Rapid Development of a Giant Cardiac Myxoma in a Patient with Carney syndrome: a Case Report
Article Information
Messi P1,2*#, Benzoni G3*#, Bonalumi G1,4, Fatti L5, Heilbron F5, Badano L3,5, Fratianni G5, Moneta A2, Donatelli F1,2
1DISCCO Department UNIMI, Milano, Italy
2IRCCS Ospedale Galeazzi – Sant’Ambrogio, Milano, Italy
3Università degli Studi Milano-Bicocca, Cardiology Residency Program, Italy
4Department of Cardiac Surgery, Centro Cardiologico Monzino, Milan, Italy
5IRCCS Istituto Auxologico-Italiano, San Luca Hospital - Cardiac Rehabilitation Unit, Italy
#Messi and Benzoni equally contributed to this article.
*Corresponding Author: Bonalumi, Department of Cardiovascular Surgery, Centro Cardiologico Monzino-IRCCS, Via Carlo Parea 4, 20100 Milan, Italy; DISCCO Department UNIMI, Milano, Italy
Received: 28 March 2023; Accepted: 12 April 2023; Published: 05 May 2023
Citation: Messi P, Benzoni G, Bonalumi G, Fatti L, Heilbron F, Badano L, Fratianni G, Moneta A, Donatelli F. Rapid Development of a Giant Cardiac Myxoma in a Patient with Carney syndrome: a Case Report. Archives of Clinical and Medical Case Reports. 7 (2023): 187-190.
View / Download Pdf Share at FacebookAbstract
Carney Complex Syndrome is a rare genetic disorder that affects multiple organs and systems, including the endocrine glands, heart, and skin. The transmission of the syndrome is autosomal dominant, the gene involved in the mutation, in more than 60% of cases, is the PRKAR1A gene. We aim to present a 38-year-old female diagnosed with Carney syndrome since 2011. During April 2022, according to the cardiac screening plan, she underwent a transthoracic echocardiography, which did not show any cardiac tumors. However, in December 2022, the patient reported symptoms like dyspnea and palpitations. Immediately a new transthoracic echocardiography was performed, which revealed a large oval mass with heterogeneous echotexture and irregular margins in the left atrium, with a broad base of implantation at the interatrial septum, measuring 5 × 5, 5 cm. The mass cause obstruction to filling because was engaging in systole in the left ventricle. Additionally, a PFO, a common anatomical variant of interatrial septum, was found. The patient underwent cardiothoracic surgery to remove the myxoma and close the PFO via right mini thoracotomy, few days after. The surgery was successful, and the patient had good results in subsequent follow-up. In this article we aim to focus attention on the need to perform echocardiographic follow-up in a short time in patient with a familiar history of neoplasm involving myxoma, to preserve the patient from major compliances, especially reduce the risk of sudden cardiac death.
Keywords
<p>Carney Complex; Myxoma; Cardiac Neoplasm; Atrial Mass; Rapid Growth Myxoma</p>
Carney Complex articles; Myxoma articles; Cardiac Neoplasm articles; Atrial Mass articles; Rapid Growth Myxoma articles
Carney Complex articles Carney Complex Research articles Carney Complex review articles Carney Complex PubMed articles Carney Complex PubMed Central articles Carney Complex 2023 articles Carney Complex 2024 articles Carney Complex Scopus articles Carney Complex impact factor journals Carney Complex Scopus journals Carney Complex PubMed journals Carney Complex medical journals Carney Complex free journals Carney Complex best journals Carney Complex top journals Carney Complex free medical journals Carney Complex famous journals Carney Complex Google Scholar indexed journals Myxoma articles Myxoma Research articles Myxoma review articles Myxoma PubMed articles Myxoma PubMed Central articles Myxoma 2023 articles Myxoma 2024 articles Myxoma Scopus articles Myxoma impact factor journals Myxoma Scopus journals Myxoma PubMed journals Myxoma medical journals Myxoma free journals Myxoma best journals Myxoma top journals Myxoma free medical journals Myxoma famous journals Myxoma Google Scholar indexed journals Cardiac Neoplasm articles Cardiac Neoplasm Research articles Cardiac Neoplasm review articles Cardiac Neoplasm PubMed articles Cardiac Neoplasm PubMed Central articles Cardiac Neoplasm 2023 articles Cardiac Neoplasm 2024 articles Cardiac Neoplasm Scopus articles Cardiac Neoplasm impact factor journals Cardiac Neoplasm Scopus journals Cardiac Neoplasm PubMed journals Cardiac Neoplasm medical journals Cardiac Neoplasm free journals Cardiac Neoplasm best journals Cardiac Neoplasm top journals Cardiac Neoplasm free medical journals Cardiac Neoplasm famous journals Cardiac Neoplasm Google Scholar indexed journals Neoplasms articles Neoplasms Research articles Neoplasms review articles Neoplasms PubMed articles Neoplasms PubMed Central articles Neoplasms 2023 articles Neoplasms 2024 articles Neoplasms Scopus articles Neoplasms impact factor journals Neoplasms Scopus journals Neoplasms PubMed journals Neoplasms medical journals Neoplasms free journals Neoplasms best journals Neoplasms top journals Neoplasms free medical journals Neoplasms famous journals Neoplasms Google Scholar indexed journals Atrial Mass articles Atrial Mass Research articles Atrial Mass review articles Atrial Mass PubMed articles Atrial Mass PubMed Central articles Atrial Mass 2023 articles Atrial Mass 2024 articles Atrial Mass Scopus articles Atrial Mass impact factor journals Atrial Mass Scopus journals Atrial Mass PubMed journals Atrial Mass medical journals Atrial Mass free journals Atrial Mass best journals Atrial Mass top journals Atrial Mass free medical journals Atrial Mass famous journals Atrial Mass Google Scholar indexed journals treatment articles treatment Research articles treatment review articles treatment PubMed articles treatment PubMed Central articles treatment 2023 articles treatment 2024 articles treatment Scopus articles treatment impact factor journals treatment Scopus journals treatment PubMed journals treatment medical journals treatment free journals treatment best journals treatment top journals treatment free medical journals treatment famous journals treatment Google Scholar indexed journals CT articles CT Research articles CT review articles CT PubMed articles CT PubMed Central articles CT 2023 articles CT 2024 articles CT Scopus articles CT impact factor journals CT Scopus journals CT PubMed journals CT medical journals CT free journals CT best journals CT top journals CT free medical journals CT famous journals CT Google Scholar indexed journals surgery articles surgery Research articles surgery review articles surgery PubMed articles surgery PubMed Central articles surgery 2023 articles surgery 2024 articles surgery Scopus articles surgery impact factor journals surgery Scopus journals surgery PubMed journals surgery medical journals surgery free journals surgery best journals surgery top journals surgery free medical journals surgery famous journals surgery Google Scholar indexed journals Pathogenesis articles Pathogenesis Research articles Pathogenesis review articles Pathogenesis PubMed articles Pathogenesis PubMed Central articles Pathogenesis 2023 articles Pathogenesis 2024 articles Pathogenesis Scopus articles Pathogenesis impact factor journals Pathogenesis Scopus journals Pathogenesis PubMed journals Pathogenesis medical journals Pathogenesis free journals Pathogenesis best journals Pathogenesis top journals Pathogenesis free medical journals Pathogenesis famous journals Pathogenesis Google Scholar indexed journals Rapid Growth Myxoma articles Rapid Growth Myxoma Research articles Rapid Growth Myxoma review articles Rapid Growth Myxoma PubMed articles Rapid Growth Myxoma PubMed Central articles Rapid Growth Myxoma 2023 articles Rapid Growth Myxoma 2024 articles Rapid Growth Myxoma Scopus articles Rapid Growth Myxoma impact factor journals Rapid Growth Myxoma Scopus journals Rapid Growth Myxoma PubMed journals Rapid Growth Myxoma medical journals Rapid Growth Myxoma free journals Rapid Growth Myxoma best journals Rapid Growth Myxoma top journals Rapid Growth Myxoma free medical journals Rapid Growth Myxoma famous journals Rapid Growth Myxoma Google Scholar indexed journals
Article Details
Abbreviations:
PFO: patent foramen ovale; CC: carney complex syndrome
1. Introduction
Carney Complex Syndrome (CNC) is a rare genetic disorder characterized by cardiac myxomas, and schwannomas, multiple endocrine neoplasms with or without endocrine hyperactivity and skin anomalies. The transmission of CNC is autosomal dominant and involve PRKAR1A gene mutation in more than 60% of cases [1]. On average, the diagnosis is made around 20 years old, while cardiac myxoma occurs on average at 30 years [2]. Cardiac myxomas are common feature of the syndrome, and early diagnosis and treatment are essential to prevent complications [3]. Finally, there are rare cases with CNC developing myxoma in organs other than the heart, such as the breasts, female genitals and skin. The most common feature observed in individuals with CNC is freckles that range from pale brown to black. It is common that the number of freckles increases with puberty [4].
The most frequent oncological manifestation of the disease is in the thyroid gland; usually it is a non-productive follicular adenoma. However, patient affected by CNC can manifest during the life different and multiple neoplasm. Oncological manifestations that involve patients with CNC are primary pigmented nodular adrenocortical disease PPNAD, related to Cushing syndrome, Large-cell calcifying Sertoli cell tumors LCCSC, pituitary adenomas producing growth hormone GH and schwannomas. Cardiac myxomas tend to arise at a young age and can be localized in any cardiac chamber with a higher rate of onset in the left atrium, adhering to the interatrial septum. It is important, according to the guidelines, a regular screening of the patients with CNC for the potential insurgence of cardiac myxoma and echocardiography is the gold standard method. Screening has the function of preventing fatal complications such as obstruction of trans-mitral blood flow, embolic phenomena, malignant arrhythmias, and heart failure [1]. In fact, cardiac myxoma is associated with a high risk of sudden death, about 16% of cases of CNC. According to the literature, cardiac myxomas should be diagnosed promptly and for this reason a color Doppler echocardiography should be performed annually o biennially [5, 6]. Otherwise, patients with a history of myxoma should be screened more frequently with echocardiography every 6 months [7].
2. Case Report
We present a case of a 38-years-old female patient with CNC (proband). She is affected by acromegaly and at the age of 27 she undergone surgery for pituitary adenoma. The patient presented the typical pathological phenotype with freckles on her face (Figure 1).
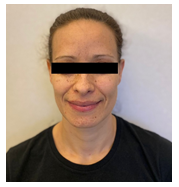
Figure 1: Typical pathological phenotype with freckles
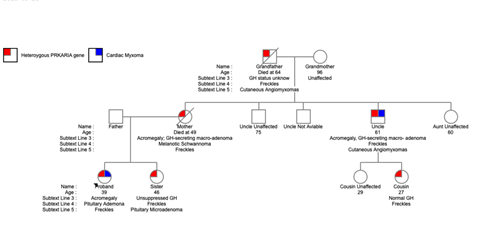
Figure 2: The dominant transmission of the disease was possible to be identified in the patient family.
Four of her closest relatives were themselves affected by the syndrome.
The maternal grandfather (GF) died at the age of 64 years, he was affected by the mutation, he presented with the cutaneous manifestations such as freckles and cutaneous angiomyxomas and Two of GF's five children were affected. One of these was the patient's uncle, who also presented with freckles, cutaneous angiomyxomas and GH-secreting pituitary adenoma. Proband's uncle underwent cardiac surgery in 2018 at our hospital to remove two atrial myxomas of the left atrium measuring 2 × 3 and 8 × 4 centimeters, respectively. The other affected relative was the proband's mother, presented with CNC manifestations such as freckles, a GH-secreting pituitary macroadenoma, and melanotic Schwannoma. The mother underwent selective transsphenoidal adenectomy at the age of 44 and died at the age of 49. The proband has a seven year older sister, also affected but has not yet had cardiac manifestations. She also presented with cutaneous phenotype and a GH-secreting adenoma, for which she underwent selective transsphenoidal adenectomy in 2009.
During April 2022, according to the regular cardiac imaging screening, proband underwent a transthoracic echocardiogram, which did not show any cardiac tumors. However, in December 2022, due to the occurrence of symptoms of dyspnea and palpitations she undergone to a new echocardiography, which revealed a large oval mass with heterogeneous echotexture and irregular margins in the left atrium, with a broad base of implantation at the interatrial septum, measuring 5 × 5, 5 cm. Additionally, a PFO, a common anatomical variant of interatrial septum, was also found (Figure 3). The mass engaged the left ventricle through the mitral valve in systole, causing filling obstruction (Figure 4).
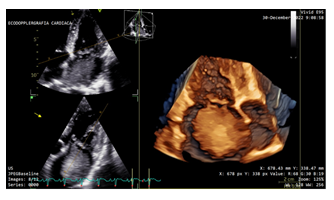
Figure 3: Preoperative echocardiography: PFO, a common anatomical variant of interatrial septum
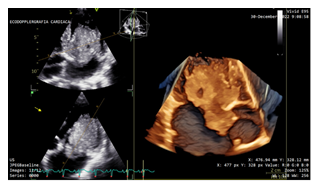
Figure 4: Preoperative echocardiography: the mass engaged the left ventricle through the mitral valve in systole, causing filling obstruction
The patient underwent cardiothoracic surgery to remove the myxoma and close the PFO via right mini thoracotomy and with extracorporeal circulation support (Figures 5, 6). The postoperative course was regular without minor and major complications, and the patient being transferred from the intensive care unit to the ward on the first postoperative day, and then transferred to cardiac rehabilitation on the fifth postoperative day.
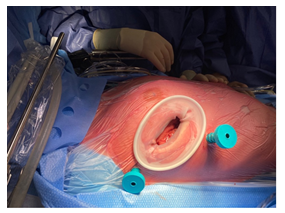
Figure 5: Surgical access, right mini thoracotomy
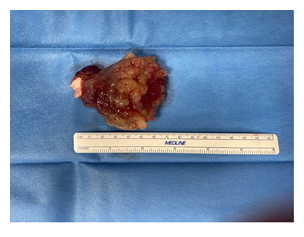
Figure 6: Myxoma after removal
3. Conclusion
This case report highlights the importance of speed up echocardiographic screenings in patients with Carney Complex Syndrome, even if they have not yet shown any cardiac manifestations. The rapid development of a large cardiac myxoma in our patient shows that such population is at risk of embolization and sudden cardiac events.
Therefore, we recommend that patients with CNC should undergo echocardiographic screenings every 6-9 months to allow for early diagnosis and treatment of any cardiac manifestations.
No Conflict of Interest to Declare
The patient has been informed and has given his consent to the use of the data and images.
References
- Pecori Giraldi F, Fatti LM, Bertola G, et al. Carney's complex with acromegaly as the leading clinical condition. Clin Endocrinol (Oxf) 68 (2008): 322-324.
- Sakuta H. [ATP-sensitive potassium channel and hormone/neuropeptide]. Nihon Naibunpi Gakkai Zasshi 71 (1995): 579-586.
- Stratakis CA, Raygada M. Carney Complex (2003). In: Adam MP, Mirzaa GM, Pagon RA, et al, editors. GeneReviews®[Internet]. Seattle (WA): University of Washington, Seattle 1993-2023.
- Stratakis CA, Carney JA, Lin JP, et al. Carney complex, a familial multiple neoplasia and lentiginosis syndrome. Analysis of 11 kindreds and linkage to the short arm of chromosome 2. J Clin Invest 97 (1996): 699-705.
- Bertherat J. Carney complex (CNC). Orphanet J Rare Dis 1 (2006): 21.
- Espiard S, Vantyghem MC, Assié G, et al. Frequency and Incidence of Carney Complex Manifestations: A Prospective Multicenter Study with a Three-Year Follow-Up. J Clin Endocrinol Metab 105 (2020): dgaa002.
- Samanidis G, Khoury M, Balanika M, et al. Current challenges in the diagnosis and treatment of cardiac myxoma. Kardiol Pol 78 (2020): 269-277.
