Sensitivity of Diagnostic Methods for Detecting S. stercoralis Infection and Comparison of the Total Global Number of Strongyloidiasis Cases with Other Recognized NTDs: A Systematic Review
Article Information
Heron Gezahegn*
School of Global Health and Bioethics, Euclid University, Banjul, Gambia
*Corresponding Author: Heron Gezahegn Gebretsadik, School of Global Health and Bioethics, Euclid University, Banjul, Gambia.
Received: 29 March 2023; Accepted: 05 April 2023; Published: 07 April 2023
Citation:
Heron Gezahegn. Sensitivity of Diagnostic Methods for Detecting S. Stercoralis Infection and Comparison of the Total Global Number of Strongyloidiasis Cases with Other Recognized NTDs: A Systematic Review. Journal of Orthopedics and Sports Medicine. 5 (2023): 181-187.
View / Download Pdf Share at FacebookAbstract
Background: Since 2005, global health policy makers have recognized and designated widespread poverty-related neglected diseases as NTDs (neglected tropical diseases). However, Strongyloides stercoralis infection is not included in the list of these recognized NTDs. A systematic review was conducted to estimate the total number of S. stercoralis infections in humans worldwide and compare the result with the prevalence data of other recognized NTDs. In addition, the sensitivity of diagnostic methods that were used for detecting Strongyloides stercoralis infection was evaluated. Methods: An electronic search of the PubMed, WHOLIS, and ISI Web of Science databases was performed for articles published between January 1990 and May 2017. Articles with quantitative data on prevalence, incidence, duration of infection, remission/cure, and mortality in humans were included. After obtaining the raw data from the systematic review, adjustments were made for diagnostic accuracy, selection of the reference population, and adjustments for age and reference year 2017 as a prerequisite for estimating the total number of strongyloidiasis cases in humans worldwide. The estimated number was then compared with other recognized neglected tropical diseases. The sensitivity of diagnostic methods for detecting Strongyloides stercoralis infection was also systematically investigated.
Findings: The electronic search included 166 articles to estimate the total number of strongyloidiasis infections. Data analysis yielded 159,542,655 and 260,710,055 strongyloidiasis infections in the best- and worst-case scenarios, respectively. Based on this estimate, the lowest estimated number of infections resulting from this review is between the fourth highest number of Schistosoma and the fifth highest number of foodborne trematodes, while the highest estimate is between the third highest number of hookworms and the fourth highest number of Schistosoma neglected tropical diseases recorded by GBD in terms of global prevalence cases. On the other hand, 47% of the diagnostic techniques identified in this review fall in the low sensitivity range, 45.2% in the medium sensitivity range, and 7.8% in the high sensitivity range.
Conclusions: The global number of human strongyloidiasis infections estimated in this report was higher than estimates for 13 of the 17 recognized Neglected Tropical Diseases (NTDs). In fact, the number of strongyloidiasis infections would have been higher if highly sensitive diagnostic tools had been used. In this context, it is important to note that only less than 10% of the diagnostic instruments used to diagnose the disease were classified as highly sensitive. Therefore, the current information emphasizes that S. stercoralis should not be neglected. Accordingly, the results of this study reflect a friendly appeal to the responsible agency to recognize human strongyloidiasis infection and add it to the list of other neglected tropical diseases.
Keywords
<p>Strongyloidiasis; Neglected; Sensitivity; S. stercoralis; Systematic; Comparison</p>
Strongyloides articles Strongyloides Research articles Strongyloides review articles Strongyloides PubMed articles Strongyloides PubMed Central articles Strongyloides 2023 articles Strongyloides 2024 articles Strongyloides Scopus articles Strongyloides impact factor journals Strongyloides Scopus journals Strongyloides PubMed journals Strongyloides medical journals Strongyloides free journals Strongyloides best journals Strongyloides top journals Strongyloides free medical journals Strongyloides famous journals Strongyloides Google Scholar indexed journals Nematodes articles Nematodes Research articles Nematodes review articles Nematodes PubMed articles Nematodes PubMed Central articles Nematodes 2023 articles Nematodes 2024 articles Nematodes Scopus articles Nematodes impact factor journals Nematodes Scopus journals Nematodes PubMed journals Nematodes medical journals Nematodes free journals Nematodes best journals Nematodes top journals Nematodes free medical journals Nematodes famous journals Nematodes Google Scholar indexed journals Strongyloidiasis infection articles Strongyloidiasis infection Research articles Strongyloidiasis infection review articles Strongyloidiasis infection PubMed articles Strongyloidiasis infection PubMed Central articles Strongyloidiasis infection 2023 articles Strongyloidiasis infection 2024 articles Strongyloidiasis infection Scopus articles Strongyloidiasis infection impact factor journals Strongyloidiasis infection Scopus journals Strongyloidiasis infection PubMed journals Strongyloidiasis infection medical journals Strongyloidiasis infection free journals Strongyloidiasis infection best journals Strongyloidiasis infection top journals Strongyloidiasis infection free medical journals Strongyloidiasis infection famous journals Strongyloidiasis infection Google Scholar indexed journals Skin articles Skin Research articles Skin review articles Skin PubMed articles Skin PubMed Central articles Skin 2023 articles Skin 2024 articles Skin Scopus articles Skin impact factor journals Skin Scopus journals Skin PubMed journals Skin medical journals Skin free journals Skin best journals Skin top journals Skin free medical journals Skin famous journals Skin Google Scholar indexed journals Trachea articles Trachea Research articles Trachea review articles Trachea PubMed articles Trachea PubMed Central articles Trachea 2023 articles Trachea 2024 articles Trachea Scopus articles Trachea impact factor journals Trachea Scopus journals Trachea PubMed journals Trachea medical journals Trachea free journals Trachea best journals Trachea top journals Trachea free medical journals Trachea famous journals Trachea Google Scholar indexed journals Anorexia articles Anorexia Research articles Anorexia review articles Anorexia PubMed articles Anorexia PubMed Central articles Anorexia 2023 articles Anorexia 2024 articles Anorexia Scopus articles Anorexia impact factor journals Anorexia Scopus journals Anorexia PubMed journals Anorexia medical journals Anorexia free journals Anorexia best journals Anorexia top journals Anorexia free medical journals Anorexia famous journals Anorexia Google Scholar indexed journals Arthritis articles Arthritis Research articles Arthritis review articles Arthritis PubMed articles Arthritis PubMed Central articles Arthritis 2023 articles Arthritis 2024 articles Arthritis Scopus articles Arthritis impact factor journals Arthritis Scopus journals Arthritis PubMed journals Arthritis medical journals Arthritis free journals Arthritis best journals Arthritis top journals Arthritis free medical journals Arthritis famous journals Arthritis Google Scholar indexed journals Cardiac arrhythmias articles Cardiac arrhythmias Research articles Cardiac arrhythmias review articles Cardiac arrhythmias PubMed articles Cardiac arrhythmias PubMed Central articles Cardiac arrhythmias 2023 articles Cardiac arrhythmias 2024 articles Cardiac arrhythmias Scopus articles Cardiac arrhythmias impact factor journals Cardiac arrhythmias Scopus journals Cardiac arrhythmias PubMed journals Cardiac arrhythmias medical journals Cardiac arrhythmias free journals Cardiac arrhythmias best journals Cardiac arrhythmias top journals Cardiac arrhythmias free medical journals Cardiac arrhythmias famous journals Cardiac arrhythmias Google Scholar indexed journals Nephrotic syndrome articles Nephrotic syndrome Research articles Nephrotic syndrome review articles Nephrotic syndrome PubMed articles Nephrotic syndrome PubMed Central articles Nephrotic syndrome 2023 articles Nephrotic syndrome 2024 articles Nephrotic syndrome Scopus articles Nephrotic syndrome impact factor journals Nephrotic syndrome Scopus journals Nephrotic syndrome PubMed journals Nephrotic syndrome medical journals Nephrotic syndrome free journals Nephrotic syndrome best journals Nephrotic syndrome top journals Nephrotic syndrome free medical journals Nephrotic syndrome famous journals Nephrotic syndrome Google Scholar indexed journals Stercoralis infections articles Stercoralis infections Research articles Stercoralis infections review articles Stercoralis infections PubMed articles Stercoralis infections PubMed Central articles Stercoralis infections 2023 articles Stercoralis infections 2024 articles Stercoralis infections Scopus articles Stercoralis infections impact factor journals Stercoralis infections Scopus journals Stercoralis infections PubMed journals Stercoralis infections medical journals Stercoralis infections free journals Stercoralis infections best journals Stercoralis infections top journals Stercoralis infections free medical journals Stercoralis infections famous journals Stercoralis infections Google Scholar indexed journals soil-transmitted articles soil-transmitted Research articles soil-transmitted review articles soil-transmitted PubMed articles soil-transmitted PubMed Central articles soil-transmitted 2023 articles soil-transmitted 2024 articles soil-transmitted Scopus articles soil-transmitted impact factor journals soil-transmitted Scopus journals soil-transmitted PubMed journals soil-transmitted medical journals soil-transmitted free journals soil-transmitted best journals soil-transmitted top journals soil-transmitted free medical journals soil-transmitted famous journals soil-transmitted Google Scholar indexed journals Helminthiasis articles Helminthiasis Research articles Helminthiasis review articles Helminthiasis PubMed articles Helminthiasis PubMed Central articles Helminthiasis 2023 articles Helminthiasis 2024 articles Helminthiasis Scopus articles Helminthiasis impact factor journals Helminthiasis Scopus journals Helminthiasis PubMed journals Helminthiasis medical journals Helminthiasis free journals Helminthiasis best journals Helminthiasis top journals Helminthiasis free medical journals Helminthiasis famous journals Helminthiasis Google Scholar indexed journals Tropical diseases articles Tropical diseases Research articles Tropical diseases review articles Tropical diseases PubMed articles Tropical diseases PubMed Central articles Tropical diseases 2023 articles Tropical diseases 2024 articles Tropical diseases Scopus articles Tropical diseases impact factor journals Tropical diseases Scopus journals Tropical diseases PubMed journals Tropical diseases medical journals Tropical diseases free journals Tropical diseases best journals Tropical diseases top journals Tropical diseases free medical journals Tropical diseases famous journals Tropical diseases Google Scholar indexed journals Hookworm infection articles Hookworm infection Research articles Hookworm infection review articles Hookworm infection PubMed articles Hookworm infection PubMed Central articles Hookworm infection 2023 articles Hookworm infection 2024 articles Hookworm infection Scopus articles Hookworm infection impact factor journals Hookworm infection Scopus journals Hookworm infection PubMed journals Hookworm infection medical journals Hookworm infection free journals Hookworm infection best journals Hookworm infection top journals Hookworm infection free medical journals Hookworm infection famous journals Hookworm infection Google Scholar indexed journals S. stercoralis articles S. stercoralis Research articles S. stercoralis review articles S. stercoralis PubMed articles S. stercoralis PubMed Central articles S. stercoralis 2023 articles S. stercoralis 2024 articles S. stercoralis Scopus articles S. stercoralis impact factor journals S. stercoralis Scopus journals S. stercoralis PubMed journals S. stercoralis medical journals S. stercoralis free journals S. stercoralis best journals S. stercoralis top journals S. stercoralis free medical journals S. stercoralis famous journals S. stercoralis Google Scholar indexed journals
Article Details
Abbreviations:
GBD: Global burden of disease study; WHO: World Health; NTDs: Neglected Tropical Diseases; MeSH: Medical Subject Headings; PubMed: U.S. National Library of Medicine, National Institute of Health; WHOLIS: World Health Organization Library Information System; ISI: Institute for Scientific Information; URL: Uniform Resource Locator; UNSD: United Nations Statistics Division; PCR: Polymerase Chain Reaction; ELISA: Enzyme-Linked Immunosorbent Assays; K-K: Kato – Katz; KAP: Koga Agar Plate; PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-analyses
1. Introduction
Strongyloides species are slender, cylindrical nematodes with long esophagus and uterus that are intertwined, and because they resemble a twisted thread, they are also called "nematodes" [1]. The adult female worm is nearly transparent, measures 2.2 to 2.5 mm in length, is 50 µm in diameter, and lives in tunnels between enterocytes in the small intestine [2-4]. Although males exist, they play no role in human infections and are readily excreted from the intestine [1,5].
The signs and symptoms of strongyloidiasis infection may be related to the condition of infection as acute or chronic strongyloidiasis [6,7]. The first sign and symptom of patients with acute strongyloidiasis, if noticed at all, is a localized pruritic, erythematous rash at the skin injection site [8,9]. Patients may then develop tracheal irritation and a dry cough as the larvae migrate upward from the lungs through the trachea [10]. After the larvae are ingested into the gastrointestinal tract, patients may develop diarrhea, constipation, abdominal pain, and anorexia [11]. Chronic strongyloidiasis is generally asymptomatic, but gastrointestinal and cutaneous manifestations are most common in patients with clinical disease [12]. In rare cases, patients with chronic strongyloidiasis have complained of arthritis, cardiac arrhythmias, and signs and symptoms associated with chronic malabsorption, duodenal obstruction, nephrotic syndrome, and recurrent asthma [13-15].
Detection of eggs or larvae in host feces is the usual method for diagnosis of strongyloidiasis [16]. However, serology has the disadvantage of cross-reactivity with other helminthic infections, such as ascariasis and slow seroreversion, making it impossible to distinguish between active and expired infections [18,19]. In addition, serology and polymerase chain reaction are diagnostic procedures requiring great expertise and resources [20,21]. Hyperinfection is more easily diagnosed by the detection of filariform larvae because of the high worm burden [22]. Larvae have also been detected in sputum, bronchoalveolar fluid, cerebrospinal fluid, and other fluids or tissues [23]. Eggs and adult worms are rarely detected. Duodenal fluid or biopsy may also be helpful in diagnosis [24]. Peripheral eosinophilia is common in chronic infections but is not a feature of hyperinfection syndrome [25]. However, the diagnostic tools most commonly used in middle- and low-income countries are not very sensitive [4]. This may be due in part to a lack of attention to the disease and its consequences by key stakeholders such as WHO. The objective of the current systematic review was to estimate the total number of S. stercoralis infections in humans worldwide and to compare the result with prevalence data of other recognized NTDs. In addition, the sensitivity of widely used diagnostic methods to detect the infection was evaluated.
2. Methodology
2.1 Systematic review: search strategy
A systematic review was conducted to collect the raw data for this project. An electronic search was conducted on May 23, 2017, in the PubMed, WHOLIS, and ISI Web of Science databases. The keywords used were "Strongyloides" and "strongyloidiasis" combined with the Boolean operator "OR." For searches in the PubMed and WHOLIS databases, the terms were used as Medical Subject Headings (MeSH). Because the ISI Web of Science database does not provide a thesaurus, a simple keyword search was performed there with the "all databases" option. The review included published literature between January 1, 1990, and May 23, 2017. Literature published before 1990 was excluded because the accessibility of older references was often problematic. The systematic review was conducted strictly in accordance with PRISMA guidelines.
2.2 Systematic review: extraction of data
All data finally deemed relevant were extracted using a simple Microsoft Excel template. The extracted information included the bibliographic details of the reference, the study location, the study design, the sampling strategy, the study population, the sample size, the period of data collection, the sex and age of the studied population, the diagnostic techniques used to diagnose Strongyloides spp: infections, and the provided quantitative information on key epidemiological parameters.
2.3 Data analysis
The prevalence data obtained from the systematic review was used as a starting point. Afterward, these raw prevalence data were adjusted for diagnostic accuracy, reference population selection, population growth, and age structure. Then, the total number of infections was calculated and compared with estimates for other NTDs. The total globally estimated number of human strongyloidiasis cases was juxtaposed to estimates for other NTDs to explore the relative importance of strongyloidiasis. Furthermore, the sensitivity of the different diagnostic techniques reported to be used for detecting the Strongyloides spp. infections was determined. Correcting the sensitivity of the diagnostic instruments with a correction factor and adjusting the population to 2017 in the respective studies were critical for estimating the global number of human strongyloidiasis cases and comparing the disease with other recognized NTDs.
2.4 Ethical consideration
Ethical clearance was not needed.
2.5 Findings
2.5.1 Databases search result
The initial electronic search yielded a total of 8441 hits. 2022 and 6419 articles were obtained from PubMed and ISI Web of Science using MeSH term and keywords search strategy, respectively. No study was identified from WHOLIS. A total of 2410 articles were found to be duplicated and excluded through EndNote (1678) and Manual (732) duplicate search strategies. From the 6031 articles without duplicates, 545 were considered potentially relevant based on their title and abstract. Nine papers were additionally identified by searching the bibliographies of included full-texts, personal archives, and recommendations from colleagues and collaborators.
A total of 554 articles were checked for full-text accessibility through EndNote full-text search, URL search, google search, Swiss Tropical and Public Health Institute, and University of Basel library sources. Approximately 97% (n=536) of the total articles checked for full-text accessibility were found, retrieved, and finally assessed based on the inclusion criteria.
After in-depth reading of the entire full-texts available, 166 articles were finally included, containing relevant data for the analysis. The following flow chart diagram illustrates the search strategy and methodology followed in the literature review (Figure 1).
2.5.2 Characteristics in terms of the kind of diagnostic tools used
Out of the 166 articles reviewed in this study, 75.3% (n=125) used a single diagnostic tool, whereas 19.9% (n=33) and 4.8% (n=8) used two and three diagnostic tools, respectively. Twenty-eight different types and modified diagnostic tools were reported. Formol ether concentration techniques FECT was used 53 (22.1%) times, which made it the most utilized diagnostic tool, followed by 39 (16.3%), 38 (15.8%), 26 (10.8%), 20 (8.3%), 11 (4.6%), 5 (2.1%), direct microscopic, kato – katz K-K, Bearmann, koga agar plate KAP, polymerase chain reaction PCR, enzyme-linked immunosorbent assay ELISA diagnostic methods respectively. Other diagnostic techniques were used 48 (20%) times. According to the sensitivity classification considered, 78 (47%) studies used diagnostic methods with low sensitivity, 75 (45.2%) with moderate sensitivity, and 13 (7.8%) with high sensitivity.
Figure 1: PRISMA flow diagram illustrating the systematic review process.
2.5.3 Total number of infections globally
The total number of the infected population globally was calculated based on the methodology developed and for a best and worst-case scenario. Accordingly, a minimum and maximum number of infected was produced. Based on the reviewed literature and the respective assumptions and methodology, in the best scenario, 159,542,655 people were estimated to be infected with S. stercoralis, and in the worst scenario, 260,710,055 people globally.
2.5.4 Comparison of the global estimate of strongyloidiasis cases with other NTDs
The estimated number of human strongyloidiasis cases in both scenarios revealed that Strongyloides stercoralis infection affects much more people than most of the recognized neglected diseases globally (Figure 2). Only the estimates for Ascariasis, trichuriasis, and hookworm infection are estimated to be higher than the worst-case scenario estimates for strongyloidiasis. When considering the best-case scenario for human strongyloidiasis, schistosomiasis is the only additional NTD with a higher number of infected.
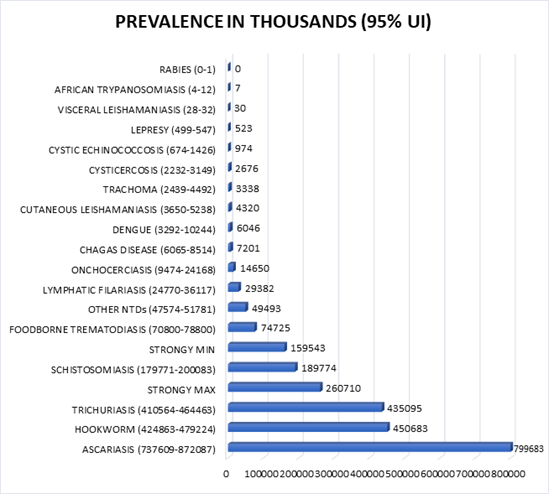
Figure 2: Describes comparison of strongyloidiasis prevalence cases with other Neglected Tropical Diseases recognized by GBD 2016.
3. Discussion
Strongyloidiasis is a harmful gastrointestinal disease caused mainly by infection with Strongyloides stercoralis. Strongyloides stercoralis is soil-transmitted helminthiasis that is common worldwide [26]. The infection affects a significant number of populations living in extreme poverty [27]. However, at the present time, Strongyloides stercoralis infection is not recognized by WHO and is included in the category of NTDs [28]. On the contrary, the estimated global total number of Strongyloidiasis cases (min = 159,542,655 and max = 260,710,055) in both scenarios indicates that more people are affected by Strongyloides stercoralis infections than by many of the 17 recognized neglected tropical diseases. These data support the argument that Strongyloidiasis is truly undermined, considering that it is not yet recognized like other NTDs. According to the Global Burden of Disease Study (GBD) 2016, ascariasis is the leading helminthic disease among the 17 neglected diseases recognized by GBD, with a prevalence of 799,683,000 cases, followed by trichuriasis, hookworm, Schistosoma, and foodborne trematode infections with 435,095,000; 450,683,000; 189,774,000; and 74,725,000 prevalence cases, respectively. Therefore, the minimum estimated number of infections resulting from this review is between the fourth-ranked Schistosoma and fifth-ranked foodborne trematode, while the maximum estimate is between the third-ranked hookworm and fourth-ranked Schistosoma, which are recorded by the GBD as neglected tropical diseases in terms of the number of prevalent cases worldwide [28].
On the other hand, identifying larvae by microscopic stool examination is usually considered a definitive diagnosis of strongyloidiasis [28-30]. However, low parasite load and irregular larvae output in the majority of subclinical infections makes it difficult [12]. As a result, the true prevalence of S. stercoralis infection is often underestimated [1]. In order to overcome the situation of underestimation of the disease, advanced diagnostic tools with high sensitivity should be applied whenever possible [29]. In this systematic review, a number of diagnostic tools were identified. The diagnostic tools used were classified into three sensitivity classes based on information gathered by another systematic review [29]. Of the diagnostic techniques identified in this review, 43.7% fall in the low, 44.9% in the medium, and 7.8% in the high sensitivity divisions. Ether concentration technique and direct microscopy were the majority, followed by Kato – Katz and agar plate culture methods. Only a few serological and molecular diagnostic tools were used. Serological tests have demonstrated high sensitivity though the extent of specificity remains a concern in other diagnostic techniques in diagnosing strongyloidiasis. The issue of specificity in the diagnosis of strongyloidiasis can be overcome by combining different diagnostic methods [26]. In the presented estimation of the total number of infected, diagnostic tools have employed a sensitivity correction factor to address the problem of low sensitivity [29].
4. Conclusion
Strongyloidiasis is a major health problem in many parts of the world. Nevertheless, little attention has been paid to the disease. The fact that the disease is not yet recognized as a neglected tropical disease and the low sensitivity of the main diagnostic tools used to detect the disease can certainly support this argument. The results of this study suggest that Strongyloides stercoralis infection should no longer be overlooked. In addition, highly sensitive diagnostic tools must be used to diagnose the disease and determine the true burden of the disease worldwide. Therefore, the disease should be recognized by the World Health Organization as a neglected tropical disease and included in Category A of the list of neglected tropical diseases to better facilitate access to highly sensitive diagnostic tools, provide more funding for research, and expand treatment options globally..
Acknowledgement
I want to thank Dr. Thomas Fuest and Prof. Peter Odermatt for their excellent supervision throughout the research work. Without their sound scientific support, this work would not have been accomplished. My thanks also go to Dr. Dora Buonfrate for her contribution during the data extraction phase of the project.
Conflict of Interest
The author declares no potential conflicts of interest concerning this article's research, authorship, and/or publication.
References
- Puthiyakunnon S, Boddu S, Li Y, et al. Strongyloidiasis—An Insight into Its Global Prevalence and Management. PLOS Negl Trop Dis 8 (2014): e3018.
- Hall A, Conway DJ, Anwar KS, et al. Strongyloides stercoralis in an urban slum community in Bangladesh: factors independently associated with infection. Trans R Soc Trop Med Hyg 88 (1994): 527-530.
- Sithithaworn P, Srisawangwong T, Tesana S, et al. Epidemiology of Strongyloides stercoralis in north-east Thailand: application of the agar plate culture technique compared with the enzyme-linked immunosorbent assay. Trans R Soc Trop Med Hyg 97 (2003): 398-402.
- Steinmann P, Zhou XN, Du ZW, et al. Occurrence of Strongyloides stercoralis in Yunnan Province, China, and comparison of diagnostic methods. PLOS Negl Trop Dis 1 (2007): e75.
- Sanprasert V, Srichaipon N, Bunkasem U, et al. Prevalence of intestinal protozoan infection among children in thailand: a large-scale screening and comparative study of three standards detection methods. Southeast Asian J Trop Med Public Health 47 (2016): 1123-1133.
- Becker SL, Piraisoody N, Kramme S, et al. Real-time PCR for detection of Strongyloides stercoralis in human stool samples from Cote d'Ivoire: Diagnostic accuracy, inter-laboratory comparison and patterns of hookworm co-infection. Acta Trop 150 (2015): 210-217.
- Assefa T, Woldemichael T, Seyoum T. Evaluation of the modified Baermann's method in the laboratory diagnosis of Strongyloides stercoralis. Ethiop Med J 29 (1991): 193-198.
- Cimino RO, Jeun R, Juarez M, et al. Identification of human intestinal parasites affecting an asymptomatic peri-urban Argentinian population using multi-parallel quantitative real-time polymerase chain reaction. Parasit Vectors 8 (2015).
- El Shazly AM, Awad SE, Sultan DM, et al. Intestinal parasites in Dakahlia governorate, with different techniques in diagnosing protozoa. J Egypt Soc Parasitol 36 (2006): 1023-1034.
- Goncalves JF, Tanabe M, Medeiros FDD, et al. Parasitological and serological studies on amebiasis and other intestinal parasitic infections in the rural sector around recife, northeast Brazil. Rev Inst Med Trop Sao Paulo 32 (1990): 428-435.
- Hall A, Conway DJ, Anwar KS, et al. Strongyloides stercoralis in an urban slum community in Bangladesh: factors independently associated with infection. Trans R Soc Trop Med Hyg 88 (1994): 527-530.
- Graczyk TK, Shiff CK, Tamang L, et al. The association of Blastocystis hominis and Endolimax nana with diarrheal stools in Zambian school-age children. Parasitol Res 98 (2005): 38-43.
- Incani RN, Homan T, Pinelli E, et al. Comparison between merthiolate-iodine-formalin and Kato-Katz methods for the diagnosis of human helminth infections in resource-limited settings. J Helminthol (2016): 1-8.
- Knopp S, Salim N, Schindler T, et al. Diagnostic accuracy of Kato-Katz, FLOTAC, Baermann, and PCR methods for the detection of light-intensity hookworm and Strongyloides stercoralis infections in Tanzania. Am J Trop Med Hyg 90 (2014): 535-545.
- Koga K, Kasuya S, Khamboonruang C, et al. An evaluation of the agar plate method for the detection of Strongyloides stercoralis in northern Thailand. J Trop Med Hyg 93 (1990): 183-188.
- Vonghachack Y, Sayasone S, Bouakhasith D, et al. Strongyloides stercoralis is highly prevalent on Mekong Islands in Southern Lao PDR. Trop Med Int Health 20 (2015): 315.
- Lopez M, Luisa Morales M, Konana M, et al. Kato Katz and Lumbreras rapid sedimentation test to evaluate helminth prevalence in the setting of a school based deworming program. Pathog Glob Health 110 (2016): 130-134.
- Schär F, Trostdorf U, Giardina F, et al. Strongyloides stercoralis: Global Distribution and Risk Factors. PLOS Negl Trop Dis 7 (2013): e2288.
- Ahmad AF, Hadip F, Ngui R, et al. Serological and molecular detection of Strongyloides stercoralis infection among an Orang Asli community in Malaysia. J Parasitol Res 112 (2013): 2811-2816.
- Krolewiecki AJ, Ramanathan R, Fink V, et al. Improved diagnosis of Strongyloides stercoralis using recombinant antigen-based serologies in a community-wide study in northern Argentina. Clin Vaccine Immunol 17 (2010): 1624-1630.
- Machicado JD, Marcos LA, Tello R, et al. Diagnosis of soil-transmitted helminthiasis in an Amazonic community of Peru using multiple diagnostic techniques. Trans R Soc Trop Med Hyg 106 (2012): 333-339.
- Mejia R, Vicuna Y, Broncano N, et al. A Novel, Multi-Parallel, Real-Time Polymerase Chain Reaction Approach for Eight Gastrointestinal Parasites' Provides Improved Diagnostic Capabilities to Resource-Limited At-Risk Populations. Am J Trop Med Hyg 88 (2013): 1041-1047.
- Meurs L, Polderman AM, Melchers NVSV, et al. Diagnosing Polyparasitism in a High-Prevalence Setting in Beira, Mozambique: Detection of Intestinal Parasites in Fecal Samples by Microscopy and Real-Time PCR. PLOS Negl Trop Dis 11 (2017).
- Mounsey K, Kearns T, Rampton M, et al. Use of dried blood spots to define antibody response to the Strongyloides stercoralis recombinant antigen NIE. Acta Trop 138 (2014): 78-82.
- Mejia R, Nutman TB. Screening, prevention, and treatment for hyperinfection syndrome and disseminated infections caused by Strongyloides stercoralis. Curr Opin Infect Dis 25 (2012): 458-463.
- Buonfrate D, Baldissera M, Abrescia F, et al. Epidemiology of Strongyloides stercoralis in northern Italy: results of a multicentre case-control study, February 2013 to July 2014. Euro Surveill 21 (2016).
- Koga-Kita K. Intestinal parasitic infections and socioeconomic status in Prek Russey Commune, Cambodia. [Nihon koshu eisei zasshi] Japanese Journal of Public Health 51 (2004): 986-992.
- Herricks JR, Hotez PJ, Wanga V, et al. The global burden of disease study 2013: What does it mean for the NTDs? PLOS Neglected Tropical Diseases 11 (2017): e0005424.
- Schaer F, Odermatt P, Khieu V, et al. Evaluation of real-time PCR for Strongyloides stercoralis and hookworm as diagnostic tool in asymptomatic schoolchildren in Cambodia. Acta Trop 126 (2013): 89-92.
- Kassalik M, Mönkemüller K. Strongyloides stercoralis Hyperinfection Syndrome and Disseminated Disease. J Gastroenterol Hepatol 7 (2011): 766-768.
