The use of SGLT2 Inhibitors in Peritoneal Dialysis Patients: a Shade of Light on Dapagliflozin
Article Information
Abdullah K Alhwiesh1*, Ibrahiem Saeed Abdul-Rahman1, Mohammed Ahmad Nasreldin1, Abdelgalil Moaz Mohammed1, Suzan Al-Oudah1, Reem Al-Thwainy1, AyatAlAwal1, Zahra Alsenpisi1, Abdulla Abdulrahman2, Sarah Al-Warthan3, Nadia Al-Oudah4
1Nephrology Division, Department of Internal Medicine, King Fahd Hospital of the University, Imam Abdulrahman Bin Faisal University, Saudi Arabia
2Department of Electrical Engineering, Queen’s University, Toronto, Kingston, Canada
3Infectious Disease Division, Department of Internal Medicine, King Fahd Hospital of the University, Imam Abdulrahman Bin Faisal University, Saudi Arabia
4Department of pathology Dammam Central Hospital, Saudi Arabia
*Corresponding Author: Abdullah Khalaf Al-Hwiesh, Professor and Consultant of Nephrology, Imam Abdulrahman Bin Faisal University, King Fahd Hospital of the University, Al-Khobar, 1952, Saudi Arabia.
Received: 08 February 2022; Accepted: 18 February 2022; Published: 23 February 2022
Citation: Abdullah K Alhwiesh, Ibrahiem Saeed Abdul-Rahman, Mohammed Ahmad Nasreldin, Abdelgalil Moaz Mohammed, Suzan Al-Oudah, Reem Al-Thwainy1, AyatAlAwal, Zahra Alsenpisi, Abdulla Abdulrahman, Sarah Al-Warthan, Nadia Al-Oudah. The Use of SGLT2 Inhibitors in Peritoneal Dialysis Patients: A Shade of Light on Dapagliflozin. Archives of Nephrology and Urology 5 (2022): 1-8.
View / Download Pdf Share at FacebookAbstract
Objectives: This article reviews the clinical and experimental data pertaining to the renal effects of SGLT2 inhibition with a particular focus on Dapagliflozin in ESRD-peritoneal dialysis patients.
Methods: Fifty type-2 diabetes patients on insulin were treated with automated peritoneal dialysis (APD), with addition of selective SGLT2 inhibitor dapagliflozin to the anti-diabetic medication. The change in peritoneal transport status (PTS) was judged by Peritoneal Equilibration Test (PET) for patients before and after Dapagliflozin use. Ultrafiltration volume and the residual renal function were recorded as well. The primary outcomes were: 1. change of the PTS with Dapagliflozin therapy, 2. Change of the mean Ultrafiltration volume (UF), 3. Change in the mean 24-hour urine volume, 4. Follow-up of the inflammatory markers in both serum and PD fluid.
Results: There was no change in PTS, but there were significant changes in insulin requirements (p = 0.0262), fasting blood sugar (p = 0.0210), hemoglobin A1-C (p =0.0265), urine volume (p < 0.001), ultrafiltration volume (p < 0.01), mean systolic blood pressure (p = 0.0431), body weight (p = 0.0337) and serum uric acid (p =0.0341). Although there was marginal augmentation of the 24-h urine uric acid, yet the change was not significant (p = 0.073). On the other hand, we did not observe significant changes in blood urea nitrogen, serum creatinine, 24-h urine creatinine clearance, Kt/V and urine protein.
Conclusion: The use of Dapagliflozin was not associated with change of the peritoneal transport status. Dapagliflozin enhances diabetic control, and its use in PD patients with type-2 diabetes mellitus was useful in improving the UF and average urine volumes. In addition, it seems that such treatment decreases mos
Keywords
APD; Dapagliflozin; Diabetes; Insulin dose; Inflammatory markers; PTS; Serum glucose; UF; Urine volume; Uric acid
APD articles; Dapagliflozin articles; Diabetes articles; Insulin dose articles; Inflammatory markers articles; PTS articles; Serum glucose articles; UF articles; Urine volume articles; Uric acid articles
APD articles APD Research articles APD review articles APD PubMed articles APD PubMed Central articles APD 2023 articles APD 2024 articles APD Scopus articles APD impact factor journals APD Scopus journals APD PubMed journals APD medical journals APD free journals APD best journals APD top journals APD free medical journals APD famous journals APD Google Scholar indexed journals Dapagliflozin articles Dapagliflozin Research articles Dapagliflozin review articles Dapagliflozin PubMed articles Dapagliflozin PubMed Central articles Dapagliflozin 2023 articles Dapagliflozin 2024 articles Dapagliflozin Scopus articles Dapagliflozin impact factor journals Dapagliflozin Scopus journals Dapagliflozin PubMed journals Dapagliflozin medical journals Dapagliflozin free journals Dapagliflozin best journals Dapagliflozin top journals Dapagliflozin free medical journals Dapagliflozin famous journals Dapagliflozin Google Scholar indexed journals Diabetes articles Diabetes Research articles Diabetes review articles Diabetes PubMed articles Diabetes PubMed Central articles Diabetes 2023 articles Diabetes 2024 articles Diabetes Scopus articles Diabetes impact factor journals Diabetes Scopus journals Diabetes PubMed journals Diabetes medical journals Diabetes free journals Diabetes best journals Diabetes top journals Diabetes free medical journals Diabetes famous journals Diabetes Google Scholar indexed journals Insulin dose articles Insulin dose Research articles Insulin dose review articles Insulin dose PubMed articles Insulin dose PubMed Central articles Insulin dose 2023 articles Insulin dose 2024 articles Insulin dose Scopus articles Insulin dose impact factor journals Insulin dose Scopus journals Insulin dose PubMed journals Insulin dose medical journals Insulin dose free journals Insulin dose best journals Insulin dose top journals Insulin dose free medical journals Insulin dose famous journals Insulin dose Google Scholar indexed journals Inflammatory markers articles Inflammatory markers Research articles Inflammatory markers review articles Inflammatory markers PubMed articles Inflammatory markers PubMed Central articles Inflammatory markers 2023 articles Inflammatory markers 2024 articles Inflammatory markers Scopus articles Inflammatory markers impact factor journals Inflammatory markers Scopus journals Inflammatory markers PubMed journals Inflammatory markers medical journals Inflammatory markers free journals Inflammatory markers best journals Inflammatory markers top journals Inflammatory markers free medical journals Inflammatory markers famous journals Inflammatory markers Google Scholar indexed journals PTS articles PTS Research articles PTS review articles PTS PubMed articles PTS PubMed Central articles PTS 2023 articles PTS 2024 articles PTS Scopus articles PTS impact factor journals PTS Scopus journals PTS PubMed journals PTS medical journals PTS free journals PTS best journals PTS top journals PTS free medical journals PTS famous journals PTS Google Scholar indexed journals Serum glucose articles Serum glucose Research articles Serum glucose review articles Serum glucose PubMed articles Serum glucose PubMed Central articles Serum glucose 2023 articles Serum glucose 2024 articles Serum glucose Scopus articles Serum glucose impact factor journals Serum glucose Scopus journals Serum glucose PubMed journals Serum glucose medical journals Serum glucose free journals Serum glucose best journals Serum glucose top journals Serum glucose free medical journals Serum glucose famous journals Serum glucose Google Scholar indexed journals UF articles UF Research articles UF review articles UF PubMed articles UF PubMed Central articles UF 2023 articles UF 2024 articles UF Scopus articles UF impact factor journals UF Scopus journals UF PubMed journals UF medical journals UF free journals UF best journals UF top journals UF free medical journals UF famous journals UF Google Scholar indexed journals Urine volume articles Urine volume Research articles Urine volume review articles Urine volume PubMed articles Urine volume PubMed Central articles Urine volume 2023 articles Urine volume 2024 articles Urine volume Scopus articles Urine volume impact factor journals Urine volume Scopus journals Urine volume PubMed journals Urine volume medical journals Urine volume free journals Urine volume best journals Urine volume top journals Urine volume free medical journals Urine volume famous journals Urine volume Google Scholar indexed journals Uric acid articles Uric acid Research articles Uric acid review articles Uric acid PubMed articles Uric acid PubMed Central articles Uric acid 2023 articles Uric acid 2024 articles Uric acid Scopus articles Uric acid impact factor journals Uric acid Scopus journals Uric acid PubMed journals Uric acid medical journals Uric acid free journals Uric acid best journals Uric acid top journals Uric acid free medical journals Uric acid famous journals Uric acid Google Scholar indexed journals
Article Details
1. Introduction
Diabetes mellitus is known of its renal complications. In the last decade, however, there has been renewed interest in role of the kidney in the development and maintenance of high glucose levels. This has led to the development of novel agents to inhibit Sodium Glucose Transporter-2 (SGLT2) as a mean to better control glucose levels and at the same time augment calorie wasting and lower insulin, blood pressure and uric acid levels. Such actions, indirectly, may also have benefits for the prevention of diabetic complications including renal disease. SGLT2 inhibitors Dapagliflozin inhibits subtype 2 of the Sodium-Glucose Transport Proteins (SGLT2) which are responsible for at least 90% of the glucose reabsorption in the kidney. Blocking this transporter mechanism causes blood glucose to be eliminated through the urine [1]. In clinical trials, dapagliflozin lowered hemoglobin A1-C by 0.6 versus placebo percentage points when added to metformin [2]. There are also data supporting the potential for direct reno-protective actions arising from inhibition of SGLT2, including actions to attenuate diabetes-associated hyperfiltration and tubular hypertrophy, as well as reduce the tubular toxicity of glucose. Some studies have demonstrated significant reductions in albumin excretion in various experimental models, independent of its effects on blood pressure or glucose control [3]. Although promising, such actions remain to be established by comprehensive clinical trials with a renal focus, many of which are currently in progress. SGLT2 transporters are responsible for glucose and sodium reabsorption in the proximal tubule. Inhibition of SGLT2 promotes the urinary excretion of glucose and sodium, leading to osmotic diuresis and natriuresis. A few studies reported acute increases in urinary volume and sodium levels, supporting the natriuretic/diuretic properties of this drug class [4, 5]. These effects dissipated during prolonged treatment, reflecting a transient natriuretic effect with a subsequent new steady state [4, 6]. Other studies reported a decrease in plasma volume and interstitial fluid volume during SGLT2 inhibition, which would be in line with their natriuretic/diuretic profile [7, 8]. Achieving and controlling optimal volume status can be a challenge in patients with diabetic kidney disease, due to the impaired net excretion of sodium. Guideline-recommended treatment for these patients consists of optimizing glucose and blood pressure control, the latter preferably with agents that intervene in the renin–angiotensin–aldosterone system. However, novel drugs that target HbA1c and, at the same time, optimize volume control are a welcome addition to the therapeutic armamentarium for these patients. Previous studies that assessed the effects of SGLT2 inhibitors on volume markers were performed in either healthy subjects or in patients with type 2 diabetes with preserved renal function, but not in patients with impaired renal function [4, 6].
In addition, the previous studies did not include End-Stage Renal Disease (ESRD) patients and particularly those on dialysis, which limits the precision of the reported effect and preclude subgroup analyses and none of those studies exceeded a treatment period of 12 weeks. Therefore, we examined the effects of six-month treatment with SGLT2 inhibitor dapagliflozin compared to pre-treatment markers on volume status and other variables in type 2 diabetes patients with end-stage kidney disease. Since peritoneal dialysis was introduced as a common therapeutic method it has been used as an increasing popular alternative to hemodialysis. Major drawbacks, however, are frequent complications such as infections and ultrafiltration failure that occur as a result of repeated injury to the peritoneal membrane. A crucial role in maintaining the integrity of the peritoneum is attributed to peritoneal mesothelial cells, which have been studied over the past years with regard to their response to various toxic stimuli [9]. Maintenance of an osmotic gradient across the peritoneum is crucial for ultrafiltration. Despite the known toxic effects of high glucose on mesothelial cells [9], it is most used to generate this osmotic gradient. Model systems of peritoneal membranes reveal a substantial gradient in glucose concentration across this membrane. While this gradient is only partially maintained by the mesothelial monolayer the primary route of glucose absorption is paracellular. Nevertheless, this gradient is significantly correlated with mesothelial cell integrity. This prospective observational study reviewed the clinical and experimental data pertaining to the renal effects of the SGLT2 inhibitor Dapagliflozin with a particular focus on the Peritoneal Transport Status (PTS) and on UF and urine volume, blood glucose, and on certain inflammatory markers in End Stage Renal Disease (ESRD) type 2 diabetes mellitus patients on Automated Peritoneal Dialysis (APD).
2. Methods
In this study, 50 type-2 diabetes patients on insulin were treated by APD (Claria, Baxter Machines) with following prescription 1.36% Physioneal 5 liter and 2.27% Physioneal 5 liter with last fill 2 liter Extraneal as needed, with addition of selective SGLT2 inhibitor dapagliflozin to the anti-diabetic medication. Standard 4-hour PET was performed one month prior to the study and then, every 2 months for 6 months as described by Twardowski, et al [10]. Patients with low, low average, high average or high PTS were included in the study. The change in membrane type as judged by the (PET) test for patients one month before and 6 months after Dapagliflozin use and the average monthly recorded ultrafiltration from peritoneal dialysis exchanges and the average urine volume were also compared. The aim of the trial is to determine whether dapagliflozin can alter PTS, glucose absorption from peritoneal fluid, and thus improve ultrafiltration with a reduction in intraperitoneal glucose dissipation. In addition, we were interested in exploring the possible anti-inflammatory and antihypertensive actions of the drug. The primary outcomes were: 1. change of the PTS with 10 mg/day Dapagliflozin therapy, 2. Change of the mean Ultrafiltration volume (UF), 3. Changes in the average urine volume, 4. Follow-up of the inflammatory markers in both serum and PD fluid. The secondary outcomes were changes in the patients’ blood pressure, mean body weight, 24-hours urine protein and creatinine clearance and alteration of serum and urine uric acid. All patients were > 18-year-old and an informed written consent was obtained from every patient.
The study was approved by both King Fahd Hospital Ethical Committee and Imam Abdulrahman Bin Faisal Ethical Committee. Exclusion Criteria: Patients with type-1 diabetes mellitus, patients with recurrent hypoglycemic episodes, patients with acute or chronic liver disease, patients with history of peritonitis during the last six months, patients who has allergic reactions to dapagliflozin, patients with any malignant disease and those who refused to participate in the study. Biometric information of all included patients was obtained from the recorded files prior to and throughout dapagliflozin treatment. The included patients were started on 10 mg oral dapagliflozin daily for six months during which PTS was estimated every two months and the following data were recorded on a monthly basis: hypoglycemia and other side-effects of the drug, mean body weight, blood pressure, serum glucose and hemoglobin A1-C, mean 24-hour urine volume, mean UF, erythrocyte sedimentation rate, C-reactive protein, serum ferritin, serum uric acid, serum and peritoneal fluid CA-125, Peritoneal fluid effluent Polymorphonuclear (PMN) leucocytes, 24-h urine protein, 24-h urine uric acid, liver function test, 24-h urine creatinine clearance and kt/V. Office blood pressure was measured every hospital visit. blood samples, urine samples, and 24-h urine collections were analyzed, using the Roche COBAS 6000 analyzer series (Basel, Switzerland).. CA125 was measured using a sandwich immunoassay with chemi-luminescence detection (Roche Diagnostics Modular Analytics E170 analyzer). Polymorphonuclear cells were recovered from a standard 2-liter 2.27 g/l dextrose peritoneal dialysate instilled for a 4-hour dwell in all APD patients included in the study. Peritoneal dialysate effluents were also sent for cytospin analysis with cell staining to confirm cell types. Serum CA125 was measured using the same immunoassay as for peritoneal CA125, and serum C Reactive Protein (CRP) by the Eurolyser CRP assay.
3. Statistical Analysis
The data are presented as mean ± SD (standard deviation) or median (IQR). The patients′ characteristics were analyzed using Student t test for continuous variables and the Chi-square test for categorical variables. All analyses were conducted using SPSS version 18 software (SPSS Inc., Chicago, IL, USA). We used correlation coefficient for comparison between some variables.
4. Results
The demographic characteristics of patients included in the study are shown in Table 1. The total number of the assigned patients was 50; all of them had type-2 diabetes mellitus for more than 10 years. Their antidiabetic medication consisted of glargine insulin; mean dose was 9.9 + 2.1 units taken as a single dose. Their median age (IQR) was 55 (47-67), 26.0% of them were females. At the beginning of the study their weight recorded 86.3 kg (79.2-89.5). After 6 months of treatment, FBS decreased by 36.4 mg/dl (from a baseline of 188.6 + 9.6 mg/dl to a mean of 152.2 + 4.5 mg/dl, p = 0.0210) and hemoglobin A1-C by 1.6% (from 7.7% to 6.1%, p = 0.0265). Furthermore, body weight decreased by ~ 2.6 kg (from 86.3 to 83.7 kg, p = 0.033) (Figure 1), and hence insulin requirements dropped by 2.6 units (from 9.9 to a mean of 7.3 + 1.5 units (p = 0.0262) relative to pre-Dapagliflozin treatment. Compared to the blood pressure recording at zero point, a significant drop was observed in the systolic blood pressure from 148 + 5.2 mmHg to 134 + 6.5 mmHg at the end of the study, (p = 0.0431), and as a result, hydralazine was discontinued in 5 (55.6%) out of 9 patients, Alpha methyldopa in 5 (41.7%) out of 12 patients, and beta-blocker dose was reduced in 4 (40.0%) out of 10 patients. Although serum uric acid decreased significantly (p = 0.0341), yet the change in urinary uric acid (although marginal) was not significant (from 238.2 to 270.6 mg/24 h, p =0.0730) at the end of study relative to pre-Dapagliflozin treatment (Table 1 and Figure 1), in addition, we failed to demonstrate a significant relation between serum uric acid and urinary uric acid excretion (r = 0.12, p = 1.0255) (Figure 4). But there was no significant change in 24-h urinary protein excretion (from 0.632 g to 610 g/24 h, p = 1.8083) (Figure 1).
We also noticed increase in the urine volume by 253.2 ml/24 h (from 445.6 to 698.8 ml/24 h, p < 0.01) (Figure 2), and significant increase in the ultrafiltration volume by 229.9 ml (from 438.5 to 668.4 ml, p < 0.01) (Table 1). After 6 months of Dapagliflozin therapy, the PTS (as judged by PET test) did not change (Table 2). In addition, serum BUN, serum creatinine and 24-hour urine creatinine clearance before and 6-month after Dapagliflozin was nearly the same without significant change (p = 1.7623, p = 2.0107, and p = 2.4223, respectively) (Table-1 & Figure 2). Furthermore, there were significant improvements in the serum inflammatory markers. ESR dropped by 37.9 mm/h (from 66.3 to 28.4 mm/h, p = 0.0127), serum CRP decreased by 2.0 mg/dl (from 3.6 to 1.6 mg/dl, p < 0.01), serum ferritin by 261.3 µg/L (from 674.2 to 412.9, p = 0.0226), and serum CA-125 decreased by 21.3 U/ml (from 58.5 to 37.2 U/ml, p = 0.0151) at the end of the study compared with zer0 point (Table-3 & Figure 3). Follow-up of the available peritoneal inflammatory markers (measured in the peritoneal fluid effluent) showed significant decrease in the polymorphonuclear leucocyte count by 5.2 x 109/L (from 12.9 to 7.7 x 109/L, p = 0.0188) but marginal decrease in peritoneal CA-125 by 2.2 U/ml (from 39.8 to 37.6 U/ml, p = 1.0131) (Table 3) at the end of the study compared with its beginning.
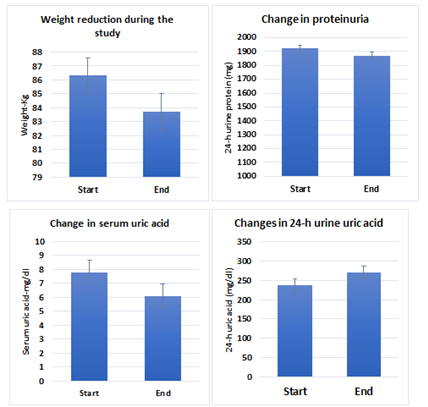
Figure 1: Changes in body eight, proteinuria, serum and urine uric acid during the study period, p = 0.033, 2.101, 0.o341, & 0.073, respectively.
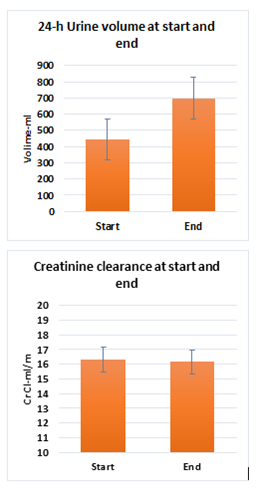
Figure 2: Progression of urinary values throughout the study. Cr Cl: creatinine clearance. p = < 0.01, 0.0144, 0.0113, 2.4223.
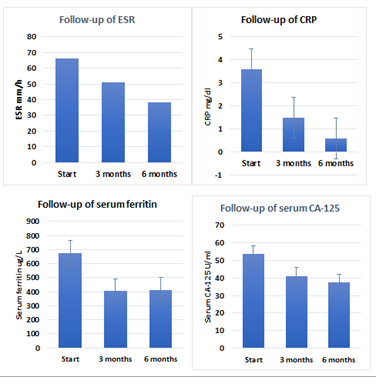
Figure 3: Follow-up of inflammatory markers. ESR: erythrocyte sedimentation rate, CRP: C-reactive protein, p = 0.022, < 0.001, 0.032, 0.015, respectively.
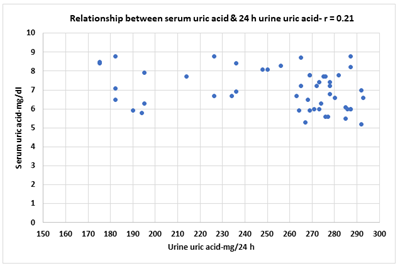
Figure 4: Correlation between serum uric acid levels (mg/dl and urine uric acid (mg/24 h), r = 0.12, p = 1.0255.
|
Characteristics |
Before Dapagliflozin |
After 6 months on Dapagliflozin |
p |
|
Age-year- Median (IQR) |
55 (47-67) |
||
|
Female sex- no % |
13 (26.0) |
||
|
Weight (Kg)- Median (IQR) |
86.3 (79.2-89.5) |
83.7 (78.6-87.8) |
0.033 |
|
Mean systolic blood pressure (mmHg) |
148 + 5.2 |
134 + 6.5 |
0.0431 |
|
FBS - mg/dl (Mean +SD) |
188.6 + 9.6 |
152.2 + 4.5 |
0.021 |
|
Hgb A1C- % median (IQR) |
7.7 (7.3-10.2) |
6.1 (5.8-6.7) |
0.0265 |
|
Anti-HTN medications |
|||
|
Beta blocker- no (%) |
10 (20.0) |
11 (22.0) |
|
|
ACEI or ARB- no (%) |
19 (38.0) |
19 (38.0) |
|
|
Aldactone-no (%) |
17 (34.0) |
17 (34.0) |
|
|
Alpha methyl dopa- no (%) |
12 (24.0) |
7 (14.0) |
|
|
Hydralazine- no (%) |
9 (18.0) |
4 (8.0) |
|
|
Furosemide- no (%) |
24 (48.0) |
17 (34.0) |
|
|
Insulin dose (glargine), mean (units) |
9.9 + 2.1 |
7.3 + 1.5 |
0.0262 |
|
BUN, mg/dl- Mean + SD |
58 + 9.3 |
55 + 10.5 |
1.7623 |
|
Creatinine, mg/dl- Mean + SD |
6.1 + 0.8 |
5.8 + 0.7 |
2.0107 |
|
Serum uric acid, mean (mg/dl) |
7.8 + 0.4 |
6.1 + 0.6 |
0.0341 |
|
24-h urine volume (mean- ml) |
445.6 + 102.3 |
698.8 + 211.5 |
< 0.01 |
|
Kt/V (mean) |
1.7 + 0.2 |
1.8 + 0.3 |
3.565 |
|
24-h urine uric acid, mean (mg/24h) |
238.2 + 18.8 |
270.6 + 20.1 |
0.073 |
|
24-h urine protein, mean (gram) |
0.632 |
0.611 |
1.8083 |
|
UF (liter/day) (mean) |
0.438.5 + 0.68.5 |
0.668.4 + 0.57.3 |
< 0.01 |
|
Cr Cl ml/min/1.73 m2 (mean) |
11.32 +2.2 |
10.96 + 2.9 |
2.4223 |
|
IQR: interquartile ratio, FBS: fasting blood sugar, Hgb A1C: hemoglobin A1C, ACEI: angiotensin converting enzyme inhibitors, ARB: angiotensin receptor blocker, BUN: blood urea nitrogen, UF: ultrafiltration volume, Cr Cl: urine creatinine clearance. |
|||
Table 1: Baseline demographic characteristics of patients.
|
Membrane type |
Before |
After |
P |
|
Low, number (%) |
3 (6.0) |
3 (6.0) |
NS |
|
Low average, number (%) |
20 (40.0) |
18 (36.0) |
NS |
|
High average, number (%) |
23 (46) |
25 (50.0) |
NS |
|
High, number (%) |
4 (8.0) |
4 (8.0) |
NS |
Table 2: Membrane type before & after the study
|
Inflammatory marker |
Beginning |
End |
P |
|
ESR mm/h (mean) |
66.3 + 9.5 |
28.4 + 5.4 |
0.0127 |
|
CRP mg/dl (mean) |
3.6 + 3.3 |
1.6 + 0.4 |
< 0.01 |
|
Serum ferritin µg/L |
674.2 + 78.7 |
412.9 + 63.6 |
0.0226 |
|
CA-125 U/ml |
58.5 + 11.1 |
37.2 + 4.4 |
0.0151 |
|
Peritoneal PMN |
12.9 + 2.4 x 109/L |
7.7 + 1.8 x 109/L |
0.0188 |
|
Peritoneal CA-125 U/ml |
39.8 + 5.1 |
37.6 + 6.6 |
1.0131 |
|
ESR: erythrocyte sedimentation rate, CRP: C-reactive protein, PMN: polymorphonuclear leucocytes |
|||
Table 3: Inflammatory markers at the beginning and at the end of the study
5. Discussion
Dapagliflozin selectively inhibits renal glucose reabsorption by inhibiting Sodium–Glucose Cotransporter-2 (SGLT2). It was developed as an insulin-independent treatment approach for Type 2 Diabetes Mellitus (T2DM). Dapagliflozin exhibits dose-proportional plasma concentrations with a half-life of ~17 h. Dapagliflozin demonstrates pharmacokinetic characteristics and dose-dependent glucosuria that are sustained over 24 hours [11]. By reducing the capacity for tubular glucose reabsorption and augmenting urinary glucose wasting, inhibition of SGLT2 results in improved glycemic control [12-16]. There is consistent evidence across many trials that simply improving control, regardless of how it is achieved, is associated with a lower risk of new onset microalbuminuria and a lower rate of progression to overt nephropathy [17, 18]. Moreover, these benefits appear to be sustained even if optimal glycemic control is subsequently lost [18]. In addition, both the United Kingdom Prospective Diabetes Study (UKPDS) and the Action in Diabetes and Vascular Disease Controlled Evaluation (ADVANCE) study reported a significant reduction in patients requiring renal replacement therapy [19-21] following intensification of glycemic control. In addition, reduction in glucose levels also leads to a compensatory fall in insulin secretion. Both result in a significant decline in insulin levels in patients with type 2 diabetes. This reduction may contribute to beneficial effects in many organs subject to the ravages of hyperinsulinism, including the kidneys [22]. In our study, we observed significant reduction of systolic blood pressure after 6-month of Dapagliflozin treatment. In several studies, however, a mild natriuretic effect has been observed following treatment with dapagliflozin associated with a reduction in systolic blood pressure of ~4 mmHg [23]; greater reductions were observed in our study. Although modest, these effects on blood pressure may contribute to the potential reno-protective actions of SGLT2 inhibition. It is well established that improvements in blood pressure control in hypertensive patients are associated with improved renal outcomes. For example, in the UKPDS study of patients with type 2 diabetes, a reduction of blood pressure from 154 to 144 mm Hg was associated with a 30% reduction in microalbuminuria [24]. In the large ADVANCE study, treatment with the Angiotensin-Converting Enzyme (ACE) inhibitor, perindopril and the natriuretic, indapamide (4 mg/1.25 mg daily) lowered the systolic blood pressure by ~7 mmHg and reduced new onset microalbuminuria by 26% compared with conventional antihypertensive treatment [25].
On the contrary, reduction of blood pressure in our patients was not associated with significant lowering of urinary protein excretion. The reason for that, in our opinion, was the fact that our subjects were ESRD patients. Diabetic nephropathy is associated with impaired autoregulation of renal blood flow, meaning that drops in blood pressure will be more likely to be associated with reduction in renal perfusion in the diabetic kidney. This is particularly the case in patients with established renal impairment. In some cases, volume depletion and blood pressure lowering with dapagliflozin as well as other SGLT2 inhibitors have been associated with acute-on-chronic renal impairment [26]. For this reason, as well as the lack of glucose lowering efficacy in this setting, SGLT2 inhibitors have not been used in patients with moderate to severe renal impairment. Interestingly, the effect on blood pressure and weight among our patients remained preserved in the setting of advanced renal impairment, findings that were consistent with Kohan et al. work 2014 [27]. The effectiveness of SGLT2 inhibitors in patients with stage-3 or higher CKD has been demonstrated in previous work [28]. Although SGLT2 inhibitors can induce a reno-protective effect in CKD patients with improvement of proteinuria, they have a reduced glucosuric effect and pose a renal risk through aggravation of the Glomerular Filtration Rate (GFR) [27, 29, 30].
The GFR aggravation can be explained by glucosuria and natriuresis, leading to blood volume depletion through osmotic diuresis [27, 30]. In our study we report another benefit of the SGLT2 inhibitor Dapagliflozin. Many of our patients complained of weight gain after the initiation of insulin. According to Henry et al [31], who investigated this issue in 1993, insulin causes a dose-related weight gain. SGLT2 inhibitors can overcome the problem of weight gain by increasing excretion of glucose and reducing the required insulin dose. As a result, our patients experienced further weight loss compared with their average weight prior to Dapagliflozin treatment. A dramatic weight reduction can improve a patient’s quality of life, in addition to achieving glucose control. Also, a recent study reported that even a short-term intensive weight reduction intervention resulted in improvements of GFR, glucose control, and physical activity [32]. Although we did not observe improvement of proteinuria in our study, yet there was significant reduction in serum uric acid without expanding uricosuric effect. It has been argued that uric acid and its metabolic effects may contribute to microvascular injury, especially in the kidney [33]. Consequently, recent attention has turned to whether lowering uric acid levels may have direct reno-protective effects in diabetes. In two small clinical studies, inhibition of xanthine oxidase has achieved a modest reduction in albuminuria [34, 35]. Whether this is due to the antioxidant effects of inhibition of xanthine oxidase or secondary to lowering uric acid is unclear but uricosuria following SGLT2 inhibition was not consistent in different studies [36]. All patients included in our study were able to reduce their insulin dose significantly. Although promising, such actions remain to be established by comprehensive clinical trials with a renal focus, many of which are currently in progress [9]. Balzer, et al demonstrated (a) the existence of SGLT2 at the murine and human peritoneum, (b) differential regulatory effects of a high-glucose environment on peritoneal glucose transporter expression, and (c) the effect of pharmacological intervention on the expression of SGLT2. Those authors further evaluated the effects of pharmacological SGLT2 inhibition on the development of structural and functional changes in the peritoneal membrane. As expected, pronounced submesothelial thickening and fibrosis developed after a 5-week exposure to PD fluid. Most importantly, Ultrafiltration (UF) improved, as evaluated after a 120 min intraperitoneal dwell time of 4.25% glucose PD fluid. All aforementioned changes were substantially mitigated by pharmacological SGLT2 inhibition with dapagliflozin [9]. It should be noted, however, that there was a trend toward high-glucose-independent structural profibrotic changes in animals receiving saline + dapagliflozin. Although such changes were expected to have some effect on the PTS, our study on real patients did not confirm that, as we did not observe changes in PTS with prolonged dapagliflozin treatment. In our study, dapagliflozin increased UF capacity, which is consistent with findings from other reports demonstrating the effect of dapagliflozin in reducing peritoneal fibrotic changes, resulting in amelioration of PD fluid-induced ultrafiltration failure [9]. In animal studies, dapagliflozin was shown to exert antioxidant and anti-inflammatory actions in the kidney, liver, and lungs [37, 38].
There is also data supporting a CRP-lowering effect of dapagliflozin in humans [39]. In this context, dapagliflozin 10mg/day combined with metformin significantly lowered CRP (from 6.2±1.1 to 3.1±0.7mg/L, p<0.05) in 59 T2DM patients at 12 weeks [40] as did dapagliflozin 5mg/day after 12weeks (from 2410±2814 to 1607±1960ng/mL, p<0.01) in 27 T2DM patients [41]. In a randomized, placebo-controlled study (n=32 T2DM patients), 8weeks of dapagliflozin (10mg/day) therapy led to a significant reduction in IL-6 levels (by −1.87pg/mL, p<0.05) [42]. A post-hoc analysis of another randomized, double-blind, placebo-controlled trial involving 33 T2DM patients, found that urinary IL-6 excretion was significantly decreased by 23.5% (p=0.04) [43]. With regard to ferritin, there are studies reporting that dapagliflozin 5mg/day significantly decreased ferritin levels in T2DM patients with either NASH or Non-Alcoholic Fatty Liver Disease (NAFLD) [41, 44]. Similarly, ferritin was significantly lowered after 12weeks of dapagliflozin (10mg/day) therapy in 26 obese T2DM patients [45]. Other studies showed that dapagliflozin can modulate the intraperitoneal Inflammatory response and inhibit the development of peritoneal fibrosis, angiogenesis, and UF failure in a high-glucose milieu [9]. We, therefore, analyzed the composition of intraperitoneal cell influx in the effluents obtained after a 4-hour dwell time of PD fluid. Consistent with previous findings from Wang, et al [46], adding dapagliflozin to the anti-diabetic regimen in our patients was associated with a significant reduction in the polymorphonuclear neutrophils in the effluent PD fluid. Concurrently, intraperitoneal cytokine levels measured in effluents by ELISA demonstrated significant decrease in proinflammatory markers CA-125 and ferritin after 6-month treatment with dapagliflozin [9]. It is important, however, to mention that despite the multitude of reports, the data analyzed fall short of providing good evidence for a clinically significant anti-inflammatory effect of dapagliflozin for several reasons. Firstly, studies were frequently small or detecting small differences. Secondly, studies of similar design yielded contrasting results for one or the other biomarker, or inconsistent findings across biomarkers. Thirdly, with all these limitations, perhaps the most suggestive data are those on ferritin for the SGLT2 inhibitors, which may be related to enhanced erythropoiesis rather than tissues inflammation. Finally, and perhaps more importantly, most studies (including ours) did not control, or were not equipoised, for the anti-hyperglycemic effect, so that a bona fide pharmacologic effect of a given drug cannot be distinguished from a non-specific effect of improved glycemia. In any event, large, multiethnic, equipoised trials would be required to determine whether certain antidiabetic drugs exert inherent anti-inflammatory properties above and beyond their antihyperglycemic efficacy. In addition, our results indicated that dapagliflozin increased urine volume. The diuretic effect of dapagliflozin can be attributed to both osmotic diuresis and natriuretic diuresis. We noted that the natriuretic effects of dapagliflozin appeared to be weaker compared to traditional diuretics. This was previously evidenced by smaller increases in renin and aldosterone in head-to-head studies [47-49]. The osmotic diuresis, thus, seems to be the driving component of the diuretic effects of SGLT2 inhibitors [49-52].
Similarly, the improvement of ultrafiltration volume in our patients might be due to better glycemic control, sustained osmotic gradient across peritoneal membrane (although we did not record changes in PTS) and significant reduction in inflammatory markers. Our study suggested additional beneficial effects of dapagliflozin on serum uric acid reduction, which was consistent with previously published studies [53, 54]. A mean reduction of 1.7 mg/dl of serum uric acid levels was observed with dapagliflozin in our patients. The considerable reduction of serum uric acid levels was similar to that shown in a meta-analysis, which revealed a significant reduction in uric acid levels with dapagliflozin 10 mg treatment [54]. Potential mechanisms behind hypouricemic effect of dapagliflozin have not yet been established; however, the renal SLC2A9 transporter that transports both uric acid and D-glucose may be involved [55, 56]. Our study failed to detect a correlation between changes in serum uric acid levels and the 24-hour urinary uric acid (Figure 4). This may be due to a small sample or short duration of follow-up. Besides, other mechanisms possibly played a role in the hypouricemic effect of dapagliflozin. Further trials and fundamental studies are needed to clarify such phenomena. Our study had certain limitations; firstly, the sample size was small, secondly, measurement of other inflammatory markers as interleukin-6, TNF α and peritoneal ferritin was not done, thirdly, measurements of plasma volumes were not available and finally the histopathological changes in the peritoneal membrane before and after commencing dapagliflozin therapy were not performed, however to the best of our knowledge, the present study is the first one that is designed to examine the effects of oral dapagliflozin on various blood and urine parameters in ESRD patients on APD with inadequately controlled T2DM, that have not been collectively touched before.
6. Conclusion
The use of Dapagliflozin was not associated with change of the peritoneal transport status. Dapagliflozin enhances diabetic control, and its use in APD patients with type-2 diabetes mellitus was useful in improving the UF and average urine volumes. In addition, it seems that such treatment decreases most of the inflammatory markers, both in the serum and PD effluent. Dapagliflozin did not affect the urine creatinine clearance or proteinuria, but it may have some hypouricemic effect, its pathogenesis needs further investigations.
Declarations
Ethics approval and consent to participate and to publish
After obtaining study-related approvals from the Ethics committee of King Fahd Hospital of the University, written informed consents to participate in and to publish the study was also obtained from all patients or their legal guardians. Study protocols conformed to the ethical principles of medical research involving human subjects based on the Helsinki Declaration. The authors also declare their consent to publication of this manuscript in “BMC Nephrology”
Availability of data and materials
All data and materials are presented in detail within the manuscript.
Declaration of conflicting (competing) interests
The author (s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author (s) received no financial support for the research, authorship, and/or publication of this article.
Acknowledgements
The authors would like to express their sincere regards to all staff of peritoneal dialysis unit and to extend their gratitude to the staff of the cardiology unit at King Fahd Hospital of the University, Saudi Arabia.
References
- Gerich J, Bastien A. Development of the sodium-glucose co-transporter 2 inhibitor dapagliflozin for the treatment of patients with type 2 diabetes mellitus. Expert Rev Clin Pharmacol 4 (2011): 669-83.
- Ferrannini E, Ramos S, Salsali A, et al. Dapagliflozin monotherapy in type 2 diabetic patients with inadequate glycemic control by diet and exercise: a randomized, double-blind, placebo-controlled, phase 3 trial. Diabetes Care 33 (2010): 2217-2224.
- Gilbert R. Sodium-glucose linked transporter-2 inhibitors: potential for renoprotection beyond blood glucose lowering? Kidney Int 86 (2013): 693-700
- Heise T, Jordan J, Wanner C, et al. Pharmacodynamic Effects of Single and Multiple Doses of Empagliflozin in Patients with Type 2 Diabetes. Clin Ther 38 (2016): 2265-2276.
- Wilcox CS, Shen W, Boulton DW, et al. Interaction Between the Sodium-Glucose-Linked Transporter 2 Inhibitor Dapagliflozin and the Loop Diuretic Bumetanide in Normal Human Subjects. J. Am. Heart Assoc 7 (2018): 007046.
- Solini A, Giannini L, Seghieri M, et al. Dapagliflozin acutely improves endothelial dysfunction, reduces aortic stiffness and renal resistive index in type 2 diabetic patients: A pilot study. Cardiovasc. Diabetol 16 (2017): 138.
- Hallow KM, Helmlinger G, Greasley PJ, et al. Why do SGLT2 inhibitors reduce heart failure hospitalization? A differential volume regulation hypothesis. Diabetes Obes. Metab 20 (2018): 479-487.
- Lambers Heerspink HJ, de Zeeuw D, Wie L, et al. Dapagliflozin a glucose-regulating drug with diuretic properties in subjects with type 2 diabetes. Diabetes Obes. Metab 15 (2013): 853-862.
- Balzer MS, Rong S, Nordlohne J, et al. SGLT2 inhibition by intraperitoneal dapagliflozin mitigates peritoneal fibrosis and ultrafiltration failure in a mouse module of chronic peritoneal exposure to high glucose dialysate. Biomolecules 10 (2020): 1573-1592.
- Twardowski ZJ, Nolph KD, Khanna R. Peritoneal equilibration test. Perit Dial Bull 7 (1987): 138-147.
- Komoroski B, Vachharajani N, Boulton D, et al. Dapagliflozin, a novel SGLT2 inhibitor, induces dose-dependent glucosuria in healthy subjects. Clin Pharmacol Therap 85 (2009): 520-526.
- Yang L, Li H, Bui A, et al. Pharmacokinetic and pharmacodynamic properties of single- and multiple-dose of dapagliflozin, a selective inhibitor of SGLT2, in healthy Chinese subjects. Clin Tharap 35 (2013): 1211-1222.
- Komoroski B, Vachharajani N, Feng Y, et al. Dapagliflozin, a novel, selective SGLT2 inhibitor, improved glycemic control over 2 weeks in patients with type 2 diabetes mellitus. Clin Pharamacol Therap 85 (2009): 513-519.
- Henry RR, Rosenstock J, Edelman S, et al. Exploring the potential of the SGLT2 inhibitor dapagliflozin in type 1 diabetes: a randomized, double-blind, placebo-controlled pilot study. Diabetes Care 38 (2015): 412-419.
- Albarrán OG, Ampudia-Blasco FJ. Dapagliflozin, the first SGLT-2 inhibitor in the treatment of type 2 diabetes. Med Clin (Barc) 141 (2013): 336-343.
- Davidson JA, Kuritzky L. Sodium glucose co-transporter 2 inhibitors and their mechanism for improving glycemia in patients with type 2 diabetes. Postgrad Med 126 (2014): 33-48.
- Petrykiv SI, Laverman GD, de Zeeuw D, et al. The albuminuria-lowering response to dapagliflozin is variable and reproducible among individual patients. Diabetes Obes. Metab 19 (2017): 1363-1370.
- Tsimihodimos V, Filippatos TD, Elisaf MS. SGLT2 inhibitors and the kidney: Effects and mechanisms. Diabetes Metab Syndr 12 (2018): 1117-1123.
- Adler AI, Stevens RJ, Manley SE, et al. Development and progression of nephropathy in type 2 diabetes: the United Kingdom Prospective Diabetes Study (UKPDS 64). Kidney Int 63 (2003): 225-232.
- Knudsen ST, Cooper ME. Renal protection: What have we learnt from ADVANCE about kidney disease in type 2 diabetes? Diabetes Obes Metab 22 (2020): 12-18.
- Merlin C Thomas. Renal effects of dapagliflozin in patients with type 2 diabetes. Ther adv Endocrinol Metab 5 (2014): 53-67.
- Han S, Susztak K. The hyperglycemic and hyperinsulinemic combo gives you diabetic kidney disease immediately. Focus on ‘combined acute hyperglycemic and hyperinsulinemic clamp induced profibrotic and proinflammatory responses in the kidney’. Am J Physiol Cell Physiol 306 (2012): 198-199.
- Musso G, Gambino R, Cassader M, et al. A novel approach to control hyperglycemia in type 2 diabetes: Sodium Glucose Co-Transport (SGLT) inhibitors: systematic review and meta-analysis of randomized trials. Ann Med 44 (2012): 375-393.
- UK Prospective Diabetes Study Group. Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. Br Med J 317 (1998): 703-713.
- Patel A. Advance Collaborative Group. Effects of a fixed combination of perindopril and indapamide on macrovascular and microvascular outcomes in patients with type 2 diabetes mellitus (the ADVANCE trial): a randomized controlled trial. Lancet; 370 (2007): 829-840.
- Scheen A. Evaluating SGLT2 inhibitors for type 2 diabetes: pharmacokinetic and toxicological considerations. Expert Opin Drug Metab Toxicol 10 (2014): 647-663.
- Kohan D, Fioretto P, Tang W, et al. Long-term study of patients with type 2 diabetes and moderate renal impairment shows that dapagliflozin reduces weight and blood pressure but does not improve glycemic control. Kidney Int 85 (2014): 962-971.
- Hyun Sun Park, Youn Joo Jung, Dong-Young Lee, et al. Use of dapagliflozin in patients with advanced diabetic kidney disease. Kidney Res Clin Pract 37 (2018): 292-297.
- Heerspink HJ, Perkins BA, Fitchett DH, et al. Sodium glucose cotransporter 2 inhibitors in the treatment of diabetes mellitus: cardiovascular and kidney effects, potential mechanisms, and clinical applications. Circulation 134 (2016): 752-772.
- Yale JF, Bakris G, Cariou B, et al. Efficacy and safety of canagliflozin in subjects with type 2 diabetes and chronic kidney disease. Diabetes Obes Metab 15 (2013): 463-473.
- Henry RR, Gumbiner B, Ditzler T, et al. Intensive conventional insulin therapy for type II diabetes. Metabolic effects during a 6-mo outpatient trial. Diabetes Care 16 (1993): 21-31.
- Friedman AN, Chambers M, Kamendulis LM, et al. Short-term changes after a weight reduction intervention in advanced diabetic nephropathy. Clin J Am Soc Nephrol 8 (2013): 1892-1898.
- Kang D, Nakagawa T, Feng L, et al. A role for uric acid in the progression of renal disease. J Am Soc Nephrol 13 (2002): 2888-2897.
- Goicoechea M, De Vinuesa S, Verdalles U, et al. Effect of allopurinol in chronic kidney disease progression and cardiovascular risk. Clin J Am Soc Nephrol 5 (2010): 1388-1393.
- Momeni A, Shahidi S, Seirafian S, et al. Effect of allopurinol in decreasing proteinuria in type 2 diabetic patients. Iran J Kidney Dis 4 (2010): 128-132.
- Cheeseman C. Solute carrier family 2, member 9 and uric acid homeostasis. Curr Opin Nephrol Hypertens 18 (2009): 428-432.
- Kingir ZB, Ozdemir Kural ZN, Cam ME, et al. Effects of dapagliflozin in experimental sepsis model in rats. Ulus Travma Acil Cerrahi Derg 25 (2019): 213-221.
- Leng W, Ouyang X, Lei X, et al. The SGLT-2 inhibitor dapagliflozin has a therapeutic effect on atherosclerosis in diabetic ApoE (−/−) mice. Mediators Inflamm 2016 (2016):6305735.
- Katsiki N, Papanas N, Mikhailidis DP. Dapagliflozin: more than just another oral glucose-lowering agent? Expert Opin Investig Drugs 19 (2010): 1581-1589.
- Di Fusco D, Dinallo V, Monteleone I, et al. Metformin inhibits inflammatory signals in the gut by controlling AMPK and p38 MAP kinase activation. Clin Sci (Lond) 132 (2018): 1155-1168.
- Tobita H, Sato S, Miyake T, et al. Effects of dapagliflozin on body composition and liver tests in patients with nonalcoholic steatohepatitis associated with type 2 diabetes mellitus: a prospective, open-label, uncontrolled study. Curr Ther Res Clin Exp 87 (2017): 13-19.
- Latva-Rasku A, Honka MJ, Kullberg J, et al. The SGLT2 inhibitor dapagliflozin reduces liver fat but does not affect tissue insulin sensitivity: a randomized, double-blind, placebo-controlled study with 8-week treatment in type 2 diabetes patients. Diabetes Care 42 (2019): 931-937.
- Dekkers CCJ, Petrykiv S, Laverman GD, et al. Effects of the SGLT-2 inhibitor dapagliflozin on glomerular and tubular injury markers. Diabetes Obes Metab 20 (2018): 1988-1993.
- Aso Y, Kato K, Sakurai S, et al. Impact of dapagliflozin, an SGLT2 inhibitor, on serum levels of soluble dipeptidyl peptidase-4 in patients with type 2 diabetes and non-alcoholic fatty liver disease. Int J Clin Pract 73 (2019): 13335
- Ghanim H, Abuaysheh S, Hejna J, et al. Dapagliflozin Suppresses Hepcidin and Increases Erythropoiesis. J Clin Endocrinol Metab 105 (2020): 57.
- Wang L, Balzer MS, Rong S, et al. Protein kinase C alpha inhibition prevents peritoneal damage in a mouse model of chronic peritoneal exposure to high-glucose dialysate. Kidney Int 89 (2016): 1253-1267.
- Wilcox CS, Shen W, Boulton DW, et al. Interaction Between the Sodium-Glucose-Linked Transporter 2 Inhibitor Dapagliflozin and the Loop Diuretic Bumetanide in Normal Human Subjects. J. Am. Heart Assoc 7 (2018): 007046.
- Solini A, Giannini L, Seghieri M, et al. Dapagliflozin acutely improves endothelial dysfunction, reduces aortic stiffness and renal resistive index in type 2 diabetic patients: A pilot study. Cardiovasc. Diabetol 16 (2017): 138.
- Lambers Heerspink HJ, de Zeeuw D, Wie L, et al. Dapagliflozin a glucose-regulating drug with diuretic properties in subjects with type 2 diabetes. Diabetes Obes. Metab 15 (2013): 853-862.
- Hallow KM, Helmlinger G, Greasley PJ, et al. Why do SGLT2 inhibitors reduce heart failure hospitalization? A differential volume regulation hypothesis. Diabetes Obes. Metab 20 (2018): 479-487.
- Sha S, Polidori D, Heise T, et al. Effect of the sodium glucose co-transporter 2 inhibitor canagliflozin on plasma volume in patients with type 2 diabetes mellitus. Diabetes Obes. Metab 16 (2014): 1087-1095.
- Ohara K, Masuda T, Murakami T, et al. Effects of the sodium-glucose cotransporter 2 inhibitor dapagliflozin on fluid distribution: A comparison study with furosemide and tolvaptan. Nephrology (Carlton) 24 (2018): 904-911.
- Ahmadieh H, Azar S. Effects of sodium glucose cotransporter-2 inhibitors on serum uric acid in type 2 diabetes mellitus. Diabetes Technol Ther 19 (2017): 507-512.
- Zhao Y, Xu L, Tian D. Effects of sodium-glucose co-transporter 2 (SGLT2) inhibitors on serum uric acid level: a meta-analysis of randomized controlled trials. Diabetes Obes Metab 20 (2018): 458-462.
- McGill JB. The SGLT2 inhibitor empagliflozin for the treatment of type 2 diabetes mellitus: a bench to bedside review. Diabetes Ther 5 (2014): 43-63.
- Li S, Sanna S, Maschio A, et al. The GLUT9 gene is associated with serum uric acid levels in Sardinia and Chianti cohorts. PLoS Genet 3 (2007): 194.
