Systemic Light Chain Amyloidosis with Cardiac and Renal Involvements in a Patient with Heart Failure
Article Information
Cong Zhang, Hongxing Luo*, Yu Xu
Department of Cardiology, Zhengzhou University People’s Hospital, Zhengzhou, Henan, China
*Corresponding Author: Hongxing Luo, Department of Cardiology, Zhengzhou University People’s Hospital, Zhengzhou, Henan, China
Received: 07 April 2017; Accepted: 10 April 2017; Published: 14 April 2017
View / Download Pdf Share at FacebookAbstract
Amyloidosis is a protein-folding disorder with more than one organ infiltrated by proteinaceous deposits, the clinical syndromes and the management strategies of the disease are determined both by the organs involved and the type of amyloidosis. In light-chain amyloidosis, the vital organs often infiltrated by the deposition of immunoglobulin light chains, kidney is the most commonly involved organ [1], whereas cardiac amyloidosis carries the worst prognosis of any involved organ [2].
Keywords
Amyloidosis; Heart Failure
Article Details
Case Presentation
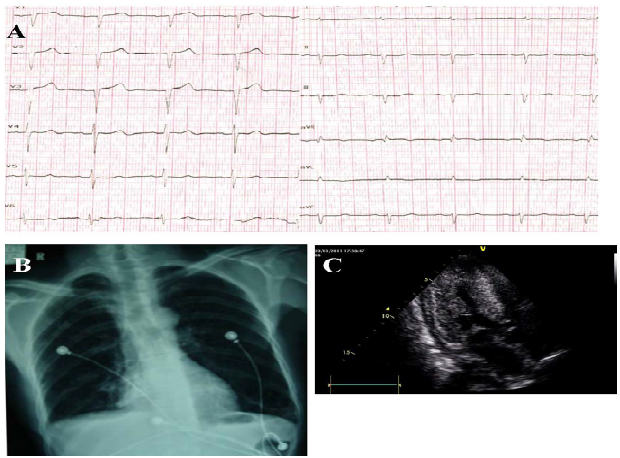
A 55-year-old man with 1-year history of exertional fatigue and lower extremity oedema was transferred to our hospital due to chest distress and dyspnoea occurring since 10 days ago. He had no history of hypertension, hyperlipidaemia or diabetes mellitus. On physical examination, the heart rate was 80 beats/minute and the blood pressure was 100/67 mmHg. Systolic murmur grade II was found on the apical area. The electrocardiogram showed low-voltage ST segments on the limb leads I and aVL, and QS-type waves on the precordial leads V1 to V3 (Figure 1A). Chest radiography showed the normal cardiac contour and bilateral lower pleura (Figure 1B).
The echocardiography revealed the left ventricular ejection fraction 59%, left ventricular end-diastolic diameter 36 mm, and interventricular septum 15 mm. (Figure 1C). The B-type natriuretic peptide was 1940 pg/ml and the serum albumin was 27.5 g/L. The 24-hour urine protein was 3.69 g. Renal biopsy was performed to show amyloid
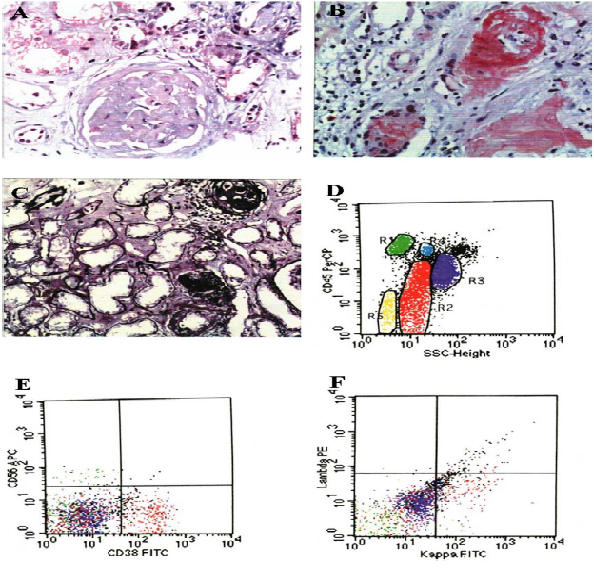
deposition in glomeruli and mesangial cells and the granular and vacuolar degenerations of tubular epithelial cells (Figure 2A). The Congo red staining and potassium permanganate method were both positive (Figure 2B, C). Bone marrow aspiration showed the plasmablasts were 0.4%, proplasmacytes were 6.8% and plasmacytes were 17.6%, indicative of multiple myeloma hyperplasia, but there was no M protein in serum or urine protein electrophoresis. Flow cytometry showed 24.36% abnormal plasma cells including 7.2% of proplasmacytes, the abnormal plasma cell expressed kappa light chain protein, which indicated multiple myeloma (Figure 2D-F). The patient was finally diagnosed with systemic light chain amyloidosis with cardiac and renal involvements. The patient was given melphalan and prednisone and the recovery was uneventful.
Figure 1: (A) Electrocardiogram shows a pseudo-infarct pattern, first-degree atrioventricular block (AVB) and low voltage on the limb leads. (B) Chest radiograph shows normal heart border, blunted bilateral rib diaphragm angle and thickened bilateral lower pleura. (C) Echocardiogram shows diffuse thickening and stippled high echo of the left ventricle with pericardial effusion.
Figure 2: (A) Hematoxylin and eosin staining shows amyloid deposition in glomeruli and mesangial cells. (B) Congo red staining is positive. (C) Potassium permanganate staining was positive. D-Flow cytometry shows R2 (red) 24.36% abnormal cells. E-Flow cytometry shows the CD38 expression was positive. F-Flow cytometry shows the positive expression of kappa light chain protein.
Source of Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
References
- Wechalekar AD, Gillmore JDand Hawkins PN. Systemic amyloidosis. The Lancet 387(2016): 2641-2654.
- Falk RH, et al. AL (Light-Chain) Cardiac Amyloidosis. Journal of the American College of Cardiology 68 (2016): 1323-1341.