Protection against Oxidation of Omega-3 Fatty Acids with Natural Antioxidants in Clove (Szygium aromaticum) Water Extracts during Storage of Sun dried Sardines (Ratrineobola argentea)
Article Information
Davis Chaula1*, Charlotte Jacobsen2, Henry Laswai1, Bernard Chove1, Anders Dalsgaard3, Robinson Mdegela4 and Grethe Hyldig2
1Department of Food Technology, Nutrition and Consumer Sciences, Sokoine University of Agriculture, Morogoro, Tanzania
2National Food Institute, Division for Food Technology, Technical University of Denmark, Søltofts Plads, Lyngby, Denmark
3Faculty of Health and Medical Sciences, Section of Food Safety and Zoonoses, Department of Veterinary and Animal Sciences, University of Copenhagen, Frederiksberg C, Denmark
4College of veterinary and medical sciences, Department of Veterinary Medicine and Public Health, Sokoine University of Agriculture, Morogoro, Tanzania
*Corresponding Author: Davis Chaula, Department of Food Technology, Nutrition and Consumer Sciences, Sokoine University of Agriculture, P.O. Box 3006, Chuo Kikuu, Morogoro, Tanzania
Received: 12 July 2019; Accepted: 29 July 2019; Published: 26 August 2019
Citation:
Davis Chaula, Charlotte Jacobsen, Henry Laswai, Bernard Chove, Anders Dalsgaard, Robinson Mdegela and Grethe Hyldig. Protection against Oxidation of Omega-3 Fatty Acids with Natural Antioxidants in Clove (Szygium aromaticum) Water Extracts during Storage of Sun dried Sardines (Ratrineobola argentea). Journal of Food Science and Nutrition Research 2 (2019): 174-187.
View / Download Pdf Share at FacebookAbstract
Omega-3 fatty acids are a family of polyunsaturated fatty acids (PUFAs) with beneficial health effects to humans if consumed in required amounts. Fatty fish species are known to be rich in marine-based omega-3 fatty acids, eicosapentaenoic acid (EPA, 20:5n-3), docosahexaenoic acid (DHA, C22:6n-3) and docosapentaenoic acid (DPA, C22:5n-3). Sardines (Rastrineobola argentea) from Lake Victoria are a good source of health promoting omega-3 fatty acids. Open sun drying is a common and traditional sardine processing and preservation method. Due to their chemical instability omega-3 fatty acids in sun dried sardines are prone to lipid oxidation during processing and subsequent storage. This study investigated the use of clove (Syzygium aromaticum) water extracts as natural antioxidants to protect omega-3 fatty acids against oxidative damage during storage of sun dried sardines. Lipid oxidation was assessed by peroxide value, volatile secondary oxidation products and fatty acid profiles. The antioxidant capacity of extracts was evaluated by total phenolic content, 1, 1-diphenyl-2-picrylhydrazyl (DPPH) radical scavenging and iron (Fe2+) chelating ability. Clove extracts resulted into significantly higher retention of total PUFAs, DHA, EPA and DPA at the end of 30 days storage period and lower concentrations of secondary lipid oxidation products. This is an evidence of their enhanced oxidative stability in the real food system due to presence of natural clove antioxidants.
Keywords
<p>Omega-3 fatty acids, Lipid oxidation, Natural antioxidants, Sardines, Lake Victoria</p>
Omega-3 fatty acids articles, Lipid oxidation articles, Natural antioxidants articles, Sardines articles, Lake Victoria articles
Omega-3 fatty acids articles Omega-3 fatty acids Research articles Omega-3 fatty acids review articles Omega-3 fatty acids PubMed articles Omega-3 fatty acids PubMed Central articles Omega-3 fatty acids 2023 articles Omega-3 fatty acids 2024 articles Omega-3 fatty acids Scopus articles Omega-3 fatty acids impact factor journals Omega-3 fatty acids Scopus journals Omega-3 fatty acids PubMed journals Omega-3 fatty acids medical journals Omega-3 fatty acids free journals Omega-3 fatty acids best journals Omega-3 fatty acids top journals Omega-3 fatty acids free medical journals Omega-3 fatty acids famous journals Omega-3 fatty acids Google Scholar indexed journals Lipid oxidation articles Lipid oxidation Research articles Lipid oxidation review articles Lipid oxidation PubMed articles Lipid oxidation PubMed Central articles Lipid oxidation 2023 articles Lipid oxidation 2024 articles Lipid oxidation Scopus articles Lipid oxidation impact factor journals Lipid oxidation Scopus journals Lipid oxidation PubMed journals Lipid oxidation medical journals Lipid oxidation free journals Lipid oxidation best journals Lipid oxidation top journals Lipid oxidation free medical journals Lipid oxidation famous journals Lipid oxidation Google Scholar indexed journals Natural antioxidants articles Natural antioxidants Research articles Natural antioxidants review articles Natural antioxidants PubMed articles Natural antioxidants PubMed Central articles Natural antioxidants 2023 articles Natural antioxidants 2024 articles Natural antioxidants Scopus articles Natural antioxidants impact factor journals Natural antioxidants Scopus journals Natural antioxidants PubMed journals Natural antioxidants medical journals Natural antioxidants free journals Natural antioxidants best journals Natural antioxidants top journals Natural antioxidants free medical journals Natural antioxidants famous journals Natural antioxidants Google Scholar indexed journals Sardines articles Sardines Research articles Sardines review articles Sardines PubMed articles Sardines PubMed Central articles Sardines 2023 articles Sardines 2024 articles Sardines Scopus articles Sardines impact factor journals Sardines Scopus journals Sardines PubMed journals Sardines medical journals Sardines free journals Sardines best journals Sardines top journals Sardines free medical journals Sardines famous journals Sardines Google Scholar indexed journals Lake Victoria articles Lake Victoria Research articles Lake Victoria review articles Lake Victoria PubMed articles Lake Victoria PubMed Central articles Lake Victoria 2023 articles Lake Victoria 2024 articles Lake Victoria Scopus articles Lake Victoria impact factor journals Lake Victoria Scopus journals Lake Victoria PubMed journals Lake Victoria medical journals Lake Victoria free journals Lake Victoria best journals Lake Victoria top journals Lake Victoria free medical journals Lake Victoria famous journals Lake Victoria Google Scholar indexed journals antioxidants articles antioxidants Research articles antioxidants review articles antioxidants PubMed articles antioxidants PubMed Central articles antioxidants 2023 articles antioxidants 2024 articles antioxidants Scopus articles antioxidants impact factor journals antioxidants Scopus journals antioxidants PubMed journals antioxidants medical journals antioxidants free journals antioxidants best journals antioxidants top journals antioxidants free medical journals antioxidants famous journals antioxidants Google Scholar indexed journals preservation articles preservation Research articles preservation review articles preservation PubMed articles preservation PubMed Central articles preservation 2023 articles preservation 2024 articles preservation Scopus articles preservation impact factor journals preservation Scopus journals preservation PubMed journals preservation medical journals preservation free journals preservation best journals preservation top journals preservation free medical journals preservation famous journals preservation Google Scholar indexed journals polyunsaturated fatty acids articles polyunsaturated fatty acids Research articles polyunsaturated fatty acids review articles polyunsaturated fatty acids PubMed articles polyunsaturated fatty acids PubMed Central articles polyunsaturated fatty acids 2023 articles polyunsaturated fatty acids 2024 articles polyunsaturated fatty acids Scopus articles polyunsaturated fatty acids impact factor journals polyunsaturated fatty acids Scopus journals polyunsaturated fatty acids PubMed journals polyunsaturated fatty acids medical journals polyunsaturated fatty acids free journals polyunsaturated fatty acids best journals polyunsaturated fatty acids top journals polyunsaturated fatty acids free medical journals polyunsaturated fatty acids famous journals polyunsaturated fatty acids Google Scholar indexed journals
Article Details
1. Introduction
Omega-3 fatty acids are a family of polyunsaturated fatty acids (PUFAs) that have a double bond on the third carbon from the omega (ω) position of the carbon chain [1]. Omega-3 fatty acids attract great attention because of their beneficial health effects [2-4]. Studies have shown that omega-3 fatty acids may play a role in prevention of cardiovascular diseases, reducing inflammation, illnesses such as high blood pressure, atherosclerosis, thrombogenesis, cancer, skin diseases and have been found to be necessary for the brain development in fetuses [5, 6, 7]. There are several sources of omega-3 fatty acids including fishes. In particular, fatty fish species are known to be rich in marine-based omega-3 fatty acids, eicosapentaenoic acid (EPA, 20:5n-3), docosahexaenoic acid (DHA, C22:6n-3) and docosapentaenoic acid (DPA, C22:5n-3).
Rastrineobola argentea (sardines, popular as dagaa in Tanzania) is one of the three leading commercial fish species of Lake Victoria, together with the Nile perch (Lates niloticus) and Nile tilapia (Oreochromis niloticus). The freshwater fatty dagaa are a richer source of omega-3 PUFAs than Lates niloticus and Oreochromis niloticus [8, 9]. Sardines have high crude protein content (47.9-58.8%), rich in iron (8.18-10.91 mg/100 g), zinc (4.07-10.25 mg/100 g) and calcium (1556.4-1866.5 mg/100 g) [10]. The stock of sardines, given by its biomass and catches, has increased steadily in the last ten years, being the largest fishery by volume with contribution of about 72% of the total catch weight in the Lake. Its annual biomass in the Lake is estimated at over 1.3 million tonnes. The rapid growth and short life cycle of sardines allow their utilization at an annual level of 70% without overexploitation [11, 12].
Despite its economic and nutritional value sardines are perceived negatively and considered as an inferior food for poor and pro-poor communities. This may be attributed to poor handling and processing technologies along the sardine value chain. The low cost, low-technology and weather dependant open sun drying is the dominant method used for processing and preservation of sardines. The method result into oxidized products [9] with characteristic off flavours, which discourage consumption of dry dagaa and limit product diversification. Chemical indicators show that lipid oxidation reactions are pronounced in sun dried dagaa with production of secondary oxidation products beyond acceptable levels [9]. Furthermore, changes in lipid contents associated with significant decrease in omega-3 fatty acids in sun dried dagaa occur during storage.
The drawback in protection of omega-3 fatty acid during processing and storage of omega-3 rich products like sardines is that PUFAs are readily oxidized in the presence of oxygen, heat, light, and metal ions [13-15]. Oxidation of PUFAs, such as EPA and DHA not only results in nutritional loss and offensive odours due to formation of volatile secondary oxidation products, but is also a safety concern [16, 17]. Commercially available synthetic antioxidants like butylated hydroxytoluene (BHT), tert-butylhydroquinone (TBHQ) and butylated hydroxyanisole (BHA) are commonly used to hinder lipid oxidation in food systems. Conversely, there are studies questioning their application in foods due to their potential carcinogenic effects and toxicity [18-20]. Due to safety concerns and increased consumer interest in natural products, efforts are directed to replacing synthetic antioxidants with ones of natural origin.
Clove is among spices with potential to protect PUFAs against oxidation because they contain anti-oxidative compounds (e.g. polyphenols and flavonoids) that may exert anti-oxidative effect by different mechanisms like scavenging of free radicals, singlet oxygen quenching, oxygen scavenging, metal chelation and inhibition of oxidizing enzymes [21, 22]. The major phenolic compounds in clove are phenolic acids such as flavonol glucosides, phenolic volatile oils and tannins, recovery of which is highly dependent on extraction conditions, type of solvent and extraction method [22-24]. Previous studies have shown that successful recovery of phenolic compounds from plant matrices by aqueous extraction depend on factors such as temperature, extraction time and solvent to solid ratio [25]. Elsewhere, the clove water extract has been found to contain substantial amounts of phenolic compounds and powerful antioxidant activity in linoleic acid emulsion [26]. Clove water extracts are reported to result in improved retention of nutritionally valued long chain PUFAs DHA, EPA and DPA during open sun and oven drying of sardines [27, 28]. The aim of the current study was to evaluate the efficacy of clove (Szygium aromaticum) water extracts in protecting omega-3 fatty acids during extended storage of sun dried sardines.
2. Materials and Methods
2.1 MaterialsFresh whole dagaa (20 Kg) were collected directly from fishermen at Kijiweni landing site at the shore of Lake Victoria, Tanzania. The dagaa were placed in ice in insulated boxes and immediately transported to the National Fish Quality Control Laboratory, Nyegezi, Mwanza for experiment. Dry clove (Szygium aromaticum) buds were obtained from a local market in Zanzibar, transported at ambient temperature to Mwanza and kept at 5 to 10°C in a refrigerator.
2.2 Preparation of clove extracts
For water extraction, 5, 10 and 20 g grounded powder (to pass through a 250 µm sieve) of clove buds were mixed with 1 L boiling water with continuous stirring to make 5, 10 and 20 gL-1 concentrations of clove extracts. The mixtures were boiled for 15 min and subsequently cooled to 0 to 5°C in a refrigerator thereafter gravity filtered to remove the particles present.
2.3 Preparation of sun dried dagaa
For each concentration of clove extract, 1 kg of dagaa (wet basis) was blanched in boiling water for 10 sec. Blanched dagaa were soaked in cooled clove extracts (1:1 w/w) for 40 min. at room temperature. After that, the fish were removed from the extracts, spread on wire mesh and sun-dried on raised platform as traditionally done by the local fish processors. Dagaa samples without clove pre-treatment were prepared in a similar way and used as control. All samples were packed in polyethylene bags each containing 200 g fish and stored in temperature controlled room at 25°C for 30 days. Analyses were done at the start (day 0) and the end of storage experiment (day 30). The room temperature was monitored by using Tinytag temperature data logger (Gemini data loggers Ltd, West Sussex, UK). For each treatment 100 g portion of whole fish was made into mince using a mixer (Moulinex Moulinette S type 643 02 210, Hamburg, Germany) for analysis.
2.4 Methods
2.4.1 Dry matter content and lipid extraction: The dry matter content for fish and clove powder samples was determined by weighing after drying a sample of approximately 2 g of homogeneous fish mince and powder at 105°C for 18 h according to the AOAC [29] and results expressed as a percentage dry matter. Lipids were extracted following the Bligh and Dyer method [30] with modifications according to Iverson et al. The sample (5 g of fish mince) was homogenized in chloroform, methanol, and water mixture (1:1:0.8 v/v) at the speed of 15000 rpm (226 g) for 90 sec using an Ultra Turrax homogenizer (T25 Homogenizer, Staufen, German). The homogenate was centrifuged at 2800 rpm (1595 g) at 18°C for 10 min using a centrifuge (Sigma 4K15, Osterode am Harz, German) to obtain the extract (Chloroform phase). The lipid content was determined by gravimetry after evaporation of chloroform and expressed as percentage of the dried fish sample.
2.4.2 Primary and secondary lipid oxidation products: Peroxide values (PV) of the lipid extracts were determined according to the method of Shantha et al. [31] based on the formation of an iron-thiocyanate complex. The colored complex was measured by spectrophotometer (Shimadzu UV1800, Shimadzu Scientific Instruments, Columbia, MD) at 500 nm. The analysis was done in duplicate, and the results were expressed in millequivalent peroxides/Kg oil (meq O2/Kg oil). The volatile compounds from fish mincewere collected using the dynamic headspace technique. The procedure was carried out using 1 g of fish mince in which 30 mg of internal standard, 4-methyl-1-pentanol were added and mixed with 15 mL of distilled water. The volatiles were collected in Tenax GR tubes at 37°C by purging with nitrogen for 30 min at 150 mL/min. The tubes were flushed with nitrogen at 50 mL/min for 20 min to remove water.
The trapped volatiles were desorbed from the Tenax tubes by heat (200°C) using an automatic thermal desorber (ATD-400, PerkinElmer, Norwalk, CT), cryofocused on a cold trap (−30°C), released again at 220°C, and led to a GC an Agilent 5890IIA model (Palo Alto, CA, USA) equipped with a HP 5972 mass selective detector. Separation was done on a DB1701 column (30 m × ID 0.25 mm × 0.5 μm film thickness, (J&W Scientific, Folsom, CA). The carrier gas used was helium at flow rate of 1.3 mL/min. The oven temperature was rising by 2.0°C/min from initial temperature of 45 to 80°C followed by an increase of 3.0°C/min to 150°C and finally increased by 12.0°C/min to 240°C. The individual compounds were identified by MS-library searches and addition of the internal standard. Quantification was done through calibration curve made by adding the standard directly on the Tenax tubes as described by Hartvigsen et al. [32]. For the quantification, a stock solution of 19 volatiles was prepared and a calibration curve was conducted in a range from 0 to 1.2 mg/g. The analysis was carried out in triplicate.
2.4.3 Free fatty acids and fatty acid composition (Fatty Acid Methyl Esters, FAME): Free fatty acids (FFAs) content was determined by acidometric titration of the lipid extract using NaOH (0.1 M). The FFAs content was calculated as oleic acid according to the AOCS [33] and the results were reported as % oleic acid. The fatty acid composition of the oil phase was determined after fatty acid methylation and analysis by GC-FID according to the American Oil Chemists’ Society (AOCS) official method; Ce 1i-07 [34] with some modification as follows. Approximately 1 g of extract was weighed in a methylation glass tube and evaporated to dryness under a gentle stream of nitrogen. Thereafter, 100 µL of internal standard solution (2% w/v C23:0 in heptane), 200 µL of heptane including 0.01% w/v butylated hydoxy toluene (BHT) as antioxidant, 100 µL of toluene and 1 mL of boron trifluoride in methanol (BF3-MeOH) was added.
Samples were mixed and methylated in the microwave oven (Microwave 3000 SOLV, Anton Paar) for 10 min at 100°C and power of 500Wand then cooled down for 5 min. Then, 1 mL of saturated salt water (NaCl) and 0.7 mL of heptane with BHT were added. After the separation of heptane, the upper phase of the sample (around 0.7 mL) was transferred into vials. Samples were analyzed by gas chromatography system (HP-5890 A, Agilent Technologies, Santa Clara, CA, USA). Fatty acid methyl esters were separated and detected by the GC column Agilent DB-wax (10 m × 100 µm × 0.1 µm), from Agilent Technologies (CA, USA). The carrier gas was helium with a flow rate of 0.38 mL/min and an inlet pressure of 51psi. The oven temperature program for separation was from 160 to 200°C, then from 200 to 220°C and from 220 to 240°C at 10.6°C /min. All analyses were done in duplicate. The result of each fatty acid was expressed as g fatty acid/100 g lipid.
2.4.4 Antioxidant Activity of Clove Water Extracts:
2.4.4.1 Total phenolic content: The total phenolic compounds of the extracts were determined using Folin–Ciocalteu reagent by a procedure described by Farvin and Jacobsen [35] in which gallic acid was used as a standard. The standard curve was prepared in distilled water at a concentration range of 0-125 µg/mL. The original extracts were diluted with water as necessary to fit within the standard curve. The absorbance was read at 725 nm using UV-vis spectrophotometer and results reported in µg gallic acid equivalent (µg GAE)/mL of clove water extracts. All measurements were performed in duplicate.
2.4.4.2 Free radical scavenging ability: The free radical scavenging activities of clove water extracts were measured by utilizing the stable radical, 1,1-diphenyl-2-picryl-hydrazil (DPPH) as described by Yang et al. [36]. The solutions of prepared extracts were diluted with water (1:1 v/v). Diluted solutions (100 µL) were added to the microplate and mixed with 100 µL of 0.1 mM DPPH in ethanol (96%). The mixtures were shaken vigorously and maintained for 30 min at ambient temperature in the dark. The absorbance of mixtures and the control (100 µL DPPH solution + 100 µL BHT) was measured at 517 nm against a reagent blank by using a UV-V is spectrophotometer. The scavenging activity was calculated as inhibition percent by using the following equation:
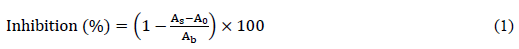
Where As is the absorbance of DPPH after reaction with antioxidant, A0 is the absorbance of antioxidant and ethanol (blank) and Ab is the absorbance of water and DPPH (blind).
2.4.4.3 Iron (Fe2+) chelating ability: The ferrous ion chelating activity of clove extracts was measured as described by Farvin et al. [37] with 20 µL of 0.5 mM ferrous chloride and 20 µL of 2.5 mM ferrozin being mixed with 100 µL of clove extracts. The mixture was allowed to equilibrate in the darkness at room temperature for 10 min before measuring the absorbance. The decrease in the absorbance at 562 nm of the iron (II)-ferrozin complex was measured. EDTA was used as the positive control and the ability of the extracts to chelate Fe2+ was calculated using the equation:
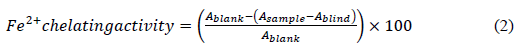
Ablank is the absorbance of blank (only iron chloride and Ferrozin), Asample is the absorbance of sample and Ablind is the absorbance of blind (only antioxidant).
2.5 Statistical analysis
Data were analyzed using IBM SPSS (SPSS for Windows Version 20.0, 2013, IBM, Bethesda, MD, USA). Data were reported as mean ± standard deviation. Differences between means were determined using one-way analysis of variance (one-way ANOVA) with Tukey’s HSD post hoc test, according to the equal variance of different groups. A p-value <0.05 was considered statistically significant.
3. Results and Discussion
3.1 Antioxidant activity of clove and seaweed water extractsThe clove (Szygium aromaticum) water extracts had total phenolic content in the range 18.18 to 28.75 µgGAE/mL (Table 1). The 20 gL-1 extract had significantly higher total phenolic content than 5 and 10 gL- 1. This observation concurs with previous studies which showed that recovery of phenolic compounds from plant matrices by aqueous extraction depends on temperature, extraction time and solvent to solid ratio [25]. The DPPH of extracts was in the range 93 to 95% inhibition. The lack of linear relationship between total phenolic content and DPPH of clove extracts suggests that compounds other than phenolics (e.g. flavonoids) might have contribution to free radical scavenging activity of clove extracts. The DPPH decreased from 95.59 to 94.34% when the amounts of clove extracted in one litre of hot water was increased from 10 to 20 g. This could be due to decrease in extraction efficiency of clove phenolics in boiling water at concentration above 10 g/L as reported in another study [28]. Elsewhere, the clove water extract has been found to contain substantial amounts of phenolic compounds and powerful antioxidant activity in linoleic acid emulsion [26].
|
Extracts (g/L) |
Total phenolic content (µgGAE/mL) |
DPPH scavenging activity (% inhibition) |
Fe2+ ion chelating activity (%) |
|
CL 5 |
18.18a ± 1.29 |
93.33u ± 0.21 |
14.74n ± 4.60 |
|
CL 10 |
25.94b ± 2.62 |
95.59v ± 1.44 |
20.87p ± 5.98 |
|
CL 20 |
28.75c ± 1.35 |
94.34w ± 0.38 |
22.24q ± 4.08 |
CL: Clove, GAE: gallic acid, 5, 10 and 20: grams of seaweed or clove extracted in 1 L of boiling water. Means marked with different letters in a column statistically significant (p<0.05), n=3
Table 1: Antioxidant capacity of different doses of clove water extracts.
The Fe2+ chelating activity increased (though not linearly) with the amount of clove powder extracted. The dependence of iron chelating capacity on concentration of clove water extracts and type of solvent used was reported by Gulcin et al. [26]. Essential oils of clove have been tested in omega-6 and omega-3 fatty acids enriched food supplements and found to have high iron-chelating properties and higher hydrogen donating power than the standard antioxidants BHT and α-tocopherol.
3.2 Fat, free fatty acids and dry matter content
The dry matter content of clove was 86.40%, whereas the mean dry matter content of clove pre-treated sardines was 92.19% and there was no significant difference between pre-treated and control samples (Table 2). For all samples the fat content decreased significantly after 30 days of storage at 25°C. The substantial decrease in fat content of fish during processing and storage has been associated with oxidative changes in fish lipids.
Owaga et al. previously reported a significant difference (P<0.05) in the total fat content of fresh sardine fish (14.8% dry weight basis [dwb]) and sun-dried sardine fish (13.9% dwb) during the sun-drying process which may result from rapid lipid oxidation. Elsewhere, another study reported 82.64 and 56.68% decrease in lipid content during the 21 days of chilled and frozen storage of fish (Mystus seenghala) muscles respectively.
|
Sample
|
DM (%)
|
Fat content (%) |
Free fatty acids (%) |
PV (meq.O2/Kg oil) |
|||
|
Day 0 |
Day 30 |
Day 0 |
Day 30 |
Day 0 |
Day 30 |
||
|
POO |
92.27x ± 0.64 |
17.30a ± 0.19 |
16.18d ± 0.28 |
9.50g ± 0.96 |
12.57k ± 1.15 |
20.32p ± 0.26 |
11.81t ± 1.38 |
|
CL 5 |
91.63x ± 0.92 |
17.65b ± 0.13 |
16.76e ± 0.73 |
12.84h ± 2.06 |
15.49l ± 0.35 |
11.54q ± 0.11 |
6.29u ± 0.67 |
|
CL 10 |
92.61x ± 0.48 |
17.52b ± 0.43 |
16.72e ± 0.77 |
11.27i ± 2.01 |
13.73m ± 1.49 |
8.56r ± 1.97 |
5.74v ± 0.41 |
|
CL 20 |
92.34x ± 0.25 |
16.28c ± 0.10 |
16.05f ± 0.05 |
12.08j ± 0.20 |
14.68n ± 0.58 |
8.28s ± 0.28 |
5.01w ± 0.14 |
POO: Control sample; CL: Clove; GAE: gallic acid; 5, 10 and 20: grams of clove powder extracted in 1 L of boiling water. Means marked with different letters in a column statistically significant, n=3.
Table 2: Dry matter (DM), fat, free fatty acid and hydroperoxide (PV) content of sun dried sardines stored at 25°C for 30 days.
Free fatty acid levels increased by 32.32% in the control sample and by 20.64, 21. 83 and 21.52% in samples treated with 5, 10 and 20 g/L clove extracts, respectively at the end of storage experiment. Increased levels of free fatty acids suggest activity of lipase and phospoholipase found in fish or which could be produced by certain microorganisms therein contributing to lipolytic breakdown of fish lipids. Lower percentage increase of free fatty acids in clove treated samples than the control sample is indicative of antimicrobial activity of clove that might have suppressed lipolytic enzymes producing microorganisms in treated samples. For ages clove has been known to have strong antimicrobial activity [38].
3.3 Primary and secondary lipid oxidation products
Both, at the start and end of storage experiment the control sample had significantly higher levels of hyroperoxides (indicated by PV) than the clove pre-treated samples (Table 3). The PV among clove treated samples decreased as the amount of clove extracted in 1 L of water increased which shows the dependence of antioxidant activity on concentration of clove extracts. Concentrations of volatile secondary oxidation products after 30 days of storage are presented in Table 3. The control samples had significantly higher concentrations of individual volatile compounds than pre-treated samples, indicating that oxidation of PUFAs was more pronounced in untreated sardines than in clove treated samples. From Table 3 it can be seen that concentrations of compounds such as 1-penten-3-ol, t-2-penten-1-ol, Hexanal and 1-octene-3-ol in clove treated samples were approximately 50% less than in the control (untreated). This observation suggests that phenolic compounds in clove extracts were still active as anti-oxidants throughout the storage period. The compounds 1-penten-3-ol, 4-heptanal and t, t-2, 4-heptadienal are recognized as decomposition products of EPA and DHA [39].
|
Compound |
POO |
CL 5 |
CL 10 |
CL 20 |
|
4-heptenal |
86.19a ± 13.22 |
62.97b ± 0.88 |
72.50c ± 3.99 |
49.83d ± 1.80 |
|
2-heptanal |
34.55e ± 10.97 |
6.66f ± 0.54 |
5.35g ± 0.04 |
ND |
|
2-methyl furan |
ND |
ND |
ND |
ND |
|
2,4-heptadienal |
23.69h ± 1.16 |
22.78i ± 0.44 |
17.01j ± 2.37 |
11.51k ± 0.74 |
|
2-pentanal |
87.99l ± 22.34 |
53.34m ± 3.45 |
48.25n ± 5.86 |
28.99o ± 1.28 |
|
1-penten-3-ol |
1403.41p ± 292.00 |
641.01q ± 64.82 |
875.85r ± 80.89 |
509.17s ± 14.53 |
|
Benzaldehyde |
213.50 ± 25.49 |
147.17 ± 5.97 |
273.68 ± 12.32 |
150.02 ± 3.61 |
|
Butanal |
269.50t ± 74.33 |
132.42u ± 9.84 |
205.53v ± 13.02 |
138.78w ± 8.92 |
|
2-methyl butanal |
366.00x ± 12.19 |
280.53y ± 28.41 |
543.29z ± 38.96 |
399.47ab ± 8.49 |
|
t-2-penten-1-ol |
838.89ac ± 62.68 |
339.68ad ± 2.35 |
429.17ae ± 23.98 |
258.95af ± 3.85 |
|
Hexanal |
1827.77ag ± 373.51 |
725.64ah ± 2.97 |
917.40aj ± 54.68 |
536.73ak ± 8.91 |
|
Pentanal |
893.97al ± 27.19 |
509.12am ± 34.47 |
682.98an ± 37.35 |
495.52ap ± 21.23 |
|
1-octene-3-ol |
642.77aq ± 60.98 |
258.24ar ± 6.65 |
299.35as ± 19.06 |
209.03at ± 7.59 |
|
3-methyl butanal |
1041.67au ± 293.11 |
737.71av ± 72.07 |
1321.13aw ± 67.42 |
997.02ax ± 24.94 |
|
Heptanal |
508.62ay ± 97.63 |
228.47az ± 6.11 |
231.78bc ± 16.34 |
179.00bd ± 8.21 |
Values are expressed in mean ± standard deviation (n=3). CL: clove, 5, 10 and 20: grams of clove extracted in 1 L of boiling water, ND: not detected. POO: Control sample. Means marked with different letters in a row are statistically significant (p<0.05).
Table 3: Concentration of volatile compounds (ng/g fish mince) in sun dried sardines pre-treated with extracts and stored at 25°C for 30 days.
Lower concentrations of such compounds in pre-treated samples imply that the nutritionally valued PUFAs DHA and EPA were protected against oxidative damage during storage. Clove water extracts have been reported to retard lipid oxidation during sun-dry processing of sardines resulting into lower concentrations of volatile compounds in dry sardines [27]. Phenolic compounds are known to exert anti-oxidative effect by different mechanisms such as scavenging of free radicals, singlet oxygen quenching, oxygen scavenging, metal chelation and inhibition of oxidizing enzymes [21, 22]. In literature, it has been reported that use of the whole spices and herbs or their extracts with strong antioxidant activity [40], can control lipid oxidation in muscle food such as mullet fish, frozen chub mackerel and smoked rainbow trout [41].
3.4 Polyunsaturated fatty acids
After 30 days of storage the retention of total PUFAs was significantly higher in clove pre-treated samples than the control (Table 4). The total PUFAs ranged from 29.58 to 30.61% in clove pre-treated sardines and it was 25.95% in the control (untreated). Correspondingly, pre-treated samples retained higher amounts of total omega-3 fatty acids (21.36-22.18%) than the control sample which had 17.31%. The same trend was observed for contents of individual nutritionally valued omega-3 PUFAs, DHA (13.22-13.38%), DPA (1.95-2.11%) and EPA (5.21-5.72%) in pre-treated against 8.71, 1.37 and 3.62% respectively in the control. In previous study, the total PUFAs in untreated sun dried sardines decreased from 29.29 to 11.49% and DHA was beyond detectable levels after 21 days of storage at ambient temperatures.
|
PUFAs |
POO |
CL5 |
CL10 |
CL20 |
|
16:2 (n-4) |
0.51 ± 0.004 |
0.65 ± 0.02 |
0.58 ± 0.02 |
0.62 ± 0.01 |
|
16:3 (n-4) |
2.52 ± 0.02 |
2.41 ± 0.08 |
2.40 ± 0.04 |
2.32 ± 0.09 |
|
18:3 (n-4) |
2.84 ± 0.04 |
3.65 ± 0.02 |
3.64 ± 0.02 |
3.65 ± 0.17 |
|
Total (n-4) |
5.87a ± 1.27 |
6.71b ± 1.53 |
6.63b ± 1.54 |
6.58b ± 1.52 |
|
18:3 (n-3) |
0.33 ± 0.04 |
0.45 ± 0.02 |
0.42 ± 0.01 |
0.44 ± 0.04 |
|
18:4 (n-3) |
2.84 ± 0.01 |
0.05 ± 0.001 |
ND |
ND |
|
20:3 (n-3) |
0.43 ± 0.01 |
0.28 ± 0.03 |
0.54 ± 0.02 |
0.57 ± 0.02 |
|
20:5 (n-3)EPA |
3.62 ± 0.01 |
5.72 ± 0.10 |
5.21 ± 0.03 |
5.69 ± 0.03 |
|
22:5 (n-3)DPA |
1.37 ± 0.03 |
2.11 ± 0.01 |
1.95 ± 0.03 |
2.10 ± 0.03 |
|
22:6 (n-3)DHA |
8.71 ± 0.01 |
13.22 ± 0.02 |
12.25 ± 0.02 |
13.38 ± 0.04 |
|
Total (n-3) |
17.31c ± 3.76 |
21.83d ± 4.84 |
20.36e ± 4.51 |
22.18f ± 4.93 |
|
18:2 (n-6) |
0.12 ± 0.01 |
0.13 ± 0.02 |
0.16 ± 0.02 |
0.18 ± 0.02 |
|
18:3 (n-6) |
0.34 ± 0.01 |
0.33 ± 0.001 |
0.32 ± 0.02 |
0.38 ± 0.08 |
|
20:2 (n-6) |
0.21 ± 0.01 |
0.25 ± 0.01 |
0.24 ± 0.001 |
0.25 ± 0.01 |
|
20:4 (n-6) |
2.11 ± 0.04 |
1.36 ± 0.07 |
2.68 ± 0.01 |
2.82 ± 0.04 |
|
Total (n-6) |
2.77g ± 0.95 |
2.07h ± 0.57 |
3.41i ± 1.23 |
3.64j ± 1.28 |
|
Total PUFAs |
25.95k ± 2.36 |
30.61l ± 3.67 |
30.41m ± 3.38 |
29.58n ± 3.95 |
*Fish lipid was extracted from whole fish. Values are expressed in mean ± standard deviation (n=3). PUFAs, polyunsaturated fatty acids; CL; sample treated with clove, 5, 10 and 20: grams of clove powder extracted in 1 L of boiling water, POO: Control sample, ND, not detected; Means marked with different letters in the same row are statistically significant (p<0.05).
Table 4: Polyunsaturated fatty acid content (% fatty acid of total fatty acids)* in sun dried sardine pre treated with clove water extracts and stored at 25°C for 30 days.
Higher proportion of DHA, DPA and EPA in lipid fractions of clove treated sardine after 30 days of storage is an evidence of their enhanced oxidative stability in the presence of natural antioxidants in clove extracts. Of the three clove extract concentrations, 20 g/L is the best as it resulted in retention of higher amounts of both total omega-3 and omega-6 fatty acids. Antioxidant activity of clove water extracts is attributed to the presence of phenolic compounds with strong hydrogen donating ability, metal chelating ability and free radicals scavenging activity. Use of spices like clove as a natural antioxidant to protect lipids against oxidation in meat and fish oil has been demonstrated. Protective effect of clove water extracts against oxidation of polyunsaturated omega-3 fatty acids during oven and sun drying of sardines has been reported [27, 28]. Clove oil and clove water extracts are also known to have as strong peroxidation inhibitory effect as ethanol extract in linoleic acid emulsion [26]. Nevertheless, the major phenolic compounds in clove are phenolic acids such as flavonol glucosides, phenolic volatile oils and tannins, recovery of which is highly dependent on extraction conditions, type of solvent used and extraction method [22-24].
4. Conclusion
The present study evaluated the protective effect of natural antioxidants in clove water extracts against oxidation of omega-3 fatty acids in real food system (sun dried sardines) after 30 days storage at 25°C. Generally, clove treated sardines retained significantly higher amounts of total polyunsaturated fatty acids at the end of storage experiment. The pre-treatment also resulted into lower amounts of volatile secondary lipid oxidation products. It was found that the chemically unstable omega-3 fatty acids DHA, EPA and DPA were retained in products, an evidence of their enhanced oxidative stability in presence of natural clove antioxidants in extracts. The small size and weight of sardine fish might have been a key to successful protection of omega-3 fatty acids. These findings would be of interest during incorporation of sardines into other food product formulation at industrial scale for product diversification. Nevertheless, for product diversification further investigation on possible alterations in products’ sensory acceptability are needed.
Acknowledgements
The authors acknowledge for the financial support provided by the Danish Development Argency (DANIDA) through the project “Innovations and Markets for Lake Victoria Fisheries (IMLAF) DFC 14 –P01 –TAN)”. National Food Institute, Technical University of Denmark is acknowledged for granting permission to carryout laboratory analyses and for technical support during laboratory work. The authors acknowledge Inge Holmberg, Rie Sørensen, Lis Berner, Thi Thu Trung Vu for their technical support and day to day assistance during laboratory analyses.
References
- Minihane AM, Givens DI, Gibbs RA. Health benefits of organic food. In Eds.: Givens I, Baxter S, Minihane AM, et al. Effects of the Environment. Commonwealth for Agricultural Bureau International, Oxford, United Kingdom (2008): 19-49.
- Kapoor R and Patil UK. Importance and production of omega-3 fatty acids from natural sources. International Food Research Journal 18 (2011): 493-499.
- Ramadeen A, Connelly KA, Leong-Poi H, et al. Docosahexaenoic acid, but not eicosapentaenoic acid, supplementation reduces vulnerability to atrial fibrillation. Circulation Arrhythmia and Electrophysiololgy 5 (2012): 978-983.
- Virtanen JK, Siscovick DS, Lemaitre RN, et al. Circulating omega-3 polyunsaturated fatty acids and subclinical brain abnormalities on MRI in older adults: The cardiovascular health study. Journal of the American Heart Association 2 (2013): 1-11.
- Finley JW and Shahidi F. The chemistry, processing, and health benefits of highly unsaturated fatty acids. An overview. American Chemical Society Symposium Series 788 (2001): 2-11.
- Sidhu KS. Health benefits and potential risks related to consumption of fish or fish oil. Regulatory Toxicology and Pharmacology 38 (2003): 336-344.
- Gladyshev MI, Lepskaya EV and Sushchik NN. Comparison of polyunsaturated fatty acids content in fillets of anadromous and locked Sockeye salmon Oncorhynchusnerka. Journal of Food Science 77 (2012): 1303-1310.
- Robert A, Mfilinge P, Limbu SM, et al. Fatty acid composition and levels of selected polyunsaturated fatty acids in four commercial important freshwater fish species from Lake Victoria, Tanzania. Journal of Lipids (2014): 1-7.
- Chaula D, Laswai L, Chove B, et al. Fatty acid profiles and lipid oxidation status of sun dried, deep fried and smoked sardine (Rastrineobolaargentea) from Lake Victoria, Tanzania. Journal of Aquatic Food Product Technology 28 (2019a): 165-176.
- Kabahenda MK, Amega R, Okalany E, et al. Protein and Micronutrient Composition of Low-Value Fish Products Commonly Marketed in the Lake Victoria Region. World Journal of Agricultural Sciences 7 (2011): 521-526.
- Damien L and Luomba J. Dagaa value chain analysis and proposal for trade development. IOC-Smart Fish Programme Technical Document (2011).
- Moenieba I. The humble sardine (small pelagics): fish as food or fodder. Agriculture and Food Security 5 (2016): 1-14.
- Azhar KF and Nisa K. Lipids and their oxidation in seafood. Journal of Chemistry Society Pakistan 28 (2006): 298-305.
- Maqsood S and Benjakul S. Effect of bleeding on lipid oxidation and quality changes of Asian seabass (Latescalcarifer) muscle during iced storage. Food Chemistry 124 (2011): 459-467.
- Maqsood S, Benjakul S and Kamal-Eldin A. Haemoglobin mediated lipid oxidation in the fish muscle: A review. Trends in Food Science and Technology 28 (2012): 33-43.
- Jacobsen C and Nielsen NS. Optimization of oxidative stability of omega-3 enriched foods. In Eds.: Breivik H. Long-Chain Omega-3 Speciality Oils. The Oily Press Bridgewater, United Kingdom (2007): 197-217.
- Hermund DB, Karadag A, Andersen U, et al. Oxidative stability of granola bars enriched with multilayered fish oil emulsion in the presence of novel brown seaweed based antioxidants. Journal of Agricultural and Food Chemistry 64 (2016): 8359-8368.
- Branen A. Toxicology and biochemistry of butylatedhydroxyanisole and butylatedhydroxytolulene. Journal of American Oil Chemistry Society 52 (1975): 59-63.
- Lindenschmidt R, Tryka A, Goad M, et al. The effects of dietary butylatedhydroxytoluene on liver and colon tumor development in mice. Toxicology 38 (1986): 151-160.
- Zheng W and Wang SY. Antioxidant activity and phenolic compounds in selected herbs. Journal of Agricultural Food Chemistry 49 (2001): 5165-5170.
- Shobana S and Akhilender NK. Antioxidant activity of selected Indian spices.Prostaglandins, Leukotrienes and Essential Fatty Acids 62 (2000): 107-110.
- Dudonné S, Vitrac X, Coutiere P, et al. Comparative study of antioxidant properties and total phenolic content of 30 plant extracts of industrial interest using 1,1-diphenyl-2-picryl-hydrazil (DPPH) and oxygen radical absorbance capacity assays. Journal of Agricultural and Food Chemistry 57 (2009): 1768-1774.
- Wu X, Beecher GR, Holden JM, et al. Lipophilic and hydrophilic antioxidant capacities of common foods in the United States. Journal of Agricultural and Food Chemistry 52 (2004): 4026-4037.
- Shan B, Cai YZ, Sun M, et al. Antioxidant capacity of 26 spice extracts and characterization of their phenolic constituents.Journal of Agricultural and Food Chemistry 53 (2005): 7749-7759.
- Cam M and Aaby K.Optimization of extraction of apple pomacephenolics with water by response surface methodology. Journal of Agricultural and Food Chemistry 58 (2010): 9103-9111.
- Gulcin I, Sat IG, Beydemir S, et al. Comparison of antioxidant activity of clove (Eugenia caryophylataThunb) buds and lavender (Lavandulastoechas L.). Food Chemistry 87 (2004): 393-400.
- Chaula D, Laswai H, Chove B, et al. Effect of clove (Syzygium aromaticum) and seaweed (Kappapycusalvarezii) water extracts pre-treatment on lipid oxidation in sun dried sardines (Rastrineobola argentea) from Lake Victoria, Tanzania. Journal of Food Science and Nutrition 7 (2019b): 1406-1416.
- Slavin M, Dong M and Gewa C. Effect of clove extract pre-treatment and drying conditions on lipid oxidation and sensory discrimination of dried omena (Rastrineobolaargentea) fish. International Journal of Food Science and Technology 51 (2016): 2376-2385.
- Official Methods of Analysis (19th Edn.), Association of Official Analytical Chemists, Gaithersburg, Maryland, United States of America (2012).
- Bligh EG and Dyer WJ. A rapid method of total lipid extraction and purification.Canadian Journal of Biochemistry and Physiology 37 (1959): 911917.
- Shantha NC and Decker EA. Rapid, Sensitive, Iron-based spectrophotometric methods for determination of peroxide values of food lipids. Journal of Association of Official Analytical Chemists 77 (1994): 421-424.
- Vartvigsen K, Lund P, Hansen LF, et al. Dynamic headspace gas chromatography/mass spectrometry characterization of volatiles produced in fish oil enriched mayonnaise during storage. Journal of Agricultural and Food Chemistry 48 (2000): 4858-4867.
- Official Method Ca5a-40: Free Fatty Acids.Official Methods and Recommended Practices of the American Oil Chemists’ Society. Champaign, United States of America (1998).
- Official Method Ce 1i-07: Saturated, cis-Monounsaturated, and cis-Polyunsaturated Fatty Acids in Marine and Other Oils Containing Long Chain Polyunsaturated Fatty Acids. Official Methods and Recommended Practices of the American Oil Chemists’ Society, Urbana, United States of America (2009).
- Farvin KS and Jacobsen C. Phenolic compounds and antioxidant activities of selected species of seaweeds from Danish coast. Food Chemistry 138 (2013): 1670-1681.
- Yang J, Guo J and Yuan J. In vitro antioxidant properties of rutin.Food Science and Technology 41 (2008): 1060-1066.
- Farvin KS, Baron CP, Nielsen NS, et al. Antioxidant activity of yoghurt peptides: Part 1-in vitro assays and evaluation in ω-3 enriched milk. Food Chemistry 123 (2010): 1081-1089.
- Yu-Feng W, Jin-Xia J, You-Qiu T, et al. Antifungal effects of clove oil microcapsule on meat products. Food Science and Technology 89 (2018): 604-609.
- Venkateshwarlu G, Let MB, Meyer AS, et al. Chemical and Olfactometric Characterization of Volatile Flavour Compounds in a Fish Oil Enriched Milk Emulsion. Journal of Agricultural and Food Chemistry 52 (2004): 311-317.
- Gachkar L, Yadegari D, Rezaei MB, et al. Chemical and biological characteristics of Cuminumcyminum and Rosmarinusofficinalis essential oils. Food Chemistry 102 (2007): 898-904.
- Emir CO, Patir B and Yilmaz O. Protective effect of essential oils on the shelf life of smoked and vacuum packed rainbow trout (OncorhynchusmykissW.1792) fillets. Journal of Food Science Technology 51 (2014): 2741-2747.
