Radiochemotherapy and Immunotherapy for A Tracheal Carcinoma
Article Information
Larnaudie*, H. Orliac , N. Deny, C. Naessens, R. Serre, P. Clavere
Department of Radiation Oncology, Dupuytren University Hospital, Limoges, France
*Corresponding Author: Larnaudie A, Department of Radiation Oncology, Dupuytren University Hospital, Limoges, France
Received: 15 September 2022; Accepted: 20 October 2022; Published: 15 November 2022
Citation: A. Larnaudie, H. Orliac, N. Deny, C. Naessens, R. Serre, P. Clavere. Radiochemotherapy and Immunotherapy for A Tracheal Carcinoma. Archives of Clinical and Medical Case Reports 6 (2022): 730-733.
View / Download Pdf Share at FacebookAbstract
Tracheal cancer is a rare localization with a lack of treatment consensus. A 45-year-old man presented a tracheal squamous cell carcinoma with mediastinal nodes and oesophagus involvement. Treatment consisted in induction chemotherapy, radiochemotherapy and immunotherapy. The different PET realised before, during and after induction chemotherapy helped to precise the radiotherapy volumes. Three months after radiochemotherapy, PET CT showed a partial response. We report choices done in our service in terms of radiotherapy and systemic treatment.
Keywords
Radiotherapy; Tracheal Cancer; Squamous Cell Carcinoma
Radiotherapy articles; Tracheal Cancer articles; Squamous Cell Carcinoma articles
Radiotherapy articles Radiotherapy Research articles Radiotherapy review articles Radiotherapy PubMed articles Radiotherapy PubMed Central articles Radiotherapy 2023 articles Radiotherapy 2024 articles Radiotherapy Scopus articles Radiotherapy impact factor journals Radiotherapy Scopus journals Radiotherapy PubMed journals Radiotherapy medical journals Radiotherapy free journals Radiotherapy best journals Radiotherapy top journals Radiotherapy free medical journals Radiotherapy famous journals Radiotherapy Google Scholar indexed journals COVID-19 articles COVID-19 Research articles COVID-19 review articles COVID-19 PubMed articles COVID-19 PubMed Central articles COVID-19 2023 articles COVID-19 2024 articles COVID-19 Scopus articles COVID-19 impact factor journals COVID-19 Scopus journals COVID-19 PubMed journals COVID-19 medical journals COVID-19 free journals COVID-19 best journals COVID-19 top journals COVID-19 free medical journals COVID-19 famous journals COVID-19 Google Scholar indexed journals Tracheal Cancer articles Tracheal Cancer Research articles Tracheal Cancer review articles Tracheal Cancer PubMed articles Tracheal Cancer PubMed Central articles Tracheal Cancer 2023 articles Tracheal Cancer 2024 articles Tracheal Cancer Scopus articles Tracheal Cancer impact factor journals Tracheal Cancer Scopus journals Tracheal Cancer PubMed journals Tracheal Cancer medical journals Tracheal Cancer free journals Tracheal Cancer best journals Tracheal Cancer top journals Tracheal Cancer free medical journals Tracheal Cancer famous journals Tracheal Cancer Google Scholar indexed journals Ultrasound articles Ultrasound Research articles Ultrasound review articles Ultrasound PubMed articles Ultrasound PubMed Central articles Ultrasound 2023 articles Ultrasound 2024 articles Ultrasound Scopus articles Ultrasound impact factor journals Ultrasound Scopus journals Ultrasound PubMed journals Ultrasound medical journals Ultrasound free journals Ultrasound best journals Ultrasound top journals Ultrasound free medical journals Ultrasound famous journals Ultrasound Google Scholar indexed journals Squamous Cell Carcinoma articles Squamous Cell Carcinoma Research articles Squamous Cell Carcinoma review articles Squamous Cell Carcinoma PubMed articles Squamous Cell Carcinoma PubMed Central articles Squamous Cell Carcinoma 2023 articles Squamous Cell Carcinoma 2024 articles Squamous Cell Carcinoma Scopus articles Squamous Cell Carcinoma impact factor journals Squamous Cell Carcinoma Scopus journals Squamous Cell Carcinoma PubMed journals Squamous Cell Carcinoma medical journals Squamous Cell Carcinoma free journals Squamous Cell Carcinoma best journals Squamous Cell Carcinoma top journals Squamous Cell Carcinoma free medical journals Squamous Cell Carcinoma famous journals Squamous Cell Carcinoma Google Scholar indexed journals Radiotherapy articles Radiotherapy Research articles Radiotherapy review articles Radiotherapy PubMed articles Radiotherapy PubMed Central articles Radiotherapy 2023 articles Radiotherapy 2024 articles Radiotherapy Scopus articles Radiotherapy impact factor journals Radiotherapy Scopus journals Radiotherapy PubMed journals Radiotherapy medical journals Radiotherapy free journals Radiotherapy best journals Radiotherapy top journals Radiotherapy free medical journals Radiotherapy famous journals Radiotherapy Google Scholar indexed journals CT articles CT Research articles CT review articles CT PubMed articles CT PubMed Central articles CT 2023 articles CT 2024 articles CT Scopus articles CT impact factor journals CT Scopus journals CT PubMed journals CT medical journals CT free journals CT best journals CT top journals CT free medical journals CT famous journals CT Google Scholar indexed journals Prognosis articles Prognosis Research articles Prognosis review articles Prognosis PubMed articles Prognosis PubMed Central articles Prognosis 2023 articles Prognosis 2024 articles Prognosis Scopus articles Prognosis impact factor journals Prognosis Scopus journals Prognosis PubMed journals Prognosis medical journals Prognosis free journals Prognosis best journals Prognosis top journals Prognosis free medical journals Prognosis famous journals Prognosis Google Scholar indexed journals Seasonality articles Seasonality Research articles Seasonality review articles Seasonality PubMed articles Seasonality PubMed Central articles Seasonality 2023 articles Seasonality 2024 articles Seasonality Scopus articles Seasonality impact factor journals Seasonality Scopus journals Seasonality PubMed journals Seasonality medical journals Seasonality free journals Seasonality best journals Seasonality top journals Seasonality free medical journals Seasonality famous journals Seasonality Google Scholar indexed journals Teledentistry articles Teledentistry Research articles Teledentistry review articles Teledentistry PubMed articles Teledentistry PubMed Central articles Teledentistry 2023 articles Teledentistry 2024 articles Teledentistry Scopus articles Teledentistry impact factor journals Teledentistry Scopus journals Teledentistry PubMed journals Teledentistry medical journals Teledentistry free journals Teledentistry best journals Teledentistry top journals Teledentistry free medical journals Teledentistry famous journals Teledentistry Google Scholar indexed journals
Article Details
1. Introduction
Tracheal cancers are a rare tumour localisation, estimated at less than 0.1 per 100,000 inhabitants [1]. Tobacco is the main risk factor [2]. Histological differences are described - the three most common being squamous cell carcinoma, adenocarcinoma and adenoid cystic carcinoma. The princeps treatment is based on surgery, radiotherapy being reserved either for the inoperable case or as an adjuvant in case of histopathologic poor prognostic factors [2,3]. Various systemic treatments have been combined concomitantly to radiochemotherapy, mostly cisplatin derivatives [4,5].
2. Case Presentation
2.1 Characteristics and Treatment Decisions
A 45-year-old man, performance status 0, with no particular medical history except smoking and asthma, developed a squamous cell carcinoma of the trachea, discovered by pharyngeal pain. A CT scan found thickening of the left wall of the trachea on 12 and 19 mm. The fibroscopy showed a budding tracheal lesion located 2 cm below the vocal cords by 3 cm in height with a halving of the caliber of the trachea. Biopsies were in favor of a PDL1 negative squamous cell carcinoma. The multidisciplinary panel (MTP) recommended a chemotherapy first line associating carboplatin and paclitaxel due to a respiratory aggravation. The patient refused a tracheal prosthesis. After two cures, a CT and endoscopic reassessment found a partial response at the level of the tracheal lesion and lymphadenopathy. Radio-chemotherapy was decided because of the oesophageal invasion and the surgery patient's refusal.
2.1 Imaging
The positron emission tomography/ computed tomography (PET CT) before chemotherapy described a hypermetabolism of the lateral wall of the left trachea with a max SUV of 13 without hypermetabolism at the mediastinal level. A second one performed after the end of the 1st chemotherapy found the initial tracheal involvement with a max SUV of 11 and mediastinal involvement not previously present with lymphadenopathy located in the upper left paratracheal, lower right laterotracheal and paratracheal regions. The PET CT performed just before radiotherapy found a partial response with disappearance of the tracheal focus and persistence of a moderate-intensity focus remaining suspicious involving Barety's space. The figure 1 shows the visible changes between the different PET CT.
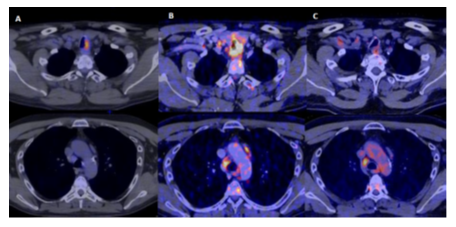
Figure 1: Evolution of PET CT scans before radiotherapy treatment.
From column left to right: A. Before chemotherapy-; B. After the 1st chemotherapy and C. After completion of chemotherapy, for radiotherapy treatment.
3. Treatments
3.1 Radiotherapy
A CT scan with a 5-point mask was performed. A PET scan in the treatment position was done within 48 hours to help delineation and comparison with previous targets. After consulting a Cancer Control Center, the total dose consisted of 66 Gy in 33 fractions. The volumes included the initial tumour, taking in consideration the evolution and barriers with a CTV margin of 5 mm. Initial mediastinal lymph nodes still morphologically or metabolically present were included with a margin of 5 mm. This volume was adapted to the affected areas and to the initial volumes of the lymph node CTV. A 5mm PTV was taken. Figure 2 shows the delineation made.
3.2 Systemic Treatment
Systemic treatment with Carboplatin AUC Paclitaxel 175 mg/m2 was pursued during radiotherapy. Durvalumab started after completion of radiochemotherapy.
3.3 Acute Tolerance
Radiotherapy and chemotherapy were well tolerated with grade 1 asthenia, grade 2 esophagitis. Swallowing disorders were reported, compatible with silent aspiration but not found using endoscopic exam.
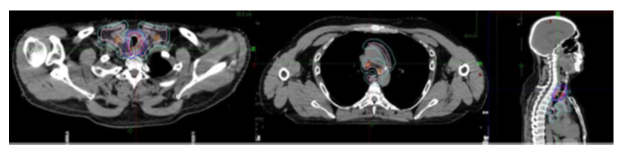
Figure 2: Delineation. P: Primary Tumour – N: Lymph Nodes
Red: GTV P; pink: CTV P; Dark blue PTV – P; light pink CTV N; light blue PTV N.
4. Treatment Evaluation
At 3 months after radiochemotherapy, an improvement of breathing and silent aspiration were described. PET CT and CT scan showed a partial response with stability of the Barety's space node, previously in the area of radiotherapy treatment receiving 66 Gy. Due to the partial response, the multidisciplinary panel decided continuation of Durvalumab with a close follow up. Figure 3 illustrates the PET and CT scan aspect.
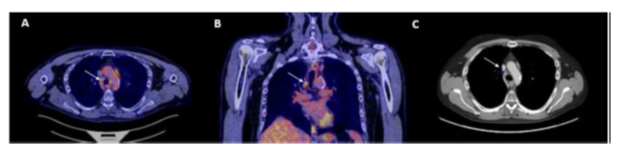
Figure 2: Delineation. P: Primary Tumour – N: Lymph Nodes
PET CT: A et B and CT: C – partial response on node ( arrow).
5. Discussion
This is a rare case of tracheal tumour, with esophageal and lymph node involvement, touching a young patient with few comorbidities. Treatment with chemotherapy, radiochemotherapy and immunotherapy was well tolerated without requiring dose reduction or discontinuation. The mediastinal involvement not found in imaging before chemotherapy was not documented and generated an additional volume. The second advice at our centre was not in favor of an infectious etiology. The persistence, at least morphological, of the macroscopic adenopathies led to the choice to consider them as pathological. The delay between the first imaging and the start of chemotherapy could explain an unknown progression. The verification of the evolution by the different modalities of imaging and endoscopy examinations helped to precise radiotherapy volume with major information on the targets. The good general condition of the patient had permitted to carry out a treatment with concomitant chemotherapy. Most of the published series and case reports have instead used cisplatin alone 100 mg/m2 or weekly 40 mg/m2 [5–7] compared to carboplatin paclitaxel [8]. The extension limited to the esophagus did not lead to the placement of a prophylactic gastrostomy. Careful monitoring was done due to the increased risk of esophagitis and fistula as acute and late side effects respectively. The choice of volumes and margins in the case of squamous cell carcinoma is not consensual. Those performed by the department were consistent with the practices described in two articles [4,7] and approached the volumes used in Ear Nose and Throat ( ENT) and pulmonary radiotherapy [9,10]. As locoregional progressions are described in tracheal squamous cell carcinomas, lymph node coverage seemed important by taking at least the initial affected areas. Prognostic of tracheal cancer remains low, especially if no surgery. Overall survivals ( OS) were around 14-47% at 5 years on published cohorts [3–5,11–15]. A case report found a complete response after immunotherapy Pembrolizumab [16]. To our knowledge, it is the first case report published with adjuvant Durvalumab like Pacific Trial [17].
6. Conclusion
This case report shows the importance of the complementarity of endoscopic, morphological and metabolic examinations in the management of tracheal cancers. The choice of lymph node areas can lead to discussion, especially in the absence of consensus on the volumes. A benefit of immunotherapy is a possible hypothesis, as responses are described for ENT and lung squamous cell carcinoma. Local control and survival remain low in the absence of surgery, supporting a close follow up.
Conflict of Interest
None.
References
- Junker K. Pathology of Tracheal Tumors. Thoracic Surgery Clinics 24 (2014): 7-11.
- Madariaga MLL, Gaissert HA. Overview of malignant tracheal tumors. Ann Cardiothorac Surg 7 (2018): 244-254.
- Xie L, Fan M, Sheets NC, et al. The Use of Radiation Therapy Appears to Improve Outcome in Patients With Malignant Primary Tracheal Tumors: A SEER-Based Analysis. International Journal of Radiation Oncology*Biology*Physics 84 (2012): 464-470.
- Zeng R, Wang H, Cai X, et al. Radiotherapy for Primary Tracheal Carcinoma: Experience at a Single Institution. Technol Cancer Res Treat 20 (2021): 153303382110342.
- Napieralska A, Miszczyk L, Blamek S. Tracheal cancer – treatment results, prognostic factors and incidence of other neoplasms. Radiology and Oncology 50 (2016): 409-417.
- Kovacs AC, Vodanovich D, Mogridge EK, et al. A case of primary tracheal squamous cell carcinoma arising from malignant transformation of recurrent respiratory papillomatosis, with a complete response to concurrent chemoradiotherapy. SAGE Open Medical Case Reports 9 (2021): 2050313X2110546.
- Yathiraj PH, Ail S, Singh A, et al. Unresectable squamous cell carcinoma of upper trachea with long-term survival after concurrent chemoradiotherapy. BMJ Case Reports (2017): bcr-2017-221284.
- Haresh K, Das P, Kumar R, et al. Unresectable basaloid squamous cell carcinoma of the trachea treated with concurrent chemoradiotherapy: A case report with review of literature. J Can Res Ther 6 (2010): 321.
- Grégoire V, Evans M, Le QT, et al. Delineation of the primary tumour Clinical Target Volumes (CTV-P) in laryngeal, hypopharyngeal, oropharyngeal and oral cavity squamous cell carcinoma: AIRO, CACA, DAHANCA, EORTC, GEORCC, GORTEC, HKNPCSG, HNCIG, IAG-KHT, LPRHHT, NCIC CTG, NCRI, NRG Oncology, PHNS, SBRT, SOMERA, SRO, SSHNO, TROG consensus guidelines. Radiother Oncol 126 (2018): 3-24.
- Khalifa J, Lerouge D, Le Péchoux C, et al. Radiotherapy for primary lung cancer. Cancer/Radiothérapie 26 (2022): 231-43.
- Hararah MK, Stokes WA, Oweida A, et al. Epidemiology and treatment trends for primary tracheal squamous cell carcinoma. The Laryngoscope 130 (2020): 405-412.
- Agrawal S, Jackson C, Celie KB, et al. Survival trends in patients with tracheal carcinoma from 1973 to 2011. American Journal of Otolaryngology 38 (2017): 673-677.
- Jiang M, Lei Q, Lv X, et al. Clinical features and prognosis analysis of 57 patients with primary tracheal tumors. Transl Cancer Res TCR 9 (2020): 613-619.
- Yusuf M, Gaskins J, Trawick E, et al. Effects of adjuvant radiation therapy on survival for patients with resected primary tracheal carcinoma: an analysis of the National Cancer Database. Japanese Journal of Clinical Oncology 49 (2019): 628-638.
- Li Y, Peng A, Yang X, et al. Clinical manifestation and management of primary malignant tumors of the cervical trachea. Eur Arch Otorhinolaryngol 271 (2014): 225-235.
- Maller B, Kaszuba F, Tanvetyanon T. Complete Tumor Response of Tracheal Squamous Cell Carcinoma After Treatment With Pembrolizumab. The Annals of Thoracic Surgery 107 (2019): e273-4.
- Spigel DR, Faivre-Finn C, Gray JE, et al. Five-Year Survival Outcomes From the PACIFIC Trial: Durvalumab After Chemoradiotherapy in Stage III Non–Small-Cell Lung Cancer. JCO 40 (2022):1301-1311.
