Prevalence and Associated Risk Factors of Preterm and Post-term Births in Northern Ghana: A Retrospective Study in Savelugu Municipality
Article Information
Silas Adjei-Gyamfi1,2*, Abigail Asirifi3, Hirotsugu Aiga1
1School of Tropical Medicine and Global Health, Nagasaki University, Nagasaki, Japan
2Savelugu Municipal Hospital, Ghana Health Service, Savelugu, Northern Region, Ghana
3Department of Midwifery, Garden City University College, Kumasi, Ashanti Region, Ghana
*Corresponding Author: Silas Adjei-Gyamfi, Savelugu Municipal Hospital, Ghana Health Service, Savelugu, Northern Region, Ghana.
Received: 04 November 2023; Accepted: 16 November 2023; Published: 01 December 2023
Citation: Silas Adjei-Gyamfi, Abigail Asirifi, Hirotsugu Aiga. Prevalence and Associated Risk Factors of Preterm and Post-term Births in Northern Ghana: A Retrospective Study in Savelugu Municipality. Journal of Pediatrics, Perinatology and Child Health. 7 (2023): 235-248.
View / Download Pdf Share at FacebookAbstract
Introduction: Preterm and post-term births are prominent leading causes of neonatal mortalities and significant contributors to long-term adverse health outcomes. Although preterm and post-term births are disproportionately rampant in most parts of Ghana, the magnitude and underlying predictors are not well comprehended which necessitates more evidence for appropriate interventions. This study assessed the prevalence and identified vital risk factors of preterm and post-term births in Northern Ghana.
Methods: This study is a retrospective cross-sectional design conducted on 356 postnatal mothers from February to March 2022 in Savelugu Municipality of Northern Region, Ghana. Anthropometric, clinical, obstetric, and sociodemographic data were collected from antenatal records using structured questionnaires. Multinomial logistic regression was used to identify independent factors of preterm and post-term births at 95% confidence interval.
Results: Prevalence of preterm and post-term births were 19.4% and 6.5% respectively. Anaemia in the first trimester of pregnancy (AOR: 2.205; 95%CI: 1.011−4.809), non-use of insecticide-treated bed nets (ITNs) during pregnancy (AOR: 1.979; 95%CI: 0.999−3.920), maternal age less than 20 years (AOR: 12.95; 95%CI: 2.977−56.34), and mothers with junior high school education (AOR: 0.225; 95%CI: 0.065−0.797) were independently associated with preterm births. Predictors for post-term births were macrosomia deliveries (AOR: 8.128; 95%CI: 1.777−37.18) and mothers with senior high school education (AOR: 0.001; 95%CI: 0.0001−0.125).
Conclusion: Preterm births are very prevalent, while post-term births are becoming crucial in the municipality. These predictors (gestational anaemia, ITNs use, teenagers, maternal education, and macrosomic births) of preterm and post-term births are modifiable and preventable. Therefore, interventions should be targeted at intensified community education on nutrition and lifestyle modifications, in addition to vigorous promotion of girls’ child education through parental empowerment.
Keywords
<p>Northern Ghana; Preterm births; Prevalence; Post-term births; Risk factors; Savelugu municipal</p>
Preterm births articles Preterm births Research articles Preterm births review articles Preterm births PubMed articles Preterm births PubMed Central articles Preterm births 2023 articles Preterm births 2024 articles Preterm births Scopus articles Preterm births impact factor journals Preterm births Scopus journals Preterm births PubMed journals Preterm births medical journals Preterm births free journals Preterm births best journals Preterm births top journals Preterm births free medical journals Preterm births famous journals Preterm births Google Scholar indexed journals Post-term births articles Post-term births Research articles Post-term births review articles Post-term births PubMed articles Post-term births PubMed Central articles Post-term births 2023 articles Post-term births 2024 articles Post-term births Scopus articles Post-term births impact factor journals Post-term births Scopus journals Post-term births PubMed journals Post-term births medical journals Post-term births free journals Post-term births best journals Post-term births top journals Post-term births free medical journals Post-term births famous journals Post-term births Google Scholar indexed journals Midwifery articles Midwifery Research articles Midwifery review articles Midwifery PubMed articles Midwifery PubMed Central articles Midwifery 2023 articles Midwifery 2024 articles Midwifery Scopus articles Midwifery impact factor journals Midwifery Scopus journals Midwifery PubMed journals Midwifery medical journals Midwifery free journals Midwifery best journals Midwifery top journals Midwifery free medical journals Midwifery famous journals Midwifery Google Scholar indexed journals Gestational weight gain articles Gestational weight gain Research articles Gestational weight gain review articles Gestational weight gain PubMed articles Gestational weight gain PubMed Central articles Gestational weight gain 2023 articles Gestational weight gain 2024 articles Gestational weight gain Scopus articles Gestational weight gain impact factor journals Gestational weight gain Scopus journals Gestational weight gain PubMed journals Gestational weight gain medical journals Gestational weight gain free journals Gestational weight gain best journals Gestational weight gain top journals Gestational weight gain free medical journals Gestational weight gain famous journals Gestational weight gain Google Scholar indexed journals Postnatal care articles Postnatal care Research articles Postnatal care review articles Postnatal care PubMed articles Postnatal care PubMed Central articles Postnatal care 2023 articles Postnatal care 2024 articles Postnatal care Scopus articles Postnatal care impact factor journals Postnatal care Scopus journals Postnatal care PubMed journals Postnatal care medical journals Postnatal care free journals Postnatal care best journals Postnatal care top journals Postnatal care free medical journals Postnatal care famous journals Postnatal care Google Scholar indexed journals Chronic morbidities articles Chronic morbidities Research articles Chronic morbidities review articles Chronic morbidities PubMed articles Chronic morbidities PubMed Central articles Chronic morbidities 2023 articles Chronic morbidities 2024 articles Chronic morbidities Scopus articles Chronic morbidities impact factor journals Chronic morbidities Scopus journals Chronic morbidities PubMed journals Chronic morbidities medical journals Chronic morbidities free journals Chronic morbidities best journals Chronic morbidities top journals Chronic morbidities free medical journals Chronic morbidities famous journals Chronic morbidities Google Scholar indexed journals Neonatal mortalities articles Neonatal mortalities Research articles Neonatal mortalities review articles Neonatal mortalities PubMed articles Neonatal mortalities PubMed Central articles Neonatal mortalities 2023 articles Neonatal mortalities 2024 articles Neonatal mortalities Scopus articles Neonatal mortalities impact factor journals Neonatal mortalities Scopus journals Neonatal mortalities PubMed journals Neonatal mortalities medical journals Neonatal mortalities free journals Neonatal mortalities best journals Neonatal mortalities top journals Neonatal mortalities free medical journals Neonatal mortalities famous journals Neonatal mortalities Google Scholar indexed journals Child articles Child Research articles Child review articles Child PubMed articles Child PubMed Central articles Child 2023 articles Child 2024 articles Child Scopus articles Child impact factor journals Child Scopus journals Child PubMed journals Child medical journals Child free journals Child best journals Child top journals Child free medical journals Child famous journals Child Google Scholar indexed journals Biological articles Biological Research articles Biological review articles Biological PubMed articles Biological PubMed Central articles Biological 2023 articles Biological 2024 articles Biological Scopus articles Biological impact factor journals Biological Scopus journals Biological PubMed journals Biological medical journals Biological free journals Biological best journals Biological top journals Biological free medical journals Biological famous journals Biological Google Scholar indexed journals Psychological articles Psychological Research articles Psychological review articles Psychological PubMed articles Psychological PubMed Central articles Psychological 2023 articles Psychological 2024 articles Psychological Scopus articles Psychological impact factor journals Psychological Scopus journals Psychological PubMed journals Psychological medical journals Psychological free journals Psychological best journals Psychological top journals Psychological free medical journals Psychological famous journals Psychological Google Scholar indexed journals Obstetric articles Obstetric Research articles Obstetric review articles Obstetric PubMed articles Obstetric PubMed Central articles Obstetric 2023 articles Obstetric 2024 articles Obstetric Scopus articles Obstetric impact factor journals Obstetric Scopus journals Obstetric PubMed journals Obstetric medical journals Obstetric free journals Obstetric best journals Obstetric top journals Obstetric free medical journals Obstetric famous journals Obstetric Google Scholar indexed journals Pre-eclampsia articles Pre-eclampsia Research articles Pre-eclampsia review articles Pre-eclampsia PubMed articles Pre-eclampsia PubMed Central articles Pre-eclampsia 2023 articles Pre-eclampsia 2024 articles Pre-eclampsia Scopus articles Pre-eclampsia impact factor journals Pre-eclampsia Scopus journals Pre-eclampsia PubMed journals Pre-eclampsia medical journals Pre-eclampsia free journals Pre-eclampsia best journals Pre-eclampsia top journals Pre-eclampsia free medical journals Pre-eclampsia famous journals Pre-eclampsia Google Scholar indexed journals Multiple pregnancy articles Multiple pregnancy Research articles Multiple pregnancy review articles Multiple pregnancy PubMed articles Multiple pregnancy PubMed Central articles Multiple pregnancy 2023 articles Multiple pregnancy 2024 articles Multiple pregnancy Scopus articles Multiple pregnancy impact factor journals Multiple pregnancy Scopus journals Multiple pregnancy PubMed journals Multiple pregnancy medical journals Multiple pregnancy free journals Multiple pregnancy best journals Multiple pregnancy top journals Multiple pregnancy free medical journals Multiple pregnancy famous journals Multiple pregnancy Google Scholar indexed journals Preterm babies articles Preterm babies Research articles Preterm babies review articles Preterm babies PubMed articles Preterm babies PubMed Central articles Preterm babies 2023 articles Preterm babies 2024 articles Preterm babies Scopus articles Preterm babies impact factor journals Preterm babies Scopus journals Preterm babies PubMed journals Preterm babies medical journals Preterm babies free journals Preterm babies best journals Preterm babies top journals Preterm babies free medical journals Preterm babies famous journals Preterm babies Google Scholar indexed journals Antenatal care articles Antenatal care Research articles Antenatal care review articles Antenatal care PubMed articles Antenatal care PubMed Central articles Antenatal care 2023 articles Antenatal care 2024 articles Antenatal care Scopus articles Antenatal care impact factor journals Antenatal care Scopus journals Antenatal care PubMed journals Antenatal care medical journals Antenatal care free journals Antenatal care best journals Antenatal care top journals Antenatal care free medical journals Antenatal care famous journals Antenatal care Google Scholar indexed journals
Article Details
List of Abbreviations:
ANC: Antenatal Care; GWG: Gestational Weight Gain; ITNs: Insecticide-Treated Bed Nets; MCHRBs: Maternal and Child Health Record Books; ODK: Open Data Kit; PNC: Postnatal Care; SDGs: Sustainable Development Goals; SES: Socioeconomic Status; SP: Sulphadoxine Pyrimethamine; STATA: Statistics and Data
1. Introduction
Preterm and post-term births are regarded as the two forms of abnormal gestational age at birth responsible for chronic morbidities and neonatal mortalities worldwide [1,2]. Preterm births have predominantly been established as the principal cause of mortalities among children under five years of age, thereby accounting for 35.0% of neonatal deaths and about 16.0% of all worldwide deaths [3,4]. Similarly, post-term births have been evidenced to be associated with long-term child maladies and mortalities [5,6]. The World Health Organization denotes preterm births (also known as prematurity) as deliveries or births that occur before 37 weeks of gestation whilst post-term births (also called post-maturity/post-date) are defined as deliveries that happen at a gestational age equal to or above 42 weeks [4].
The multi-factorial determinants of abnormal gestational age at birth are largely related to biological, psychological, obstetric, demographic, and social contributions [1,6]. Therefore, there is a need to investigate clinical, nutritional, economic, geographic, and other related predictors of these preterm and post-term births globally, especially in developing countries. Some significant elements associated with preterm births encompass antepartum haemorrhage, pre-eclampsia, multiple pregnancy [7-11], gestational anaemia, rural residence, and short birth spacing [1,12-15]. Some retrospective studies in Africa also identified multiparity, adolescent mothers, low socioeconomic status (SES) [11,16], married mothers, low parity, and inadequate antenatal visits [16,17] as vital determinants of preterm babies. Alternatively, other studies revealed large babies [18], maternal overweight, low gestational weight gain [19,20], high SES, unmarried mothers [5], and pregnancy-related psycho-emotional problems [21] to be the predictors of post-term infants.
The global action for preterm births which is particularly centered on policies including the Sustainable Development Goals (SDGs) and Global Strategy for Women’s, Children’s and Adolescents’ Health is still on course to improve newborn and child survival. However, post-term births have not been accorded similar needed attention. Notwithstanding, the prevalence rates of preterm and post-term births are still on the rise as preterm births have significantly upsurged spanning over 20 years [22]. Globally, an estimated 13.4 million (9.9%) babies were born too soon in 2020 with the highest burden in Southern Asia (13.2%) and sub-Saharan Africa (10.1%) [4]. The prevalence of preterm deliveries in Brazil [16], Ethiopia [9,12], Nigeria [11], Kenya [10], and Southern Ghana [17] were 11.5%, 13.3%, 16.0%, 18.3%, and 18.9% respectively. Additionally, in the Kassena-Nankana district in the Upper East Region of Ghana, the prevalence of preterm births was 32.0% [23]. On the other hand, despite data paucity on post-matured births in Ghana, the incidence was found to be 4.5%, 6.0%, 6.5%, and 11.4% among the American [20], Ethiopian [5], British [24], and South African [25] mothers correspondingly in some prospective and retrospective reports.
The entire globe has unacceptable gaps in perinatal health outcomes by virtue of low SES (poor), ethnic or racial differences, and most marginalized groups in society. Nonetheless, preterm and post-date births have higher survival rates in developed nations after delivery while low-resource settings like Ghana are still recording huge perinatal and neonatal deaths [1,4]. Therefore, identifying and comprehending the risk factors for preterm and post-term births have the potential to help tackle this problem and also aid in achieving SDG 3 through the reduction of neonatal mortalities and morbidities in developing countries like Ghana. Globally, most maternal and child health information on risk factors for abnormal gestational age at birth are always focused on preterm births without paying much attention to post-term ones. Nevertheless, this study considers both. Although preterm and post-term births are disproportionately rampant in most parts and peri-urban belts of Northern Ghana, the magnitude and underlying predictors are also not well comprehended which necessitates more evidence for apt interventions. Currently, there is a scarcity of published data on the prevalence of abnormal gestational age at birth and its possible determining factors in the Savelugu municipality and Northern Ghana. This study therefore assessed the prevalence of preterm and post-term births and their associated risk factors among mothers attending postnatal care services in the Savelugu municipality of Northern Ghana.
2. Materials and Methods
2.1 Study design
The study employed a retrospective and cross-sectional design to determine the magnitude of preterm and post-term births among mothers attending postnatal care (PNC) services and also identify the predictors of these adverse pregnancy outcomes in the study area.
2.2 Study area
Savelugu municipality is situated in the northern hemisphere of Northern Region of Ghana. The major source of income in the municipal is farming while the inhabitants are predominantly from the Dagomba ethnic group. Five major public health facilities with 21 operational CHPS zones provide health services to a populace of 125,469 in this municipality. The total number of reproductive-aged women is more than 40,000 which places the municipal at a higher risk of frequent pregnancies with routine increased rate of PNC and antenatal care (ANC) attendance. The municipal has a skilled delivery rate of 93% whereas monthly skilled delivery is estimated between 300 and 400. Moreover, maternal and child health record books (MCHRBs) have been consistently used by mothers when seeking antenatal, postnatal, and child welfare services since the inception of a Japanese project on MCHRBs [26].
2.3 Study population
This study recruited postnatal mothers living in the municipality and having a baby of less than 29 days (neonatal period). Mothers who owned MCHRBs and also attended first-day PNC services at the municipal public health facilities were sampled while mothers with heart diseases, twin deliveries, and home deliveries were not included in the study.
2.4 Sample size and sampling methods
The sample size (n) was attained by the formula; n = . Due to the scarcity of Ghanaian data on post-term births, the study employed prevalence (p) of preterm births in Southern Ghana which was reported as 33% [8]. By applying margin of error (e) of 5%, and standard normal variate (z) at 95% confidence level of 1.96, the sample size was initially estimated at 339. After adding 5% non-response rate [7], the final sample size was rounded off to 356.
During sampling, all the five major public health facilities namely Savelugu municipal hospital, Diare, Moglaa, Pong-Tamale, and Savelugu RCH health centres in the municipality were selected. Probability proportional to size technique was used to estimate the sample sizes for the five major public health facilities in the study area (Table 1). A coin was tossed to randomly select all the 356 postnatal mothers, using the daily PNC attendance book from the respective public health facilities.
|
Health facilities |
U: Total first PNC visits at targeted health facilities in 2020 |
V: Facility coverage [=(U)/8042] |
X: Number of mothers to be drawn for each facility [= (V) x 356] |
|
Diare health centre |
2220 |
27.6% |
98 |
|
Moglaa health centre |
346 |
4.3% |
15 |
|
Pong-Tamale health centre |
852 |
10.6% |
38 |
|
Savelugu RCH health centre |
1198 |
14.9% |
53 |
|
Savelugu municipal hospital |
3426 |
42.6% |
152 |
|
Total |
8042 |
100.0% |
356 |
Table 1: Approximated facility sample sizes in the study area.
2.5 Study variables
All the study variables and/or data were collected from MCHRBs and/or ANC records except some sociodemographic information which was collected through structured interviews from February to March 2022. Data collection was done using structured questionnaires designed onto Open Data Kit (ODK) version 2021.2.4 (Get ODK Inc., San Diego, USA), and pre-installed onto handheld tablets.
The outcome variable for this study is gestational age at birth which is multinomial recorded as preterm (< 37 weeks of gestation) = 1, normal term (≥ 37–41 weeks of gestation) = 2, and post-term (≥ 42 weeks of gestation) = 3. The “normal term” was used as the base outcome (reference point) during analyses.
From the collected data, 39 exposure variables were selected and categorized based on biological plausibility, existing literature, and the potential to influence gestational age at birth [18,27-29] (Tables 2-4). Some of these exposure variables included marital status, educational level, ethnic group, religion, sex of neonate, household size, job status, birthweight, birth length, parity, gravidity, frequency of ANC visits, tetanus-diphtheria immunization, iron-folic acid intake, anthelminthic drugs intake, and sulphadoxine-pyrimethamine (SP) doses intake, gestational weight gain (measured within one week prior to delivery and the one recorded at the first trimester ANC visit), and pre-gestational body mass index (mother’s body weight at first trimester of pregnancy during antenatal visit was used as a proxy for pre-pregnancy weight since foetal weight is low). Other variables were diagnosis of hypertension, diabetes, hepatitis B, and malaria during pregnancy, rhesus type, Glucose-6-phosphate dehydrogenase deficiency (G6PD) status, haemoglobin levels at first, second, and third trimesters of pregnancy (classified as anaemia [< 11g/dL], normal [≥ 11–13.1g/dL], and high [≥ 13.2g/dL]), and wealth index [18,27,28].
Wealth index was assessed based on possession of household assets, housing quality, and availability of household utilities among others which were used as proxy indicators for SES of mothers. By using principal component analysis, the SES of mothers was then categorized into five groups (thus, wealth quintiles: poorest, poorer, poor, less poor, least poor) [29].
2.6 Statistical analysis
The data attained was first transferred from ODK platform onto Microsoft excel version 17.0 before moving them onto STATA version 17.0 (Stata Corporation, Texas, USA). All analyses were conducted in STATA and the results were presented in the form of Tables, Figure, summary statistics in odds ratios (OR), and p-values at 95 % confidence intervals (CI). Associations between each exposure (background) variable and outcome variable (gestational age at birth) were explored at the bivariate level by using Chi-square or Fisher’s exact test (Tables 2-4). Variance inflation factor (VIF) was used to address potential multicollinearity between all exposure variables significant at p < 0.05 before employing them in logistic analyses. During the testing, the variables with a VIF of less than 10 were selected for the logistic analyses. After addressing multicollinearity, the significant exposure variables were simultaneously entered into a multinomial logistic regression model to identify the associated risk factors of preterm and post-term births (Table 5).
3. Results
3.1 Background characteristics among study participants
The average age of the mothers was 27 years with a standard deviation of ±5. Larger proportion of the mothers had marriage partners (90%) and 88% were Muslims. Approximately, one-third of the mothers had no formal education (34%) (Table 2). Slightly above one-half of the respondents were informally employed (51%), attended ANC less than eight times (54%), took more than three doses of sulphadoxine-pyrimethamine (SP) drugs (55%), and used insecticide-treated bed nets (ITNs) (61%) during gestation (Table 3). The incidence of gestational anaemia in the first, second, and third trimesters were 45%, 56%, and 44% respectively. Less than one-quarter of the mothers were diagnosed with gestational diabetes (6%), gestational hypertension (12%), and hepatitis B (11%) infections during pregnancy. Most mothers experienced two or more childbirths (73%) and had become pregnant for more than one time (75%). A greater proportion of the neonates were born with small weights (22%) as compared to macrosomic births (9%) (Table 4).
3.2 Prevalence of preterm and post-term births
Out of the total study participants, the prevalence of preterm and post-term births was 19.4% (95%CI: 15.4%-23.8%) and 6.5% (95%CI: 4.1%-9.5%) respectively (Figure 1).
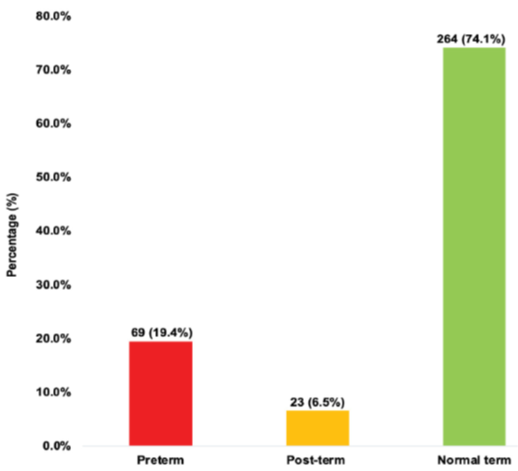
Figure 1: Prevalence of preterm and post-term births.
3.3 Association of preterm and post-term births with background characteristics
As presented in Tables 2-4, maternal age (p=0.001), educational level (p=0.009), gestational weight gain (p=0.012), neonatal birth length (p=0.041), neonatal birth weight (p<0.001), frequency of ANC visits (p<0.001), use of ITNs (p=0.037), frequency of SP intake (p=0.009), number of pregnancies (p=0.002), number of deliveries (p=0.001), haemoglobin levels in each trimester of pregnancy (p<0.001), gestational diabetes (p=0.014), and G6PD status (p=0.034) showed bivariate relationship with abnormal gestational age at birth (preterm and post-term births).
|
Background variables |
Bivariate analysis |
|||||
|
Total (%) |
Preterm (%) |
Post-term (%) |
Normal term (%) |
p–value* |
||
|
n = 356 |
n = 69 |
n = 23 |
n = 264 |
|||
|
Maternal age category |
||||||
|
16 to 19 years |
18 (5.1) |
10 (14.5) |
0 (0.0) |
8 (3.0) |
0.001† |
|
|
20 to 35 years |
308 (86.5) |
54 (78.2) |
23 (100.0) |
231 (87.5) |
||
|
Above 35 years |
30 (8.4) |
5 (7.3) |
0 (0.0) |
25 (9.5) |
||
|
Mean (SD) = 27 (5) |
||||||
|
Marital status |
||||||
|
Married |
319 (89.6) |
59 (85.5) |
21 (91.3) |
239 (90.5) |
0.492 |
|
|
Unmarried |
37 (10.4) |
10 (14.5) |
2 (8.7) |
25 (9.5) |
||
|
Highest level of education |
||||||
|
None |
121 (34.0) |
24 (34.8) |
13 (56.5) |
84 (31.8) |
0.009† |
|
|
Primary |
46 (12.9) |
14 (20.3) |
3 (13.0) |
29 (11.0) |
||
|
Junior high |
46 (12.9) |
7 (10.1) |
3 (13.0) |
36 (13.6) |
||
|
Senior high |
105 (29.5) |
14 (20.3) |
1 (4.5) |
90 (34.1) |
||
|
Tertiary |
38 (10.7) |
10 (14.5) |
3 (13.0) |
25 (9.5) |
||
|
Ethnic group |
||||||
|
Dagomba |
273 (76.7) |
52 (75.4) |
15 (65.2) |
206 (78.0) |
0.432 |
|
|
Frafra |
60 (16.8) |
14 (20.3) |
6 (26.1) |
40 (15.2) |
||
|
Others a |
23 (6.5) |
3 (4.4) |
2 (8.7) |
18 (6.8) |
||
|
Religious affiliation |
||||||
|
Christian |
44 (12.4) |
4 (5.8) |
3 (13.0) |
37 (14.0) |
0.181 |
|
|
Muslim |
312 (87.6) |
65 (94.2) |
20 (87.0) |
227 (86.0) |
||
|
Occupation status |
||||||
|
Unemployed |
142 (39.9) |
24 (34.8) |
7 (30.4) |
111 (42.1) |
0.334 |
|
|
Informal |
181 (50.8) |
40 (57.9) |
15 (65.2) |
126 (47.7) |
||
|
Formal |
33 (9.3) |
5 (7.3) |
1 (4.4) |
27 (10.2) |
||
|
Fuel type |
||||||
|
Firewood |
148 (41.6) |
32 (46.4) |
7 (30.4) |
109 (41.3) |
0.397 |
|
|
Charcoal |
162 (45.5) |
31 (44.9) |
14 (60.9) |
117 (44.3) |
||
|
Gas |
46 (12.9) |
6 (8.7) |
2 (8.7) |
38 (14.4) |
||
|
Wealth index |
||||||
|
Poorest |
72 (20.2) |
13 (18.8) |
3 (13.0) |
56 (21.2) |
0.630 |
|
|
Poorer |
75 (21.0) |
19 (27.5) |
5 (21.7) |
51 (19.3) |
||
|
Poor |
70 (19.7) |
15 (21.7) |
4 (17.4) |
51 (19.3) |
||
|
Less Poor |
70 (19.7) |
13 (19.0) |
7 (30.4) |
50 (19.0) |
||
|
Least Poor |
69 (19.4) |
9 (13.0) |
4 (17.4) |
56 (21.2) |
||
|
Family size |
||||||
|
< 10 occupants |
283 (79.5) |
58 (84.1) |
19 (82.6) |
206 (78.0) |
0.505 |
|
|
≥ 10 occupants |
73 (20.5) |
11 (15.9) |
4 (17.4) |
58 (22.0) |
||
|
Neonatal age category |
||||||
|
< 1 week |
106 (29.8) |
20 (29.0) |
6 (26.1) |
80 (30.3) |
0.902 |
|
|
2 – 4 weeks |
250 (70.2) |
49 (71.0) |
17 (73.9) |
184 (69.7) |
||
|
Gender of neonates |
||||||
|
Male |
200 (56.2) |
38 (55.1) |
10 (43.5) |
152 (57.6) |
0.417 |
|
|
Female |
156 (43.8) |
31 (44.9) |
13 (56.5) |
112 (42.4) |
||
SD: standard deviation †p-value < 0.05 *Chi-square/Fisher’s exact test aBulsa, Dagaati, Ewe
Table 2: Association of preterm and post-term births with sociodemographic characteristics among study participants.
|
Background variables |
Bivariate analysis |
|||||
|
Total (%) |
Preterm (%) |
Post-term (%) |
Normal term (%) |
p–value * |
||
|
n = 356 |
n = 69 |
n = 23 |
n = 264 |
|||
|
Maternal body mass index (BMI) |
||||||
|
Underweight (< 18.5 kg/m2) |
25 (7.0) |
4 (5.8) |
0 (0.0) |
21 (8.0) |
0.540 |
|
|
Normal BMI (≥ 18.5 to 24.9 kg/m2) |
229 (64.3) |
47 (68.1) |
18 (78.3) |
164 (62.1) |
||
|
Overweight (≥ 25.0 kg/m2) |
102 (28.7) |
18 (26.1) |
5 (21.7) |
79 (29.9) |
||
|
First-trimester gestational weight |
||||||
|
< 50 kg |
42 (11.8) |
9 (13.0) |
0 (0.0) |
33 (12.5) |
0.192 |
|
|
≥ 50 kg |
314 (88.2) |
60 (87.0) |
23 (100.0) |
231 (87.5) |
||
|
Maternal height |
||||||
|
< 150 cm |
13 (3.7) |
2 (2.9) |
0 (0.0) |
11 (4.2) |
1.000 |
|
|
≥ 150 cm |
343 (96.7) |
67 (97.1) |
23 (100.0) |
253 (95.8) |
||
|
Gestational weight gain |
||||||
|
< 6 kg |
94 (26.4) |
19 (27.5) |
0 (0.0) |
75 (21.2) |
0.012† |
|
|
≥ 6 kg |
262 (73.6) |
50 (72.5) |
23 (100.0) |
189 (71.6) |
||
|
Neonatal birth length |
||||||
|
≤ 47.5 cm |
72 (20.2) |
16 (23.2) |
0 (0.0) |
56 (21.2) |
0.041† |
|
|
> 47.5 cm |
284 (79.8) |
53 (76.8) |
23 (100.0) |
208 (78.8) |
||
|
Neonatal birth weight (BW) |
||||||
|
Low birth weight (< 2.5 kg) |
79 (22.2) |
29 (42.0) |
2 (8.7) |
48 (18.2) |
< 0.001† |
|
|
Normal BW (≥ 2.5 kg – 3.9 kg) |
246 (69.1) |
37 (53.6) |
11 (47.8) |
198 (75.0) |
||
|
Macrosomia (≥ 4.0 kg) |
31 (8.7) |
3 (4.4) |
10 (43.5) |
18 (6.8) |
||
|
Frequency of ANC visits |
||||||
|
< 8 visits |
191 (53.7) |
48 (69.6) |
4 (17.4) |
139 (52.6) |
< 0.001† |
|
|
≥ 8 visits |
165 (46.3) |
21 (30.4) |
19 (82.6) |
125 (47.4) |
||
|
Facility type for ANC visits |
||||||
|
District hospital |
175 (49.2) |
32 (46.4) |
10 (43.5) |
133 (50.4) |
0.716 |
|
|
Health centre |
181 (50.8) |
37 (53.6) |
13 (56.5) |
131 (49.6) |
||
|
Family planning use before last pregnancy |
||||||
|
No |
232 (65.2) |
45 (65.2) |
12 (52.2) |
175 (66.3) |
0.395 |
|
|
Yes |
124 (34.8) |
24 (34.8) |
11 (47.8) |
89 (33.7) |
||
|
Insecticide-treated nets (ITNs) use |
||||||
|
No |
138 (38.8) |
36 (52.2) |
9 (39.1) |
93 (35.2) |
0.037† |
|
|
Yes |
218 (61.2) |
33 (47.8) |
14. (60.9) |
171 (64.8) |
||
|
Frequency of sulphadoxine-pyrimethamine (SP) intake |
||||||
|
None |
18 (5.1) |
6 (8.7) |
2 (8.7) |
10 (3.8) |
0.009† |
|
|
1 to 3 times |
141 (39.6) |
34 (49.3) |
3 (13.0) |
104 (39.4) |
||
|
Above 3 times |
197 (55.3) |
29 (42.0) |
18 (78.3) |
150 (56.8) |
||
|
Anthelminthics intake |
||||||
|
No |
218 (61.2) |
44 (63.8) |
15 (65.2) |
159 (60.2) |
0.797 |
|
|
Yes |
138 (38.8) |
25 (36.2) |
8 (34.8) |
105 (39.8) |
||
|
Iron/folic acid intake |
||||||
|
No |
13 (3.7) |
1 (1.5) |
0 (0.0) |
12 (4.6) |
0.298 |
|
|
Yes |
343 (96.3) |
68 (98.5) |
23 (100.0) |
252 (95.4) |
||
|
Tetanus-diphtheria vaccination |
||||||
|
No |
26 (7.3) |
4 (5.8) |
2 (8.7) |
20 (7.6) |
0.850 |
|
|
Yes |
330 (92.7) |
65 (94.2) |
21 (91.3) |
244 (92.4) |
||
|
Nutrition education received |
||||||
|
No |
47 (13.2) |
5 (7.3) |
6 (26.1) |
36 (13.6) |
0.064 |
|
|
Yes |
309 (86.8) |
64 (92.7) |
17 (73.9) |
228 (86.4) |
||
†p-value < 0.05 *Chi-square test/Fisher’s exact test
Table 3: Association of preterm and post-term births with antenatal and anthropometric characteristics among study participants.
|
Background variables |
Bivariate analysis |
|||||
|
Total (%) |
Preterm (%) |
Post-term (%) |
Normal term (%) |
p-value * |
||
|
n = 356 |
n = 69 |
n = 23 |
n = 264 |
|||
|
Number of pregnancies |
||||||
|
0 – 1 pregnancy |
88 (24.7) |
25 (36.2) |
0 (0.0) |
63 (23.9) |
0.002† |
|
|
≥ 2 pregnancies |
268 (75.3) |
44 (63.8) |
23 (100.0) |
201 (76.1) |
||
|
Number of deliveries |
||||||
|
0 – 1 delivery |
97 (27.3) |
27 (39.1) |
0 (0.0) |
70 (26.5) |
0.001† |
|
|
≥ 2 deliveries |
259 (72.7) |
42 (60.9) |
23 (100.0) |
194 (73.5) |
||
|
Haemoglobin (Hb) levels at first trimester |
||||||
|
Low Hb/Anaemia (< 11g/dL) |
160 (44.9) |
49 (71.0) |
4 (17.4) |
107 (40.5) |
< 0.001† |
|
|
Normal Hb (≥ 11 – 13.1g/dL) |
177 (49.7) |
20 (29.0) |
12 (52.2) |
145 (54.9) |
||
|
High Hb (≥ 13.2g/dL) |
19 (5.4) |
0 (0.00) |
7 (30.4) |
12 (4.6) |
||
|
Hb levels at second trimester |
||||||
|
Low Hb/Anaemia |
200 (56.2) |
56 (81.2) |
5 (21.7) |
139 (52.6) |
< 0.001† |
|
|
Normal Hb |
152 (42.7) |
13 (18.8) |
16 (69.6) |
123 (46.6) |
||
|
High Hb |
4 (1.1) |
0 (0.00) |
2 (8.7) |
2 (0.8) |
||
|
Hb levels at third trimester |
||||||
|
Low Hb/Anaemia |
158 (44.4) |
46 (66.6) |
3 (13.0) |
109 (41.3) |
< 0.001† |
|
|
Normal Hb |
192 (53.9) |
22 (31.9) |
19 (82.6) |
151 (57.2) |
||
|
High Hb |
6 (1.7) |
1 (1.5) |
1 (4.4) |
4 (1.5) |
||
|
Malaria infection during pregnancy |
||||||
|
No |
254 (71.3) |
44 (63.8) |
20 (87.0) |
190 (72.0) |
0.094 |
|
|
Yes |
102 (28.7) |
25 (36.2) |
3 (13.0) |
74 (28.0) |
||
|
HIV infection |
||||||
|
No |
350 (98.3) |
67 (97.1) |
22 (95.6) |
261 (98.9) |
0.354 |
|
|
Yes |
6 (1.7) |
2 (2.9) |
1 (4.4) |
3 (1.1) |
||
|
Hepatitis B infection |
||||||
|
No |
318 (89.3) |
58 (84.1) |
21 (91.3) |
239 (90.5) |
0.286 |
|
|
Yes |
38 (10.7) |
11 (15.9) |
2 (8.7) |
25 (9.5) |
||
|
Gestational diabetes status |
||||||
|
Not diagnosed |
335 (94.1) |
69 (100.0) |
20 (87.0) |
246 (93.2) |
0.014† |
|
|
Diagnosed |
21 (5.9) |
0 (0.00) |
3 (13.0) |
18 (6.8) |
||
|
Gestational hypertension status |
||||||
|
Not diagnosed |
312 (87.6) |
58 (86.1) |
20 (87.0) |
234 (88.6) |
0.586 |
|
|
Diagnosed |
44 (12.4) |
11 (15.9) |
3 (13.0) |
30 (11.4) |
||
|
Sickle cell status |
||||||
|
Negative |
301 (84.5) |
54 (78.3) |
18 (78.3) |
229 (86.7) |
0.153 |
|
|
Positive |
55 (15.5) |
15 (21.7) |
5 (21.7) |
35 (13.3) |
||
|
G6PD status |
||||||
|
Normal |
333 (93.5) |
61 (88.4) |
20 (87.0) |
252 (95.4) |
0.034† |
|
|
Complete/partial |
23 (6.5) |
8 (11.6) |
3 (13.0) |
12 (4.6) |
||
†p-value < 0.05 *Chi-square test/Fisher’s exact test
Table 4: Association of preterm and post-term births with clinical and obstetric characteristics among study participants.
3.4 Associated risk factors of preterm and post-term births
Fifteen exposure variables exhibited significant bivariate association (p<0.05) with preterm and post-term births as shown in Tables 2-4. However, one variable (thus, number of pregnancies) was excluded from the significant exposure variables due to multicollinearity before all the other variables were entered into the final logistic model. In the final model, maternal age < 20 years, educational level (junior high school), non-use of ITNs, and anaemia in the first trimester of pregnancy remained significant for preterm births while senior high school level of education and macrosomia (large birth weight) showed significant relationship with post-term births (Table 5).
Mothers aged less than 20 years (thus teenage mothers) were 12.95 times more likely to give birth to preterm babies (AOR: 12.95; 95%CI: 2.977-56.34). Mothers who completed junior high school were less likely to give birth to preterm babies, showing 77.5% [=(1-0.225)-100] reduction in delivering preterm babies (AOR: 0.225; 95%CI: 0.065-0.797). Mothers who didn’t use ITNs during pregnancy were 97.9% more likely to have preterm births (AOR: 1.979; 95%CI: 0.999-3.920). The odds of a mother with anaemia in the first trimester of pregnancy having a preterm birth was 2.205 times (AOR: 2.205; 95%CI: 1.011-4.809).
Furthermore, mothers who completed senior high school were 99.9% less likely to give birth to post-term babies (AOR: 0.001; 95%CI: 0.0001-0.125). Mothers who delivered at an advanced age (post-term) were also more likely to give birth to macrosomic (large weight) babies (AOR: 8.128; 95%CI: 1.777-37.18).
†p-value < 0.05 AOR: Adjusted odds ratio COR: Crude odds ratio
Table 5: Multivariate analysis of risk factors for preterm and post-term births.
4. Discussion
Abnormal gestational age at birth and its associated complications are one of the setbacks in reducing children under-five mortality in most developing countries like Ghana [1]. The study found the prevalence of preterm births to be 19.4%. The prevalence of preterm births is greater than the worldwide incidence of 10.6% [30]. Additionally, greater preterm prevalence was found in this study than the prevalence from developing countries including Brazil (11.5%) [16], Ethiopia (13.3%) [9,12], Iran (12.7%) [31], Kenya (18.3%) [10], and Southern Nigeria (16.0%) [11]. This places the municipality at higher risk of neonatal deaths since preterm births are responsible for 35.0% of all neonatal mortalities and about 16.0% of all global deaths [3]. These aforementioned countries (Brazil, Ethiopia, Iran, Kenya, and Nigeria) might be effectively implementing the recommended interventions on premature births and may have comprehensive healthcare system for pregnant women leading to their relatively low preterm prevalence. Moreover, despite the diverse healthcare systems across those countries, the study designs, and larger sample sizes employed in those earlier studies [9-12,16,31] could be accountable for these variations. The present study finding was also lower than some retrospective studies in Northern India and a teaching hospital in Southern Ghana with prevalence of 35.0% [32] and 37.3% [7] respectively. The increased rate of multiple gestations in India and Southern Ghana could be responsible [7,32], as this may cause an overly stretched uterus which might precipitate preterm deliveries [12] as multiple gestations are established predictors of preterm births [30].
The prevalence of post-term births in this study was estimated at 6.5%. Our study’s post-term prevalence was lesser as compared to some findings in the Northern Ethiopia [5], and South Africa [25] at corresponding rates of 8.0%, and 11.4%. The differences could be explained in terms of demographic and geographical features of the study populations, as well as the timing of these studies. Furthermore, the lesser post-term births in our study may be linked to the cultural beliefs of the Savelugu citizenry. Culturally, the people of Savelugu municipal regard post-term births as a curse. Due to this cultural belief, pregnant women usually ingest some local oxytocin (kalgu-tim) to increase uterine contractions whenever they reach term gestational age [26], leading to early delivery of their babies and causing a possible reduction of post-term births.
In the study, 5.1% (95%CI: 3.0% - 7.9%) of the mothers were teenagers (adolescents). Teenage mothers (thus, mothers aged less than 20 years) were 13 times more likely to deliver prematurely. An analogous pattern was found among Brazilian [16] and Nigerian [11] mothers aged less than 20 years old with an increased risk of preterm births. However, our study was incongruent with some retrospective studies in Southern Ghana [7,17], Kenya [10], and Ethiopia [12]. Teenagers (young mothers) are still in the active phase of physiological, psychological, emotional, biological, and physical maturity and development. These young women (teenagers) may not cope with the changing demands of pregnancy and also identify the significance of feto-maternal nutrition, childbearing, and self-care activities among others during pregnancy [33] which may lead to adverse pregnancy outcomes like preterm births.
The current study demonstrated that mothers who completed junior high school had a reduced risk for preterm births. This is different from the findings of Wagura and colleagues [10] but similar to that of Spanish and Japanese cohort studies [34,35] which indicated that maternal lower educational level (junior high school) was associated with premature births. Approximately 13.0% (95%CI: 9.6% – 16.9%) of the mothers completed junior high school while 34.0% (95%CI: 29.1% – 39.2%) had no formal education in the Savelugu municipality. Junior high school education has been linked to lower the incidence of pregnancy complications in women including delivery of preterm babies [36,37]. This is in agreement with our study findings. It is found in most developing countries that unsatisfactory antenatal visits which predict preterm births are higher in settings where junior high school enrollment rate is low [35,36]. Hence, this gives an explanation of why mothers who completed junior high school tend to be protective against preterm births.
Gestational anaemia is a global challenge as well as the most prodigious haematological manifestation of pregnancy and a heralded cause of gestational complications and adverse reproductive outcomes [6]. Mothers with anaemia in the first trimester of pregnancy were twice more likely to give birth to preterm babies in this study. This finding is consistent with some meta-analyses [38,39] which concluded that first-trimester anaemia increases the risk of premature births. Many prospective and retrospective studies were also consistent with our study findings [13-15] while others were incongruent [40,41]. Despite the complex and multifactorial aetiological mechanisms such as nutritional deficiencies ( iron, folic acid, vitamin A, or vitamin B12), infections (malaria, worm infestations, and HIV), and haemoglobinopathies (sickle cell) underlying the relationship between low gestational haemoglobin levels and prematurity, iron deficiency (which is a major contributor to gestational anaemia) tend to be the most reported trigger of preterm births [39,42]. Thus, anaemia in the first and/or early trimester of gestation may designate pre-existing, early onset, and/or persistent iron deficiency which precipitates preterm births. Additionally, the current study revealed significant crude association between first-trimester gestational anaemia and non-intake of antihelminthics (OR: 1.55, p = 0.038). This evidence supports the mechanism of infections like worm infestations on gestational anaemia leading to the delivery of preterm babies [42]. Anaemia including iron deficiency could induce maternal infection, hypoxia, and oxidative stress that may trigger the spontaneous onset of preterm labour [32]. This highlights the need for early prevention and treatment of anaemia during pregnancy due to the relatively high preterm prevalence in the Savelugu municipality.
About 40% (95%CI: 33.7% – 44.0%) of mothers didn’t use ITNs during pregnancy in our study which is comparable to a Nigerian cross-sectional study [43]. Non-use of ITNs during gestation was significantly associated with an increased risk of preterm births in our study. This is confirmatory to some studies in the Sahelian Africa [44,45]. Strengthening community and antenatal education on the use of ITNs during pregnancy is crucial for preventing malaria infection [43,44] which predisposes pregnant women to premature deliveries. Likewise, health facilities must constantly supply these ITNs to pregnant mothers and adequately monitor ITNs use to prevent malaria infections from leading to preterm births in the municipality.
On the other hand, post-term births have not been accorded similar recognition as preterm births. Hence, there are very rare studies on post-term births globally. From our study, senior high school leavers were protective factor for post-term births. Thus, mothers who completed senior high school were less likely to deliver post-mature babies. The proportion of mothers with senior high school education were 29.5% (95%CI: 24.8% – 34.5%) in our present study. The present finding is unparallel to a prospective study in Ethiopia where no association was found between post-term births and maternal educational level [5]. By virtue of relatively higher education among mothers who completed senior high school, we conjecture that mothers with senior secondary education are relatively knowledgeable and aware of their reproductive health and pregnancy outcomes and always adhere to antenatal care education and counseling which lead to a reduction in the birth of post-term babies.
With an approximate macrosomia incidence of 9.0% (95%CI: 5.0% – 12.0%) found in our study, it was revealed that macrosomia was significantly associated with post-maturity. This finding is in contrast to that of meta-analysis [19] and longitudinal studies in the United States of America [20] but mirrored a cross-sectional study by Adjei-Gyamfi and his colleagues [18]. Maternal, child, and neonatal mortalities are associated with advanced gestational age (post-term) in most developing countries [46]. Physiologically, large-weight (macrosomic) foetus tends to experience weekly intra-uterine weight gain estimated between 0.12 to 0.24 kilograms as the foetus continuously stays in the uterus after 37 weeks of gestation [47]. As the uterus promotes growth processes, the foetus enjoys a substantial increase in weight as it stays longer in the uterus leading to late gestation. In our study, out of the 31 mothers who delivered macrosomic babies, larger proportion were diagnosed with gestational diabetes (26; 83.9%) as few mothers were not diagnosed with gestational diabetes during pregnancy (p=0.011 in χ2/Fisher’s exact test). Concurrently, of the same 31 mothers, those who had gestational weight gain (GWG) of at least 6kg (28; 90.3%) were greater than those who had GWG less than 6kg (p=0.027 in χ2/Fisher’s exact test). As supported by previous studies [5,18-20], gestational diabetes, and increased GWG surge the risk of macrosomia births [5,19,20], which could subsequently lead to higher post-term deliveries in the municipality.
5. Limitations of the Study
This study identified some limitations. Selection bias could happen. The study did not recruit mothers without antenatal record books, delivered at home, and were absent from first-trimester antenatal services. Furthermore, information bias could occur due to the use of previous obstetric and anthropometric data from antenatal records including gestational age, birthweight, birth length, ANC visits, and mother's weight among others. This is because mismeasurement and mis-recording could occur rendering some of the data incorrect. Finally, since the study design is a cross-sectional type, it may require regional or national studies to attain generalizable findings and conclusions.
6. Conclusion
Preterm births are very prevalent while post-term births are increasingly becoming more important in the Savelugu municipality. Preterm births were independently associated with anaemia in the first trimester of pregnancy, non-use of ITNs during pregnancy, teenage mothers, and mothers with junior high school education. Alternatively, while macrosomic deliveries increased the risk of post-term births, mothers with senior high school levels of education appeared to be a protective factor. These risk factors are modifiable and preventable. Ghanaian Ministry of Health through the Ghana Health Service should give greater attention to first-trimester anaemia during pregnancy (due to its effect on preterm births) through the consistent creation of community and health workers' awareness. Also, promoting and encouraging girl child education through parental empowerment in Ghana (especially in Savelugu municipality) in addition to regular community education on nutrition together with lifestyle modification are commended.
7. Declarations
Ethical Standards Disclosure
This study was conducted according to the guidelines laid down in the Declaration of Helsinki and all procedures involving research study participants were approved by the Ethical Committee of Graduate School of Tropical Medicine and Global Health, Nagasaki University, Japan (approval no.: NU_TMGH_2021_194_1) and Ghana Health Service Ethics Review Committee, Ghana (approval no.: GHS-ERC 026/12/21). Written informed consent/assent was obtained from all study participants and/or legal representatives.
Consent for publication
Not required
Availability of data and materials
The datasets generated, and analyzed during the present study will be made available by the corresponding author, without undue reservation. Kindly email: adjeigyamfis@yahoo.com
Conflict of Interest
None
Authorship
SAG, HA, and AA formulated the research questions, designed, and conceptualized the study. SAG and AA carried out the data collection, data analysis, and interpretations. All authors contributed to drafting the manuscript, reviewing, and approving the final manuscript.
Authors’ acronyms
Silas Adjei-Gyamfi (SAG); Abigail Asirifi (AA); Hirotsugu Aiga (HA)
Funding
This study was supported by The Project for Human Resource Development Scholarship - Japan International Cooperation Agency (JICA), Nagasaki University School of Tropical Medicine and Global Health - Japan, and Savelugu Municipal Hospital - Ghana Health Service.
Acknowledgements
The authors are grateful to The Project for Human Resource Development Scholarship - Japan International Cooperation Agency (JICA), Nagasaki University School of Tropical Medicine and Global Health - Japan, and Savelugu Municipal Hospital - Ghana Health Service for supporting this study. We are also thankful to all research assistants, study participants, and volunteers who contributed in diverse ways to the successful implementation of the study.
References
- Profile of Preterm and Low Birth Weight Prevention and Care (2021): 1-3. www.everypreemie.org
- Annamraju H, Pavord S. Anaemia in pregnancy. British Journal of Hospital Medicine. MA Healthcare Ltd. 77 (2016): 584-588.
- You D, New JR, Wardlaw T. Levels and Trends in Child Mortality Report 2017 | United Nations Population Division Department of Economic and Social Affairs (2017).
- PMNCH, WHO, UNICEF, UNFPA. Born too Soon: Decade of Action on Preterm Birth. Findings and actions. Geneva (2023).
- Mengesha HG, Lerebo WT, Kidanemariam A, et al. Pre-term and post-term births: Predictors and implications on neonatal mortality in Northern Ethiopia. BMC Nurs 15 (2016): 1-11.
- World Health Organization. WHO recommendations on interventions to improve preterm birth outcomes (2015).
- Anto EO, Ofori Boadu WI, Opoku S, et al. Prevalence and Risk Factors of Preterm Birth Among Pregnant Women Admitted at the Labor Ward of the Komfo Anokye Teaching Hospital, Ghana. Front Glob Women’s Heal 3 (2022): 1-8.
- Aseidu EK, Bandoh DA, Ameme DK, et al. Obstetric determinants of preterm delivery in a regional hospital, Accra, Ghana. BMC Pregnancy Childbirth 15 (2019).
- Mulualem G, Wondim A, Woretaw A. The effect of pregnancy induced hypertension and multiple pregnancies on preterm birth in Ethiopia: A systematic review and meta-analysis 12, BMC Research Notes. BioMed Central Ltd. (2019).
- Wagura P, Wasunna A, Laving A, et al. Prevalence and factors associated with preterm birth at kenyatta national hospital. BMC Pregnancy Childbirth 18 (2018): 107.
- Zini ME, Omo-Aghoja LO. Clinical and sociodemographic correlates of preterm deliveries in two tertiary hospitals in southern Nigeria. Ghana Med J 53 (2019): 20-28.
- Aregawi G, Assefa N, Mesfin F, et al. Preterm births and associated factors among mothers who gave birth in Axum and Adwa Town public hospitals, Northern Ethiopia, 2018. BMC Res Notes 12 (2019): 1-6.
- Beckert RH, Baer RJ, Anderson JG, et al. Maternal anemia and pregnancy outcomes: a population-based study. J Perinatol 39 (2019): 911-919.
- Çakmak BD, Türker ÜA, Öztas S, et al. The effect of first trimester hemoglobin levels on pregnancy outcomes. Turk Jinekoloji ve Obstet Dern Derg. 15 (2018): 165-170.
- Zhang X, Xu Q, Yang Y, et al. Preconception Hb concentration and risk of preterm birth in over 2•7 million Chinese women aged 20-49 years: A population-based cohort study. Br J Nutr 120 (2018): 508-516.
- Do Carmo Leal M, Esteves-Pereira AP, Nakamura-Pereira M, et al. Prevalence and risk factors related to preterm birth in Brazil. Reprod Health 13 (2016): 163-174.
- Adu-Bonsaffoh K, Gyamfi-Bannerman C, Oppong SA, et al. Determinants and outcomes of preterm births at a tertiary hospital in Ghana. Placenta, W.B. Saunders Ltd. 79 (2019): 62-67.
- Adjei-Gyamfi S, Musah B, Asirifi A, et al. Maternal risk factors for low birthweight and macrosomia: a cross-sectional study in Northern Region, Ghana. J Heal Popul Nutr 42 (2023): 1-16.
- Heslehurst N, Vieira R, Hayes L, et al. Maternal body mass index and post-term birth: a systematic review and meta-analysis. Obesity Reviews 18 (2017): 293-308.
- Zhu Y, Zhang J, Li Q, et al. Association between gestational weight gain and preterm birth and post-term birth: a longitudinal study from the National Vital Statistics System database. BMC Pediatr 23 (2023): 127.
- El marroun H, Zeegers M, Steegers EA, et al. Post-term birth and the risk of behavioural and emotional problems in early childhood. Int J Epidemiol 41 (2012): 773-781.
- Vogel JP, Chawanpaiboon S, Watananirun K, et al. Global, regional and national levels and trends of preterm birth rates for 1990 to 2014: Protocol for development of World Health Organization estimates. Reprod Health 13 (2016): 1-9.
- Welaga P, Moyer CA, Aborigo R, et al. Why Are Babies Dying in the First Month after Birth? A 7-Year Study of Neonatal Mortality in Northern Ghana. PLoS One 8 (2013).
- Slack E, Best KE, Rankin J, et al. Maternal obesity classes, preterm and post-term birth: A retrospective analysis of 479,864 births in England. BMC Pregnancy Childbirth 19 (2019): 1-9.
- Majola L, Budhram S, Govender V, et al. Reliability of last menstrual period recall, an early ultrasound and a Smartphone App in predicting date of delivery and classification of preterm and post-term births. BMC Pregnancy Childbirth 21 (2021): 1-9.
- Savelugu Municipal Health Directorate. Annual performance report 2019. Savelugu, Northern Region; (2020).
- Rasmussen KM, Catalano PM, Yaktine AL. New guidelines for weight gain during pregnancy: what obstetrician/gynecologists should know. Curr Opin Obs Gynecol 21 (2009): 521-526.
- Falcão IR, Ribeiro-Silva RDC, De Almeida MF, et al. Factors associated with low birth weight at term: A population-based linkage study of the 100 million Brazilian cohort. BMC Pregnancy Childbirth 20 (2020): 1-11.
- Manyeh AK, Kukula V, Odonkor G, et al. Socioeconomic and demographic determinants of birth weight in southern rural Ghana: Evidence from Dodowa Health and Demographic Surveillance System. BMC Pregnancy Childbirth 16 (2016): 1-9.
- Chawanpaiboon S, Vogel JP, Moller AB, et al. Global, regional, and national estimates of levels of preterm birth in 2014: a systematic review and modelling analysis. Lancet Glob Heal 7 (2019): e37-46.
- Jaberi E, Roksana M. A study on preterm births during 2013–2015, Shiraz, Iran. J Obstet Gynaecol (Lahore) 38 (2018): 22-26.
- Kumari S, Garg N, Kumar A, et al. Maternal and severe anaemia in delivering women is associated with risk of preterm and low birth weight: A cross sectional study from Jharkhand, India. One Heal 8 (2019).
- Shrivastava S, Shrivastava P. A longitudinal study of maternal and socioeconomic factors influencing neonatal birth weight in pregnant women attending an urban health center. Saudi J Heal Sci 2 (2013): 87.
- Granés L, Torà-Rocamora I, Palacio M, et al. Maternal educational level and preterm birth: Exploring inequalities in a hospital-based cohort study. PLoS One 18 (2023): 1-15.
- Okui T. Correction to: Analysis of an Association between Preterm Birth and Parental Educational Level in Japan Using National Data (Children, (2023), 10, 2, (342). Children (Basel) 10 (2023): 1034.
- Muyunda B, Makasa M, Jacobs C, et al. Higher Educational Attainment Associated with Optimal Antenatal Care Visits among Childbearing Women in Zambia. Front Public Heal 4 (2016): 1-7.
- Cantarutti A, Franchi M, Monzio Compagnoni M, et al. Mother’s education and the risk of several neonatal outcomes: An evidence from an Italian population-based study. BMC Pregnancy Childbirth 17 (2017): 1-10.
- Rahmati S, Azami M, Badfar G, et al. The relationship between maternal anemia during pregnancy with preterm birth: a systematic review and meta-analysis. J Matern Neonatal Med 33 (2019): 2679-2689.
- Young MF, Oaks BM, Rogers HP, et al. Maternal low and high hemoglobin concentrations and associations with adverse maternal and infant health outcomes: an updated global systematic review and meta-analysis. BMC Pregnancy Childbirth 23 (2023): 1-16.
- Masukume G, Khashan AS, Kenny LC, et al. Risk factors and birth outcomes of anaemia in early pregnancy in a nulliparous cohort. PLoS One 10 (2015): e0122729.
- Mocking M, Savitri AI, Uiterwaal CSPM, et al. Does body mass index early in pregnancy influence the risk of maternal anaemia? An observational study in Indonesian and Ghanaian women. BMC Public Health 18 (2018).
- Di Renzo GC, Spano F, Giardina I, et al. Iron deficiency anemia in pregnancy. Women’s Heal 11 (2015): 891-900.
- Ugwu EO, Ezechukwu PC, Obi SN, et al. Utilization of insecticide treated nets among pregnant women in enugu, South Eastern Nigeria. Niger J Clin Pract 16 (2013): 292-296.
- Mahamar A, Andemel N, Swihart B, et al. Malaria Infection Is Common and Associated with Perinatal Mortality and Preterm Delivery Despite Widespread Use of Chemoprevention in Mali: An Observational Study 2010 to 2014. Clin Infect Dis 73 (2021): 1355-1361.
- Manu G, Boamah-Kaali EA, Febir LG, et al. Low utilization of insecticide-treated bed net among pregnant women in the middle belt of Ghana. Malar Res Treat (2017).
- Andersson CB, Petersen JP, Johnsen SP, et al. Risk of complications in the late vs early days of the 42nd week of pregnancy: A nationwide cohort study. Acta Obstet Gynecol Scand 101 (2022): 200-211.
- Capital Institute of Pediatrics. Nine Cities Children’s Physical Development Survey Collaboration Group. Growth standard curves of birth weight, length and head circumference of Chinese newborns of different gestation. Chinese J Pediatr 58 (2020): 738-746.
