Pulmonary Vein Stenosis in an Infant with Bronchopulmonary Dysplasia and Partial Anomalous Pulmonary Venous Circulation: Is This Double Trouble?
Article Information
Renjini Lalitha1,4*, Kambiz Norozi2,3,4, Soume Bhattacharya1,4
1University of Western Ontario, Department of Pediatrics, Division of Neonatal-Perinatal Medicine, London, Ontario, Canada
2University of Western Ontario, Department of Pediatrics, Division of Pediatric Cardiology, London, Ontario, Canada
3Department of Pediatric Cardiology and Intensive Care Medicine, Hannover Medical School, Hannover, Germany
4Children Health Research Institute, London, ON, Canada
*Corresponding Author: Renjini Lalitha, The Division of Neonatal-Perinatal Medicine, London Health Sciences Centre, 800 Commissioners Road East, London, ON, N6A 5W9, Canada
Received: 14 April 2023; Accepted: 24 April 2023; Published: 12 May 2023
Citation: Renjini Lalitha, Kambiz Norozi, Soume Bhattacharya. Pulmonary Vein Stenosis in an Infant with Bronchopulmonary Dysplasia and Partial Anomalous Pulmonary Venous Circulation: Is This Double Trouble?. Archives of Clinical and Medical Case Reports Reports. 7 (2023): 191-196.
View / Download Pdf Share at FacebookAbstract
Pulmonary vein stenosis (PVS) in preterm infants is a rare emerging complication that is associated with bronchopulmonary dysplasia (BPD) which has an unpredictable outcome. Though PVS can be associated with congenital heart defects, its concurrent occurrence in a preterm infant with partial anomalous pulmonary venous return (PAPVR) and severe BPD has not been documented yet. This case shows the potential progressive nature of PVS that might have been aggravated by the presence of two disease condition associated with it and the need for heighted vigilance when an unusual clinical trajectory is demonstrated in a high-risk preterm infant.
Keywords
Pulmonary vein stenosis, preterm infants, bronchopulmonary dysplasia, partial anomalous pulmonary venous return
Pulmonary vein stenosis articles, preterm infants articles, bronchopulmonary dysplasia articles, partial anomalous pulmonary venous return articles
Pulmonary vein stenosis articles Pulmonary vein stenosis Research articles Pulmonary vein stenosis review articles Pulmonary vein stenosis PubMed articles Pulmonary vein stenosis PubMed Central articles Pulmonary vein stenosis 2023 articles Pulmonary vein stenosis 2024 articles Pulmonary vein stenosis Scopus articles Pulmonary vein stenosis impact factor journals Pulmonary vein stenosis Scopus journals Pulmonary vein stenosis PubMed journals Pulmonary vein stenosis medical journals Pulmonary vein stenosis free journals Pulmonary vein stenosis best journals Pulmonary vein stenosis top journals Pulmonary vein stenosis free medical journals Pulmonary vein stenosis famous journals Pulmonary vein stenosis Google Scholar indexed journals stenosis articles stenosis Research articles stenosis review articles stenosis PubMed articles stenosis PubMed Central articles stenosis 2023 articles stenosis 2024 articles stenosis Scopus articles stenosis impact factor journals stenosis Scopus journals stenosis PubMed journals stenosis medical journals stenosis free journals stenosis best journals stenosis top journals stenosis free medical journals stenosis famous journals stenosis Google Scholar indexed journals preterm infants articles preterm infants Research articles preterm infants review articles preterm infants PubMed articles preterm infants PubMed Central articles preterm infants 2023 articles preterm infants 2024 articles preterm infants Scopus articles preterm infants impact factor journals preterm infants Scopus journals preterm infants PubMed journals preterm infants medical journals preterm infants free journals preterm infants best journals preterm infants top journals preterm infants free medical journals preterm infants famous journals preterm infants Google Scholar indexed journals bronchopulmonary dysplasia articles bronchopulmonary dysplasia Research articles bronchopulmonary dysplasia review articles bronchopulmonary dysplasia PubMed articles bronchopulmonary dysplasia PubMed Central articles bronchopulmonary dysplasia 2023 articles bronchopulmonary dysplasia 2024 articles bronchopulmonary dysplasia Scopus articles bronchopulmonary dysplasia impact factor journals bronchopulmonary dysplasia Scopus journals bronchopulmonary dysplasia PubMed journals bronchopulmonary dysplasia medical journals bronchopulmonary dysplasia free journals bronchopulmonary dysplasia best journals bronchopulmonary dysplasia top journals bronchopulmonary dysplasia free medical journals bronchopulmonary dysplasia famous journals bronchopulmonary dysplasia Google Scholar indexed journals dysplasia articles dysplasia Research articles dysplasia review articles dysplasia PubMed articles dysplasia PubMed Central articles dysplasia 2023 articles dysplasia 2024 articles dysplasia Scopus articles dysplasia impact factor journals dysplasia Scopus journals dysplasia PubMed journals dysplasia medical journals dysplasia free journals dysplasia best journals dysplasia top journals dysplasia free medical journals dysplasia famous journals dysplasia Google Scholar indexed journals treatment articles treatment Research articles treatment review articles treatment PubMed articles treatment PubMed Central articles treatment 2023 articles treatment 2024 articles treatment Scopus articles treatment impact factor journals treatment Scopus journals treatment PubMed journals treatment medical journals treatment free journals treatment best journals treatment top journals treatment free medical journals treatment famous journals treatment Google Scholar indexed journals CT articles CT Research articles CT review articles CT PubMed articles CT PubMed Central articles CT 2023 articles CT 2024 articles CT Scopus articles CT impact factor journals CT Scopus journals CT PubMed journals CT medical journals CT free journals CT best journals CT top journals CT free medical journals CT famous journals CT Google Scholar indexed journals surgery articles surgery Research articles surgery review articles surgery PubMed articles surgery PubMed Central articles surgery 2023 articles surgery 2024 articles surgery Scopus articles surgery impact factor journals surgery Scopus journals surgery PubMed journals surgery medical journals surgery free journals surgery best journals surgery top journals surgery free medical journals surgery famous journals surgery Google Scholar indexed journals Pathogenesis articles Pathogenesis Research articles Pathogenesis review articles Pathogenesis PubMed articles Pathogenesis PubMed Central articles Pathogenesis 2023 articles Pathogenesis 2024 articles Pathogenesis Scopus articles Pathogenesis impact factor journals Pathogenesis Scopus journals Pathogenesis PubMed journals Pathogenesis medical journals Pathogenesis free journals Pathogenesis best journals Pathogenesis top journals Pathogenesis free medical journals Pathogenesis famous journals Pathogenesis Google Scholar indexed journals partial anomalous articles partial anomalous Research articles partial anomalous review articles partial anomalous PubMed articles partial anomalous PubMed Central articles partial anomalous 2023 articles partial anomalous 2024 articles partial anomalous Scopus articles partial anomalous impact factor journals partial anomalous Scopus journals partial anomalous PubMed journals partial anomalous medical journals partial anomalous free journals partial anomalous best journals partial anomalous top journals partial anomalous free medical journals partial anomalous famous journals partial anomalous Google Scholar indexed journals
Article Details
1. Introduction
Pulmonary vein stenosis is a rare but emerging condition in ex-preterm infants who are diagnosed with bronchopulmonary dysplasia (BPD) with or without pulmonary hypertension (PHTN). This new entity has an unpredictable natural history with worse outcome when its earlier in onset and bilateral in nature [1]. While primary pulmonary vein stenosis has a known association with other cardiac defects especially septal defects [2], progressive pulmonary vein stenosis in the setting of a partial anomalous pulmonary venous return (PAPVR) is a rare occurrence. Here, we highlight a case of progressive pulmonary vein stenosis in an ex-preterm infant with bronchopulmonary dysplasia who also had PAPVR. With markedly variable diagnostic threshold for pulmonary vein stenosis (PVS), for this case report we defined PVS as mean pressure gradient greater than 2mmHg using doppler echocardiogram [3].
2. Case Presentation
We present a female infant born at 24weeks of gestational age to a 31year of G10A9. This spontaneously conceived pregnancy was complicated by essential hypertension and preterm labor that was managed with betamethasone and magnesium sulphate. The infant’s birth weight was 700grams (82nd centile). Her Apgar scores were 4, 7 and 8 at 1, 5 and 10 minutes respectively. She required intubation by 7minutes of life due to increasing oxygen (FiO2) needs of 0.8.
2.1 Clinical Course in Neonatal Period
In the Neonatal Intensive Unit (NICU) the infant was managed for issues related to extreme prematurity. She was initiated on high frequency oscillation (HFO) ventilation and given surfactant with good response. She has umbilical arterial line and a low-lying umbilical vein (UV) catheter. On 5th day of life she developed progressive abdominal distension, with hemodynamic instability consisting of diastolic hypotension, oliguria (0.8-1.1ml/kg/hr), worsening metabolic acidosis along with anemia and thrombocytopenia.
A neonatal hemodynamic consultation was requested. A targeted neonatal echocardiogram (TNE) discovered a large hepatic cystic mass with possible inferior vena cava (IVC) compression consistent with intrahepatic UVC extravasation. This UV catheter was replaced with an alternate central line and supportive management with normal saline bolus, packed red blood cell and platelet transfusion which resulted in satisfactory stabilization of hemodynamic parameters. The other TNE findings included a normal cardiac structure, a large patent ductus arteriosus (PDA) 2.1mm in size, shunting left to right with a peak gradient of 4.6mmHg suggesting near systemic pulmonary pressures. There were no echocardiographic markers suggesting hemodynamic significance of PDA. In this initial study - all four pulmonary veins appeared to drain into left atrium.
Her respiratory status remained an ongoing concern requiring escalation of support to a high frequency jet ventilator (HFJV) by second week of life. She received two courses of Dexamethasone (Course 1 at DOL 14 and course 2 at DOL 43). She was extubated and gradually weaned to high flow nasal canula at 5 litres/minute on 0.25-0.30 FiO2 by 36+5 weeks gestational age. She received a diagnosis of moderate to severe BPD at 36 weeks corrected and continue to demonstrate significant respiratory support needs till term corrected.
2.2 Echocardiographic Assessments
The infant had three echocardiographic assessments in the first 3 weeks of life, including the first one mentioned above, during her acute deterioration. The subsequent assessments continued to demonstrate a PDA and estimated pressures based on PDA gradient to be 80-90% of systemic. She was assessed for BPD associated pulmonary hypertension at 36 weeks corrected which revealed systemic-suprasystemic pulmonary hypertension as evidenced by moderate bidirectional PDA (71% of cardiac cycle is left to right shunt) with flattened septal curvature and mild right atrial and ventricular dilation with preserved cardiac systolic function. Note was made of left upper pulmonary vein (LUPV) stenosis with mean gradient of 7mmHg. Rest 3 pulmonary veins appeared to be normal. Pediatric Cardiology closely followed clinical and echocardiographic course of this infant. Following a course of Aldactazide, she was eventually weaned to Low flow oxygen by DOL#95 at 37+4weeks corrected gestational age. Due to C02 retention and rising needs to maintain oxygen saturations above 92%, the infant needed escalation of respiratory support to NIPPV/CPAP. She required multiple courses of diuretics for management of pulmonary hypertension in the setting of PVS associated with severe BPD.
|
Pulmonary vein gradient prior to surgical intervention |
DOL# 5 |
36weeks CGA |
38weeks CGA |
40weeks CGA |
|
LUPV, mmHg |
0.21 |
7 |
12.5 |
~ 1, almost atretic with minimal flow |
|
LLPV, mmHg |
0.27 |
1 |
2.2 |
2.4 |
|
Presumed RUPV, mmHg |
0.18 |
0.89 |
0.77 |
|
|
RLPV, mmHg |
0.24 |
0.7 |
0.82 |
0.35 |
|
PDA size and shunt direction |
Large PDA with left to right shunt |
Moderate PDA bidirectional shunt |
Moderate PDA bidirectional shunt |
Moderate PDA bidirectional shunt |
|
Right chambers |
RA and RV normal size |
RA/RV mild dilatation |
RA/RV moderate dilatation |
RA/RV moderate dilatation |
|
TAPSE, cm |
0.5 |
1.1 |
n/a |
n/a |
|
RV FAC, % |
37 |
44 |
45 |
37 |
Table 1: Serial trend in the Echocardiographic parameters prior to procedural intervention.
Abbreviations: CGA- corrected gestational age, DOL- Day of life, LUPV- Left Upper Pulmonary vein, LLPV- Left Lower Pulmonary vein, RUPV- Right Upper Pulmonary vein, RLPV- Right Lower Pulmonary vein, PDA- Patent Ductus Arteriosus, RA- Right Atrium, RV- Right Ventricle. TAPSE - Tricuspid Annular Plane Systolic Excursion, FAC-Fractional Area Change
Serial echocardiogram showed progression of pulmonary vein stenosis (Table 1) with LUPV becoming almost atretic (Figure 1A) by 40weeks corrected age and additional development of left lower pulmonary vein stenosis (LLPV) (Figure 1B). Quaternary referral center with surgical cardiac unit was consulted and she was deemed eligible for immediate intervention with dilatation of the stenotic veins.
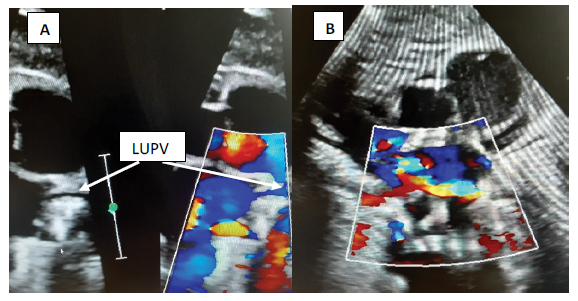
Figure 1: 1A shows severe stenotic almost atretic LUPV and 1B is the crab view showing almost absent flow in the LUPV and turbulent flow in LLPV suggesting PVS. There appears to be a flow where RUPV should drain into Left atrium.
2.3 Cardiac Catheterization Findings and Procedural Interventions
At the quaternary referral center, she underwent cardiac catheterization at 42+5 weeks corrected age which revealed severely stenosed and almost atretic LUPV that failed dilatation as well as mild to moderate stenosis of LLPV with successful balloon dilatation. The Doppler flow indicating RUPV draining into LA that was noted in initial echoes could not be confirmed by cardiac catheterization. Instead a moderate-severe stenotic right upper pulmonary vein was found to drain into the Superior Vena Cava (SVC) making it PAPVR anomaly. Additionally, an accessory right pulmonary vein was noted to drain into the left atrium. The right lower pulmonary vein was noted to have a normal laminar flow. Parents were counselled on the guarded nature of the prognosis given that three pulmonary veins were significantly affected along with PAPVR anomaly and early presentation with significant pulmonary hypertension. Palliative care options were discussed given the significant pathology. Parents chose to pursue a ‘Do Not Resuscitate (DNR) ‘plan that excluded use of chest compression and medication in the event of a cardiac arrest. Infant continued to have serial echocardiographic assessment with stable PVS and pulmonary hypertension. She had a reattempt for balloon dilatation of the atretic LUPV which was unsuccessful again. She was subsequently discharge home after 7months stay in the hospital on High Flow 4litres/minute at 25% FiO2. She never received pulmonary vasodilators given her lower FiO2 requirements to maintain saturations above 92% and relatively stable pulmonary hypertension.
2.4 Post Discharge Clinical Course
Following her hospital discharge, she had three hospital visits to the local emergency department due to respiratory distress needing escalation in respiratory support within the first 9months of her life. She was found to have frequent hypoxemia to 80% oxygen saturations on High Flow of 4l/min that was associated with agitation/crying/clinical care as well as tachypnea and increased work of breathing leading to re-admission to Pediatric Critical Care Unit (PCCU) each time. Other than the significant pulmonary hypertension suggested by bidirectional PDA shunt, echocardiogram also demonstrated re-stenosis of LLPV and RUPV along with SVC stenosis with the first two re-admissions. She was transferred to quaternary center for procedural intervention consisting of balloon dilatation and stenting of LLPV and balloon dilatation of both RUPV and SVC. The last admission at 7months corrected age and subsequent follow up cardiac clinic visits revealed stable PVS but persistence of significant pulmonary hypertension with adequate cardiac function. There were no infectious etiology or chest radiograph abnormalities associated with any of these readmissions. She had 2-3weeks stay at PCCU with each of those admissions. At present, she is 9months corrected age with history of procedural interventions for PVS on four occasions thus far and has been actively followed by home palliative care team with stable clinical course and remains on High Flow 4l/min. She continues to have adequate weight gains following enteral nutrition through G-tube. She has monthly cardiac appointments and weekly follow up by palliative care team.
3. Discussion
Pulmonary vein stenosis (PVS) is a rare but serious complication of premature infants with Bronchopulmonary dysplasia (BPD). It is a progressive disease with 5-year survival rate being less than 50% [1, 4]. This has led to increasing vigilance by the neonatal physicians towards its earlier detection despite its variable presentation. The median age of diagnosis of PVS in preterm infants ranged from 5-7.4months across literature with only 20% diagnosed before NICU discharge [5, 6, 7]. The widespread availability of bedside echocardiogram has made detailed screening for PVS in the high-risk preterm infants possible with increasing detection rate over the last one decade. Approximately half of the pediatric patients with PVS also have associated cardiac defect [2]. Here we have described a rare association of extreme prematurity with moderate-severe BPD and PAPVR contributing to the increased risk for development of PVS. To the best of our knowledge this association has not been reported previously in literature. Our report, summarizes echocardiographic findings, details of clinical course, management plans associated with this rare disease entity as there is limited information about the clinical trajectory of this disease process.
It is estimated that in 30-80% of cases of PVS, there are other associated cardiac defects [8-10]. This is mostly left to right shunting lesions like Atrial Septal defect, Ventricular Septal defect, Atrio-Ventricular septal defect, and PDA. But it can also be seen in conjunction with all major type of congenital cardiac malformations [2]. An association of pulmonary venous agenesis with PAPVR is postulated to result from abnormal incorporation of the common pulmonary vein into the left atrium in the later stages of cardiac development [11]. During the 4th week of the embryonic period, there is a common pulmonary venous out pouching that develops from the posterior part of the primordial left atrium. This out pouching engages both pulmonary and systemic venous system initially. With normal cardiac development this connection separates into two independent venous systems namely pulmonary and systemic venous system and eventual formation of 2 right-sided and 2 left-sided pulmonary veins that enter the smooth portion of the posterior left atrium [11]. Failure of this process may result in persistence of the connections of the pulmonary veins to the systemic venous system leading to the various forms of partial or total anomalous pulmonary venous return. It has been postulated that abnormal incorporation of the common pulmonary vein into the left atrium in the later stages of cardiac development can result in primary PVS. This is suggested to be a neo-proliferative process and therefore may not be evident at birth but could progress later [11, 12].
Our case showed progressive nature of PVS needing multiple procedural interventions. Both PAPVR and BPD could have simultaneously accelerated the progression of PVS in our case. Bilateral, multiple vein involvement, presentation before 6months of age and recurrent stenosis despite intervention suggest aggressive disease progression and poor prognosis for survival [1, 13]. The one-year mortality rate reaches 80% when ≥ 3veins are affected. [1, 2, 14-18]. The upstream effect of the progressive pulmonary vein disease leads to pulmonary hypertension and right ventricular dysfunction. This can manifest as persistent and frequent hypoxemia, tachypnea, increased work of breathing, unexplained increase in ventilatory and oxygen support [5, 14]. These were the presentations that our case manifested at each of the emergency visit leading up to re-admissions. The lability of the oxygen saturations is more in keeping with the clinical manifestation of pulmonary hypertension in our case though the exact cause for this sudden lability could not be delineated.
There is increasing evidence that the left sided pulmonary veins are more affected than the right sided ones, which is noted in our case as well [6, 21-23]. The explanation for this observation is unclear. But it’s being proposed that since supine position is the most preferred position in infants, the possible compression of the left pulmonary veins against the heart and the spine could potentially lead to stenosis. While others suggest an increased volume of blood shunting to the left pulmonary artery due to PDA and the resultant increased blood circulating in the left lung leading up to vascular endothelial injury could be a contributing factor [1].
Echocardiogram is generally sufficient to diagnosis PVS and remains the first line imaging modality. Although echocardiogram allows bedside assessment and is easily accessible in many centers, inadequate acoustic windows in these extreme preterm infants on ventilatory support limits its diagnostic yield both in the diagnosis of PVS and PAPVR (6). Furthermore, less than 20% of PVS is typically detected before discharge from the NICU and most infants have a median of 3-5 echocardiograms before diagnosis is made [7]. Hence cardiac catheterization/angiogram remains the gold standard for diagnostic purposes. In our case, the right accessory pulmonary vein was mistaken for RUPV in all of the echocardiograms performed to assess pulmonary veins missing the draining of RUPV into the SVC and [21] the development of stenosis in RUPV. This again highlights the limitation of echocardiography in diagnosis of PAPVR spectrum. Both Pediatric Pulmonary Hypertension Network and American Heart Association and American Thoracic society collectively recommends evaluation of pulmonary veins during echocardiographic assessment for BPD associated pulmonary hypertension at 36weeks corrected age in all preterm infants with established BPD [24, 25]. Detailed re-evaluation of pulmonary veins especially in the context of ongoing pulmonary hypertension in neonates-with established BPD, atypical clinical trajectory or poor response to medical therapy for pulmonary hypertension cannot be over emphasized.
Current medical therapy for PVS is mainly focused on symptomatic management of cardiorespiratory symptoms. Diuretic therapy is the mainstay of treatment for pulmonary venous congestion that ensues due to PVS. Use of pulmonary vasodilators for pulmonary hypertension are controversial in PVS due to the fixed nature of obstruction ad therefore potential worsening of pulmonary congestion secondary to its use [3]. It is prudent that the use of pulmonary vasodilators should be in conjunction with diuretic therapy in such cases. Definitive management of PVS is surgical and Coles procedure which is suture less technique is the preferred method [2, 6]. The goal of this method is to reduce surgical trauma and hence reduce any stimulus that can worsen the fibroproliferative process leading to re-stenosis. Other procedural interventions include balloon dilatation and stent implantation [2, 26, 27]. In refractory cases lobectomy and lung transplantation has been performed. Prognosis is guarded even after surgical intervention with less than 50% chance of survival at 5years. With bilateral vein involvement and development of restenosis needing repeat procedural intervention, and the presence of at least two aggravating factors, namely BPD and PAPVR, that can contribute to progression of PVS and the presence of significant pulmonary hypertension, the prognosis for our case is considered poor. Commonly death is secondary to pulmonary hypertension, recurrent pulmonary infection or hemoptysis [2]. Consultation with experts and institutions that have expertise in caring for children with PVS is highly recommended to develop an individualized treatment and follow-up plan. Palliative care could be a reasonable option in cases where quality of life is deemed significantly affected.
4. Conclusion
PVS and BPD are morbid diseases that have some pathogenetic and epidemiologically association. Cardiac defects are frequent association with PVS which can further aggravate the pathogenetic process. Association of PAPVR with PVS has not been well described in literature. The combination of PAPVR and BPD could potentially accelerate the progressive nature of PVS making its course unrelenting despite surgical intervention. Routine echocardiographic evaluation has some inherent limitation when assessing PAPVR especially in the presence of accessory pulmonary veins. High index of suspicion, early screening and detection, timely referral is paramount if actual clinical course is out of keeping with the expected trajectory.
Source of Financial Support
None
Conflicts of Interest
None
References
- Mahgoub L, Kaddoura T, Kameny AR, Lopez Ortego P, Vanderlaan RD, Kakadekar A, et.al. Pulmonary vein stenosis of ex-premature infants with pulmonary hypertension and bronchopulmonary dysplasia, epidemiology, and survival from a multicenter cohort. Pediatric Pulmonology 52 (2017): 1063-1070.
- Latson LA and Prieto LR. Congenital and Acquired Pulmonary Vein Stenosis. Circulation 115 (2007): 103-108.
- Jadcherla AV, Backes CH, Cua CL, Smith CV, Levy PT, Ball MK, Primary Pulmonary Vein Stenosis: A New Look at a Rare but Challenging Disease. Neoreviews 22 (2021): e296-e308.
- Frank DB, Levy PT, Stiver CA, Boe BA, Baird CW, Callahan RM, et al. Primary pulmonary vein stenosis during infancy: State of the art review. J. Perinatol 41 (2021): 1528-1539.
- Backes CH, Nealon E, Armstrong AK, Cua C, Mitchell C, Krishnan U, et al. Pulmonary Vein Stenosis in Infants: A Systematic Review, Meta-Analysis, and Meta-Regression. J. Pediatr 198 (2018): 36-45.
- Drossner DM, Kim DW, Maher KO, Mahle WT. Pulmonary vein stenosis: prematurity and associated conditions. Pediatrics 122 (2008): e656-e661.
- Vyas-Read S, Varghese NP, Suthar D, Backes C, Lakshminrusimha S, Petit CJ, et al. Prematurity and Pulmonary Vein Stenosis: The Role of Parenchymal Lung Disease and Pulmonary Vascular Disease. Children 9 (2022): 713
- Bini RA, Cleveland DC, Ceballos R, Bargeron LM, Pacifico AD, Kirklin JW. Congenital pulmonary vein stenosis. Am J Cardiol 54 (1984): 369-75.
- Fong LV, Anderson RH, Park SC, Zuberbuhler JR. Morphologic features of stenosis of the pulmonary veins. Am J Cardiol 62 (1988): 1136-1138.
- Driscoll DJ, Hesslein PS, Mullins CE. Congenital stenosis of individual pulmonary veins: clinical spectrum and unsuccessful treatment of transvenous balloon dilation. Am J Cardiol 49 (1982): 1767-1772.
- Edwards JE. Congenital stenosis of pulmonary veins: pathologic and developmental considerations. Lab Invest 9 (1960): 46-66.
- Sadr IM, Tan PE, Kieran MW, Jenkins KJ. Mechanism of pulmonary vein stenosis in infants with normally connected veins. Am J Card 86 (2000): 577-579.
- Cory MJ, Ooki YK, Kelleman MS, Vincent RN, Kim DW, Petit CJ. Reintervention Is Associated with Improved Survival in Pediatric Patients With Pulmonary Vein Stenosis. JACC Cardiovasc Interv 10 (2017): 1788-1798.
- Holt DB, Moller JH, Larson S, Johnson MC. Primary pulmonary vein stenosis. Am. J. Cardiol 99 (2007): 568-572.
- Breinholt JP, Hawkins JA, Minich LA, Tani LY, Orsmond GS, Ritter S, et al. Pulmonary vein stenosis with normal connection: associated cardiac abnormalities and variable outcome. Ann Thorac Surg 68 (1999): 164-8.
- Song MK, Bae EJ, Jeong SI, Kang IS, Kim NK, Choi JY, et al. Clinical characteristics and prognostic factors of primary pulmonary vein stenosis or atresia in children. Ann Thorac Surg 95 (2013): 229-34.
- Lo Rito M, Gazzaz T, Wilder TJ, Vanderlaan RD, Van Arsdell GS, Honjo O, et al. Pulmonary vein stenosis: severity and location predict survival after surgical repair. J Thorac Cardiovasc Surg 151 (2016): 657-66.
- Charlagorla P, Becerra D, Patel PM, Hoyer M, Darragh RK. Congenital pulmonary vein stenosis: encouraging mid-term outcome. Pediatr Cardiol 37 (2016): 125-30.
- Balasubramanian S, Rehman M, Gauvreau K, Jenkins KJ. Bilateral disease and early age at presentation are associated with shorter survival in patients with congenital heart disease and intraluminal pulmonary vein stenosis. Congenit Heart Dis 7 (2012): 378-86.
- Un TJ, Coles JG, Konstantinov IE, Al-Radi OO, Wald RM, Guerra V, et al. Conventional and sutureless techniques for management of the pulmonary veins: evolution of indications from postrepair pulmonary vein stenosis to primary pulmonary vein anomalies 129 (2005): 167-74.
- Seale AN, Webber SA, Uemura H, Patridge J, Roughton M, Ho SY, et al. Pulmonary vein stenosis: the UK, Ireland and Sweden collaborative study. Heart (British Cardiac Society) 95 (2009): 1944-1949.
- Laux D, Rocchisani MA, Boudjemline Y, Gouton M, Bonnet D, Ovaert C. Pulmonary hypertension in the preterm infant with chronic lung disease can be caused by pulmonary vein stenosis: a must-know entity. Pediatr Cardiol 37 (2016): 313-321.
- Benjamin JT, Hamm CR, Zayek M, Eyal FG, Carlson S, Manci E. J Pediatr 154 (2009): 459-e451.
- Krishnan U, Feinstein JA, Adatia I, Austin ED, Mullen MP, Hopper RK, et al. Evaluation and Management of Pulmonary Hypertension in Children with Bronchopulmonary Dysplasia. Pediatr188 (2017): 24-34.
- Abman SH, Hansmann G, Archer SL, Ivy DD, Adatia I, Chung WK, et al. Pediatric Pulmonary Hypertension: Guidelines from the American Heart Association and American Thoracic Society. Circulation132 (2015): 2037-2099.
- Tomita H, Watanabe K, Yazaki S, Kimura K, Ono Y, Yagihara T, et al. Stent implantation and subsequent dilatation for pulmonary vein stenosis in pediatric patients: Maximizing effectiveness. Circulation 67 (2003): 187-90.
- Mendelsohn AM, Bove EL, Lupinetti FM, Crowley DC, Lloyd TR, Fedderly RT, et al. Intraoperative and percutaneous stenting of congenital pulmonary artery and vein stenosis. Circulation 88 (1993): 210-7.
