Using Cognitive Event-Related Potentials in the Management of Alcohol Use Disorder: Towards an Individual Approach
Article Information
Anaïs Ingels1, Lauriane Fabry1, Catherine Hanak1, Florence Hanard1, Hendrik Kajosch1, Charles Kornreich1, Salvatore Campanella1*
1Laboratoire de Psychologie Médicale et d’Addictologie, ULB Neuroscience Institute (UNI), CHU Brugmann-Université Libre de Bruxelles (U.L.B.), Belgium
*Corresponding Author: Salvatore Campanella, PhD, Research Director, The Belgian Fund for Scientific Research (F.N.R.S.), CHU Brugmann, Psychiatry Secretary 4, Place Vangehuchten - B-1020 Brussels, Belgium
Received: 15 September 2022; Accepted: 17 November 2022; Published: 12 December 2022
Citation: Anais Ingels, Lauriane Fabry, Catherine Hanak, Florence Hanard, Hendrik Kajosch, Charles Kornreich, Salvatore Campanella. Using Cognitive Event-Related Potentials in the Management of Alcohol Use Disorder: Towards an Individual Approach. Archives of Clinical and Medical Case Reports 6 (2022): 772-775.
View / Download Pdf Share at FacebookAbstract
Objective: Alcohol use disorder (AUD) is associated with important cognitive impairments. These deficits play a significant role in the maintenance of consumption, despite the negative effect that it has on the daily life of alcohol addicts. The main objective of this case report is to illustrate how cognitive event-related potentials (ERPs) can be an adjuvant tool to propose an individualized and effective management of this disorder.
Method: Three patients worried about their alcohol consumption were evaluated in a Go-NoGo task during an electroencephalographic recording. The peak amplitude of the NoGo N200, NoGo P300 and ERN evoked response were confronted to behavioral scores as well as to a clinical assessment.
Results: ERPs allowed to distinguish between two types of cognitive deficits associated with alcohol consumption and treatment seeking. This was not possible when investigating only the behavioral and clinical scores which merely allowed to differentiate patients with AUD from the ones without AUD.
Conclusion: Cognitive ERPs are biomarkers of great relevance in the management of AUD by identifying altered cognitive processes that are not observable at the single behavioral level.
Significance: Further longitudinal studies should be designed in order to investigate the effect of an individualized cognitive retraining program, based on the observed cognitive alterations as well as on the relapse’ rate of these individual patients.
Keywords
AUD; Biomarkers; Cognitive Processes; Complex N2-P3; ERN; ERPs; Go-NoGo Task
AUD articles; Biomarkers articles; Cognitive Processes articles; Complex N2-P3 articles; ERN articles; ERPs articles; Go-NoGo Task articles
AUD articles AUD Research articles AUD review articles AUD PubMed articles AUD PubMed Central articles AUD 2023 articles AUD 2024 articles AUD Scopus articles AUD impact factor journals AUD Scopus journals AUD PubMed journals AUD medical journals AUD free journals AUD best journals AUD top journals AUD free medical journals AUD famous journals AUD Google Scholar indexed journals COVID-19 articles COVID-19 Research articles COVID-19 review articles COVID-19 PubMed articles COVID-19 PubMed Central articles COVID-19 2023 articles COVID-19 2024 articles COVID-19 Scopus articles COVID-19 impact factor journals COVID-19 Scopus journals COVID-19 PubMed journals COVID-19 medical journals COVID-19 free journals COVID-19 best journals COVID-19 top journals COVID-19 free medical journals COVID-19 famous journals COVID-19 Google Scholar indexed journals Biomarkers articles Biomarkers Research articles Biomarkers review articles Biomarkers PubMed articles Biomarkers PubMed Central articles Biomarkers 2023 articles Biomarkers 2024 articles Biomarkers Scopus articles Biomarkers impact factor journals Biomarkers Scopus journals Biomarkers PubMed journals Biomarkers medical journals Biomarkers free journals Biomarkers best journals Biomarkers top journals Biomarkers free medical journals Biomarkers famous journals Biomarkers Google Scholar indexed journals Ultrasound articles Ultrasound Research articles Ultrasound review articles Ultrasound PubMed articles Ultrasound PubMed Central articles Ultrasound 2023 articles Ultrasound 2024 articles Ultrasound Scopus articles Ultrasound impact factor journals Ultrasound Scopus journals Ultrasound PubMed journals Ultrasound medical journals Ultrasound free journals Ultrasound best journals Ultrasound top journals Ultrasound free medical journals Ultrasound famous journals Ultrasound Google Scholar indexed journals Cognitive Processes articles Cognitive Processes Research articles Cognitive Processes review articles Cognitive Processes PubMed articles Cognitive Processes PubMed Central articles Cognitive Processes 2023 articles Cognitive Processes 2024 articles Cognitive Processes Scopus articles Cognitive Processes impact factor journals Cognitive Processes Scopus journals Cognitive Processes PubMed journals Cognitive Processes medical journals Cognitive Processes free journals Cognitive Processes best journals Cognitive Processes top journals Cognitive Processes free medical journals Cognitive Processes famous journals Cognitive Processes Google Scholar indexed journals Complex N2-P3 articles Complex N2-P3 Research articles Complex N2-P3 review articles Complex N2-P3 PubMed articles Complex N2-P3 PubMed Central articles Complex N2-P3 2023 articles Complex N2-P3 2024 articles Complex N2-P3 Scopus articles Complex N2-P3 impact factor journals Complex N2-P3 Scopus journals Complex N2-P3 PubMed journals Complex N2-P3 medical journals Complex N2-P3 free journals Complex N2-P3 best journals Complex N2-P3 top journals Complex N2-P3 free medical journals Complex N2-P3 famous journals Complex N2-P3 Google Scholar indexed journals CT articles CT Research articles CT review articles CT PubMed articles CT PubMed Central articles CT 2023 articles CT 2024 articles CT Scopus articles CT impact factor journals CT Scopus journals CT PubMed journals CT medical journals CT free journals CT best journals CT top journals CT free medical journals CT famous journals CT Google Scholar indexed journals SRAS-CoV-2 articles SRAS-CoV-2 Research articles SRAS-CoV-2 review articles SRAS-CoV-2 PubMed articles SRAS-CoV-2 PubMed Central articles SRAS-CoV-2 2023 articles SRAS-CoV-2 2024 articles SRAS-CoV-2 Scopus articles SRAS-CoV-2 impact factor journals SRAS-CoV-2 Scopus journals SRAS-CoV-2 PubMed journals SRAS-CoV-2 medical journals SRAS-CoV-2 free journals SRAS-CoV-2 best journals SRAS-CoV-2 top journals SRAS-CoV-2 free medical journals SRAS-CoV-2 famous journals SRAS-CoV-2 Google Scholar indexed journals ERPs articles ERPs Research articles ERPs review articles ERPs PubMed articles ERPs PubMed Central articles ERPs 2023 articles ERPs 2024 articles ERPs Scopus articles ERPs impact factor journals ERPs Scopus journals ERPs PubMed journals ERPs medical journals ERPs free journals ERPs best journals ERPs top journals ERPs free medical journals ERPs famous journals ERPs Google Scholar indexed journals Treatment articles Treatment Research articles Treatment review articles Treatment PubMed articles Treatment PubMed Central articles Treatment 2023 articles Treatment 2024 articles Treatment Scopus articles Treatment impact factor journals Treatment Scopus journals Treatment PubMed journals Treatment medical journals Treatment free journals Treatment best journals Treatment top journals Treatment free medical journals Treatment famous journals Treatment Google Scholar indexed journals
Article Details
1. Introduction
This case report aims at investigating how cognitive Event-Related Potentials (ERPs) can be used in clinical practice in order to refine the diagnosis, prognostic and care proposed to patients suffering from Alcohol Use Disorder (AUD). To date, despite a significant amount of ERP research which has tried to provide a better understanding of alcohol abuse, ERPs still need to be implemented in the clinical management of AUD [1]. Indeed, besides technical difficulties [2], the main reason why ERPs are still absent in clinical practice is that most of these ERPs studies mainly focused on the evaluation of “grand-average data” rather than on the evaluation of “individual parameters”. Yet, this step is necessary for a meaningful utilization of these data in clinical practice [3]. Through this paper, we would like to highlight how individual ERPs data could instruct us more about cognitive efficiency and therefore provide a better management of AUD patients from a clinical perspective. AUD remains a major public health concern, with 283 billion people suffering from excessive consumption problem worldwide [4], a relapse rate which remains high (around 80% of relapse within one year of treatment [5]), and a delay of approximately one to four years after the onset of AUD for people to receive treatment (i.e., the treatment gap), because of the strong resistance and the frequent denial of their conditions [6]. Altogether, we state that it is crucial to reinforce prevention (an earlier and more sensible detection) and care for AUD. To reduce the treatment gap, the Brugmann’s hospital (Brussel, Belgium) has developed an innovative project in which a thorough evaluation is proposed to people worried about their alcohol consumption. The main objective of this program is to strengthen the insights into their disorder and to help patients to have a better understanding of their medical conditions. During two days, the participants notably undergo a clinical and an electrophysiological evaluation. The clinical evaluation comprised some important medical parameters (blood test, Fibroscan®, liver echography, dietetic evaluation) as well as a clinical psychological interview and classical questionnaires (described below).
The electrophysiological screening comprised an EEG recording during a Go-Nogo task (described below). In this paper, we will focus on the electrophysiological evaluation which is aimed at assessing the integrity of the main cognitive functions known to be altered by AUD: the inhibition and the monitoring of actions. As a matter of fact, it is now well agreed that substance use disorders are associated with dysfunction of cognitive processes [7]. According to the Dual Process Model theory, addicted patients present a default in the balance between a strengthened automatic system of impulsive habits (rewarding system) and a weakened executive processes system (goal-directed behavior) [8]. On the one hand, we observe an abnormal bottom-up system generating a “craving” response (attentional biases towards alcohol cues). On the other hand, we find a weakened top-down process (cognitive control processes allowing goal-directed behavior) [9] that is currently observed among AUD studies (medium effect size) [10]. This leads to a vicious circle [11]: cognitive control processes, such as inhibitory control or error processing, are impacted in AUD by alcohol-related urges known as craving, and conversely, weakened top-down processes do not allow a sufficient control of impulsive behaviors related to craving. These altered processes are therefore described as good indicator and predictors of substance use disorders (SUD) treatment outcomes [10].
Among the methods used to evaluate these cognitive processes, ERPs stand out compared to classical clinical approaches, as ERPs are known as valuable endophenotypic markers of greater sensitivity and sensibility. Indeed, their ability to reflect post synaptic released neurotransmitters (like GABA [12], Glutamate [13]) and neuromodulators (Acetylcholine [14], Dopamine [15]) plays a significant role in cortical functions altered by addictive substances [16]. Moreover, as successful inhibition processes and error monitoring produce little or no observable behavior, ERPs are of great interest to understand the internal processes [17]. During a cognitive task, the time-locked electro-encephalographic signals to specific events, giving us access to these ERPs which are consequently considered as brain’s response to the stimuli. Those ERPs inform us about the different stages of information processing, allowing us to achieve specific performances [18]. This way, they allow us to identify the “onset” of a dysfunction, and then to infer the impaired cognitive stages. Thus, the analyses of different ERPs can provide information regarding the functional impairments associated with excessive alcohol consumption, even for minor neurocognitive dysfunctions (e.g. when behavioral impairments are still not detected [19]). This way, they are good indicators of dysfunctional cognitive processes which are not accessible to the classical clinical approach [3] and can be considered, therefore, as interesting first line tools for the differential diagnosis, the monitoring of treatment and the therapeutic course’ evaluation of psychiatric disorders [20]. A limit to the use of ERPs is the lack of specificity that represents the evaluation of one single component. Indeed, a similar electrophysiological pattern of alteration can be found in various forms of mental disorders. It is consequently recommended to use ERPs in a different way. By taking into consideration several ERPs, we can reveal specific and individualized electrophysiological profiles reflecting the disturbed cognitive functions. This will also, ultimately, increase the specificity of the assessment and allow the prediction of a clinical trajectory of individual patients [3,21].
The Go-NoGo task is commonly used to measure inhibitory control. This task has the advantage of eliciting three major components: the N2d and P3d (observed on NoGo minus Go Wave) which reflect the inhibitory control processes [22,23] and the Error-Related Negativity (ERN) reflecting the error processing [24]. The N2d is a negative going wave emerging 200-300 ms after the onset of a stimulus. This component is mainly generated by the anterior cingulate cortex (ACC) and refers to the conflict monitoring processes and the cognitive parts of inhibition [25]. The following P3d, is a positive wave that peaks 300-600 ms after the presentation of a stimulus. This component is subtended by the inferior frontal gyrus (IFG) and the supplementary motor area and reflects the motor aspect of inhibition [26]. Although, the inhibitory N2-P3 complex has been predominantly studied in clinical trials [27-30], a third component deserves our attention in the field of AUD: the ERN. This component presents a negative deflection following an erroneous response after 50-150 ms an erroneous and is subtended by the ACC. It reflects early error detection processing [31], and it has been reported to be impacted by excessive alcohol consumption [32]. Excessive alcohol intake can affect these electrophysiological components in two different ways. Firstly, altered inhibitory skills can be associated with a poor performance associated with a decrease in neural activation indexed by decreased amplitude. This effect is supported by evidence of a dysfunction in inhibition-related receptor like GABA that, by affecting the level of neural excitability, enhances the risk of developing alcohol dependence [33]. Secondly, it is also possible to find a preserved performance associated with an enhanced amplitude in heavy drinkers reflecting a compensatory neurofunctional mechanism allowing drinkers to inhibit responses similarly to control subjects, despite latent cognitive impairments [22,34,35]. These subjects seem to still be able to activate the inhibitory system, but this is more demanding in terms of cognitive resources allocated to do so. The identification of these two types of alteration is important because it informs us about the objective that must be reached by cognitive retraining programs (CRP): allowing the patient to allocate more resources to realize this cognitive function or decreasing the amount of cognitive resources needed to perform the task adequately [36,37]. This could also be a potential explanation as to why some treatments work for some patients but don’t for others, in a perspective of stratified psychiatry [38]. For example, in a recent follow-up study involving AUD inpatients, researchers found that a preserved ERN at the onset of a detoxification program seems to be a protective factor against relapse one month after the cure. In other words, it seems that AUD patients with distorted metacognitive processes are more likely to quickly relapse (and even if they recover an intact ERN at the end of the detoxification program). This is suggesting that, as a consequence of their inability to benefit from “insight metacognitive properties”, they show a high risk of relapse. Therefore, other cognitive processes, such as cue reactivity or inhibitory [39,40] skills, should be rehabilitated through neurocognitive programs to reduce this risk of relapse [23].
The aim of the present study is to explore the way ERPs can be used in such a clinical perspective. Supporting the idea that an added value can be found in the use of ERPs in psychiatric care, how could such ERPs data be relevant at a clinical and individual level? Our main hypothesis is that relevant ERPs data can help to identify cognitive vulnerabilities and the appropriate neurocognitive program, predict the risk of relapse of specific patients, evaluate the treatment outcomes and follow up psychiatric disabilities. Obviously, such ERPs data should be complementary add-on tools to individual psychological, social and pharmacological care.
2. Materials and Methods
2.1 Participants
In this case report, we compare three patients presenting different type of alcohol consumption. These patients were matched for gender (female), age (55 years ±9 years), educational level (master’s degree) and history of detoxification cure (none). Each of these three patients has been seen in the context of the “Alcool et vous” (“You and Alcohol”) outpatient module, which is designed for subjects displaying hazardous drinking behavior and wishing to reduce their alcohol consumption. During this program, a physical evaluation including blood testing, fibroscan®, and liver echography is carried out, and no significant differences were observed between our 3 patients. In the present paper, we focus on the psychological evaluation including an electroencephalogram (EEG) and a clinical assessment (psychological interview and some questionnaires). The Alcohol Use Disorder Identification Test (AUDIT [41]) is a self-reported 10 items assessment of alcohol related behavior that maps out the problems of harmful alcohol consumption (items 1 to 3), drinking behavior (items 4 to 6), adverse reactions to alcohol (items 7 to 8), and alcohol-related problems (items 9 to 10). The score on items 1 to 8 can range from 0 to 4, and that on items 9 to 10 can be 0, 2 or 4). Subjects with a score between 8 to 12 are considered as having a hazardous to harmful alcohol consumption, while a score of 13 to 40 indicates a likely alcohol dependence [42]. The Beck Depression Inventory (BDI-II [43]) is a self-reported 21 items assessment of depression symptoms and severity. The items are rated from 0 (not at all) to 3 (extreme form) for each symptom. A score above 13 is indicative of depression (14 to 19 = mild depression; 20 to 28 = moderate depression; 29 to 63 = severe depression) [44]. The Spielberger State and Trait Anxiety Inventory (STAI [45]) is a self-reported 20 items assessment of the general propensity to be anxious. The items are rated from 1 (almost never) to 4 (almost always). A score above 40 is considered high [46]. The UPPS-c impulsive behavior scale [47] is a short version of the UPPS scale that includes 20 items. Normative data are, to our knowledge, not available but a higher score is indicative of a higher level of impulsivity. The Obsessive Compulsive Drinking Scale (OCDS [48]) is self-reported 14 items scale that measures various aspects of craving for alcohol. A score of 7 and higher discriminates alcohol-dependent patients from social drinkers. Furthermore, a score from 7 to 15 is representative of non-treatment seeking alcoholics, a score from 15 to 20 is representative of treatment seeking patients and a score from 20 to 25 is representative of inpatient alcoholics [48]. The patients signed an informed consent allowing us to follow up their clinical situation. The local ethics committee of Brugmann Hospital (“Comité d’Ethique Hospitalière CE2014/73) provided its approval for this study.
2.2 ERPs Task and Procedure
During the ERPs Go/Nogo, the patients were seated in a darkened room on a comfortable chair. The person who instructed the patients and managed the recording was alongside the patient during the complete registration procedure. Patients were seated at one meter from a screen, on which was centrally displayed the letter M (“Go” trials; button click with the right forefinger, as quickly and accurate as possible) and the letter W (“No-go” trial; no button click required). A block, repeated twice, comprised 133 letters displayed in a semi-random order to avoid the consecutive presentation of 2 “No-go” trials and was divided into 93 “Go” (70%) and 40 “No-go” (30%) letters. Each block started with the presentation of a background screen (for 500 ms), and the letters M or W (Arial yellow capital letters; 500x400 mm in size) were the displayed on this background for 200 ms, followed by a return to the initial background display (for 1300 ms). Thus, the participant had a maximum of 1500 ms to press the button before the next letter was displayed. Characteristics of stimuli presentation are presented in figure 1. This task was consistently used in our laboratory: more details can be found in these papers [49,29,36].
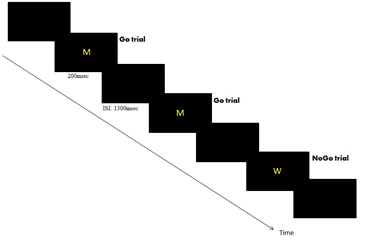
Figure 1: Illustration of the Go-NoGo task.
2.3 EEG Recording and Analysis
Electroencephalograms (EEG) were recorded by 21 electrodes fixed in an electrode Quick-cap with a linked mastoid physical reference (M1, M2). The electrode positions included the standards 10 to 20 system location (Fpz, Fp1, Fp2, Fz, F3, F4, F7, F8, Cz, C3, C4, T7, T8, P3, Pz, P4, P7, P8, O1, Oz, O2). The amplification of the EEG was realized by a battery-operated A.N.T. ® amplifier with a gain of 30,000 and a band-pass of 0.01-100 Hz. The ground electrode (AFz) was positioned between FPz and Fz along the midline. The impedence of the electrodes was maintained below 10kW during the experiments. The recording of the EEG was continuously at a sampling rate of 256 Hz with A.N.T. ® Cognitrace software. A cutoff of 30 mV was used to define trials that were contaminated either by eye movement or muscular artifacts, which were cut out offline in order to only analyze the artifact-free trials. Once acquired, a band-pass filter 0.3 to 30 Hz was applied. To generate the “Go” and “No-go” ERPs components, a stimulus-locked epochs of 1000 ms (200 ms before and 800 ms after the stimulus onset) were created with a baseline of -200 to 0 ms. Incorrect response (i.e., missed hit for “Go”, or a hit for “Nogo”) were excluded from further analysis. Specific “inhibitory No-go effect” tagging inhibitory control skills were obtained by computing the difference “No-go minus Go” wave form giving rise to the No-go N2d and the No-go P3d (d for difference) waves [36,37,50,51]. As ERPs latencies provide information about the processing speed, the individual latency has been quantified, according to the peak latency method [52]. The time after the stimulus onset at which the peak reaches the maximum negative amplitude for the N2d (within the 150- to 300- ms) and then the maximum positive amplitude for the P3d component (within the 300- to 500- ms), were identified in a cluster of classical frontal and central electrode (Fz, F3, F4 and Cz). As ERPs amplitudes provide information concerning the intensity of processing, the individual amplitude has been recorded at the first maximal amplitude value observable on the classical frontal and central electrode (Fz, F3, F4 and Cz) [36, 37]. To generate the “ERN” and “CRN” waveform, response-locked epochs of 700 ms (200 ms before and 500 ms after the response onset) were created. Only trials related to correct hits for targets and hits for non-targets (commission errors) were included in these averages. The types of response (key press for targets, key press for non-targets) were coded differently in order to compute different averages of ERP: (1) the ERN produced in association to key press on non-targets (= errors); and (2) the correct-related negativity (CRN) produced in association to correct key press on targets. The averages were computed for each patient individually. The process of error detection, tagging the difference in conflict between correct and error trials, was obtained by computing the difference “ERN-CRN” waveform giving rise to the DERNsubstract waveform [24,53]. As the ERP’s latency and amplitude provides information about the processing speed and the intensity of processing respectively, the individual latency and amplitude have been quantified according to the peak latency method (within the range of 0 to 100 ms) for the ERN, CRN and DERNsubstract. This time window has been chosen regarding the recommendations established in previous research [53,54].
3. Results
3.1 Clinical Data
Here we report the clinical data of the three patients concerning (1) the history of alcohol consumption and, (2) the scores on the clinical questionnaires.
3.1.1 Patient K.C: K.C is 53-year-old woman. She has been drinking alcohol since she is twelve years old. She came to the program because she reported experienced what she called “a loss of control” since one year ago. Indeed, since that time, she reports “not being able to experience any period of abstinence”. She has never been in treatment for her alcohol consumption before. Concerning her scores on the different scales, we observe a result of 13 on the AUDIT, indicating a heavy consumption [41,42]; a score of 13 on the BDI-II which is indicative of a mild depression [43,44]; a score of 65 on the STAI-state which revealed a high level of anxiety [45,46]; a total score of 45 on the UPPS-c; a score of 10 on the OCDS scale, indicating a moderate concern about alcohol (infrequent thought but frequent craving) [48]. She consumes five units of alcohol per day. On this basis, patient K.C. is the participant with the least risky alcohol consumption in the present paper.
3.1.2 Patient S.V: S.V. is a 47-year-old woman. She has been drinking alcohol since the age of 16. She reports a loss of control that occurred a few years ago and she drinks approximately 14 units of alcohol per day. She has not been able to experience any period of abstinence for 2 years. At the time of the assessment, she had never received a treatment for her alcohol consumption, but we know that she entered in a three-week detoxication cure 9 months later. Concerning her scores on the different scales, we observe a result of 34 on the AUDIT, which is indicative of a high risk of alcohol dependence [41,42]; a score of 13 on the BDI-II which is indicative of a mild depression [43,44]; a score of 71 on the STAI-state scale, indicating a high level of anxiety [45,46]; a total score of 42 on the UPPS-c and a score of 25 on the OCDS scale, indicating persistent thoughts about alcohol and frequent craving [48].
3.1.3 Patient R.M: R.M. is a 65-year-old woman who drinks alcohol since the age of 4. She reports a loss of control that occurred at the age of 18. Since that time, she has been able to experience only three months of complete abstinence. She is treated by a psychiatrist as outpatient and has an anti-craving medication composed of Campral® (333 mg) three times a day and Solian® two times a day. But she has never been in detoxification cure for her alcohol consumption. She currently drinks 8 units of alcohol per day. Concerning her scores on the different scales, the patient scored 26 on the AUDIT scale which is indicative of a high risk of alcohol dependence [41,42]; a score of 8 on the BDI-II, indicating mild depression [43,44]; a score of 48 on the STAI-state scale, indicating a moderate anxiety [45,46]; a total score of 46 on the UPPS-c and a score of 15 on the OCDS scale indicating moderate concern towards alcohol but frequent craving [48]. At this stage, from a clinical point of view, these 3 patients present very similar clinical characteristics (same level of depression, anxiety, education (master’s degree), no previous detoxification cure). They only differ as regards their alcohol consumption and pharmacological treatment. Patient K.C. has a less problematic intake than the two others, who both present a different hazardous consumption pattern. Indeed, Patient S.V. has a more recent but intense loss of control than Patient R.M. who has a long history of drinking problems. Please refer to table 1 for a more global overview of the clinical data.
|
Patient K.C. |
Patient S.V. |
Patient R.M. |
|
|
Age |
53 year-old |
47 year- old |
65 year-old |
|
Educational level |
Master degree |
Master degree |
Master degree |
|
1st consumption |
12 years old |
16 years old |
4 years old |
|
Onset of the problem |
1 year ago |
Few years |
From 18 years old |
|
Unit of Alcohol / day |
5 |
14 |
8 |
|
Detoxification Cure |
0 |
1 |
0 |
|
AUDIT |
13 |
34 |
26 |
|
BDI-II |
13 |
13 |
8 |
|
STAI-state |
65 |
71 |
48 |
|
UPPS-c tot |
45 |
42 |
46 |
|
OCDS |
10 |
25 |
15 |
Table 1: Clinical Data. Abbreviations: AUDIT- Alcohol Use Disorders Identification Test; BDI-II- Beck depression Inventory; STAI- State and Trait Anxiety Inventory; UPPS- Urgence Premeditation Perseverance Sensation seeking impulsivity scale; OCDS- Obsessive Compulsive Drinking Scale.
3.2 Behavioral Data
Here we report behavioral data of the three patients, concerning (1) performance: omissions (missing response for Go trials), commissions errors (false alarms for NoGo trials); and (2) reactions times (click on Go trials).
3.2.1 Patient K.C: She omitted no answers (0 omission errors for Go trials) and committed only 2 errors (2 commission errors). She had a reaction time of 424 ms.
3.2.2 Patient S.V. vs KC: Like patient K.C., Patient S.V. omitted no answers, but she committed much more errors (17 commission errors) showing difficulties in inhibition. She was also faster to respond (357 ms) attesting of a pre-potency for the dominant response.
3.2.3 Patient R.M. vs KC: Compared to patient K.C., patient R.M. omitted more responses (2 omission errors) and committed also more errors (12 commission errors), showing difficulties in inhibition of the dominant response. She was also the slowest (661 ms).
3.2.4 Patient S.V. vs R.M: Compared to patient S.V., patient R.M omitted more responses but committed less errors. This can be explained by the slower reaction times observed for patient R.M. which is suggesting some behavioral compensatory mechanism aiming at facilitating the task. Overall, from a behavioral point of view, we observe that the two patients with the most problematic alcohol consumption are also those who present more inhibition errors. Patient R.M., presenting a consumption period of 47 years, presents a lengthening compared to the 2 other patients. Interestingly, the lengthening of the reaction time does not permit her to inhibit more efficiently the automated response, suggesting an altered inhibitory process. Such data are important, because it allows us to distinguish between the two patients with inhibition difficulties (S.V. and R.M.) and the patient without (K.C.). Please, refer to table 2 for a larger overview of the behavioral data. The next section will help us to check whether using ERPs may add clinically relevant aspects to the care of these patients.
|
Patient K.C. |
Patient S.V. |
Patient R.M. |
|
|
Reaction Time |
424 ms |
357 ms |
661 ms |
|
Correct Response |
186/186 |
186/186 |
184/186 |
|
Commission Error |
Feb-80 |
17/80 |
Dec-80 |
Table 2: Behavioral data concerning the reaction time, the rate of correct response for Go Trials and the rate of commission errors (response for NoGo trials).
3.3 ERPs Data
In this section, we will verify whether the evaluation of the N2d, P3d and ERN components can add complementary information to the clinical and behavioral data analysis allowing us a conclusion which can have an impact on the further treatment procedure for the patient(s).
3.3.1 Patient K.C: Concerning the N2d component, patient K.C. shows a peak of -2,2 mV amplitude which appears at a latency of 329 ms. Then, concerning the P3d component, she shows a peak of 3,2 mV amplitude which appears at a latency of 500 ms. Finally, the DERNsubstract could not be achieved due to the low rate of errors committed by the patient. We thus dispose of any information concerning her metacognitive processes.
3.3.2 Patient S.V. vs KC: The N2d amplitude of Patient S.V. appears slightly reduced compared to the one of Patient K.C. suggesting that she allocates less cognitive resources for the cognitive part of inhibition. Conversely, the P3d component appears higher (5,3 mV) suggesting that a larger amount of cognitive resources is needed to perform the task. We thus observed a compensation at the level of the motor (part of) inhibition which is not sufficient regarding the patient’s behavioral performances. The latencies of these components are very close to those observed with Patient K.C. The DERNsubstract could be achieved for patient S.V. Since the DERNsubstract could not be recorded in patient K.C., a comparison is not possible. We will therefore compare it, in the next section, with the one observed for the patient R.M.
3.3.3 Patient R.M. vs K.C: In the same way as for Patient S.V., we observed, for patient R.M., a reduction in the amplitude for the N2d component compared to the one observed for patient K.C. But in contrast, the amplitude of the P3d components appeared to be reduced compared to the one detected for patient K.C., suggesting that patient R.M. does not allocate more cognitive resources to the motor part of the inhibition to compensate a weakened cognitive part of inhibition processes. We also observed a delayed N2d and P3d latency which seems to mirror the behavioral data and provides a certain facilitation in the task achievement. However, this compensatory mechanism is again not sufficient to compensate the initial cognitive impairments. Concerning the DERNsubstract, we observe a smaller amplitude for this patient comparing to patient S.V., suggesting reduced metacognitive abilities.
3.3.4 Patient S.V. vs R.M: Patient S.V shows faster a reaction time compared to patient R.M. and much a more reduced amplitude for the N2d and P3d components. This is suggesting that Patient R.M. is not able to allocate enough cognitive resources to the cognitive and motor part of inhibition even when using a compensatory mechanism such as slowing down the reaction times (please refer to Figure 2 for more details).
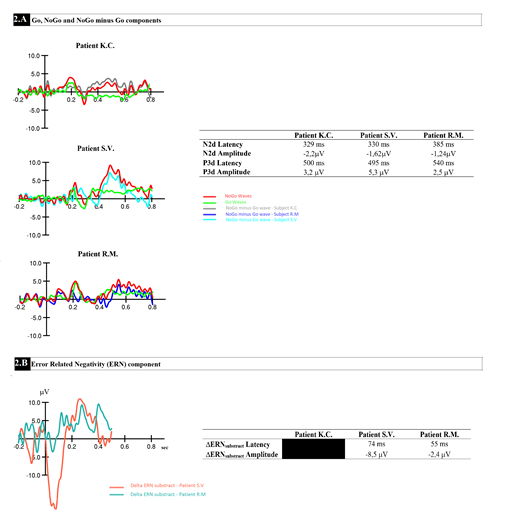
Figure 2: Grand averaged event-related potential waveforms obtained on Fz (as the best signal quality illustration) for the three patients during the Go/NoGo task for (A) the Go, NoGo and NoGo minus Go and (2) the DERN substract.
4. Discussion
The first aim of this study was to explore the way ERPs can inform us about the cognitive state of a patient presenting a risky alcohol consumption. Therefore, we have evaluated three patients presenting a problematic alcohol consumption and who underwent two days in the “Alcool et vous” program of the CHU Brugmann. They were matched for their age, gender and educational level and were selected based on their different patterns of alcohol consumption. The first main result is that the behavioral scores allow us only to distinguish the patients with inhibition difficulties from the one who had no inhibition difficulties. Indeed, compared to patient K.C., patient S.V. and patient R.M. show a higher rate of commission errors. But only the ERPs allow to distinguish different subgroups among the patients and which kind of support would be the most beneficial for them. This constatation is in line with previous research attesting that ERPs are a useful tool to detect neural vulnerabilities reflecting latent inhibitory restrictions, even in the absence of significant behavioral modifications. These modifications can influence the clinical trajectory and guide the care [29]. To highlight these modifications, we focused on two components that have been predominantly studied and which are indicative of alterations of the inhibitory processes, the N2d and P3d [31-34], as well as the ERN which is showing some promises as vulnerability factor for risky behavior (substance abuse) [40] and to influence clinical trajectory [24]. Specific behavioral and electrophysiological alterations can be observed in the two patients with excessive alcohol consumption attesting to their inhibition difficulties. Indeed, the more important rate of commission errors observed in S.V. and R.M., is accompanied by different kind of electrophysiological modification. First, S.V. and R.M. are presenting a decreased N2d amplitude compared to K.C., which indicates an absence of the “NoGo N2 effect”. Indeed, while the Patient K.C. showed a larger N2d amplitude compared to Go N2, patients S.V. and R.M. didn’t show this effect or a reduced version of this effect [32]. Since the N2d is derived from the subtraction of the “NoGo Curve minus Go curve”, it directly reflects the “NoGo N2 effect”. This indicates a lower activation when it comes to detecting response conflict and recognizing the need for inhibition. As a result, they need to engage more cognitive resources to complete the inhibition task, as recorded by a higher P3d amplitude [35,51]. It is at this level that we were able to observe a difference between the Patient S.V. and R.M. Patient S.V. presents, as expected, an enhanced amplitude of the P3d, reflecting a compensatory effect of the impaired detection of inhibitory need and inhibition preparation. She needs to engage more cognitive resources to inhibit a prepotent response. Despite this compensation, she presents a higher rate of commission errors compared to the Patient K.C. Furthermore, her relatively fast reaction time is also indicative of a prepotency to the dominant response. The inhibition difficulties are therefore also caused by a priming of the motor system compared to the executive one, which makes any withholding attempts even more difficult [55].
In contrast, we do not observe this compensatory effect for the Patient R.M., recorded by the lowest P3d amplitude. Accordingly, we understand that this patient also presents an impaired conflict detection and inhibition preparation, but in contrast to Patient S.V., she is no more able to allocate more cognitive resource in the motor aspect of inhibition. Hence, this patient extends her reaction time in order to reduce the difficulty of the task, and despite this, she maintains a higher number of commission errors and also omits to respond, sometimes, when necessary. This could be explained by the neurotoxic effects of her long-term alcohol consumption on the central nervous system, such as brain atrophy and/or dysfunction that are correlated with the lifetime dose of ethanol consumed [56,57] or by her current anti-craving medication. However, the impact of these anti-craving drugs on the ERP components had not yet been demonstrated to our knowledge. Further studies should investigate the effect of these treatments on the electrophysiological components in order to promote the use of ERPs as biomarkers of treatment efficiency or to guide practitioners towards the more efficient pharmacological treatment. Now focusing on the ERN, we observe that our results are consistent with those found by Claus and colleagues [55]. Patient R.M. who has been consuming alcohol for a longer period of time, and thus exhibits a more severe alcohol consumption, seems to invest less resources in the detection of committed errors compared to the patient S.V., who has been consuming alcohol for a shorter period of time. This could be explained by the neurotoxic effect of a long-term alcohol consumption on the anterior cingulate cortex [45] and could be interpreted as a lower error-monitoring skills. This ultimately leads to the inability to adjust behavior after an error has been committed. In a more clinical and ecological perspective, we can thus imagine that patients with a reduced ability to adjust behavior, will be less likely to seek treatment to diminish their alcohol consumption.
To our knowledge, no study has yet investigated the way in which diminished error-monitoring skills can influence the treatment seeking in AUD. This is however of greater interest as we already know that alcohol-dependent patients who, at the beginning of their detoxification program, did not show a preserved ERN component are also at high risk of relapse [24]. A lack of error monitoring skills could be an additional factor for explaining the delay to start with a detoxification program in addition to the high rate of relapse as it is an indicator of risky behavior (substance abuse) [54]. It also reinforces the belief that an earlier detoxification program is a perquisite for reducing the relapse rate, since a better metacognitive property will help patients to take more benefit from the proposed clinical care. Accordingly, a neurocognitive program tagging these altered cognitive processes could also be proposed to patients presenting cognitive alterations due to their long-term alcohol consumption [25,58-61].
5. Conclusion
In conclusion, the presented cases support the aim of a rational use of event-related potentials in a clinical perspective, not as a diagnostic tool, but as a relevant indicator of cognitive vulnerabilities. The principle behind this is that within a single nosological entity, there exists different subgroups of patients presenting different patterns of cognitive difficulties that may influence the maintenance of AUD. The identification of these difficulties and of cognitive alterations as biomarkers is crucial in a perspective of stratified psychiatry [33]. This case report seems to indicate that ERPs can deliver an important support regarding their ability to (1) inform us about individual cognitive deficits even when no behavioral difficulties are visible; (2) investigate the complete stream of information processing (e.g. N2d, P3d, ERN) in order to determine the stages that need to be trained in a specific cognitive function (here, inhibition); (3) follow the individual characteristics of a patient in order to evaluate the benefits of the proposed care or the evolution of a specific disease; (4) predict the relapse risk of one patient in order to orient him/her towards social supports or neurocognitive programs well-known to trigger relapse [38,47,48].
In addition, (in this case report) we were able to distinguish, thanks to ERPs, two different patterns of cognitive alterations in patients presenting similar clinical characteristics. These patterns of deficits were mostly linked with a history of consumption and metacognitive abilities. Indeed, longer alcohol consumption causes greater cognitive impairments, which manifest themselves sat the electrophysiological level by an inability to recruit more cognitive resources. Moreover, better metacognitive abilities appear as a protective factor leading the subject who is facing difficulties to take steps to receive the support that s/he needs. Taking all this into consideration and given the cognitive alterations that can be observed by the discussed ERPs, different kinds of rehabilitation programs seem to be necessary in order to reduce the risk of relapse. A longitudinal study concept following patients from a first electrophysiological evaluation towards an individualized cognitive retraining program, followed by second evaluation at the end of the detoxification program and finally a follow up procedure attesting of the relapse state, should be of great relevance for future research and open further perspectives. In addition, such a study should allow us to replicate the first results presented in this case report. These single subject data should also be completed and validated by a group study of patients presenting similar and different cognitive impairments within the same AUD condition.
References
- Verdejo-Garcia A, Lorenzetti V, Manning V, et al. A roadmap for integrating neuroscience into addiction treatment: a consensus of the neuroscience interest group of the international society of addiction medicine. Frontiers in psychiatry 10 (2019): 877.
- Luck SJ. Event-related potentials. In: Cooper H, Camic PM, Long DL, planter AT; Rindskopf D, Sher KJ, editors. APA Handbook of Research Methods in Psychology. Washington DC. USA.: American Psychology Association 2 (2012): 523-546.
- Campanella S, Schroder E, Kajosch H, et al. Why cognitive event-related potentials (ERPs) should have a role in the management of alcohol disorders. Neuroscience & biobehavioral Reviews 106 (2019): 234-244.
- World Health Organization, Management of substance Abuse Team, &World Health Organization. Global status report on Alcohol and health (2018).
- Sinha R. New findings on biological factors predicting addiction relapse vulnerability. Current psychiatry reports 13 (2011): 389-405.
- Ten Have M, de Graaf R, van Doorselaer S, et al. Lifetime treatment contact and delay in treatment seeking after first onset of a mental disorder. Psychiatric Services 64 (2013): 981-989.
- Bruijnene CJ, Dijkstra BA, Walvoort SJ, et al. Prevalence of cognitive impairments in patients with substance use disorder. Drug and alcohol review 38 (2019): 435-442.
- Stacy A, Wiers RW. Implicit cognition and addiction: a tool for explaining paradoxical behavior. Annual Review of Clinical Psychology 6 (2010): 551-575.
- Wiers RW, Gladwin TE, Hofmann W, et al. Cognitive bias modification and cognitive control training in addiction and related psychopathology: Mechanisms, clinical perspectives, and ways forward. Clinical Psychological Science 1 (2013): 192-212.
- Smith JL, Mattick RP, Jamadar SD, et al. Deficits in behavioural inhibition in substance abuse and addiction: a meta-analysis. Drug and alcohol dependence 145 (2014): 1-33.
- Mahre R, Luijten M, Franken IH. The clinical relevance of neurocognitive measures in addiction. Frontiers in Psychiatry 4 (2014): 185.
- Koob G. A role for GABA mechanisms in the motivational effects of alcohol. Biochemical pharmacology 68 (2004): 1515-1525.
- Gonzales R, Jaworski JN. Alcohol and Glutamate. Alcohol health and research world 2 (1997): 120.
- Feduccia AA, Chatterjee S, Bartlett SE. Neuronal nicotinic acetylcholine receptors: neuroplastic changes underlying alcohol and nicotine addictions. Frontiers in molecular neuroscience 5 (2012): 83.
- Di Chiara G. Alcohol and Dopamine. Alcohol Health and research world 21 (1997): 108.
- Pogarell O, Mulert C, Heherl U. Event Related potentials in psychiatry. Clinical EEG Neurosci 38 (2007): 25-34.
- Smith J, Jamadar S, Provost A, et al. Motor and non-motor inhibition in the GO/NoGo task: An ERP and fMRI study. International journal of Psychophysiology 87 (2013): 244-253.
- W, et al. Electrophysiological correlates of anterior cingulate function in a Go/no-go task: effects of response conflict Hansenne M. Event-related brain potentials in psychopathology: clinical and cognitive perspectives. Psychologica Belgical (2006).
- Maurage P, Pesenti M, Philippot P, et al. Latent deleterious effect of binge drinking over a short period of time revealed only by electrophysiological measures. Journal of Psychiatry and Neuroscience 34 (2009): 111-118.
- Biasiucci A, Franceschiello B, Murray MM. Electroencephalography. Current Biology 29 (2019): R80-R85.
- Campanella S. Use of cognitive event-related potentials in the management of psychiatric disorders: Towards an individual follow-up and multi-component clinical approach. World J. Psychiatry 11 (2021): 153.
- Petit, G, Cimochowska A, Cevallos C, et al. Reduced processing of alcohol cues predicts abstinence in recently detoxified alcoholic patients in a three-month follow-up period: and ERP study. Neuropsychiatric Disease and Treatment 10 (2014): 1025.
- Bell RP, Foxe JJ, Ross LA. Intact inhibitory control processes in abstinent drug abusers (I): a functional neuroimaging study in former cocaine addicts. Neuropharmacology 82 (2014): 143-150.
- Dousset C, Schroder E, Ingels A, et al. Intact Error-Related-negativity at the start of a three-week detoxification program reflects a short-term protective factor against relapse in Alcoholic patients: Some preliminary evidence from a follow-up Event-Related Potentials study. Clinical EEG and Neuroscience (2022): 15500594221076579.
- Nieuwenhuis S, Yeung N, Van Den Wildenberg and trial type frequency. Cognitive, affective, & behavioural Neuroscience 3 (2003): 17-26.
- Smith JL, Johnstone SJ, Barry RJ. Movement-related potentials in the go/nogo task: the P3 reflects both cognitive and motor inhi- bition. Clin Neurophysiol 119 (2008): 704-714.
- Campanella S. Neurocognitive rehabilitation for addiction medicine: from neurophysiological markers to cognitive rehabilitation and relapse prevention. Progress in Brain Research 224 (2016): 85-103.
- Pandey AK, Kamarajan C, Tang Y, et al. Neurocognitive deficits in male alcoholics: an ERP/sLORETA analysis of the N2 component in an equal probability. Biological psychology 89 (2012):170-182.
- Petit G, Cimochowska A, Kornreich C, et al. Neurophysiological correlates of response inhibition predict relapse in detoxified alcoholic patients: some preliminary evidence from event-related potentials. Neuropsychiatric Disease and Treatment 10 (2014): 1025.
- Batschelet HM, Tschuemperlin RM, Moggi F, et al. Neurophysiological correlates of alcohol-specific inhibition in alcohol use disorder and its association with craving and relapse. Clinical neurophysiology 132 (2021): 1290-1301.
- Olvet DM, Hajack G. The error-related-negativity (ERN) and psychopathology. Toward an endophenotype. Clinical psychology review 28 (2008): 1343-1354.
- Ridderinkof KR, DeVlugt Y, Bramlage A, et al. Alcohol consumption impairs detection of performance errors in mediofrontal cortex. Science 298 (2002): 2209-2221.
- Volkow ND, Wang GJ, Hitzemann R, et al. Decreased cerebral response to inhibitory neurotransmission in alcoholics. The American Journal of Psychiatry 150 (1993): 417-422.
- Géczy I, Czigler I, Balazs L. Effect of cue information on response production and inhibition measured by event-related potentials. Acta Physiol. Hung 86 (1999): 37-44.
- Lopez-Caneda E, Cadaveira F, Crego A. Hyperactivation of right inferior frontal cortex in young binge drinkers during response inhibition: a follow-up study. Addiction. 2012; 107 (2012):1796-1808.
- Schroder E, Dubuson M, Dousset C, et al. Training inhibitory control induced robust neural changes when behavior is affected: a foolow-up study using cognitive event-related potentials. Clinical EEG Neururosci 51 (2020): 303-316.
- Dousset C, Ingels A, Schroder E, et al. Transcranial direct current stimulation combined with cognitive training induces response inhibition facilitation through distinct neural responses according to the stimulation site: a follow–up event-related potentials study. Clinical EEG Neurosci 52 (2021): 181-192.
- Arns M, van Dijk H, Luykx JJ, et al. Stratified Psychiatry: Tomorrow’s Precision psychiatry? European Neuropsychophramacology 55 (2022): 14-19.
- Wiers RW, Eber C, Rinck M, et al. Retraining automatic action tendencies changes alcoholic patients’ approach bias for alcohol and improves treatment outcomes. Psychological science 22 (2011): 490-497.
- Dubuson M, Kornreich C, Vanderhasselt M, et al. Transcranial direct current stimulation combined with alcohol cue inhibitory control training reduces the risk of early alcohol relapse: a randomized-placebo-controlled clinical trial. Brain stimulation 14 (2021): 1531-1543.
- Saunders JB, Aasland OG, Babor TF, et al. Development of the acohol use disorders identification test (AUDIT): WHO collaborative project on early detection of persons with harmful alcohol consumption-II. Addiction 88 (1993): 791-804.
- Dolman JM, Hawkes ND. Combining the AUDIT questionnaire and biochemical markers to assess alcohol use and risk alcohol withdrawal in medical patient. Alcohol Alcohol 40 (2005): 515-519.
- Beck AT, Steer RA, Brown GK. Beck depression inventory (BDI-II). Pearson (1996).
- Smaar KL, Keefer AL. Measure of depression and depressive symptoms: Beck depression inventory-II (BDI-II), center for epidemiologic studies depression scale (CES-D), geriatric depression scale (GDS), hospital anxiety and depression scale (HADS), and patient health questionnaire-9 (PHQ-9). Arthritis care & research 63 (2011): S454-S466.
- Spielberger CD. State trait anxiety inventory for adults. Palo Alto CA: mind garden (1983).
- Knight RG, Waal-Manning HJ, Spears GF. Some norms and reliability data for the State -Trait Anxiety inventory and the Zung Self rating depression Scale. Br J. Clin. Psychol 22 (1983): 245-249.
- Van Der Linden M, d’Acremont M, Zermatten A, et al. A french adaptation of the UPPS Impulsive Behavior Scale: Confirmatory factor analysis of in a sample of undergraduates. European Journal of Psychological Assessment 22 (2006): 38-42.
- Anton RF. Obsessive compulsive aspects of craving: development of the obsessive Compulsive Drinking Scale. Addiction 95 (2000): 211-217.
- Campanella S, Schroder E, Kajosch H, et al. Neurophysiological markers of cue reactivity and inhibition subtend a three-month period of complete alcohol abstinence. Clinical neurophysiology 13 (2020): 555-565.
- Petit G, Kornreich C, Noël X, et al. Alcohol related context modulates performance of social drinkers in a visual Go/No-Go task: a preliminary assessment of event-related potentials. PloS one 7 (2012): e 37466.
- Falkenstein M, Hoorman J, Hohnsbein J. ERP components in go/nogo task and their relation to inhibition.Acta Psychol (Amst.) 101 (1999): 267-291.
- Clayson PE, Baldwin SA, Larson MJ. How does noise affect amplitude and latency measurement of event-related potentials (ERPs)? A methodological critique and simulation study. Psycholphysiology 50 (2012): 174-186.
- Klawohn J, Meyer A, Weinberg A, et al. Methodological choices in Event-related potential (ERP) research and their impact on internal consistency reliability and individual differencies: an examination of the Error-related negativity (ERN) and anxiety.J Abnorm Psychol 129 (2020): 29.
- Gehring W, Himle J, Nisenson LG. Action-monitoring dysfunction in obsessive- compulsive disorder.Psychol. Sci 11 (2000): 1-6.
- Claus ED, Feldstein ESW, Filbey FM, et al. Behavioral control in alcohol use disorders: relationships with severity. Journal of studies on alcohol and drugs 74 (2013): 141-151.
- Dong G, Lu Q, Zhou H, et al. Impulse inhibition in people with internet addiction disorder. Electrophysiological evidence from a Go/NoGo study. Neuroscience letters 485 (2010): 138-142.
- Nicolas JM, Estuch R, Salamero M, et al. Brain impairment in well-nourished chronic alcoholics is related to ethanol intake. Annals of neurology 41 (1997): 590-598.
- Fadardi JS, Cox WM. Reversing the sequence: reducing alcohol consumption by overcoming alcohol attentional bias. Drug Alcohol Depend 101 (2009): 137-145.
- Houben K, Nederkoorn C, Wiers RW, et al. Resisting temptation: decreasing alcohol-related affect and drinking behavior by training response inhibition. Drug Alcohol Depend 116 (2011): 132-136.
- Riesel A, Klawohn J, Grützmann R, et al. Error-related brain activity as a transdiagnostic endophenotype for obsessive-compulsive disorder, anxiety and substance use disorder. Psychol Med 49 (2019): 1207-1217.
- Batschelet HM, Tschuemperlin RM, Moggi F, et al. Neurophysiological correlates of alcohol specific inhibition in alcohol use disorder and its association with craving and relapse. Clinical neurophysiology 132 (2021): 1290-1301.
