Severe Acute Thrombocytopenia After Biosimilar Trastuzumab Administration: A Case Report
Article Information
Ibtihal Ahalli*, Samia Arifi, Soumia Berradi, Raihana Boujarnija, Kaouthar Messoudi , Lamiaa Amaadour, Karima Oualla, Zineb Benbrahim, Nawfel Mellas
Department of Medical Oncology, Hassan II University Hospital, Fez, Morocco
*Corresponding author: Ibtihal Ahalli, Department of Medical Oncology, Hassan II University Hospital, Fez, Morocco
Received: 04 March 2020; Accepted: 31 March 2020; Published: 03 April 2020
Citation: Ibtihal Ahalli, Samia Arifi, Soumia Berradi, Raihana Boujarnija, Kaouthar Messoudi , Lamiaa Amaadour, Karima Oualla, Zineb Benbrahim, Nawfel Mellas. Severe Acute Thrombocytopenia After Biosimilar Trastuzumab Administration: A Case Report. Journal of Pharmacy and Pharmacology Research 4 (2020): 015-020.
View / Download Pdf Share at FacebookAbstract
Drug-induced immune thrombocytopenia may occur secondary to several chemotherapeutic agents or new targeted monoclonal antibodies, but thrombocytopenia induced by trastuzumab is a very rare occurrence. We report a case of severe thrombocytopenia related to the first administration of a biosimilar trastuzumab, trastuzumab rechallenge was undertaken with the reference trastuzumab without other problems.
Keywords
<p>Acute Thrombocytopenia; Biosimilar; Trastuzumab</p>
Administration articles
Article Details
Introduction
Trastuzumab is a humanized monoclonal antibody directed against the human epidermal growth factor receptor 2 (HER2). The combination of trastuzumab with chemotherapy has been shown to improve survival in both the metastatic and adjuvant settings [1,2,4].
Thrombocytopenia induced by trastuzumab is a very rare incident, we report one case of this severe and potentially life-threatening complication associated to trastuzumab biosimilar.
Several trastuzumab biosimilars are gradually entering the mondial market. These biosimilars represent an important opportunity for society in terms of cost savings and for patients by opening up treatment access. The different trastuzumab biosimilar candidates have followed diverse pathways in their clinical development, with differences in clinical trial design, these differences in approach in phase 3 testing must be viewed in the totality of evidence demonstrating biosimilarity [3].
Case presentation
A 64-year-old woman was diagnosed with hormone receptors-positive, HER2- positive breast cancer in March 2019, she underwent patey's operation for left breast.
Histopathological examination showed invasive ductal carcinoma, with grade SBR III, size 3 cm, ductal carcinoma in situ and lymphovascular invasion were found, she had 23 lymph nodes removed from the right axilla and one of them was positive, diagnosis of stage II B (p T2N1M0) was made.
In September 2019, she started on adjuvant trastuzumab with taxane, with a loading dose of 8 mg/kg of trastuzumab as an IV infusion. Her full blood count 1 day before her loading dose revealed a hemoglobin level of 12.8 g/dL, white blood cell count of 4.3 × 10*3/ mm3, and platelet count of 260 × 10*3/mm3.
During the first infusion of trastuzumab, she presented an allergic reaction: dyspnea and hot flashes, with stable hemodynamic et respiratory parameters, infusion was interrupted, she was given 100 mg IV hydrocortisone and 5 mg IV dexchlorphéniramine. After 30 min, the symptoms had resolved and the infusion was restarted, the rest of infusion of trastuzumab was uneventful until 6 hours after infusion, and the patient was planned to receive the taxane the next day.
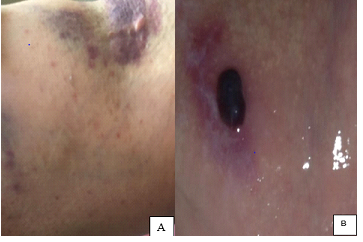
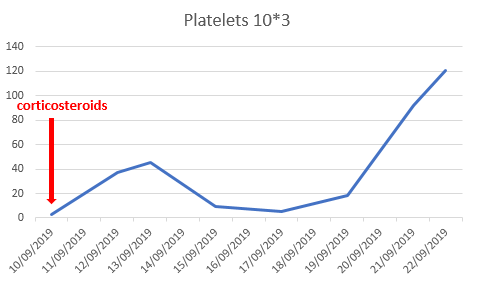
The next day, the patient turned to the department of oncology with generalized petechiae and oral hemorrhagic blisters (Figure 1). She was afebrile and cardiovascularly stable, Investigation revealed a hemoglobin level of 7.7 g/dL, white blood cell count of 9.2 × 10*3/ mm3, and platelet count of 2 × 10*3/ mm3. She was hospitalized and multiple platelet transfusion was administered. On the next day, her platelet count was 37×10*3/ mm3.
She was treated with intravenous corticosteroids (Methylprednisolone 2mg/kg dialy) for 5 days.
The platelet counts were recovered 10 days later, their number was within normal limits (Figure 2).
In view of her complete recovery of platelets, she did not undergo a bone marrow investigation.
Diagnosis of trastuzumab-induced thrombocytopenia was made, we proposed the patient re-exposure to trastuzumab while explaining the risk benefit and she accepted.
The patient was re-exposed to trastuzumab using the reference product subcutaneously without a change in the platelet count.
Discussion
Drug-induced thrombocytopenia can be caused by many different medications [5,7], diagnosis is usually made by exclusion. Since their first description, seven cases of trastuzumab related thrombocytopenia have been reported to date [6,8,9,10]. The exact pathogenesis of drug-induced thrombocytopenia is unknown. However, there are several models that try to explain this phenomen and implicate hapten-induced antibodies, drug-dependent antibodies, glycoprotein IIb/IIIa inhibitors or direct bone marrow toxicity [11,12].
George et al establish criteria to evaluate the etiologic role of the drug to thrombocytopenia [15]. In our case, therapy with trastuzumab preceded thrombocytopenia, recovery from thrombocytopenia was complete and sustained after discontinuation of trastuzumab, it was the only drug used before the onset of the thrombocytopenia, and other causes of thrombocytopenia were excluded, but re-exposure to the trastuzumab don’t resulted in recurrent thrombocytopenia, this can be explained by the fact that we used the reference trastuzumab product subcutaneously.
Based on these criteria it is apparent that the thrombocytopenia was probably a result of the loading dose of trastuzumab.
This is a rare incident with trastuzumab, in the Hannah study, only one patient had thrombocytopenia with febrile neutropenia in population with subcutaneous trastuzumab, who died with septic shock [13], and the final analysis of this trial published on april 18, 2019 at jama oncology did not noted an unexpected safety signal [14].
The demonstration of similar immunogenicity is a critical requirement for the demonstration of similarity between a biosimilar candidate and the respective reference product. The clinical development of a biosimilar includes head-to head clinical studies to assess potential differences in immunogenicity between the two products.
The development and validation of Anti-drug Antibodies ADA assays to assess the immunogenicity of a biosimilar should follow the same guidance set by the FDA and EMA for any therapeutic protein. However, during assay development and subsequently the assay validation, the originator should also be tested in select experiments to ensure the assay’s similar ability to detect antibodies against biosimilar and originator, and similar reactivity of the two drug products with the positive control [16,17].
Trastuzumab biosimilar used in our patient have been tested in phase 3 trial, and the equivalence in efficacy, immunogenicity and safety was comparable to Herceptin [18].
The immunogenicity profile was low, similar between the 2 products and consistent with published data with trastuzumab showing a low immunogenic potential. Baseline positivity for antidrug antibody in a small number of patients is expected.
Management of drug-induced thrombocytopenia includes immunoglobulin therapy, or corticosteroids and platelets transfusion in cases of severe hemorrhage. Our patient had a course of corticosteroids at the episode of thrombocytopenia and continued on trastuzumab treatment without other problems after rechallenge by the reference trastuzumab.
Conclusion
In conclusion, our patient showed a trastuzumab biosimilar-related thrombocytopenia, among the reported cases of trastuzumab-induced thrombocytopenia, this patient is the second one who did not interrupt trastuzumab treatment.
Given the increasing use of this agent, it is important that clinicians are aware of this rare toxicity.
References
- Perez EA, Romond EH, Suman VJ, Jeong JH, Davidson NE, Geyer CE Jr, Martino S, Mamounas EP, Kaufman PA, Wolmark N: Four-year follow-up of trastuzumab plus adjuvant chemotherapy for operable human epidermal growth factor receptor 2-positive breast cancer: joint analysis of data from NCCTG N9831 and NSABP B-31. J Clin Oncol 29 (2011): 3366–3373.
- Slamon DJ, Leyland-Jones B, Shak S, Fuchs H, Paton V, Bajamonde A, Fleming T, Eiermann W, Wolter J, Pegram M, Baselga J, Norton L: Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med 344 (2001): 783–792.
- Liese Barbier, Paul Declerck, Steven Simoens: The arrival of biosimilar monoclonal antibodies in oncology:clinical studies for trastuzumab biosimilars. British journal of cancer 2019: 1.
- Hudis CA: Trastuzumab-mechanism of action and use in clinical practice. N Engl J Med 357 (2007): 39–51.
- Cathomas R, Goldhirsch A, Von Moos R. Drug induced thrombocytopenia. Correspondence. N Engl J Med 357 (2007): 1870–1871.
- Parikh O, Neave F, Palmieri C. Severe thrombocytopenia induced by a single infusion of trastuzumab. Clin Breast Cancer 8 (2008): 285–286.
- Aster RH, Bougie DW. Drug-induced immune thrombocytopenia. N Engl J Med 357 (2007): 580–587.
- Mantzourani M, Gogas H, Katsandris A, Meletis J. Severe thrombocytopenia related to trastuzumab infusion. Med Sci Monit 17 (2011): CS85–CS87.
- Maria Simona Pino, Catia Angiolini and Luisa Fioretto. Severe thrombocytopenia after trastuzumab retreatment: a case report. BMC research notes 6 (2013): 400.
- Elena Aguirre, Teresa Taberner, Armando Luaña : Severe thrombocytopenia related to long-term trastuzumab exposure. Tumori Journal 99 (2013): e1-e2.
- Warkentin TE: Drug-induced immune-mediated thrombocytopenia-from purpura to thrombosis. N Engl J Med 356 (2007): 891–93.
- Aster RH: Drug-induced immune thrombocytopenia: an overview of pathogenesis. Semen Hematol 36 (1999): 2–6.
- Jackisch, S.-B. Kim, V. Semiglazov, B. Melichar. Subcutaneous versus intravenous formulation of trastuzumab for HER2-positive early breast cancer: updated results from the phase III HannaH study. Annals of Oncology 26 (2015): 320-325.
- Jackisch C, Stroyakovskiy D. Subcutaneous vs Intravenous Trastuzumab for Patients With ERBB2-Positive Early Breast Cancer: Final Analysis of the HannaH Phase 3 Randomized Clinical Trial. JAMA oncology 5 (2019): e190339-e190339.
- George JN, Raskob GE, Shah S, et al. Drug-induced thrombocytopenia: a systematic review of published case reports. Ann Intern Med 129 (1998): 886-90.
- Chamberlain P, Kurki P. Immunogenicity assessment of biosimilars: a multidisciplinary perspective. American Association of Pharmaceutical Scientists 2018 Series 34, Chapter 19. H. J. Gutka et al. (eds.), Biosimilars, AAPS Advances in the Pharmaceutical Sciences.
- Francesca Civoli,1,15 Aparna Kasinath,2 Xiao-Yan Cai. Recommendations for the Development and Validation of Immunogenicity Assays in Support of Biosimilar Programs 18 September 2019
- Rugo H. S., Barve A., Waller C. F., Bronchud M. H., Herson J., Yuan J. et al. Heritage: a phase III safety and efficacy trial of the proposed trastuzumab biosimilar Myl1401O versus Herceptin. J Clin Oncol 34 (2016): S18.