Role of D-Dimer in Stroke: A Systematic Review
Article Information
Akram M Eraky1*, Felicity Osula2, Abiodun O. Aboaba3, Mahrukh Rasheed4, Komal Naz Khalid5, Obianuju Efobi6, Ahmad Mashlah7, Mahwish Ahmed8, Mehnaz Fatima9, Franca Ofudu10, Hina Rmsha Alfia Khan9, Suganya Giri Ravindran11, Oluwafunsho Oreniyi12, Syed Hassan Tanvir Ramzi13, and Stephen A Akinfenwa14
1University of Wisconsin School of Medicine and Public Health, Madison, Wisconsin, USA
2Kharkiv National Medical University (KNMU), Ukraine
3Avalon University School of Medicine, Curacao
4Dow University of Health and Sciences (DMC), Pakistan
5Shifa College of Medicine, Pakistan
6Danylo Halytsky Lviv National Medical University, Ukraine
7Alfaisal University, Saudi Arabia
8Islam Medical College (IMC), Pakistan
9Deccan College of Medical Sciences, India
10University of Benin, Nigeria
11Kanyakumari Government Medical College, India
12Saint James School of Medicine, Saint Vincent and the Grenadines
13Multan Medical and Dental College (MMDC), Pakistan
14Caribbean Medical University, School of Medicine (CMUSOM), Curacao
*Corresponding author: Akram M Eraky, Department of Neurological Surgery, University of Wisconsin School of Medicine and Public Health, Madison, Wisconsin, USA
Received: 20 December 2021; Accepted: 27 December 2021; Published: 05 January 2022
Citation:
Eraky AM, Osula F, Aboaba AO, Rasheed M, Khalid KN, Efobi O, Mashlah A, Ahmed M, Fatima M, Ofudu F, Khan HRA, Ravindran SG, Oreniyi O, Ramzi SHT, Akinfenwa SA. Role of D-Dimer in Stroke: A Systematic Review. Archives of Internal Medicine Research 5 (2022): 018-026.
View / Download Pdf Share at FacebookAbstract
Introduction and Objective: cute cerebral strokes lead to complex chronic disabilities worldwide, bearing high morbidity and mortality. Around 7 in 10 strokes occur in low- and middle-income countries (LMIC). D-dimer is a commonly performed laboratory test that is easily accessible in LMIC. This systematic review aims to evaluate the effectiveness of D-dimer as a diagnostic predictor of stroke within the 6- and 24-hour time period.
Methods: This systematic review adhered to PRISMA guidelines. Keywords including stroke, D-dimer, laboratory testing, and indicators were used. PubMed, Scopus, and CINAHL Plus were searched. Quality appraisal was conducted using the Newcastle Ottawa Scale.
Results: A total of nine studies were included in the review. Studies were conducted in Spain (n=3), Germany (n=1), China (n=1), Turkey (n=1), USA (n=1), Korea (n=1), and Italy (n=1). Statistical significance of D-dimer values was found in 6 of the nine studies (66.6%). The overall quality of evidence is considered to be at the upper-moderate level.
Conclusion: D-dimer is a promising biomarker that may be utilized and fully scaled as a rapid biochemical test to diagnose stroke. As the lab test is already conducted across many healthcare settings, the extension to testing in patients with acute cerebrovascular ischemic events will help predict the exact stroke type and quicken treatment formalities. This systematic review identifies statistically signi-ficant (P<0.05) differences of plasma D-dimer values within 6 and 24 hours among stroke and stroke-mimicking patients. No optimal cut-off value was determined due to the dearth of data. An optimal cut-off value of plasma D-dimer levels must be determined in future clinical studies to estimate the sensitivity and specificity of D-dimer in diagnosing stroke.
Keywords
Cerebrovascular, Diagnostic, D-dimer, Stroke, Neurological Burden
Cerebrovascular articles; Diagnostic articles; D-dimer articles; Stroke articles; Neurological Burden articles
Article Details
1. Background
Acute cerebral strokes are one of the leading causes of mortality and morbidity, leading to complex chronic disabilities across the world [1]. Stroke survivors often suffer from long-term neurological disabilities leading to reduced ability to integrate into society effectively, leading to health and economic burden for the family and society [2]. The narrow time index required for successful treatment is the central reason for the need to integrate out-of-hospital and in-hospital systems of care beyond traditional imaging modalities used in the current standard of care [2]. From establishing the penumbra concept, ischemic strokes are known to be dynamic processes [3]. When the healthcare provider re-establishes the patency of the occluded artery, preventing ischemia quickly, the contribution of CT/MRI imaging evidence of the cerebral infarction leads to good outcomes [4]. The most commonly used treatment that is effective is thrombolysis with intravenous administration of recombinant tissue plasminogen activator, which is limited due to the narrow window of use, usually less than 4.5 hours post-onset of symptoms [4]. The earlier the thrombolysis procedure is performed, the better the clinical outcomes for the patient, leading to a higher quality of life. This prompts clinicians and best practice evidence researchers to correctly and quickly diagnose ische-mic stroke in the super-acute stage before clear imaging evidence manifests, and this is the key step in successfully treating patients with thrombolysis in the acquired < 4.5 hours' time window [5].
Age-standardized stroke incidence and mortality are reducing largely in high-income countries (HIC) [6]. However, around 70% of all strokes and 87% of stroke-related morbidity and mortality occur in low- and middle-income countries (LMIC) [6]. A common laboratory test available across LMIC is D-dimer, the specific degradation product of cross-linked fibrin hydrolyzed by fibrinolysin. In general, the hyper-coagulable state and secondary fibrinolysis exist in-vivo in acute cerebral ischemic stroke, with high D-dimer levels. In this systematic review, we seek to develop and evaluate the effectiveness of the plasma biomarker (i.e., D-dimer) because it may be a useful adjuvant in diagnosing stroke in the super-acute stage, thus increasing the proportion of patients eligible for thrombolysis.
2. Methods
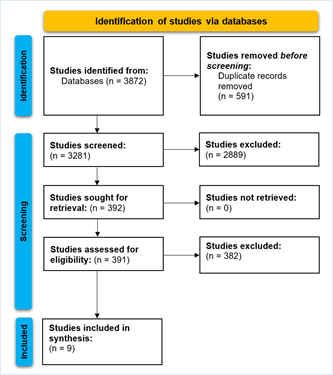
This systematic review was conducted in accordance with PRISMA guidelines [7, 8]. The following keywords were combined across databases, including PubMed (MEDLINE), Scopus, and CINAHL Plus: Stroke, D-dimer, Neurological Burden, Cerebrova-scular, Diagnostic. Only peer-reviewed publications, including clinical trials, cohorts, and case series with ten or more patients, were included. Letter to editors, systematic reviews, and single patient reports were omitted. The articles were required to report D-dimer values of patients with stroke, stroke mimics, or controls to be considered eligible. Studies were screened in accordance with the inclusion criteria. The PRISMA flowchart is depicted in (Figure 1).
The titles and abstracts were screened before the full-text review phase was initiated. The articles were appraised for quality using the Newcastle Ottawa Scale (NOS). The quality rating is done under three major sections viz: (1) Selection, (2) Comparability, and (3) Outcome. A score with a range of 0-9 was allocated to each study, and those with a score of 6 or more were considered to be high-quality studies. The data acquired from the studies was tabulated onto a shared spreadsheet under the following headings: 1) Author, year, 2) Country, 3) Different conditions demarcated, 4) time of sample collection, 5) method-logy used, 6) D-dimer values, 7) P-value, 8) Quality appraisal scores. No funding was obtained for this study. An ethical review committee was not required as only secondary clinical data were analyzed.
3. Results
Of the 3872 studies identified from the databases, 591 duplicates were removed before the screening. During this screening phase, 2889 studies did not meet the inclusion criteria and were omitted. During the full-text reviewal phase, 392 studies were assessed, of which nine studies were included in the final synthesis. The characteristics of included studies are listed in (Table 1). Three studies (33.3%) were conducted in Spain [9-11]. One each (11.1%) was conducted in Germany [12], China [13], Turkey [14], USA [15], Korea [16], and Italy [17] (Table 2). Different conditions were demarcated, including ischemic stroke, intracerebral hemorrhage, transient ischemic attacks, and stroke mimics. The time of sample collection was conducted at the 6-hour mark for three studies [9, 12, 16], within the 4.5-hour period for one study by Meng et al., [13], and within the 24 hours for five studies [10, 11, 14, 15, 17]. The different kinds of methodologies used to collect/ analyze the D-dimer values included Immunoassays technology, ELISA techniques, triage stroke panels, immune-turbidometry.
The statistically significant relevance of D-dimer values was presented by firstly, Bustamante et al., 2017, where patients with stroke had a higher mean value of 6.75 μg/mL compared to 6.24 μg/mL in stroke mimics (P<0.0001). Secondly, Montaner et al. presented a value of 0.81 μg/mL in patients with stroke compared to 0.56 μg/mL in the stroke mimicking group (P=0.002). Kavalci et al., 2011 found that patients with HS and IS had a respective value of 1780 and 5741 ng/ml compared to 150 ng/ml in patients who were mimicking stroke (P=0.0002), suggesting a large, quantitative difference. Glickman et al., 2011 found that patients with stroke had a value of 2.4 ng/ml, whereas patients that mimicked stroke had a lab value of 1 ng/ml (P=0.0002). Kim et al., 2012 measured an estimated four times higher value of D-dimer with 888.1 ng/ml in those with stroke and 188.6 ng/ml in mimics (P<0.001). Lastly, Ageno et al. 2002 find that stroke mimics had a D-dimer lab value of 0.53 μg/ml, whereas patients with stroke had a value of 1.74 μg/ml (P<0.05). Data were unavailable to determine the cutoff value of D-dimer in predicting stroke. Furthermore, not enough studies provided data for different stroke subtypes such as IS or HS. Of all 9 studies, two were high quality (score 7-9) as per the NOS scoring system employed in this study. Six out of 9 were of moderate quality (score 4-6), and one was of low quality (score 0-3) (Table 1). The overall quality of evidence is considered to be at the upper-moderate level.
Table 1: Characteristics of included studies in the systematic review.
|
Country |
Income Classification |
|
Spain (n=3, 33.3%) |
High Income Country |
|
Germany (n=1, 11.1%) |
High Income Country |
|
China (n=1, 11.1%) |
Upper-Middle-Income Country |
|
Turkey (n=1, 11.1%) |
Upper-Middle-Income Country |
|
USA (n=1, 11.1%) |
High Income Country |
|
Korea (n=1, 11.1%) |
High Income Country |
|
Italy (n=1, 11.1%) |
High Income Country |
Table 2: Countries included in this synthesis and classification as per New World Bank country classifications by income level: 2021-2022.
4. Discussion
This systematic review assessed the potential of D-dimer in nine studies for the diagnosis of ischemic stroke in the 4.5-, 6-, and 24-hour periods. We collate pertinent evidence applicable to LMIC where biomarker testing may be a useful adjuvant in differentiating total strokes such as ischemic and hemorrhage from healthy controls and patients mimicking stroke. Current evidence guidelines suggest that biomarkers ought to be analyzed within the 6-hour period to timely diagnose and differentiate ischemic stroke from other subtypes. In developing countries where neuroimaging facilities are scarce or blood biomarkers are routinely obtained at the point of care, D-dimer testing can offer a rapid diagnosis of ischemic stroke.
Using a sample size of 240 individuals, Zi and Shai (2014) found that D-dimer levels are correlated to the stroke subtypes and may be used as indicators in diagnosing cardioembolic stroke [18]. Furthermore, the authors estimated that the D-dimer levels increase with the severity of the stroke, and it is defined by the NIHSS score and infarct volume [18]. The study is in accordance with results from other studies where D-dimer was associated with brain infarct [19] and infarct volume [20]. Yuan et al. [21] conducted a meta-analysis to determine the relationship between D-dimer levels and the risk of stroke in addition to adverse clinical outcomes post-acute IS or TIA [21]. In total, the authors included 22 cohort studies [21]. The results suggested that high D-dimer levels were linked to an increased risk of total stroke (RR=1.4, 95% CI= 1.2-1.63) [21]. In addition to higher D-dimer levels on admission for stroke patients, there was also an increased risk of mortality (RR=1.77, 95% CI=1.26-2.49) and poor functional outcomes (RR=2.01, 95% CI=1.71-2.36) [21]. Overall, it may be inferred that high D-dimer levels are associated with a high risk of ischemic stroke and total stroke, but the evidence does not posit a higher risk for hemorrhagic stroke [21]. If the patient presents with high D-dimer levels on admission, it helps predict adverse events, death, and functional outcomes at the 3-month period [21].
D-dimer is derived from the cross-linked fibrin network, the final degradation product undergoing plasmin-mediated degradation [22]. Plasma D-dimer may be elevated in people with pulmonary embolism and venous thromboembolism; however, many of these central mechanisms remain unclear [22]. There are various explanations as to why plasma D-dimer levels may lead to poor outcomes in patients with stroke [22]. For example, the level increases in blood coagulation and fibrin degradation may be a marker of thrombosis [22]. Moreover, the high level of D-dimer may influence the formation of thromboembolism [22]. D-dimer also stimulates the immune system and leads to changes in IL-1, TNF-alpha, IL-6, and IL-8 [22]. While we have established the D-dimer may be a useful diagnostic tool, it is essential to understand that it may also be correlated with infarct volume, stroke severity at the start, and progression of stroke [22]. Therefore, patients with stroke may benefit from interventions that target the mechanism of D-dimer elevation in correlation to stroke subtypes and recognize the severity of stroke as per the NIHSS score and infarct volume [22]. This
ought to be explored in future studies.
5. Conclusion
The employment of D-dimer as an additional biomarker appears to be promising to achieve a rapid biochemical diagnosis of stroke. If this method is replicated across HIC and LMIC, it may be used as an urgent referral tool for stroke patients to hospitals where they may acquire thrombectomy treatment depending on the origin of the attack. While acute ischemic stroke remains a diagnosis of clinical nature, the additional discovery of biomarkers in addition to improvements of laboratory techniques must be made to achieve a rapid, molecular diagnosis of stroke. In current practice, the increased use of D-dimer assay may be extended to patients presenting with acute cerebrovascular ischemic events to help predict the stroke subtype (i.e., HS, IS) and hasten treatment. Our systematic review identifies that a statistically significant difference was present among patients with stroke compared to stroke mimics, but an optimal cut-off value could not be determined. An optimal cut-off value of plasma D-dimer levels must be determined in future clinical studies to estimate the sensitivity and specificity of D-dimer in diagnosing stroke.
References
- Persky RW, Turtzo LC, McCullough LD. Stroke in women: disparities and outcomes. Curr Cardiol Rep 12 (2010): 6-13.
- Ramos-Lima MJM, Brasileiro I de C, Lima TL de, et al. Quality of life after stroke: impact of clinical and sociodemographic factors. Clinics (2018): 73.
- Al-Buhari AR, Jan MM. Recombinant tissue plasminogen activator for acute ischemic str-oke. Neurosci J 7 (2002): 7-13.
- Stemer A, Lyden P. Evolution of the throm-bolytic treatment window for acute ischemic stroke. Curr Neurol Neurosci Rep 10 (2010): 29-33.
- Selvaratnam R, Srirangalingam U, Ahmad S, et al. Thrombolysis beyond 3 hours for acute ischaemic stroke. Emerg Med J 26 (2009): 756-757.
- Lanas F, Seron P. Facing the stroke burden worldwide. Lancet Glob Heal 9 (2021): 235-236.
- Sarfraz A, Sarfraz Z, Sanchez-Gonzalez M, et al. Randomized controlled trials of remdesivir in hospitalized coronavirus disease 2019 patients: A meta-analysis. Turkish J Emerg Med (2021).
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. The BMJ (2021).
- Bustamante A, López-Cancio E, Pich S, et al. Blood biomarkers for the early diagnosis of stroke: the stroke-chip study. Stroke 48 (2017): 2419-2425.
- Montaner J, Mendioroz M, Delgado P, et al. Differentiating ischemic from hemorrhagic stroke using plasma biomarkers: the S100B/ RAGE pathway. J Proteomics 75 (2012): 4758-4765.
- Montaner J, Mendioroz M, Ribó M, et al. A panel of biomarkers including caspase-3 and D-dimer may differentiate acute stroke from stroke-mimicking conditions in the emer-gency department. J Intern Med 270 (2011): 166-174.
- Knauer C, Knauer K, Müller S, et al. A biochemical marker panel in MRI-proven hyperacute ischemic stroke-a prospective study. BMC Neurol 12 (2012): 1-7.
- Meng R, Li Z-Y, Ji X, et al. Antithrombin III associated with fibrinogen predicts the risk of cerebral ischemic stroke. Clin Neurol Neuro-surg 113 (2011): 380-386.
- Kavalci C, Genchallac H, Durukan P, et al. Value of biomarker-based diagnostic test in differential diagnosis of hemorrhagic-ische-mic stroke. Bratisl Lek Listy 112 (2011): 398-401.
- Glickman SW, Phillips S, Anstrom KJ, et al. Discriminative capacity of biomarkers for acute stroke in the emergency department. J Emerg Med 41 (2011): 333-339.
- Kim MH, Kang SY, Kim MC, et al. Plasma biomarkers in the diagnosis of acute ischemic stroke. Ann Clin Lab Sci 40 (2010): 336-341.
- Ageno W, Finazzi S, Steidl L, et al. Plasma measurement of D-dimer levels for the early diagnosis of ischemic stroke subtypes. Arch Intern Med 162 (2002): 2589-2593.
- Zi W-J, Shuai J. Plasma D-dimer levels are associated with stroke subtypes and infarction volume in patients with acute ischemic stroke. PLoS One 9 (2014): 86465.
- Kang D-W, Yoo S-H, Chun S, et al. Inflam-matory and hemostatic biomarkers associated with early recurrent ischemic lesions in acute ischemic stroke. Stroke 40 (2009): 1653-1658.
- Matsumoto M, Sakaguchi M, Okazaki S, et al. Relationship between plasma D-dimer level and cerebral infarction volume in patients with nonvalvular atrial fibrillation. Cerebrovasc Dis 35 (2013): 64-72.
- Yuan B, Yang T, Yan T, et al. Relationships Between D-Dimer Levels and Stroke Risk as Well as Adverse Clinical Outcomes After Acute Ischemic Stroke or Transient Ischemic Attack: A Systematic Review and Meta-Ana-lysis. Front Neurol 12 (2021): 792.
- Yao T, Tian B-L, Li G, et al. Elevated plasma D-dimer levels are associated with short-term poor outcome in patients with acute ischemic stroke: a prospective, observational study. BMC Neurol 19 (2019): 1-8.