Unusual Presentation of a Metastatic Lymph Node from Giant Phyllodes Tumour of Breast– A Case Report
Article Information
Sherry Abraham*, Patricia Solomon, Rajesh B, Selvamani B
Department of Radiation Oncology Unit III, Christian Medical College, Vellore, Tamil Nadu, India
*Corresponding Author: Dr. Sherry Abraham, Senior Resident, Department of Radiation Oncology, Christian Medical College, Vellore, Tamil Nadu, India
Received: 30 September 2019; Accepted: 02 March 2020; Published: 10 March 2020
Citation: Sherry Abraham, Patricia Solomon, Rajesh B, Selvamani B. Unusual Presentation of a Metastatic Lymph Node from Giant Phyllodes Tumour of Breast– A Case Report. Archives of Clinical and Medical Case Reports 4 (2020): 253-258.
View / Download Pdf Share at FacebookAbstract
Background: Phyllodes tumours(PTs) of breast are rare fibroepithelial neoplasms which constitute 0.3% to 0.5% of female breast tumours. Malignant PTs usually develop haematogenous spread and only <1% of the patients with PT have lymph node metastasis.
Case Presentation: We report a 38 year old premenopausal lady who presented with a progressive right breast lump with intermittent pain of one year duration. She had similar history two years ago and was diagnosed with phyllodes tumour for which she underwent lumpectomy in hometown. Local examination of right breast revealed a 20 × 15cm sized lump of variegated consistency involving all the quadrants and two hard, mobile lymph nodes of size 2.5cm were palpable in the right axilla. Trucut biopsy of the right breast lump showed compressed ducts lined by benign bilayered cuboidal epithelium with stromal nuclear atypia and occasional mitotic activity suggestive of phyllodes tumour of aggressive behavior. She underwent modified radical mastectomy with right axillary lymph node clearance. Histopathology of the specimen was suggestive of malignant phyllodes tumour and seven of 11 axillary lymph nodes contained metastatic disease. She received 4 cycles of adjuvant chemotherapy with single agent Doxorubicin and radiotherapy to the chest wall following the same. She was advised to be on follow up. Four months later she presented with two weeks history of dyspnea and was found to have bilateral pleural effusion, pericardial effusion, tricuspid ball valve thrombus and acute submassive pulmonary embolism. Though effusions were thought to be secondary to malignancy, cytology did not prove the same. She succumbed to her illness within 72 hours of admission to emergency room.
Conclusion: Malignant phyllodes tumour is a rare entity which mimics benign neoplasm clinically, but behave like sarcomas with poor prognosis and haem
Keywords
Malignant phyllodes tumour; Lymph node metastasis; Axillary lymphadenopathy
Malignant phyllodes tumour articles, Lymph node metastasis articles, Axillary lymphadenopathy articles
Article Details
1. Introduction
Phyllodes tumours(PTs) of breast are rare fibroepithelial neoplasms which constitute 0.3% to 0.5% of female breast tumours [1]. Their incidence is 2.1 per million and peaks from the age of 45 to 49 years [2]. World Health Organization classified PTs as benign, borderline and malignant based on histological findings [3]. The incidence of each type of PTs has been reported by the M.D. Anderson Cancer Center based on a large case series to be 58% in benign, 12% in borderline and 30% in malignant [4]. Local recurrence rate of up to 17% is seen in benign PTs and up to 27% in malignant tumours [5]. Malignant PTs usually develop haematogenous spread and only <1% of the patients with PT have lymph node metastasis [2]. Tumors which grow larger than 10cm are designated as giant PTs and constitute 20% of the PTs. We report a giant phyllodes tumor with a metastatic lymph node.
2. Case Discussion
A 38 year old premenopausal lady presented with a progressive right breast lump with intermittent pain for one year with no other associated symptoms in March 2018. She had similar presentation 2 years earlier and was diagnosed to have benign phyllodes tumour for which she had right lumpectomy done at hometown. She noticed recurrence of lump one year later. There was no family history of malignancy. Local examination of right breast revealed a 20 × 15 cm sized lump of variegated consistency involving all the quadrants. Skin over the lump was stretched with engorged veins. Two hard, mobile lymph nodes of size 2.5 cm were palpable in the right axilla. There was no supraclavicular lymphadenopathy. Contralateral breast was normal. Though the tumour appeared to be phyllodes, carcinoma was also considered due to the presence of lymphadenopathy.
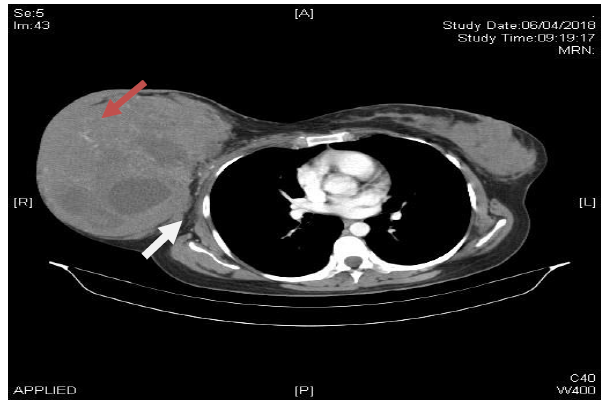
Ultrasound of right breast and axilla showed a large fairly well defined heterogenous mass with solid and cystic components showing posterior enhancement and demonstrable internal vascularity, involving all the quadrants of breast, replacing the normal parenchyma. Left breast was normal. Contrast enhanced computed tomography of thorax revealed a large soft tissue density mass lesion of size 17 × 18 × 20 cm replacing the entire right breast (Figure 1). Multiple cystic areas and linear branching vessels were noted within the lesion. Posteriorly, the pectoralis muscles were not involved. Anteriorly, the lesion reached up to the skin with significant skin thickening in the upper quadrant. There was significant right axillary lymphadenopathy, largest measuring upto 17 mm.
Trucut biopsy of the right breast lump showed compressed ducts lined by benign bilayered cuboidal epithelium with stromal nuclear atypia and occasional mitotic activity suggestive of phyllodes tumour of aggressive behavior. Her chest x-ray was normal. Ultrasound abdomen and pelvis did not show any focus of metastasis. She had modified radical mastectomy of the right breast and level 1 and 2 axillary nodal clearance with latissimus dorsi flap reconstruction. Operative findings were suggestive of a single large 5-kg tumor occupying the whole of the right breast with dilated vessels over the surface, infiltrating into the pectoralis major muscle.
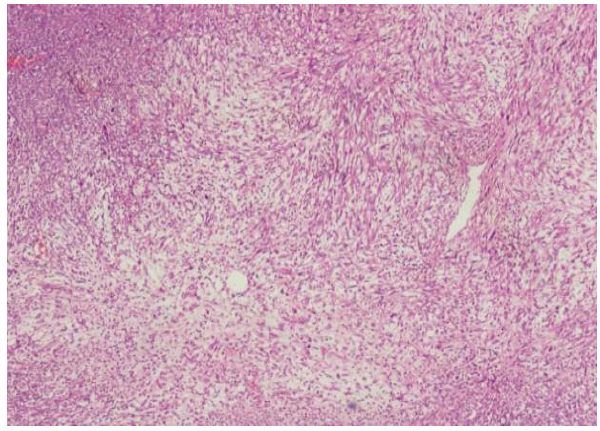
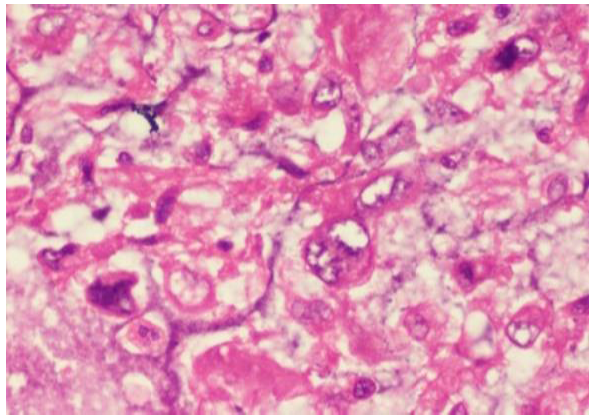
There were multiple large solid nodes at axillary levels 1 and 2. Histopathology of the specimen was suggestive of malignant phyllodes tumour of maximum dimension of 22cm, with sarcomatous overgrowth, lymphovascular invasion and poor margin clearance. Tumour was composed of sheets of round to short spindle cells with moderate to marked nuclear pleomorphism and mitotic activity of >15/10 hpf (Figure 2 and 3). Focally proliferated epithelial component forming occasional cleft like spaces and necrosis were also noted. Seven of 11 axillary lymph nodes contained metastatic disease. The tumour cells were negative for ER, HER2, HMWCK, p63 and CK5/6 with occasional foci showing faint staining for PR. The atypical proliferated stromal cells show patchy BCL2 positivity and were negative for CD34. Background epithelial components were staining positively for HMWCK and Pan Cytokeratin.
She received 4 cycles of adjuvant chemotherapy with single agent Doxorubicin. Post 4 cycles of chemotherapy, she received radiotherapy to the chest wall. She was treated with 3D conformal radiotherapy using 6MV photons to a dose of 60Gy in 30 fractions to the Planning Target Volume. She had been advised to be on follow up. Four months later, she presented with complaints of cough and dyspnea on exertion for 2 weeks to emergency room. She was found to have bilateral pleural effusion, pericardial effusion, tricuspid ball valve thrombus and acute submassive pulmonary embolism. Though effusions were thought to be secondary to malignancy, cytology did not prove the same. She succumbed to her illness within 72 hours of admission to emergency room.
3. Discussion
Malignant phyllodes tumour is a rare entity which mimics benign neoplasm clinically, but, is characterized by a typical rapid growth. Microscopic findings is the basis of subclassification of PTs. Features consistent with a malignant PT include high mitotic rate of >5/10hpf, stromal hypercellularity and pleomorphism, necrosis and infiltrative tumour edge [6] – all of which were seen in histopathology of the specimen of our patient.
Malignant PTs behave like sarcomas with poor prognosis and haematogenous spread. Local recurrence rate upto 17% or distant metastasis upto 27% has been documented by Jhang Y and Kleer CG [5]. Pleomorphism, infiltrative margins and stromal overgrowth are the poor histological findings related to recurrence in breast or distant metastasis. Recurrence rate or metastasis is usually seen within the first two years after diagnosis, ranging from 1 month to 10 years with an average of 15 to 26 months [3]. Lung is the most common site of metastasis (66-84.5%) followed by bone (28-39%) and rare sites include liver and brain [3]. Clinically palpable axillary nodes have been identified in upto 10-15% of PTs; however, pathologically positive nodes have been present in only <1% of the patients [2]. Chen et al conducted a retrospective study of 172 patients with phyllodes tumour at Mackay Memorial Hospital from January 1985 to December 2003 and reported that among the 42 patients who had modified radical mastectomy, none had lymph node involvement [7]. Confavreux et al restrospectively analysed 70 patients with malignant phyllodes tumour and primary breast sarcoma from 1966 to 2004 and found that of the 26 patients who had axillary lymph node dissection, only 2 patients had pathologically positive nodes [8]. When nodes are positive, there is also a possibility of coexistent invasive carcinoma along with phyllodes tumor as demonstrated by Korula et al.[9].
Wide local excision is the standard care for all histological types of phyllodes [10]. Hence, surgical excision with greater emphasis on obtaining adequate surgical margin (atleast 1cm) is the mainstay of treatment of malignant PTs. In terms of metastasis free survival and overall survival, studies have not shown significant difference between mastectomy and breast conserving surgery. Mastectomy should be considered in larger tumours and recurrent tumours with malignant histology. Role of axillary lymph node dissection in malignant PTs is controversial and conflicting views are documented in the literature. Most of the time, axillary lymphadenopathy is due to reactive hyperplasia. Majority of the investigators recommend against axillary lymph node dissection due to the low risk of lymph node involvement, except in patients with proven metastatic nodes or in clinically palpable axillary nodes [7, 11]. Adjuvant radiotherapy is recommended to minimize local recurrence in malignant PTs, recurrent tumours and tumours with close margins. Belkacemi et al found that radiotherapy was associated with superior 10 year local control rate from 59% to 86% for malignant and borderline PTs [12]. Role of adjuvant chemotherapy in malignant PTs is debatable. In an observational study by Morales-Vasquez in Mexico, 28 patients with malignant PT after surgical resection, from January 1993 to December 2003, reported 58% recurrence free survival with adjuvant chemotherapy with Doxorubicin and Dacarbazine as compared to 86% when in observation [13]. Turalba et al demonstrated efficacy of Doxorubicin and Ifosfamide based chemotherapy in women with metastatic phyllodes [14]. Our patient had recurrent giant PT of which, initial trucut biopsy showed PT of aggressive behavior with highly suspicious hard axillary lymph nodes. Hence, modified radical mastectomy with axillary lymph node dissection was done and the histopathology showed malignant PT with lymph node metastasis. She received adjuvant radiation therapy and chemotherapy. She died due to pulmonary embolism at 13 months from the date of diagnosis.
4. Conclusion
Malignant phyllodes tumour is a rare entity for which wide local excision with adequate margins is the mainstay of treatment. Though the most common cause of axillary lymphadenopathy is reactive hyperplasia; rarely, the patients may present with axillary node metastasis. Data regarding role of routine lymph node dissection remains controversial; however, it is indicated in palpable axillary nodes.
References
- Rowell MD, Perry RR, Hsiu JG, et al. Phyllodes tumors. Am J Surg 165 (1993): 376-379.
- Mishra SP, Tiwary SK, Mishra M, et al. Phyllodes tumor of breast: a review article. ISRN Surg 2013 (2013): 361469.
- Amir RA, Rabah RS, Sheikh SS. Malignant Phyllodes Tumor of the Breast with Metastasis to the Pancreas: A Case Report and Review of Literature. Case Rep Oncol Med 2018 (2018): 1-5.
- Chaney AW, Pollack A, McNeese MD, et al. Primary treatment of cystosarcoma phyllodes of the breast. Cancer 89 (2000): 1502-1511.
- Zhang Y, Kleer CG. Phyllodes Tumor of the Breast: Histopathologic Features, Differential Diagnosis, and Molecular/Genetic Updates. Arch Pathol Lab Med 140 (2016): 665-671.
- Tan BY, Acs G, Apple SK, et al. Phyllodes tumours of the breast: a consensus review. Histopathology 68 (2016): 5-21.
- Chen W-H, Cheng S-P, Tzen C-Y, et al. Surgical treatment of phyllodes tumors of the breast: Retrospective review of 172 cases. J Surg Oncol 91 (2005): 185-194.
- Confavreux C, Lurkin A, Mitton N, et al. Sarcomas and malignant phyllodes tumours of the breast – A retrospective study. Eur J Cancer 42 (2006): 2715-2721.
- Korula A, Varghese J, Thomas M, et al. Malignant phyllodes tumour with intraductal and invasive carcinoma and lymph node metastasis. Singapore Med J 49 (2008): e318-e321.
- Verma S, Singh RK, Rai A, et al. Extent of surgery in the management of phyllodes tumor of the breast: a retrospective multicenter study from India. J Cancer Res Ther 6 (2010): 511-515.
- Ramakant P, Chakravarthy S, Cherian JA, et al. Challenges in management of phyllodes tumors of the breast: A retrospective analysis of 150 patients. Indian J Cancer 50 (2013): 345.
- Belkacémi Y, Bousquet G, Marsiglia H, et al. Phyllodes tumor of the breast. Int J Radiat Oncol Biol Phys 70 (2008): 492-500.
- Morales-Vásquez F, Gonzalez-Angulo AM, Broglio K, et al. Adjuvant Chemotherapy with Doxorubicin and Dacarbazine has No Effect in Recurrence-Free Survival of Malignant Phyllodes Tumors of the Breast. Breast J 13 (2007): 551-556.
- Turalba CI, el-Mahdi AM, Ladaga L. Fatal metastatic cystosarcoma phyllodes in an adolescent female: case report and review of treatment approaches. J Surg Oncol 33 (1986):176-181.