dental Science Review 54 (2018): 139-149.
- Rengier F, Mehndiratta A, von Tengg-Kobligk H, et al. 3D printing based on imaging data: review of medical applications, Int J CARS 5 (2010): 335-341.
- Chepelev L, Wake N, Ryan J, Althobaity W, et al. Radiological Society of North America (RSNA) 3D printing Special Interest Group (SIG): guidelines for medical 3D printing and appropriateness for clinical scenarios. 3D Print Med 4 (2018): 11.
- Mitsouras D, Liacouras PC, Imanzadeh A, et al. Medical 3D printing for the radiologist. Radiographics 35 (2015): 1965-1988.
- Mitsouras D, Liacouras PC, Wake N, et al. RadioGraphics Update: Medical 3D Printing for the Radiologist. Radiographics. 40 (2020): e21-e23.
- Zang Q, Eagleson R, Peters TM. Volume Visualization: A technical overview with a focus on medical applications. Journal of Digital Imaging 24 (2011): 640-664.
- Salgado R, Mulkens T, Bellinck P, et al. Volume Rendering in Clinical Practice. A Pictorial Review. JBR–BTR 86 (2003): 215-220.
- Mitsouras D, Liacouras P, Imanzadeh A, et al. Medical 3D Printing for the Radiologist. RadioGraphics 35 (2015): 1965-1988.
- Zhang X, Zhang K, Pan Q, et al. Three-dimensional reconstruction of medical images based on 3D slicer. Journal of Complexity in Health Sciences 2 (2019): 1-12.
- Mohiyuddin SMA, Padiyar BV, Suresh TN, et al. Clinicopathological study of surgical margins in squamous cell carcinoma of buccal mucosa. World Journal of Otorhinolaryngology-Head and Neck Surgery 2 (2016): 17-21.
- Funayama A, Kojima T, Yoshizawa M, et al. A simple technique for repositioning of the mandible by a surgical guide prepared using a three-dimensional model after segmental mandibulectomy. Maxillofacial Plastic and Reconstructive Surgery 39 (2017): 16.
- Ellis III E, Walker RV. Treatment of Malocclusion and TMJ Dysfunction Secondary to Condylar Fractures. Craniomaxillofac Trauma Reconstr 2 (2009): 1-18.
- Ferreira JJ, Zagalo CM, Oliveira ML, et al. Mandible reconstruction: History, state of the art and persistent problems. Prosthetics and Orthotics International 39 (2015): 182-189.
- Wilkman T, Husso A, Lassus P. Clinical Comparison of Scapular, Fibular, and Iliac Crest Osseal Free Flaps in Maxillofacial Reconstructions. Scandinavian Journal of Surgery 108 (2019): 76-82.
- Carvalho PS, Carvalho MC, Ponzoni D. Reconstruction of alveolar bone defect with autogenous bone particles and osseointegrated implants: histological analysis and 0 years monitoring. Ann Maxillofac Surg 51 (2015):135-139.
- Chepelev L, Hodgdon T, Gupta A, et al. Medical 3D printing for vascular interventions and surgical oncology: a primer for the 2016 Radiological Society Of North America (RSNA) hands-on course in 3D printing. 3D Print Med 2 (2016): 5.
- Meyer C, Kahn JL, Boutemi P, et al. Photoelastic analysis of bone deformation in the region of the mandibular condyle during mastication. J Craniomaxillofac Surg 30 (2002): 160-169.
- Meyer C, Sehir L, Boutemi P. Experimental evaluation of three osteosynthesis devices used for stabilizing condylar fractures of the mandible. J Craniomaxillofac Surg 34 (2006): 173-181.
- Mazzoni S, Badiali G, Lancellotti L, et al. Simulation guided navigation: a new approach to improve intraoperative three-dimensional reproducibility during orthognathic surgery. J Craniofac Surg 21 (2010): 1698-1705.
- Battulga B, Konishi T, Tamura Y, et al. The effectiveness of an interactive 3- dimensional computer graphics model for medical education. Interact J Med Res 1 (2012): e2.
- de Montbrun SL, MacRae H. Simulation in Surgical Education. Clin Colon Rectal Surg 25 (2012): 156-165.
- Valverde I, Gomez G, Coserria JF, et al. 3D printed models for planning endovascular stenting in transverse aortic arch hypoplasia. Catheter Cardiovasc Interv 85 (2015): 1006-1012.
- Valverde I. Three-dimensional printed cardiac models: applications in the field of medical education, cardiovascular surgery, and structural heart interventions. Rev Esp Cardiol 70 (2017): 282-291.
