Study of Hypoxia Induced Factor-1Alpha (HIF-1A) and Carbonic Anhydrase 9 (CAIX) in clear cell Renal Cell Carcinoma (ccRCC)
Article Information
Anna Tassoudi1, Ioannis Stefanidis2, Theodoros Eleftheriadis3, Vasilios Tzortzis4, Vasilios Tassoudis5*and Maria Ioannou6
1Undergraduate student of Medicine, University of Thessaly, Volos, Greece
2Professor of Internal Pathology/ Nephrology, University of Thessaly, Volos, Greece
3Associate Professor of Internal Pathology/ Nephrology, University of Thessaly, Volos, Greece
4Professor of Urology, University of Thessaly, Volos, Greece
5N.H.S Director of Anesthesiology, University Hospital of Larissa, Larissa, Greece
6Professor of Pathology, University of Thessaly, Volos, Greece
*Corresponding Author: Vasilios Tassoudis, N.H.S Director of Anesthesiology, University Hospital of Larissa, Larissa, Greece
Received: 21 March 2021; Accepted: 07 April 2021; Published: 12 April 2021
Citation: Anna Tassoudi, Ioannis Stefanidis, Theodoros Eleftheriadis, Vasilios Tzortzis, Vasilios Tassoudis, Maria Ioannou. Study of Hypoxia Induced Factor-1Alpha (HIF-1A) and Carbonic Anhydrase 9 (CAIX) in clear cell Renal Cell Carcinoma (ccRCC). Archives of Nephrology and Urology 4 (2021): 050-062.
View / Download Pdf Share at FacebookAbstract
Objective: The aim of this study was to investigate the immunohistochemical expression of hypoxia inducible factor-1α (HIF-1α) and carbonic anhydrase 9 (CAIX) in renal cell carcinoma and the association with clinicopathological parameters.
Methods: We retrospectively analyzed 42 ccRCC cases immunohistochemically for their expression status of HIF-1α, correlating this status with CAIX expression and clinicopathological data. The Kruskal-Wallis H test and the Mann-Whitney U test were used for statistical analysis. A Kendall’s tau-b test was performed to find possible correlation between HIF-1 alpha and CAIX.
Results: We found significant correlation between HIF-1a nuclear staining and CAIX immunostaining. We also found a positive relation between high HIF-1alpha immunoscore and higher tumor grades (Furhman 3 and 4). In addition, we detected correlation between HIF-1alpha overexpression and angioinvasion.
Conclusion: Our results suggest that HIF-1 alpha and CAIX in clear cell renal cell carcinoma are associated with histopathological prognostic factors. Since HIF-1α inhibitors are under investigation for the treatment of many human tumors, further investigation, in larger series of patients, might elucidate the exact role of HIF1α/CAIX in clear cell renal cell carcinoma and possibly reveal novel predictive tissue markers.
Keywords
<p>HIF-1A; CAIX; Immunohistochemistry; Renal cell carcinoma</p>
HIF-1A articles; CAIX articles; Immunohistochemistry articles; Renal cell carcinoma articles
Article Details
1. Introduction
Tumor hypoxia has long been known as a fundamental property of the tumor microenvironment. It results from uncontrolled tumor growth outpacing the rate of neovascularization, as well as from architecturally defective microcirculation compromising oxygen diffusion even to tumor cells adjacent to neovessels [1]. Accumulated clinical and experimental data highlight intratumoral hypoxia not only as the main driving force of tumor-induced angiogenesis [2], but also as a major factor in treatment failure [3].
Hypoxia-inducible factor 1 (HIF-1), a heterodimeric transcription factor activated under hypoxic conditions, is the master regulator of adaptive responses to hypoxia, initiating with oxygen sensing and culminating, among other things, in increased vessel formation [2]. Under hypoxia, decreased hydroxylation of the HIF-1α rescues it from proteasome-mediated degradation. Then HIF-1α translocates into the nucleus, where it binds HIF-1β, forming the heterodimer that recognizes hypoxia-response elements in a wide array of genes. Among the various HIF-1–dependent genes are the surface transmembrane carbonic anhydrases (CAs), which are involved in the maintenance of acid-base balance in the microenvironment [4-6].
Carbonic anhydrase IX (CAIX) catalyzes the reversible hydration of carbon dioxide to the carbonic acid, thereby contributing to acidification of the extracellular environment and the maintenance of normal pH in tumor cells [7,8]. In other words, CAIX allows tumor cells to acclimatize to hypoxia and to proliferate away from blood vessels [9]. Blockade of CAIX results in increased cell death under hypoxic conditions, whereas transfection of CAIX promotes cell proliferation [9]. Transcription of the CAIX gene is regulated by the von Hippel-Lindau protein, which is part of the protein complex that ubiquitinates HIF-1α under hypoxic conditions, resulting in its proteolysis [10,11].
Given the long recognized literature regarding CAIX in renal cell carcinomas [12-14], we assessed the expression of CAIX, and HIF-1α in relation to clinicopathological parameters in a series of surgical specimens of clear cell renal cell carcinoma (ccRCC), and investigated the potential value of CAIX and HIF-1α as biologic and prognostic factors in these tumors.
2. Materials and Methods
2.1 Patients
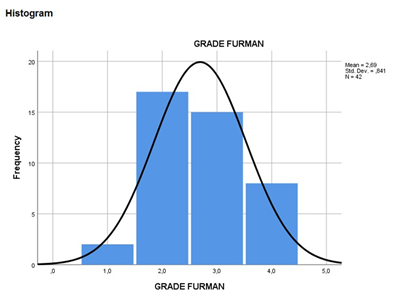
The ethics committee at our institution approved the study protocol and written informed consent was obtained from each patient enrolled in the study. In this retrospective study, 42 cases of ccRCC (27 male and 15 female patients, median age 67 years) were evaluated, for which archival specimens from the resected primary tumor were available. None of the patients had received any treatment before surgery. All surgical specimens were retrieved from the files in the Pathology Department at the University of Thessaly. In total, 2 ccRCC Furhman 1, 17 Furhman 2, 15 Furhman 3 and 8 Furhman 4 tumors were evaluated (Histogram). The samples had been routinely fixed in 10% buffered formalin, processed, and embedded in paraffin.
2.2 Immunohistochemistry
Sections were cut at 4μm using a Leica TP1020 microtome and dried overnight at 60oC. After deparaffinization in xylene, the sections were rehydrated in decreasing ethanol solutions. Antigen retrieval was achieved by microwaving tissue sections in 0.01 M citrate buffer solution (pH 6) for 20 minutes (LG WAVEDOM, 850 Watt). After antigen retrieval, the sections were cooled and washed in phosphate-buffer saline (PBS). They were incubated in 0.3% hydrogen peroxide for 10 minutes, to block endogenous peroxidase. The primary antibodies for immunostaining were specific for Η1α67 (aa432-528, IgG2b; Novus Biologicals, Littleton CO, dilution 1:25) for HIF-1 α and EPR415 (rabbit monoclonal, Epitomics CA dilution 1:250) for CAIX. Sections were incubated over night at 4oC with each antibody. Envision fluid (polymer- peroxidase method, EnVision+/ Horseradish peroxide (HRP), DAKO, Denmark) was then added, followed by incubation for 30 minutes. Bound antibodies were visualized using 0.05% 3,3-diaminobenzidine solution (DAB, DAKO). Finally, sections were counterstained with hematoxylin and mounted in DPX mounting medium (BDH Laboratory Supplies, UK). As positive control, histological sections from RCC with known VHL mutation were used. As negative control, histological sections incubated with non-antigenic serum instead of the main antibody were used.
2.3 Evaluation of ‘Final’ immunohistochemical scores
Immunohistochemistry analysis was blinded regarding the patients’ data. More than 1000 tumor cells were counted in several high-power fields, and the percentage of neoplastic cells with membrane and/or cytoplasmic CAIX or nuclear HIF-1α reactivity out of the total number of neoplastic cells was determined.
Α nuclear expression of HIF-1α in more than 10% of the total cancer cells of every section was considered positive. CAIX immunohistochemical detection was performed in the membrane or cytoplasm of cancer cells. For both of the above markers the quantification of expression level based on the staining area and intensity. The Quick score was used where Q= percentage of (nuclei for HIF-1a and cells for CAIX) multiplied by staining intensity. The intensity scores were from 0 (no staining at all) to 3 (very bright staining). Maximum value for Q was 300% and minimum was 0%.
2.4 Statistical analysis
For the following reasons, non-parametric tests were used for statistical analysis: firstly, many variables were qualitative, such as sex (0 = male, 1 = female), infiltration by renal vessels, renal vein and renal nerve (0 = absent, 1 = present), and renal tumor location (0 = left kidney, 1 = right kidney). Secondly, our data were not normally distributed so a lot of statistical tests were by definition excluded. The Furhman grade also was modified, because this was used as a grouping variable, to group 0 (Furhman grade1-2) and group 1(Furhman grade 3-4). Scale characteristics such as age were converted to ordinal data and mean was used as a cutoff point (age mean=66.9 years, group 0= 1-67 years, group 1= older than 67 years). The same when HIF-1α (mean=1.0738-group 0=1.0738 or less, other=1) or CAIX (1.5976-group 0=1.5976 or less, other=1) were used as grouping variables. The Kruskal-Wallis H test followed by the Mann-Whitney U test were used for comparison of means, whereas the Kendall’s tau-b test was used for evaluating possible correlation between HIF-1A and CAIX. The SPSS v25 statistical software package (IBM, USA) was used, with differences being considered statistically significant when P<0.05.
3. Results
3.1 HIF-1a expression
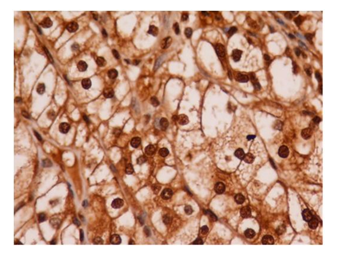
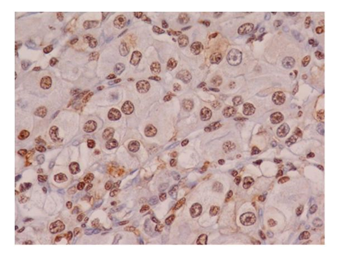
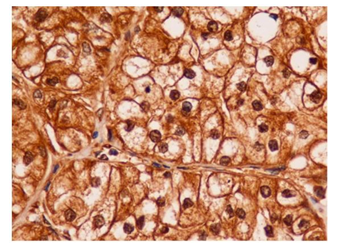
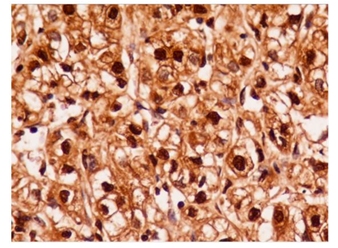
Forty-two cases were considered HIF-1α-positive (Figure 1-5). A summary of the HIF-1a immunoreactivity scores in RCC is shown in Table 1. Cytoplasmic staining was not detected in our study. The most intense immunoreaction was demonstrated in the vicinity of necrotic tumors. The overall distribution of immunoreaction in the tissue sections was diffuse.
The highest rate of intense positivity concerned tumors in Fuhrman grade 2 (40,5% - group 0, 17/42), while the highest percentage of cases with strong positivity was observed in Furhman grade 2 (40,5%- group 0) and grade 3 (35,7%- group 1, 15/42).
3.2 CAIX expression
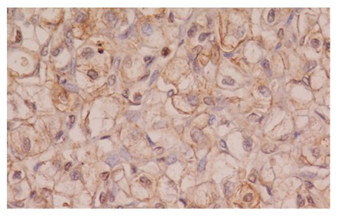
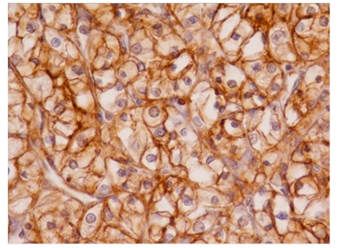
CAIX immunostaining of tumor cells was seen in 42 cases (100%). The immunoreactivity was membranous and/or cytoplasmic (Figure 6-7). The distribution of immunoreactivity was diffuse. Staining intensity was even and convincing. Case grouping according to percentages of immunostained tumor cells is shown in Figure 6-7). 45,3% (23/42) were in the category ‘intense expression’ and 44,7% (19/42) ‘weak expression’.
3.3 Immunoreactivity relation between HIF-1α and CAIX
Immunohistochemical staining results of HIF-1-α and CAIX in ccRCC are summarized in Table 2. 42 cases (100%) exhibited nuclear immunostaining and were considered HIF-1-α positive (Figure 1-5). CAIX membranous/cytoplasmic immunoreactivity was observed in 42 (100%) cases) as depicted in Figures 6-7. Higher immunohistochemical scores indicating strong expression of HIF-1α were mostly detected in Furhman grade 3 and 4 cases (histogram), whereas reduced HIF-1α expression was associated with lower histological grade of ccRCC (p=0.001) (Table 1 and Figure 1-5). Similarly, strong CAIX expression was associated with high grade cases, whereas lower grade ccRCC cases were associated with reduced CAIX immunohistochemical expression (p=0.003) (Table 1 and Figure 6-7). These findings suggest that the HIF-1α /CAIX axis is present in ccRCC, and reflects the prominent role of HIF-1α in tumor hypoxia, which is one of the histological signs of RCC (Table 1 and Figures 1-5).
|
Parameter10 |
Count |
|
Age (years) |
|
|
Mean |
66.9 |
|
Standard Deviation |
8.03 |
|
Variation |
50 -79 years |
|
Sex |
|
|
Male |
27 (64.3%) |
|
Female |
15 (35.7%) |
|
nuclear Furhman grade 1 |
2 (4.8%) |
|
nuclear Furhman grade 2 |
17 (40.5%) |
|
nuclear Furhman grade 3 |
15 (35.7%) |
|
nuclear Furhman grade 4 |
8 (19%) |
|
Tumor Location |
|
|
left |
20 (47.6%) |
|
right |
22 (52.4%) |
|
Renal vein infiltration |
|
|
negative |
36 (85.7%) |
|
positive |
6 (14.3%) |
|
Vascular Infiltration |
|
|
negative |
35 (83.3%) |
|
positive |
7 (16.7%) |
|
Perineural infiltration |
|
|
negative |
39 (92.9%) |
|
positive |
3 (7.1%) |
HIF-1-alpha and CAIX exhibited similar expression patterns, which were highly correlated (τ=0.588 p=0.01).
Table 1: Demographic and clinicopathological characteristics of 42 patients.
3.4 Correlation of HIF-1α and CAIX with tumor Grade
No difference was detected in HIF-1α or CAIX expression in different groups of gender, age, renal nerve or vein infiltration, or tumor location. On the contrary, we detected higher HIF-1α in the group with more intense angioinvasion (p=0.041).
3.5 Collectively about HIF-1 α and CAIX
Samples of 42, 27 male and 15 female patients (Table 1) of mean age 66.9 were statistically evaluated. Concerning age, sex, renal nerve infiltration, renal vein infiltration and tumor location (left or right kidney), there was no statistically significant difference when examined with HIF-1A (grouping variable). In contrast, the vascular infiltration and Furhman grade showed significant differences when examined with HIF-1A (P=0.041 and 0.006 respectively). When examined with CAIX, only Furhman grade showed any difference (P=0.032). The Man-Whitney test showed that HIF-1A and CAIX levels in Furhman grade 1 and 2 tumors (group 0) were significantly different from those in Fuhrman grade 3-4 samples (group1; p=0.001 and p=0.003 respectively, asymt. Sig. 2- tailed). A Kendall’s tau-b test showed the correlation between HIF-1A and CAIX, and with a coefficient=0.588 the correlation was significant at the 0.01 level (2- tailed).
4. Discussion
Clear cell renal cell carcinoma is pathogenetically related to the loss of the VHL tumor suppressing gene, with consequent deregulation of hypoxia pathways and HIF-1α overexpression [16]. In our study, we detected positive nuclear expression of HIF-1α in all tumors. This expression was independent of patient’s age or sex. Our findings are consistent with previous reports [17]. Since inactive HIF-1α remains in the cytoplasm, the nuclear location of HIF-1α in all cases indicates its transcriptional activity [18].
Using a semi-quantitative method for evaluation of the immunohistochemical expression, tumors in our study were separated into those with high HIF-1α expression and those with low HIF-1α expression. Higher expression was related to higher Furhman nuclear grade, as has been demonstrated by other investigators as well [19]. Considering the prognostic value of Furhman grade, our results could possibly indicate the complimentary role of HIF-1α immunohistochemical stain in histopathological examination of ccRCC and in patient stratification. In fact, a recent meta-analysis which included 14 studies and over 1258 patients with ccRCC showed that the immunohistochemical overexpression of HIF-1α is related to reduced overall survival [20].
Moreover, our study showed that the overexpression of HIF-1a is related to vascular invasion, which indicates more aggressive biological behavior. However, we did not show a similar correlation to renal vein neoplastic invasion, probably due to the small number of cases included in our study. In ccRCC hypoxia-induced CAIX expression occurs because of the VHL gene mutation [16], with CAIX being considered as a diagnostic marker, although its prognostic and predictive role remains controversial. Most studies have shown that strong immunohistochemical CAIX expression is related to a favorable prognosis in patients with ccRCC [21]. However, other investigators have reported that CAIX expression is not an independent prognostic factor [22-24].
In our study, we detected immunohistochemical expression of CAIX in all tumors. The immunoscores were related to HIF-1α expression and angioinvasion, a factor that suggests highly aggressive biological behavior. Moreover, CAIX expression was not correlated to the age or sex, a finding that is in agreement with previous studies [25]. On the contrary, we found that the immunohistochemical expression of CAIX was related to Furhman grade, since tumors of high Furhman grade presented high CAIX immunoscores. Other investigators have shown that CAIX overexpression is detected in tumors of low Furhman grade [26]. Technical differences in staining protocols, the variability of the antibodies and heterogeneity of neoplastic tissue could explain such discrepancies. However, the fact that CAIX expression is under the transcriptional control of HIF fits our results better [27].
Interestingly, CAIX expression in circulating ccRCC tumor cells obtained by liquid biopsy has been related to tumor size and clinical course [28]. Our study showed that samples with high immunoscore of CAIX expression were related with higher Furhman grade, but there was no relation with other parameters such as renal vein invasion, vascular and perineural infiltration, probably due to the limited number of cases included in this preliminary study. In addition, the detected correlation between HIF-1α and CAIX expression supports their common causal relevance. Similar findings have been detected in other studies with various types of cancer, including ccRCC [25].
In conclusion, in ccRCC, HIF-1α expression is related to established histopathological prognostic factors and increases CAIX expression in tumors of high Furhman grade. Further investigation in larger series of patients is undoubtedly called for, in order to establish the role of HIF-1α and CAIX expression as prognostic markers in patients with ccRCC.
Conflict of Interest
No conflict of interest was declared by the authors.
References
- Sowter HM, Raval R, Moore J, et al. Predominant role of hypoxia-inducible transcription factor (Hif)-1α versus Hif-2α in regulation of the transcriptional response to hypoxia. Cancer Research 63 (2003): 6130-6134.
- Semenza GL. Hypoxia-inducible factors in physiology and medicine. Cell 148 (2012): 399-408.
- Ivanov SV, Kuzmin I, Wei MH, et al. Down-regulation of transmembrane carbonic anhydrases in renal cell carcinoma cell lines by wild-type von Hippel-Lindau transgenes. Proceedings of the National Academy of Sciences 95 (1998): 12596-12601.
- Pastorek J, Pastoreková S, Callebaut I, et al. Cloning and characterization of MN, a human tumor-associated protein with a domain homologous to carbonic anhydrase and a putative helix-loop-helix DNA binding segment. Oncogene 9 (1994): 2877-2888.
- De Simone G, Supuran CT. Carbonic anhydrase IX: Biochemical and crystallographic characterization of a novel antitumor target. Biochimica et Biophysica Acta (BBA)-Proteins and Proteomics 1804 (2010): 404-409.
- Swietach P, Patiar S, Supuran CT, et al. The role of carbonic anhydrase 9 in regulating extracellular and intracellular ph in three-dimensional tumor cell growths. Journal of Biological Chemistry 284 (2009): 20299-20310.
- Neri D, Supuran CT. Interfering with pH regulation in tumours as a therapeutic strategy. Nature Reviews Drug Discovery 10 (2011): 767-777.
- Fang JS, Gillies RD, Gatenby RA. Adaptation to hypoxia and acidosis in carcinogenesis and tumor progression. InSeminars in Cancer Biology 18 (2008): 330-337.
- Woelber L, Kress K, Kersten JF, et al. Carbonic anhydrase IX in tumor tissue and sera of patients with primary cervical cancer. BMC Cancer 11 (2011): 1-10.
- Giatromanolaki A, Koukourakis MI, Sivridis E, et al. Expression of hypoxia-inducible carbonic anhydrase-9 relates to angiogenic pathways and independently to poor outcome in non-small cell lung cancer. Cancer Research 61 (2001): 7992-7998.
- Wang Y, Wang XY, Subjeck JR, et al. Carbonic anhydrase IX has chaperone-like functions and is an immunoadjuvant. Molecular Cancer Therapeutics 7 (2008): 3867-3877.
- Kaluz S, Kaluzová M, Liao SY, et al. Transcriptional control of the tumor-and hypoxia-marker carbonic anhydrase 9: A one transcription factor (HIF-1) show?. Biochimica et Biophysica Acta (BBA)-Reviews on Cancer 1795 (2009): 162-172.
- Stillebroer AB, Mulders PF, Boerman OC, et al. Carbonic anhydrase IX in renal cell carcinoma: implications for prognosis, diagnosis, and therapy. European Urology 58 (2010): 75-83.
- Tostain J, Li G, Gentil-Perret A, et al. Carbonic anhydrase 9 in clear cell renal cell carcinoma: a marker for diagnosis, prognosis and treatment. European Journal of Cancer 46 (2010): 3141-3148.
- McGough RL, Aswad BI, Terek RM. Pathologic neovascularization in cartilage tumors. Clinical Orthopaedics and Related Research® 397 (2002): 76-82.
- Ivanov SV, Kuzmin I, Wei MH, et al. Down-regulation of transmembrane carbonic anhydrases in renal cell carcinoma cell lines by wild-type von Hippel-Lindau transgenes. Proceedings of the National Academy of Sciences 95 (1998): 12596-12601.
- Minardi D, Lucarini G, Santoni M, et al. Survival in patients with clear cell renal cell carcinoma is predicted by HIF-1α expression. Anticancer Research 35 (2015): 433-438.
- Di Cristofano C, Minervini A, Menicagli M, et al. Nuclear expression of hypoxia-inducible factor-1α in clear cell renal cell carcinoma is involved in tumor progression. The American Journal of Surgical Pathology 31 (2007): 1875-1881.
- Ebru T, Fulya OP, Hakan A, et al. Analysis of various potential prognostic markers and survival data in clear cell renal cell carcinoma. International Braz J Urol 43 (2017): 440-454.
- Fan Y, Li H, Ma X, et al. Prognostic significance of hypoxia-inducible factor expression in renal cell carcinoma: a PRISMA-compliant systematic review and meta-analysis. Medicine 94 (2015).
- Zhao Z, Liao G, Li Y, et al. Prognostic value of carbonic anhydrase IX immunohistochemical expression in renal cell carcinoma: a meta-analysis of the literature. PloS One 9 (2014): e114096.
- Kim SH, Park WS, Park EY, et al. The prognostic value of BAP1, PBRM1, pS6, PTEN, TGase2, PD-L1, CA9, PSMA, and Ki-67 tissue markers in localized renal cell carcinoma: a retrospective study of tissue microarrays using immunohistochemistry. PLoS One 12 (2017): e0179610.
- Zhang BY, Thompson RH, Lohse CM, et al. Carbonic anhydrase IX (CAIX) is not an independent predictor of outcome in patients with clear cell renal cell carcinoma (ccRCC) after long-term follow-BJU International 111 (2013): 1046-1053.
- Zerati M, Leite KR, Pontes-Junior J, et al. Carbonic Anhydrase IX is not a predictor of outcomes in non-metastatic clear cell renal cell carcinoma-a digital analysis of tissue microarray. International Braz J Urol 39 (2013): 484-492.
- Büscheck F, Fraune C, Simon R, et al. Aberrant expression of membranous carbonic anhydrase IX (CAIX) is associated with unfavorable disease course in papillary and clear cell renal cell carcinoma. InUrologic Oncology: Seminars and Original Investigations 36 (2018): 531-e19.
- Genega EM, Ghebremichael M, Najarian R, et al. Carbonic anhydrase IX expression in renal neoplasms: correlation with tumor type and grade. American Journal of Clinical Pathology 134 (2010): 873-879.
- Tafreshi NK, Lloyd MC, Bui MM, et al. Carbonic anhydrase IX as an imaging and therapeutic target for tumors and metastases. Carbonic Anhydrase: Mechanism, Regulation, Links to Disease, and Industrial Applications (2014): 221-254.
- Zhou GX, Ireland J, Rayman P, et al. Quantification of carbonic anhydrase IX expression in serum and tissue of renal cell carcinoma patients using enzyme-linked immunosorbent assay: prognostic and diagnostic potentials. Urology 75 (2010): 257-261.