The Role of Exercise on Sarcopenia
Article Information
Guilherme Wesley Peixoto da Fonseca, Francis Ribeiro de Souza, Marcelo Rodrigues dos Santos, Maria Janieire de Nazaré Nunes Alves*
Heart Institute (InCor), University of São Paulo Medical School, SP 05403-900, São Paulo, Brazil
*Corresponding Author: Prof. Maria Janieire de Nazaré Nunes Alves, MD, PhD, Department of Cardiovascular Rehabilitation and Exercise Physiology, Heart Institute (InCor), University of São Paulo Medical School, Av. Dr. Enéas Carvalho de Aguiar, 44 – Cerqueira César, SP 05403-900, São Paulo, Brazil
Received: 01 June 2020; Accepted: 09 June 2020; Published: 17 October 2020
Citation: Fonseca GWPD, Francis Ribeiro de Souza, Marcelo Rodrigues dos Santos, Maria Janieire de Nazaré Nunes Alves. The Role of Exercise on Sarcopenia. Cardiology and Cardiovascular Medicine 4 (2020): 540-562.
View / Download Pdf Share at FacebookAbstract
The skeletal muscle is a highly adaptable tissue and orchestrates many responses related to a healthy status. Sarcopenia, the age-related decline in muscle mass, muscle strength, and physical performance, has shown to affect up to 36.5% of adults aged ≥60 years. Currently, muscle strength has been considered as the primary indicator of sarcopenia. In this context, resistance training and its modalities have shown to improve the parameters of sarcopenia with positive impact on reducing the incidence of falls and fractures. The outcomes of resistance training are mediated primarily by an inversely proportional relationship between intensity and volume. Muscle strength gains seem to be more pronounced with higher intensity, whereas volume may be more effective in promoting muscle mass accrual. However, muscle strength improvement must increase physical performance to reflect significant clinical outcomes and adding exercise related to activities of daily living and aerobic training can be important tools in this process. In addition, resistance training associated with nutrition interventions can have additional effect on muscle mass accrual. In conclusion, adherence and safety of exercise training are important to the success of an exercise training program and depends on instruction by qualified professionals such as personal trainers and exercise physiologists.
Keywords
Aerobic training; Muscle strength; Physical performance; Resistance training; Sarcopenia
Aerobic training articles, Muscle strength articles, Physical performance articles, Resistance training articles, Sarcopenia articles
Article Details
Abbreviations
1 RM One repetition maximum
ACSM American College of Sports and Medicine
Akt Protein kinase B
ALM Appendicular lean mass
ASM Appendicular skeletal muscle mass
BIA Bioelectrical impedance analysis
BMI Body mass index
CRP C-reactive protein
CSA Cross-sectional area
CT Computed tomography
DHEA-S Dehydroepiandrosterone sulfate
DXA Dual energy X-ray absorptiometry
EWGSOP European Working Group on Sarcopenia in Older People
GS Grip strength
IGF-1 Insulin-like growth factor 1
IL-6 Interleukin 6
MRI Magnetic resonance imaging
mTOR Mammalian target of rapamycin
NFκ-B Nuclear factor kappa-light-chain-enhancer of activated B-cells
SDStandard deviation
SMISkeletal muscle mass index
TNFTumor necrosis factor
VO2max Maximal oxygen consumption
1.Introduction
The skeletal muscle is the most abundant and malleable tissue of the human body. It comprises nearly 40% of the body weight, accommodating between 50-75% of the entire body proteins [1]. Despite its unique role on movement and locomotion, skeletal muscle, through contraction, orchestrates a variety of responses to maintain a healthy status, which includes the release of myokines with anti-inflammatory properties, glucose homeostasis control, production of heat to maintain body temperature, vascular plasticity, intrathoracic pressure during breathing, and gastrointestinal motility [2-4].
During the last three decades, since 1989 when Irwin Rosenberg coined the term sarcopenia referring to the loss of skeletal muscle mass [5], the importance of skeletal muscle to bodily functions has been receiving increased attention. The term derived from the Greek, which Sarx means “flesh” and penia means “loss”. Initially, sarcopenia referred only to the progressive reduction in skeletal muscle mass due to the “healthy” ageing process and did not include function parameters as part of the syndrome. Along the subsequent years, definitions were primarily based on muscle mass alone to identify sarcopenia [6,7]. In fact, the term dynapenia has also been introduced to differentiate reduced muscle strength from changes in muscle mass, which both seem to occur in a different rate [8,9]. In 2010, the European Working Group on Sarcopenia in Older People (EWGSOP) proposed a series of performance criteria (muscle strength and physical performance) to be added to the reduction of muscle mass to diagnose sarcopenia [10]. Then, the relevance of each criterion in the syndrome spectrum was edited in 2019, ascribing muscle strength as the main factor [11].
In essence, sarcopenia can be acknowledged as an organ failure representing a muscle insufficiency to maintain contractibility properties in the absence of contractile component (muscle mass) and/or adequate contraction efficiency (muscle strength/endurance). As the mean age of the population continues to rise, sarcopenia has become a public health issue worldwide.
Sarcopenia has been associated with physical disability [7], increased risk of falls and fractures [12], poor quality of life, increased hospital length of stay [13], and higher all-cause and cardiovascular mortality [14]. In a multi-continent study, it was estimated to affect from 12.6% (Poland) to 17.5% (India) of the general population [15], whereas, in a population-base study from the USA, the prevalence of sarcopenia reached up to 36.5% in adults aged 60 years or older [14]. The discrepancy between studies may be related to the different methods applied to measure muscle mass and criteria to define sarcopenia. Moreover, these numbers can vary even more in subpopulations with chronic disease, such as chronic heart failure [16], chronic obstructive pulmonary disease [17], chronic kidney disease [18], and cancer [19].
Although the mechanisms involved in the development of sarcopenia are still inconclusive, increased pro-inflammatory cytokines [20], reduced anabolic hormone concentrations [21], mitochondrial dysfunction [22], inadequate nutrition/digestive disorders [23], physical inactivity [24], and loss of motor neurons [25] may be key drivers of the syndrome. In addition, the proposed treatment of sarcopenia has been based on resistance exercise usually associated with aerobic exercise and nutrition intervention with a higher caloric or protein intake [26]. Several pharmacological agents that act in many aspects of the muscle wasting spectrum have also been studied, but none of them is currently approved by regulatory agencies, showing limited effect on clinical outcomes like muscle strength and physical function [27].
Therefore, considering the low cost and effectiveness of exercise training and the positive effect with low adverse events in elderly subjects with sarcopenia, the aim of this review is to outline the specific role of exercise training on sarcopenia and discuss strategies to enhance muscle strength and muscle mass.
2. Definition of sarcopenia
Sarcopenia is a syndrome defined by a global and progressive reduction in skeletal muscle mass associated with changes in muscle strength and physical performance. The muscle wasting and decreased function have a great impact on adverse outcomes, such as falls, fractures, physical disability and mortality, with a significant socioeconomic burden [11]. Sarcopenia has also been recognized as a disease entity and received ICD-10 code in 2016 (M62.84) [28], which may lead to an increased awareness of the condition by physicians and interest of pharmaceutical companies in investing on new compounds for prevention and treatment. However, a consensus about sarcopenia that encompasses research and clinical practice has not been reached yet.
In 1998, Baumgartner et al. proposed the first definition of sarcopenia using dual energy X-ray absorptiometry (DXA), which was based on the sum of lean mass in both arms and legs, termed appendicular skeletal muscle mass (ASM), divided by height squared. The cutoff values for sarcopenia were ASM/height2 ≤7.26 kg/m2 for men and ASM/height2 ≤5.45% kg/m2 for women [6]. These values represented two standard deviations (SD) below the mean value of a sex-specific population between 18-40 years of age. Indeed, it was a very similar approach applied to body mass index (BMI) to classify individuals according to body composition categories.
In 2002, Janssen et al. introduced another definition, dividing total skeletal muscle mass by total body mass, and designed the term skeletal muscle mass index (SMI). Based on a sex-specific mean of SMI in a young population aged 18-39 years, this new definition intended to categorize sarcopenia into: normal SMI (within one SD), Class I (between one and two SD) and Class II (below two SD) [7]. This type of approach has also been used to classify bone mineral density into normal, osteopenia, and osteoporosis [29].
It is worthy mention that Baumgartner’s definition used DXA, while Janssen measured muscle mass by bioelectrical impedance analysis (BIA). Although other methods to measure muscle mass, such as magnetic resonance imaging (MRI) and computed tomography (CT), have been considered gold standard tests in the assessment of muscle mass with a great reliability compared to cadaver skeletal muscle measurement [30], the high cost, the need of highly skilled personnel, and the lack of cut-off points to define sarcopenia have been limiting the application of these methods in clinical setting [31].
Moreover, the equation proposed by Baumgartner et al. underestimates body fat percentage, limiting its applicability in patients who presented sarcopenia associated with higher amount of fat mass (sarcopenic obesity) [6]. In this perspective, Newman et al. proposed an index based on the residual of a linear regression that included appendicular lean mass (ALM; initially termed ASM by Baumgartner et al.) as the dependent variable and height and total fat mass as the independent variables [32]. Thus, a negative residual indicated a sarcopenic individual, while a positive residual indicated a muscular individual. In fact, this method was able to identify patients with sarcopenia, especially those with BMI ≥25kg/m2, showing a strong correlation with Baumgartner’s definition (r=0.88 for men; r=0.71 for women).
In 2014, Studenski et al. proposed another index to identify sarcopenia that corrected ALM by BMI and also included grip strength (GS) [33]. The cut-off values for sarcopenia were ALM/BMI <0.789 + GS <26 kg for men and ALM/BMI <0.512 + GS <16 kg for women. Recently, our group showed that, by applying different definitions to detect sarcopenia in patients with heart failure, the prevalence of sarcopenia was similar across all patients regardless of BMI values (normal vs. overweight/obese categories) [16]. In other words, when appropriate definitions are used, obese patients can be as affected by sarcopenia as lean patients.
More recently, the EWGSOP revised the criteria proposed in 2010 to diagnose sarcopenia, which at the time included reduced muscle mass assessed by DXA or BIA associated with either impaired muscle strength (GS <30 kg for men and GS <20 kg for women or GS according to BMI categories and sex) or decreased physical performance (Short Physical Performance Battery ≤8 or gait speed <0.8 m/s) [10]. The new revised definition draws attention to the low muscle strength as the primary indicator of sarcopenia (GS <27 kg for men and GS <16 kg for women) because low muscle strength may have a good sensitivity to detect sarcopenia in the early stage of the syndrome, before the occurrence of reduced muscle mass [11]. This paradigm shift occurred because muscle strength has shown to be better than muscle mass at predicting adverse outcomes [34-37]. However, future studies applying these criteria are necessary to validate this new definition.
3. Relationship between the loss of muscle mass and muscle strength
Several studies have intended to quantify the decline in muscle mass across the lifespan. The relationship between age and skeletal muscle mass seems to be inversely related throughout the years, but many factors may influence the magnitude of these changes [8]. Although muscle mass has been widely quoted to decrease about 1-2% per year after the fifth decade of life [38,39], this loss may not be as linear as it has been thought, with still conflicting data. Studies have shown that skeletal muscle mass can reach its peak in the third [40] and even fourth decade of life [41] followed by a plateau or a slowly progressive decline until the sixth decade [42,43], which then a steep decline takes place [44].
One of the major factors that impact the loss of skeletal muscle mass is sex with men showing a greater absolute and relative decline when compared with women, independent of changes in body weight and height [40,41,44,45]. However, women may be more affected by the reduction in skeletal muscle mass due to a lower reached peak muscle, an earlier decline in muscle mass and a greater longevity [42,46]. Additionally, the sex difference with age may also vary according to the ethnicity group. African American men and women have shown to have the highest values of muscle mass followed by White, Hispanic and Asian [40], although the drop in muscle mass may arise in a slightly different rate for these groups. In women, after the third decade of life, there was a decline in muscle mass of 1.11 kg/decade for African Americans, 0.69 kg/decade for Asians, 0.65 kg/decade for Whites, and 0.48 kg/decade for Hispanics. In men, Hispanics showed a decline of 2.03 kg/decade, followed by African Americans (1.81 kg/decade) and Whites (1.26 kg/decade) [40]. Interestingly, a higher prevalence of sarcopenia has been found in elderly Hispanics than in non-Hispanic [6].
Moreover, the loss of muscle mass may also differ from body compartments, since lower limbs have shown to contribute to a greater extent to the wasting process in comparison to the upper limbs in both sexes [41].
Nevertheless, the loss of muscle strength seems to occur in a more rapidly fashion than the loss of muscle mass, with the peak in muscle strength appearing between the second and third decade of life followed by a significant decline after the fifth decade (12-15%/decade) [47]. In a study involving 3075 participants followed for 3 years, men have shown to lose muscle strength twice as much as women and the loss of muscle strength was 3-fold greater than the loss of leg lean mass independent of sex and race [9]. Moreover, a reduction of 6-7% in muscle mass, 30% in physical performance and 60% in muscle strength have been reported in men and women over the age of 75 years compared to a younger group aged 18-19 years [48]. Supporting this data, a longer longitudinal study that followed men and women (≥75 years old) for 5 years showed a reduction of 20 and 15% in muscle strength, whereas muscle mass assessed by BIA diminished only 3.6 and 2.1%, respectively [49].
Interestingly, quadriceps muscle strength and handgrip strength, but not muscle mass, have shown to be a predictor of mortality in a cohort of elderly men and women [35], while maintaining or improving muscle mass does not seem to delay the decline in muscle strength [9]. Thus, the changes in muscle mass have shown to explain only in part the decline in muscle strength [38].
This dissociation between muscle mass and muscle strength must be interpreted with caution because most of these studies have been conducted in “healthy” ageing subjects, so that muscle mass alterations can become more evident in subjects with chronic disease and associated comorbidities. In addition, the relevance of muscle function over muscle mass alone might also explain why the EWGSOP decided to place muscle strength as the first step in the algorithm to start screening for sarcopenia, even before submitting individuals to an assessment of muscle mass (DXA, BIA, MRI, CT) [11].
The mechanisms behind this interwoven relationship between muscle mass and muscle strength have not been completely elucidated, but it has been speculated that muscle weakness may lead to a decrease in physical activity and limited mobility, which consequently cause disuse of the muscular apparatus culminating in atrophy. A vicious cycle, therefore, is developed in which reduced muscle mass is thought to be the result and cause of the decline in muscle strength.
4. Mechanisms of sarcopenia
The etiology of sarcopenia is multifactorial and the maintenance of muscle mass depends on a fine balance between muscle protein synthesis and breakdown. Catabolic pathways prevail over anabolic metabolism in sarcopenic patients, and a blunted post-prandial muscle protein synthesis, known as anabolic resistance, has also been shown to affect older adults [50].
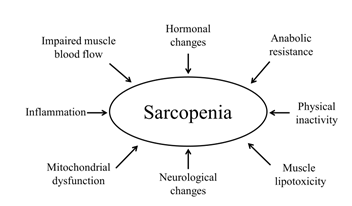
The age-related increase in intramuscular adipose tissue deposition has been associated with metabolic abnormalities (i.e., insulin resistance) and also caused alterations in performance and muscle strength [51]. The ectopic accumulation of fat within muscle, referred as muscle lipotoxicity, is believed to occur due to a reduced uptake of fat acids from adipose tissue and, as a result, skeletal muscle, a highly active tissue, captures and stores these fat acids (myosteatosis) for its own metabolic requirements [52]. This redistribution in body composition may lead to mitochondrial dysfunction reducing capability of muscle cells to oxidize free fat acids [53]. In addition, increased fat mass, especially visceral fat, can also trigger the release of pro-inflammatory cytokines, including interleukin 6 (IL-6) and tumor necrosis factor (TNF) (Figure 1) [54].
Inflammation is an acute physiological response of the immune system mediated by macrophages in the presence of infection or tissue injury, with the purpose of destroying the pathogen, repairing the damage, and restoring homeostasis again. Persistently increased levels of IL-6 and TNF even in the absence of acute infection/injury, the so-called chronic low-grade inflammation, has been negatively associated with muscle mass and muscle strength (Figure 1) [55, 56]. These inflammatory markers can act through different mechanisms. First, TNF, via the nuclear factor kappa-light-chain-enhancer of activated B-cells (NFκ-B) pathway, has shown to cause directly protein degradation in skeletal muscle myotubes [57]. Secondly, IL-6 has been reported to inhibit the mammalian target of rapamycin (mTOR) signaling pathway, resulting in reduced protein synthesis [58].
However, the sensitivity and specificity of these cytokines used as biomarkers of sarcopenia have shown to be limited, whereas C-reactive protein (CRP), another pro-inflammatory cytokine, seems to be more related to sarcopenia [59]. Longitudinal studies are necessary to determine the influence of inflammation on the development of sarcopenia and a multivariate approach, including more than one inflammatory marker, should be consider to encompass the complex pathophysiology of muscle wasting [60].
Sarcopenia can also be a result of reduced blood flow to the skeletal muscle that impairs oxygen delivery and nutrients to muscle cells (Figure 1) [61]. Interestingly, endothelial dysfunction has been linked to sarcopenia and reduced exercise capacity in patients with heart failure [62].
In addition, an age-related impairment in sex steroid hormones has been linked to sarcopenia [63]. Dehydroepiandrosterone sulfate (DHEA-S), a precursor of testosterone, is associated with muscle mass and muscle strength [64] and an imbalance between increased activity of catabolic hormones (i.e., cortisol) and impaired action of anabolic hormones (i.e., DHEA-S) has also been independently associated with sarcopenia [65]. Moreover, insulin-like growth factor 1 (IGF-1), circulating or released by muscle contraction, also plays an important role on this anabolic cascade that sustain the turnover of muscle protein synthesis via protein kinase B/mTOR (Akt/mTOR) [66] and a decline in growth hormone and IGF-1 may lead to reduced muscle mass [67].
Although the etiology of sarcopenia has been long focused on morphological changes in muscle mass, a neurogenic origin of sarcopenia based on motor neuron alterations has been proposed [68]. Indeed, reduction in the number of motor units has been reported in sarcopenic patients when compared to non-sarcopenic [25]. Interestingly, there may be prototypic disease models to study the endocrinological and neurological cause of sarcopenia (Figure 1) as suggested by Stangl et al., comprising patients suffering from diabetes, Cushing syndrome, chronic kidney disease, Klinefelter syndrome, and amyotrophic lateral sclerosis [69].
Physical inactivity, not only related to sedentary behavior, but also including prolonged bed rest and limb immobilization, has shown to contribute to the loss of muscle mass as well (Figure 1) [70-72]. Kortebein et al. showed that 10 days of bed rest caused a decrease of 1 kg in leg lean mass, a decline of 16% in knee extensor strength, and protein synthesis impairment [73]. In addition, reduced skeletal muscle mass, considered the main organ responsible for maintaining glucose homeostasis, may also lead to worsen insulin sensitivity in older subjects during bed rest, impairing anabolic responses [74].
5. Characteristics of resistance and aerobic training: intensity and volume
Regular exercise, composed mainly by resistance and aerobic training (e.g., walking, running, cycling, and swimming), is associated with numerous physical health benefits [75] and reduced risk of developing comorbidities related to sarcopenia [76]. Every program of resistance training is developed with a unique goal to improve muscle strength and muscle mass that eventually contributes to improved physical performance and quality of life, while aerobic training enhances cardiorespiratory fitness. For such purposes, a vast range of training variables can be manipulated considering that each variable can elicit a specific response in these modes of exercise.
The intensity of resistance training is the amount of weight lifted in a single exercise determined by one repetition maximum test (1 RM) of the respective movement. The volume of resistance training is characterized by the amount of time that a muscle is exposed to a certain stimulus (number of sets and repetitions). Although intensity and volume are the main variables manipulated during progressive resistance training to improve muscle strength and muscle mass, other variables, such as frequency, interval between sets, lifting cadency, type of contraction, time under tension, range of motion, and exercise grouping, have a direct impact on muscle strength and muscle mass [77].
For any mode of exercise, the relation between intensity and volume is inversely proportional so that when intensity is increased volume must be decreased, and vice-versa. In aerobic exercises, intensity is represented by a percentage of maximal oxygen consumption (VO2max), while volume consists of the exercise duration [75].
6. Resistance training, muscle strength, and physical performance
Progressive resistance training has proven to be well tolerated and safe in elderly subjects. Adequate prescription of this exercise modality can improve mobility, physical performance, muscle mass, and muscle strength, with positive results in reducing the incidence of falls, fractures, and injuries [78]. Several forms of resistance training, including weight stack, pneumatic machines, free weights, elastic bands, and body weight, have shown beneficial effects on these parameters [79,80].
Sarcopenia is thought to affect predominantly type II fibers and resistance training has shown to promote higher adaptation in this type of fibers in elderly subjects [81]. Muscle hypertrophy is one of the main benefits of resistance training mediated by satellite cells activation. In response to muscle damage, satellite cells, initially in a quiescent state, are activated and undergo proliferation and differentiation adding myonuclei to preexisting muscle cells, which in turn leads to muscle repair and regeneration [82]. Although a reduced content of satellite cells, assessed by muscle biopsy, has been found in the vastus lateralis muscle of older subjects and resistance training was able to effectively reverse this loss [83], the role of satellite cells in sarcopenia is still controversial [84].
Resistance exercise program associating flexibility, balance, and weight-lifting exercises (3 sets of 8-12 repetitions at ≥85% 1 RM) in frail elderly women and men with multiple co-morbidities showed improvement in fat-free mass as well as muscle strength in leg extension/flexion, leg press, and seated row [85]. Interestingly, as the decline in muscle mass and muscle strength throughout ageing occurs in a distinct rate, so does the improvement of these parameters due to resistance training. In a study with older subjects (≥85 years) performing progressive resistance training (3 sets of 8 repetitions at 50 to 80% 1 RM) for 12 weeks, the increment of cross-sectional area (CSA) in quadriceps muscle represents only 9.8%, whereas isokinetic knee strength improved up to 47% [81].
Other studies, applying resistance training, have shown slightly higher muscle hypertrophy [86], lower [87-89] or no changes at all [90,91], while the changes in muscle strength seem to be more pronounced and consistent across trials [86,87,92]. Even though there is a difference in absolute muscle strength between men and women at baseline and post exercise, women are still able to positively respond to resistance training [92]. In addition, sarcopenic patients may respond better to muscle strength acquisition when compared to healthy elderly subjects [93,94].
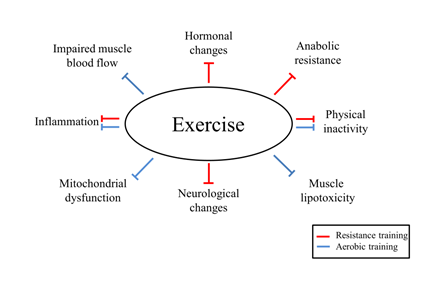
However, the increase in muscle strength is not totally dependent on the muscle mass accrual and neural adaptations play a major role in this process. Indeed, the adaptations of resistance training in older subjects seem to depend more on neural than hypertrophic adaptation [95]. The main neural adaptations of resistance training in elderly subjects may include augmented firing rate from neurons, increased motor unit recruitment, and improved agonist activation with reduced antagonist coactivation (Figure 2) [91].
The intensity of resistance training (i.e., weight lifted) has shown to be determinant in acquiring muscle strength. Comparing high intensity (>75% 1RM) to moderate (55-75% 1RM) and low intensities (<55% 1RM), higher intensity showed to be more effective in increasing muscle strength, showing a dose-dependent relationship with muscle strength in elderly subjects (≥65 years) [96].
Nonetheless, these changes in strength must reflect an enhancement of physical performance to produce significant clinical outcomes. In a study comparing supervised high intensity (80% 1RM) to low intensity (40% 1RM) resistance training in frail elderly subjects ≥70 years, the improvement in functional outcomes, assessed by six-minute walking distance test, chair-rising time, and stair climbing, were related to changes in knee extensor strength [97]. However, other authors have shown that changes in muscle strength and stair climbing occur after resistance training in spite of intensity applied [90]. A reason for this difference may be the baseline strength of the participants, considering that institutionalized subjects [97] tend to show greater improvement in the assessed parameters than community-dwelling subjects [90].
Higher intensity may elicit better function results in elderly subjects, but recent meta-analysis demonstrated substantial increases in muscle strength with low-moderate intensity (~45% 1 RM) [98]. It is important to mention that blood pressure response to high intensity resistance training (80% 1 RM) in hypertensive patients, a critical point of concern, is lower than low intensity (40% 1 RM) [99]. However, in sarcopenic patients with associated cardiovascular disease, the exercise execution should be encouraged to stop 2-3 repetitions before reaching the maximal volitional fatigue to avoid valsalva maneuver. Additionally, adherence to exercise seems to increase when intensity is moderate, whereas adding more session to the exercise program per week increases volume of exercise and does not alter adherence [100].
Other studies suggest that improving muscle power (force multiplied by velocity) may lead to greater impact on functional tasks [101]. This type of resistance training is characterized by high-velocity contraction during the concentric phase of the movement, while the eccentric phase is slower. Comparing different intensities of explosive resistance training (20% vs. 50% vs. 80% 1 RM) with pneumatic resistance machines, de Vos et al. demonstrated that muscle power was improved regardless of intensity in comparison to a non-trained control group, but there was a linear increase between intensity, muscle strength, and endurance changes [102].
Moreover, explosive resistance training based on specific exercises of mobility improved gait speed and chair rise compared to a control group that performed progressive resistance training with a consistent speed during the concentric and eccentric phase of the movement [103]. These findings indicate that the training principle of specificity, which states that a certain skill/task is performed better when it is trained, may also contribute to the improvement of strength and function observed in older subjects. Thus, adding into resistance training exercises that resemble gestures of the activities of daily living and aerobic exercises can optimize the transference of muscle strength into physical performance.
7. Resistance training and muscle mass
Resistance training has shown to be effective in producing an acute positive muscle protein balance, which can chronically leads to muscle mass accretion. Muscle hypertrophy seems to take place with an optimal combination between mechanical tension, metabolic stress, and muscle damage [77].
Intensity appears to be an important variable to promote muscle growth. Comparing sarcopenic male patients that performed high-intensity progressive resistance training to those who did not exercise, SMI improved from baseline [7.01 (6.85 to 7.16) vs. 6.89 (6.74 to 7.02) kg/m2] to one year [0.25 (0.18 to 0.33) vs. -0.08 (-.01 to -.15) kg/m2] [104]. Although the compliance with this type of exercise was very high (95%), the authors report that the prescribed intensity had not been achieved in one third of the sessions.
Apart from intensity, there are many variables that can also have a greater impact on muscle mass. In fact, low intensity resistance training (30-40% 1 RM) has proven to be as equally effective as high intensity (70-80% 1 RM) in promoting muscle protein synthesis when a volitional fatigue is reached [105]. Resistance training program with higher volume seems to elicit significant gains of lean body mass in adults ≥ 50 years (~1.1 kg) [106]. In clinical practice, the main strategies to increase volume in older populations include increased frequency of session per week and also number of sets per exercise.
However, several studies demonstrate that elderly subjects compared to young have a reduced ability to generate muscle hypertrophy [107-109] with training background, manipulation of training variables, and age itself explaining in part the impaired muscle regeneration in this population. Furthermore, though sarcopenic patients are able to increase muscle CSA with progressive resistance training (4 sets of 10 repetitions at 50 to 85% 1RM), it seems to occur to lesser degree than healthy elderly subjects, diverging from muscle strength that the adaptation is superior [93]. These findings raise attention to the importance of manipulating training variables precisely in order to optimize muscle mass gains in sarcopenia.
In elderly women with sarcopenia, resistance training with kettlebell (3 sets of 8-12 repetitions at 60-70% 1 RM) increased ALM and, consequently, ALM/height2 [110]. In addition, a reduction of CRP in the resistance exercise group was also reported in this study, without differences for IL-6 and TNF in comparison to control group (Figure 2). The impact of resistance training on reducing pro-inflammatory cytokines seems to be restricted to CRP, since studies with other populations have not demonstrated changes in IL-6 and TNF as well [111,112].
Moreover, Mero et al. showed that increased CSA of type I and II muscle fibers was more pronounced in young than in older subjects after progressive resistance training (40 to 80% 1 RM) for 21 weeks, but the authors reported a higher ingestion of protein, carbohydrates and fat per body mass in the young group, which may also explain the lower muscle hypertrophy in the older group due to a reduced caloric intake [107]. Indeed, not only total caloric intake, but the ingested amount of protein and essential amino acids in elderly may need to be higher in order to promote muscle protein synthesis [113,114], while resistance training associated with increased protein intake has shown to be more effective in increasing muscle mass than supplementation alone (Figure 2) [115]. Similar findings in patients with sarcopenia are still controversial, showing equal effect of exercise and exercise plus supplementation on ALM [116].
The impaired anabolic response in elderly subjects is thought to be related to a dysregulation of the Akt/mTOR signaling pathway [117], which is stimulated by circulating anabolic hormones. In this perspective, resistance training has shown to increase the release of testosterone associated with reduced catabolic hormones like cortisol, which leads to an improvement in muscle size and strength in older subjects (Figure 2) [118]. The differences in hormone response may also lie in types of exercise performed, considering that larger muscle groups can elicit a higher release of anabolic hormones [119]. However, other authors, despite an acute spike in anabolic hormones after a single bout of exercise, demonstrated that this acute increase was not determinant in the process of muscle hypertrophy with chronic exposure to resistance training [120]. In this regard, studies assessing the effect of anabolic hormones on muscle mass in sarcopenic patients are still lacking.
8. Aerobic exercise and sarcopenia
The multifactorial nature of sarcopenia is a great barrier to determine only a specific treatment to counteract the loss of muscle strength, muscle mass, and physical performance. In contrast to resistance training, aerobic training, such as cycling, walking, and jogging, is characterized by an enhancement of metabolic capacity [75].
To maintain the energy and oxygen supply to active skeletal muscles, metabolic adaptations take place during aerobic training, including increases in capillarization, number and size of mitochondria, and expression of oxidative enzymes (Figure 2) [121]. Paradoxically, in initial states of aerobic training, particularly with cycling, and when a subject has a very limited baseline functional capacity, modest changes in muscle mass and muscle strength may be seen [122]. However, aerobic training is effective in improving maximum aerobic power (VO2max) [123]. Despite the well-known cardiorespiratory fitness improvement documented with aerobic training, the association of aerobic and resistance training in elderly subjects showed to optimize cardiovascular benefits as well as muscle strength compared to each intervention alone [124].
Taken into consideration the limited effect of resistance training on inflammation, aerobic training can be an aligned tool to produce an anti-inflammatory state [125], as shown by reduce pro-inflammatory cytokines in rats with cachexia [126]. In fact, moderate intensity physical activity may lead to more significant reductions in CRP than those at low intensity (Figure 2) [127], suggesting that exercise intensity may also modulate inflammation.
8. Exercise prescription
The American College of Sports and Medicine (ACSM) suggests that participants engaged in resistance training should perform 1-4 sets of 8-15 repetitions at moderate (50-69% 1 RM) to vigorous intensity (70-84% 1 RM) with a rest interval of 2-3 minutes between sets [75]. The session can be comprised of 8-10 exercises for the major muscle groups and also involve balance, agility, and coordination exercises, performed with a frequency of 2-3 times per week with a 48-hour interval between sessions. It is important to mention that these variables must be manipulated according to the experience, goals, health status, and exercise response of each participant.
For aerobic training, the ACSM recommend continuous and rhythmic exercises, including walking, running, cycling, and swimming, performed either ≥5 times per week at moderate intensity (46-63% VO2max), ≥3 times per week at vigorous intensity (64-90% VO2max) or a combination of both ≥3-5 times per week [75].
9. Conclusion
In summary, the definition of sarcopenia has evolved from only considering the loss of muscle mass to including criteria of physical performance and muscle strength, with the latter being recently suggested as the primary indicator of the syndrome. In this scenario, resistance training, performed using a variety of modalities, rises as a great clinical tool to counteract the impairment of muscle mass and muscle strength. The intensity of resistance training (>70-80% 1 RM) seems to be more effective than volume in improving muscle strength, whilst reaching a maximal controllable volitional fatigue, despite the intensity (high vs. low), may be essential to promote muscle mass.
In addition, resistance training associated with nutrition interventions can have additional effect on muscle mass accrual. Transferring muscle strength into physical performance may produce better clinical outcomes, with aerobic training being an aligned therapy in this process. Finally, it is important to highlight that adherence and safety of exercise training are important to the success of an exercise training program and depends on instruction by qualified professionals such as personal trainers and exercise physiologists.
Author Contributions:
Conceptualization, M.R.S and M.J.N.N.A; Writing – Original Draft Preparation, G.W.P.D.F.; Writing – Review & Editing, M.R.S and M.J.N.N.A; Supervision, M.J.N.N.A; Funding Acquisition, M.J.N.N.A.
Funding:
This study was financed in part by the Coordenacão de Aperfeiçoamento de Pessoal de Nível Superior – Brazil (CAPES) - Finance Code 001. G.W.P.D.F. was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq; 148758/2016-9).
Conflicts of Interest:
There is no conflict of interest related to this work.
References
- Frontera WR, Ochala J. Skeletal muscle: a brief review of structure and function. Calcified Tissue International 96 (2015): 183-195.
- Pedersen BK, Febbraio MA. Muscle as an Endocrine Organ: Focus on Muscle-Derived Interleukin-6. Physiological Reviews 88 (2008): 1379-1406.
- Shulman GI, Rothman DL, Jue T, et al. Quantitation of muscle glycogen synthesis in normal subjects and subjects with non-insulin-dependent diabetes by 13C nuclear magnetic resonance spectroscopy. New England Journal of Medicine 322 (1990): 223-228.
- Macagno EO, Christensen J, Lee CL. Modeling the effect of wall movement on absorption in the intestine. American Journal of Physiology-Gastrointestinal and Liver Physiology 243 (1982): G541-G550.
- Rosenberg IH. Sarcopenia: origins and clinical relevance. The Journal of Nutrition 127 (1997): 990S-991S.
- Baumgartner RN, Koehler KM, Gallagher D, et al. Epidemiology of sarcopenia among the elderly in New Mexico. American Journal of Epidemiology 147 (1998): 755-763.
- Janssen I, Heymsfield SB, Ross R. Low relative skeletal muscle mass (sarcopenia) in older persons is associated with functional impairment and physical disability. Journal of the American Geriatrics Society 50 (2002): 889-896.
- Mitchell WK, Atherton PJ, Williams J, et al. Sarcopenia, dynapenia, and the impact of advancing age on human skeletal muscle size and strength; a quantitative review. Frontiers in Physiology 3 (2012): 260.
- Goodpaster BH, Park SW, Harris TB, et al. The loss of skeletal muscle strength, mass, and quality in older adults: the health, aging and body composition study. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 61 (2006): 1059-1064.
- Cruz-Jentoft AJ, Baeyens JP, Bauer JM, et al. Sarcopenia: European consensus on definition and diagnosisReport of the European Working Group on Sarcopenia in Older PeopleA. J. Cruz-Gentoft et al. Age and ageing 39 (2010): 412-423.
- Cruz-Jentoft AJ, Bahat G, Bauer J, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age and ageing 48 (2019): 16-31.
- Landi F, Liperoti R, Russo A, et al. Sarcopenia as a risk factor for falls in elderly individuals: results from the ilSIRENTE study. Clinical Nutrition 31 (2012): 652-658.
- Beaudart C, Zaaria M, Pasleau F, et al. Health outcomes of sarcopenia: a systematic review and meta-analysis. PloS One 12 (2017): e0169548.
- Brown JC, Harhay MO, Harhay MN. Sarcopenia and mortality among a population-based sample of community-dwelling older adults. Journal of Cachexia, Sarcopenia and Muscle 7 (2016): 290-298.
- Tyrovolas S, Koyanagi A, Olaya B, et al. Factors associated with skeletal muscle mass, sarcopenia, and sarcopenic obesity in older adults: a multi-continent study. Journal of Cachexia, Sarcopenia and Muscle 7 (2016): 312-321.
- Fonseca GW, Dos Santos MR, de Souza FR, et al. Discriminating sarcopenia in overweight/obese male patients with heart failure: the influence of body mass index. ESC Heart Failure 7 (2019): 84-91.
- Kim SH, Shin MJ, Shin YB, et al. Sarcopenia associated with chronic obstructive pulmonary disease. Journal of Bone Metabolism 26 (2019): 65-74.
- Watanabe H, Enoki Y, Maruyama T. Sarcopenia in chronic kidney disease: Factors, mechanisms, and therapeutic interventions. Biological and Pharmaceutical Bulletin 42 (2019): 1437-1445.
- Pamoukdjian F, Bouillet T, Lévy V, et al. Prevalence and predictive value of pre-therapeutic sarcopenia in cancer patients: a systematic review. Clinical Nutrition 37 (2018): 1101-1113.
- Degens H. The role of systemic inflammation in age-related muscle weakness and wasting. Scandinavian Journal of Medicine & Science in Sports 20 (2010): 28-38.
- Morley JE. Hormones and sarcopenia. Current Pharmaceutical Design 23 (2017): 4484-4492.
- Marzetti E, Calvani R, Cesari M, et al. Mitochondrial dysfunction and sarcopenia of aging: from signaling pathways to clinical trials. The International Journal of Biochemistry & Cell Biology 45 (2013): 2288-2301.
- Onishi S, Shiraki M, Nishimura K, et al. Prevalence of sarcopenia and its relationship with nutritional state and quality of life in patients with digestive diseases. Journal of Nutritional Science and Vitaminology 64 (2018): 445-453.
- Fonseca H, Powers SK, Goncalves D, et al. Physical inactivity is a major contributor to ovariectomy-induced sarcopenia. International Journal of Sports Medicine 33 (2012): 268-278.
- Drey M, Krieger B, Sieber CC, et al. Motoneuron loss is associated with sarcopenia. Journal of the American Medical Directors Association 15 (2014): 435-439.
- Bauer J, Morley JE, Schols AM, et al. Sarcopenia: a time for action. An SCWD position paper. Journal of Cachexia, Sarcopenia and Muscle 10 (2019): 956-961.
- Molfino A, Amabile MI, Rossi Fanelli F, et al. Novel therapeutic options for cachexia and sarcopenia. Expert Opinion on Biological Therapy 16 (2016): 1239-1244.
- Anker SD, Morley JE, von Haehling S. Welcome to the ICD-10 code for sarcopenia. Journal of Cachexia, Sarcopenia and Muscle 7 (2016): 512-514.
- Kanis JA. Assessment of fracture risk and its application to screening for postmenopausal osteoporosis: synopsis of a WHO report. Osteoporosis International 4 (1994): 368-381.
- Mitsiopoulos N, Baumgartner RN, Heymsfield SB, et al. Cadaver validation of skeletal muscle measurement by magnetic resonance imaging and computerized tomography. Journal of Applied Physiology 85 (1998): 115-122.
- Heymsfield SB, Adamek M, Gonzalez MC, et al. Assessing skeletal muscle mass: historical overview and state of the art. Journal of Cachexia, Sarcopenia and Muscle 5 (2014): 9-18.
- Newman AB, Kupelian V, Visser M, et al. Sarcopenia: alternative definitions and associations with lower extremity function. Journal of the American Geriatrics Society 51 (2003): 1602-1609.
- Studenski SA, Peters KW, Alley DE, et al. The FNIH sarcopenia project: rationale, study description, conference recommendations, and final estimates. Journals of Gerontology Series A: Biomedical Sciences and Medical Sciences 69 (2014): 547-558.
- Leong DP, Teo KK, Rangarajan S, et al. Prognostic value of grip strength: findings from the Prospective Urban Rural Epidemiology (PURE) study. The Lancet 386 (2015): 266-273.
- Newman AB, Kupelian V, Visser M, et al. Strength, but not muscle mass, is associated with mortality in the health, aging and body composition study cohort. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 61 (2006): 72-77.
- Schaap LA, Van Schoor NM, Lips P, et al. Associations of sarcopenia definitions, and their components, with the incidence of recurrent falling and fractures: the longitudinal aging study Amsterdam. The Journals of Gerontology: Series A 73 (2018): 1199-1204.
- Metter EJ, Talbot LA, Schrager M, et al. Skeletal muscle strength as a predictor of all-cause mortality in healthy men. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 57 (2002): B359-B365.
- Hughes VA, Frontera WR, Roubenoff R, et al. Longitudinal changes in body composition in older men and women: role of body weight change and physical activity. The American Journal of Clinical Nutrition 76 (2002): 473-481.
- Thomas DR. Loss of skeletal muscle mass in aging: examining the relationship of starvation, sarcopenia and cachexia. Clinical Nutrition 26 (2007): 389-399.
- Silva AM, Shen W, Heo M, et al. Ethnicity-related skeletal muscle differences across the lifespan. American Journal of Human Biology: The Official Journal of the Human Biology Association 22 (2010): 76-82.
- Janssen I, Heymsfield SB, Wang Z, et al. Skeletal muscle mass and distribution in 468 men and women aged 18–88 yr. Journal of Applied Physiology 89 (2000): 81-88.
- Barlett HL, Puhl SM, Hodgson JL, et al. Fat-free mass in relation to stature: ratios of fat-free mass to height in children, adults, and elderly subjects. The American Journal of Clinical Nutrition 53 (1991): 1112-1116.
- Pichard C, Kyle UG, Bracco D, et al. Reference values of fat-free and fat masses by bioelectrical impedance analysis in 3393 healthy subjects. Nutrition 16 (2000): 245-254.
- Kyle UG, Genton L, Hans D, et al. Age-related differences in fat-free mass, skeletal muscle, body cell mass and fat mass between 18 and 94 years. European Journal of Clinical Nutrition 55 (2001): 663-672.
- Gallagher D, Visser M, De Meersman RE, et al. Appendicular skeletal muscle mass: effects of age, gender, and ethnicity. Journal of Applied Physiology 83 (1997): 229-239.
- Barford A, Dorling D, Smith GD, et al. Life expectancy: women now on top everywhere. BMJ: British Medical Journal 332 (2006): 808.
- Hurley BF. Age, gender, and muscular strength. Journals of Gerontology-Biological Sciences and Medical Sciences 50 (1995): 41-44.
- Landi F, Calvani R, Tosato M, et al. Age-related variations of muscle mass, strength, and physical performance in community-dwellers: results from the Milan EXPO survey. Journal of the American Medical Directors Association 18 (2017): 88-e17.
- Dey DK, Bosaeus I, Lissner L, et al. Changes in body composition and its relation to muscle strength in 75-year-old men and women: a 5-year prospective follow-up study of the NORA cohort in Göteborg, Sweden. Nutrition 25 (2009): 613-619.
- Wall BT, Gorissen SH, Pennings B, et al. Aging is accompanied by a blunted muscle protein synthetic response to protein ingestion. PloS One 10 (2015): e0140903.
- Zoico E, Rossi A, Di Francesco V, et al. Adipose tissue infiltration in skeletal muscle of healthy elderly men: relationships with body composition, insulin resistance, and inflammation at the systemic and tissue level. Journals of Gerontology Series A: Biomedical Sciences and Medical Sciences 65 (2010): 295-299.
- Kalinkovich A, Livshits G. Sarcopenic obesity or obese sarcopenia: a cross talk between age-associated adipose tissue and skeletal muscle inflammation as a main mechanism of the pathogenesis. Ageing Research Reviews 35 (2017): 200-221.
- Guillet C, Delcourt I, Rance M, et al. Changes in basal and insulin and amino acid response of whole body and skeletal muscle proteins in obese men. The Journal of Clinical Endocrinology & Metabolism 94 (2009): 3044-3050.
- Rossi FE, Lira FS, Silva BS, et al. Influence of skeletal muscle mass and fat mass on the metabolic and inflammatory profile in sarcopenic and non-sarcopenic overfat elderly. Aging Clinical and Experimental Research 31 (2019): 629-635.
- Visser M, Pahor M, Taaffe DR, et al. Relationship of interleukin-6 and tumor necrosis factor-α with muscle mass and muscle strength in elderly men and women: the Health ABC Study. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 57 (2002): M326-M332.
- Schaap LA, Pluijm SM, Deeg DJ, et al. Higher inflammatory marker levels in older persons: associations with 5-year change in muscle mass and muscle strength. Journals of Gerontology Series A: Biomedical Sciences and Medical Sciences 64 (2009): 1183-119.
- Li YP, Reid MB. NF-kappaB mediates the protein loss induced by TNF-alpha in differentiated skeletal muscle myotubes. Am J Physiol Regul Integr Comp Physiol 279 (2000): R1165-R1170.
- White JP, Puppa MJ, Gao S, et al. Muscle mTORC1 suppression by IL-6 during cancer cachexia: a role for AMPK. American Journal of Physiology-Endocrinology and Metabolism 304 (2013): E1042-E1052.
- Bano G, Trevisan C, Carraro S, et al. Inflammation and sarcopenia: a systematic review and meta-analysis. Maturitas 96 (2017): 10-15.
- Calvani R, Marini F, Cesari M, et al. Biomarkers for physical frailty and sarcopenia: state of the science and future developments. Journal of Cachexia, Sarcopenia and Muscle 6 (2015): 278-286.
- Morley JE, Anker SD, von Haehling S. Prevalence, incidence, and clinical impact of sarcopenia: facts, numbers, and epidemiology—update 2014. Journal of Cachexia, Sarcopenia and Muscle 5 (2014): 253-259.
- dos Santos MR, Saitoh M, Ebner N, et al. Sarcopenia and endothelial function in patients with chronic heart failure: results from the studies investigating comorbidities aggravating heart failure (SICA-HF). Journal of the American Medical Directors Association 18 (2017): 240-245.
- Vitale G, Cesari M, Mari D. Aging of the endocrine system and its potential impact on sarcopenia. European Journal of Internal Medicine 35 (2016): 10-15.
- Valenti G, Denti L, Maggio M, et al. Effect of DHEAS on skeletal muscle over the life span: the InCHIANTI study. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 59 (2004): M466-M472.
- Yanagita I, Fujihara Y, Kitajima Y, et al. A high serum cortisol/DHEA-S ratio is a risk factor for sarcopenia in elderly diabetic patients. Journal of the Endocrine Society 3 (2019): 801-813.
- Barclay RD, Mackenzie RW, Burd NA, et al. The Role of the IGF-1 Signalling Cascade in Muscle Protein Synthesis and Anabolic Resistance in Ageing Skeletal Muscle. Frontiers in Nutrition 6 (2019): 146.
- Nass R. Growth hormone axis and aging. Endocrinology and Metabolism Clinics 42 (2013): 187-199.
- Kwan P. Sarcopenia, a neurogenic syndrome?. Journal of Aging Research (2013): 791679.
- Stangl MK, Böcker W, Chubanov V, et al. Sarcopenia–endocrinological and neurological aspects. Experimental and Clinical Endocrinology & Diabetes 6 (2019): 8-22.
- Paddon-Jones D, Sheffield-Moore M, Cree MG, et al. Atrophy and impaired muscle protein synthesis during prolonged inactivity and stress. The Journal of Clinical Endocrinology & Metabolism 91 (2006): 4836-4841.
- Kortebein P, Symons TB, Ferrando A, et al. Functional impact of 10 days of bed rest in healthy older adults. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 63 (2008): 1076-1081.
- Backx EM, Horstman AM, Marzuca-Nassr GN, et al. Leucine supplementation does not attenuate skeletal muscle loss during leg immobilization in healthy, young men. Nutrients 10 (2018): 635.
- Kortebein P, Ferrando A, Lombeida J, et al. Effect of 10 days of bed rest on skeletal muscle in healthy older adults. Jama 297 (2007): 1769-1774.
- Coker RH, Hays NP, Williams RH, et al. Bed rest worsens impairments in fat and glucose metabolism in older, overweight adults. Journals of Gerontology Series A: Biomedical Sciences and Medical Sciences 69 (2014): 363-370.
- Garber CE, Blissmer B, Deschenes MR, et al. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Medicine and Science in Sports and Exercise 43 (2011): 1334.
- Xu HQ, Shi JP, Shen C, et al. Sarcopenia-related features and factors associated with low muscle mass, weak muscle strength, and reduced function in Chinese rural residents: a cross-sectional study. Archives of Osteoporosis 14 (2019): 2.
- Schoenfeld BJ. The mechanisms of muscle hypertrophy and their application to resistance training. The Journal of Strength & Conditioning Research 24 (2010): 2857-2872.
- Fragala MS, Cadore EL, Dorgo S, et al. Resistance Training for Older Adults: Position Statement From the National Strength and Conditioning Association. The Journal of Strength & Conditioning Research 33 (2019).
- Sundstrup E, Jakobsen MD, Andersen CH, et al. Swiss ball abdominal crunch with added elastic resistance is an effective alternative to training machines. International Journal of Sports Physical Therapy 7 (2012): 372.
- Jakobsen MD, Sundstrup E, Andersen CH, et al. Muscle activity during leg strengthening exercise using free weights and elastic resistance: effects of ballistic vs controlled contractions. Human Movement Science 32 (2013): 65-78.
- Kryger AI, Andersen JL. Resistance training in the oldest old: consequences for muscle strength, fiber types, fiber size, and MHC isoforms. Scandinavian Journal of Medicine & Science in Sports 17 (2007): 422-430.
- Conceição MS, Vechin FC, Lixandrão M, et al. Muscle Fiber Hypertrophy and Myonuclei Addition: A Systematic Review and Meta-analysis. Medicine & Science in Sports & Exercise 50 (2018): 1385-1393.
- Verdijk LB, Snijders T, Drost M, et al. Satellite cells in human skeletal muscle; from birth to old age. Age 36 (2014): 545-557.
- Fry CS, Lee JD, Mula J, et al. Inducible depletion of satellite cells in adult, sedentary mice impairs muscle regenerative capacity without affecting sarcopenia. Nature Medicine 21 (2015): 76-80.
- Binder EF, Yarasheski KE, Steger-May K, et al. Effects of progressive resistance training on body composition in frail older adults: results of a randomized, controlled trial. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 60 (2005): 1425-1431.
- Pyka G, Lindenberger E, Charette S, et al. Muscle strength and fiber adaptations to a year-long resistance training program in elderly men and women. Journal of Gerontology 49 (1994): M22-M27.
- Fiatarone MA, Marks EC, Ryan ND, et al. High-intensity strength training in nonagenarians: effects on skeletal muscle. Jama 263 (1990): 3029-3034.
- McCartney N, Hicks AL, Martin J, et al. Long-term resistance training in the elderly: effects on dynamic strength, exercise capacity, muscle, and bone. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 50 (1995): B97-104.
- Grimby G, Aniansson A, Hedberg M, et al. Training can improve muscle strength and endurance in 78-to 84-yr-old men. Journal of Applied Physiology 73 (1992): 2517-2523.
- Vincent KR, Braith RW, Feldman RA, et al. Resistance exercise and physical performance in adults aged 60 to 83. Journal of the American Geriatrics Society 50 (2002): 1100-1107.
- Hakkinen K, Kallinen M, Izquierdo M, et al. Changes in agonist-antagonist EMG, muscle CSA, and force during strength training in middle-aged and older people. Journal of Applied Physiology 84 (1998): 1341-1349.
- Lexell J, Downham DY, Larsson Y, et al. Heavy-resistance training in older Scandinavian men and women: short-and long-term effects on arm and leg muscles. Scandinavian Journal of Medicine & Science in Sports 5 (1995): 329-341.
- Negaresh R, Ranjbar R, Baker JS, et al. Skeletal muscle hypertrophy, insulin-like growth factor 1, myostatin and follistatin in healthy and sarcopenic elderly men: the effect of whole-body resistance training. International Journal of Preventive Medicine 10 (2019).
- Stoever K, Heber A, Eichberg S, et al. Changes of Body Composition, Muscular Strength and Physical Performance Due to Resistance Training in Older Persons with Sarcopenic Obesity. The Journal of Frailty & Aging 4 (2015): 216-222.
- Walker S, Häkkinen K. Similar increases in strength after short-term resistance training due to different neuromuscular adaptations in young and older men. The Journal of Strength & Conditioning Research 28 (2014): 3041-3048.
- Steib S, Schoene D, Pfeifer K. Dose-response relationship of resistance training in older adults: a meta-analysis. Med Sci Sports Exerc 42 (2010): 902-914.
- Seynnes O, Fiatarone Singh MA, Hue O, et al. Physiological and functional responses to low-moderate versus high-intensity progressive resistance training in frail elders. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 59 (2004): M503-M509.
- Csapo R, Alegre LM. Effects of resistance training with moderate vs heavy loads on muscle mass and strength in the elderly: A meta- Scandinavian Journal of Medicine & Science in Sports 26 (2016): 995-1006.
- Nery SD, Gomides RS, Silva GV, et al. Intra-arterial blood pressure response in hypertensive subjects during low-and high-intensity resistance exercise. Clinics 65 (2010): 271-277.
- Perri MG, Anton SD, Durning PE, et al. Adherence to exercise prescriptions: effects of prescribing moderate versus higher levels of intensity and frequency. Health Psychology 21 (2002): 452.
- Hazell T, Kenno K, Jakobi J. Functional benefit of power training for older adults. Journal of Aging and Physical Activity 15 (2007): 349-359.
- De Vos NJ, Singh NA, Ross DA, et al. Optimal load for increasing muscle power during explosive resistance training in older adults. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 60 (2005): 638-647.
- Bean JF, Herman S, Kiely DK, et al. Increased Velocity Exercise Specific to Task (InVEST) training: a pilot study exploring effects on leg power, balance, and mobility in community-dwelling older women. Journal of the American Geriatrics Society 52 (2004): 799-804.
- Kemmler W, Kohl M, Fröhlich M, et al. Effects of High-Intensity Resistance Training on Osteopenia and Sarcopenia Parameters in Older Men with Osteosarcopenia—One-Year Results of the Randomized Controlled Franconian Osteopenia and Sarcopenia Trial (FrOST). Journal of Bone and Mineral Research (2020).
- Burd NA, Mitchell CJ, Churchward-Venne TA, et al. Bigger weights may not beget bigger muscles: evidence from acute muscle protein synthetic responses after resistance exercise. Applied Physiology, Nutrition, and Metabolism 37 (2012): 551-554.
- Peterson MD, Sen A, Gordon PM. Influence of resistance exercise on lean body mass in aging adults: a meta-analysis. Medicine and Science in Sports and Exercise 43 (2011): 249.
- Mero AA, Hulmi JJ, Salmijärvi H, et al. Resistance training induced increase in muscle fiber size in young and older men. European Journal of Applied Physiology 113 (2013): 641-650.
- Kosek DJ, Kim JS, Petrella JK, et al. Efficacy of 3 days/wk resistance training on myofiber hypertrophy and myogenic mechanisms in young vs. older adults. Journal of Applied Physiology 101 (2006): 531-544.
- Welle S, Totterman S, Thornton C. Effect of age on muscle hypertrophy induced by resistance training. The Journals of Gerontology Series A: Biological Sciences and Medical Sciences 51 (1996): M270-M275.
- Chen HT, Wu HJ, Chen YJ, et al. Effects of 8-week kettlebell training on body composition, muscle strength, pulmonary function, and chronic low-grade inflammation in elderly women with sarcopenia. Experimental Gerontology 112 (2018): 112-118.
- Olson TP, Dengel DR, Leon AS, et al. Changes in inflammatory biomarkers following one-year of moderate resistance training in overweight women. International Journal of Obesity 31 (2007): 996-1003.
- White LJ, Castellano V, Mc Coy SC. Cytokine responses to resistance training in people with multiple sclerosis. Journal of Sports Sciences 24 (2006): 911-914.
- Katsanos CS, Kobayashi H, Sheffield-Moore M, et al. Aging is associated with diminished accretion of muscle proteins after the ingestion of a small bolus of essential amino acids. The American Journal of Clinical Nutrition 82 (2005): 1065-1073.
- Volpi E, Mittendorfer B, Rasmussen BB, et al. The response of muscle protein anabolism to combined hyperaminoacidemia and glucose-induced hyperinsulinemia is impaired in the elderly. The Journal of Clinical Endocrinology & Metabolism 85 (2000): 4481-4490.
- Junior PS, Ribeiro AS, Nabuco HC, et al. Effects of whey protein supplementation associated with resistance training on muscular strength, hypertrophy, and muscle quality in preconditioned older women. International Journal of Sport Nutrition and Exercise Metabolism 28 (2018): 528-535.
- Hita-Contreras F, Bueno-Notivol J, Martínez-Amat A, et al. Effect of exercise alone or combined with dietary supplements on anthropometric and physical performance measures in community-dwelling elderly people with sarcopenic obesity: A meta-analysis of randomized controlled trials. Maturitas 116 (2018): 24-35.
- Guillet C, Prod'homme M, Balage M, Gachon P, Giraudet C, Morin L, Grizard J, Boirie Y. Impaired anabolic response of muscle protein synthesis is associated with S6K1 dysregulation in elderly humans. The FASEB Journal 18 (2004): 1586-1587.
- Kraemer WJ, Hakkinen K, Newton RU, et al. Effects of heavy-resistance training on hormonal response patterns in younger vs. older men. Journal of Applied Physiology 87 (1999): 982-992.
- Schroeder ET, Phillips SM, Villanueva MG, et al. Are Acute Post-Resistance Exercise Increases in Testosterone, Growth Hormone, and IGF-1 Necessary to Stimulate Skeletal Muscle Anabolism and Hypertrophy?. Medicine & Science in Sports & exercise: Official Journal of the American College of Sports Medicine 45 (2013): 2044-2051.
- Nicklas BJ, Ryan AJ, Treuth MM, et al. Testosterone, growth hormone and IGF-I responses to acute and chronic resistive exercise in men aged 55-70 years. International Journal of Sports Medicine 16 (1995): 445-450.
- Hawley JA, Lundby C, Cotter JD, et al. Maximizing cellular adaptation to endurance exercise in skeletal muscle. Cell Metabolism 27 (2018): 962-976.
- Gordon A, Sylven C, Tyni-Lennaé R, et al. Markedly improved skeletal muscle function with local muscle training in patients with chronic heart failure. Clinical Cardiology 19 (1996): 568-574.
- Strasser B, Keinrad M, Haber P, et al. Efficacy of systematic endurance and resistance training on muscle strength and endurance performance in elderly adults–a randomized controlled trial. Wiener Klinische Wochenschrift 121 (2009): 757-764.
- Wood RH, Reyes RA, Welsch MA, et al. Concurrent cardiovascular and resistance training in healthy older adults. Medicine and Science in Sports and Exercise 33 (2001): 1751-1758.
- Mikkelsen UR, Couppé C, Karlsen A, et al. Life-long endurance exercise in humans: circulating levels of inflammatory markers and leg muscle size. Mechanisms of Ageing and Development 134 (2013): 531-540.
- Lira FS, Yamashita AS, Rosa JC, et al. Hypothalamic inflammation is reversed by endurance training in anorectic-cachectic rats. Nutrition & Metabolism 8 (2011): 60.
- Nilsson A, Bergens O, Kadi F. Physical Activity Alters Inflammation in Older Adults by Different Intensity Levels. Medicine and Science in Sports and Exercise 50 (2018): 1502-1507.