Post-Operative Cerebral Oximetry for Detection of Low - Cardiac Output Syndrome after Coronary Artery Bypass Surgery in Patients with Low Pre-Operative Left Ventricular Ejection Fraction - A Protocol for an Observational Study on Simultaneous Monitoring of Cerebral Oximetry and Cardiac Index
Article Information
Miriam Silaschi1*, Markus Velten2, Marc Rohner2, Marcus Thudium2, Jacqueline Kruse1, Marwan Hamiko1, David Rowlands1, Stefan Kreyer2, Christian Putensen2, Nils Theuerkauf2, Mark Coburn2, Farhad Bakhtiary1
1Department of Cardiac Surgery, University Hospital Bonn, Germany
2Department of Anesthesiology and Intensive Care Medicine, University Hospital Bonn, Germany
*Corresponding Author: Miriam Silaschi, Department of Cardiac Surgery, University Hospital Bonn, Germany
Received: 08 May 2023; Accepted: 17 May 2023; Published: 17 August 2023
Citation: Miriam Silaschi, Markus Velten, Marc Rohner, Marcus Thudium, Jacqueline Kruse, Marwan Hamiko, David Rowlands, Stefan Kreyer, Christian Putensen, Nils Theuerkauf, Mark Coburn, Farhad Bakhtiary. Post-Operative Cerebral Oximetry for Detection of Low - Cardiac Output Syndrome after Coronary Artery Bypass Surgery in Patients with Low Pre-Operative Left Ventricular Ejection Fraction - A Protocol for an Observational Study on Simultaneous Monitoring of Cerebral Oximetry and Cardiac Index. Journal of Surgery and Research. 6 (2023): 277-282.
View / Download Pdf Share at FacebookAbstract
Background: While cerebral near infrared spectroscopy (NIRS) is a valuable diagnostic tool to monitor brain oxygenation during cardiac surgery, its value in situations of low cardiac output in critical care patients has not been thoroughly evaluated. Thus, the aim of this study is to evaluate the correlation of NIRS with cardiac index in low cardiac output syndrome (LCOS). The hypothesis of this study is that NIRS shows significant decline during LCOS earlier than routine hemodynamic measurements.
Methods: This study is a monocentric, prospective, observational study. The reference diagnostic test for comparison is continuous cardiac index measurement through Swan-Ga\gnz ca\gtheter. Patients undergoing coronary artery bypass grafting and pre-operative reduced left ventricular ejection fraction (<35%) will be included. Patients will receive non-invasive cNIRS for up to 48 hours after surgery. Swan-Ganz catheter measurements will be performed simultaneously as per institutional standard. The primary endpoint is LCOS by a standard definition and its timepoint of onset. In addition to correlation of NIRS values with cardiac index, sensitivity and specificity analyses will be performed after adjustment for confounders (systemic vascular resistance, hemoglobin). Assuming a 20% LCOS rate in patients with baseline ejection fraction below 35%, at least 68 individuals need to be included in the study to gain enough data to reach statistical significance. In case at 68 individuals, the primary endpoint is not met in enough cases to perform robust statistical analysis, enrollment will be continued. Patients admitted to intensive-care unit (ICU) on mechanical circulatory support classify as screening failures.
Discussion: This study could improve understanding the value of NIRS in LCOS. Findings of this research potentially impact guideline recommendations on routine hemodynamic monitoring on cardiac ICU.
Trial registration: DRKS00017260 (German Registry of Clinical Studies)
Keywords
<p>Low cardiac output syndrome, Hemodynamic monitoring, Cerebral oximetry, Cerebral near infrared spectroscopy</p>
Low cardiac output syndrome articles; Hemodynamic monitoring articles; Cerebral oximetry articles; Cerebral near infrared spectroscopy articles
Low cardiac output syndrome articles Low cardiac output syndrome Research articles Low cardiac output syndrome review articles Low cardiac output syndrome PubMed articles Low cardiac output syndrome PubMed Central articles Low cardiac output syndrome 2023 articles Low cardiac output syndrome 2024 articles Low cardiac output syndrome Scopus articles Low cardiac output syndrome impact factor journals Low cardiac output syndrome Scopus journals Low cardiac output syndrome PubMed journals Low cardiac output syndrome medical journals Low cardiac output syndrome free journals Low cardiac output syndrome best journals Low cardiac output syndrome top journals Low cardiac output syndrome free medical journals Low cardiac output syndrome famous journals Low cardiac output syndrome Google Scholar indexed journals Hemodynamic monitoring articles Hemodynamic monitoring Research articles Hemodynamic monitoring review articles Hemodynamic monitoring PubMed articles Hemodynamic monitoring PubMed Central articles Hemodynamic monitoring 2023 articles Hemodynamic monitoring 2024 articles Hemodynamic monitoring Scopus articles Hemodynamic monitoring impact factor journals Hemodynamic monitoring Scopus journals Hemodynamic monitoring PubMed journals Hemodynamic monitoring medical journals Hemodynamic monitoring free journals Hemodynamic monitoring best journals Hemodynamic monitoring top journals Hemodynamic monitoring free medical journals Hemodynamic monitoring famous journals Hemodynamic monitoring Google Scholar indexed journals Cerebral oximetry articles Cerebral oximetry Research articles Cerebral oximetry review articles Cerebral oximetry PubMed articles Cerebral oximetry PubMed Central articles Cerebral oximetry 2023 articles Cerebral oximetry 2024 articles Cerebral oximetry Scopus articles Cerebral oximetry impact factor journals Cerebral oximetry Scopus journals Cerebral oximetry PubMed journals Cerebral oximetry medical journals Cerebral oximetry free journals Cerebral oximetry best journals Cerebral oximetry top journals Cerebral oximetry free medical journals Cerebral oximetry famous journals Cerebral oximetry Google Scholar indexed journals Cerebral near infrared spectroscopy articles Cerebral near infrared spectroscopy Research articles Cerebral near infrared spectroscopy review articles Cerebral near infrared spectroscopy PubMed articles Cerebral near infrared spectroscopy PubMed Central articles Cerebral near infrared spectroscopy 2023 articles Cerebral near infrared spectroscopy 2024 articles Cerebral near infrared spectroscopy Scopus articles Cerebral near infrared spectroscopy impact factor journals Cerebral near infrared spectroscopy Scopus journals Cerebral near infrared spectroscopy PubMed journals Cerebral near infrared spectroscopy medical journals Cerebral near infrared spectroscopy free journals Cerebral near infrared spectroscopy best journals Cerebral near infrared spectroscopy top journals Cerebral near infrared spectroscopy free medical journals Cerebral near infrared spectroscopy famous journals Cerebral near infrared spectroscopy Google Scholar indexed journals cardiac surgery articles cardiac surgery Research articles cardiac surgery review articles cardiac surgery PubMed articles cardiac surgery PubMed Central articles cardiac surgery 2023 articles cardiac surgery 2024 articles cardiac surgery Scopus articles cardiac surgery impact factor journals cardiac surgery Scopus journals cardiac surgery PubMed journals cardiac surgery medical journals cardiac surgery free journals cardiac surgery best journals cardiac surgery top journals cardiac surgery free medical journals cardiac surgery famous journals cardiac surgery Google Scholar indexed journals hemodynamic monitoring articles hemodynamic monitoring Research articles hemodynamic monitoring review articles hemodynamic monitoring PubMed articles hemodynamic monitoring PubMed Central articles hemodynamic monitoring 2023 articles hemodynamic monitoring 2024 articles hemodynamic monitoring Scopus articles hemodynamic monitoring impact factor journals hemodynamic monitoring Scopus journals hemodynamic monitoring PubMed journals hemodynamic monitoring medical journals hemodynamic monitoring free journals hemodynamic monitoring best journals hemodynamic monitoring top journals hemodynamic monitoring free medical journals hemodynamic monitoring famous journals hemodynamic monitoring Google Scholar indexed journals postoperative low-cardiac output syndrome articles postoperative low-cardiac output syndrome Research articles postoperative low-cardiac output syndrome review articles postoperative low-cardiac output syndrome PubMed articles postoperative low-cardiac output syndrome PubMed Central articles postoperative low-cardiac output syndrome 2023 articles postoperative low-cardiac output syndrome 2024 articles postoperative low-cardiac output syndrome Scopus articles postoperative low-cardiac output syndrome impact factor journals postoperative low-cardiac output syndrome Scopus journals postoperative low-cardiac output syndrome PubMed journals postoperative low-cardiac output syndrome medical journals postoperative low-cardiac output syndrome free journals postoperative low-cardiac output syndrome best journals postoperative low-cardiac output syndrome top journals postoperative low-cardiac output syndrome free medical journals postoperative low-cardiac output syndrome famous journals postoperative low-cardiac output syndrome Google Scholar indexed journals post surgery articles post surgery Research articles post surgery review articles post surgery PubMed articles post surgery PubMed Central articles post surgery 2023 articles post surgery 2024 articles post surgery Scopus articles post surgery impact factor journals post surgery Scopus journals post surgery PubMed journals post surgery medical journals post surgery free journals post surgery best journals post surgery top journals post surgery free medical journals post surgery famous journals post surgery Google Scholar indexed journals
Article Details
1. Background
Cerebral oximetry is a non-invasive tool to monitor regional oxygen saturation of the frontal cortex. Using basic physical principles of different absorption and dispersion of infrared light, oxygenated and deoxygenated hemoglobin can be distinguished. Thus, cerebral oximetry can estimate oxygenation of blood approximately 25mm behind the skull [1]. In this region, it represents mostly venous oxygenation of blood, thus reflecting balance or imbalance of oxygen supply and demand. However, any given value by cerebral near infrared spectroscopy (NIRS) is mainly, but not exclusively a trend value, always dependent on its individual baseline. Average baseline value for cardiac surgical patients is 62%, ranging from 56 to 67% [1,2]. Also, there are various covariates influencing cerebral oxygenation, such as arterial blood oxygenation, concentration of hemoglobin, partial pressure of carbon dioxide, mean arterial pressure, depth of sedation and body temperature. Nevertheless, cerebral oxygenation also has a close relation to cardiopulmonary function, as systemic supply of oxygen is also depending on cardiac output [1]. Any reduction in cardiac output, due to systolic failure or venous congestion, may lead to reduced cerebral oxygenation. Currently, intra-operative use of NIRS during cardiac surgery is recommended by German guidelines to detect cerebral desaturation early and improve neurological outcome of patients. Guidelines also acknowledge the close correlation of cerebral oxygenation and central-venous/ mixed-venous oxygenation [2,3]. Yet, there is no recommendation or evidence base for routine use of NIRS in the postoperative course on intensive-care-units (ICU) after cardiac surgery in adult patients [2]. In theory, NIRS may limit the gap in early recognition of potential life-threatening complications present on cardiac ICU. This is suggested by three case series: van Roest and colleagues reported a case of pericardial tamponade during catheter ablation, in which a sudden drop in NIRS was the earliest indicator of hemodynamic compromise before other hemodynamic parameters (central venous pressure, mean arterial pressure) showed any obvious alteration [4]. Woodford was able to present a case series of nine patients undergoing cardiac surgery with NIRS monitoring [5]. From these patients, two developed a cardiac tamponade on intensive care unit, in both cases a sudden drop in cerebral saturation was the first indicator [5]. Kopp and colleagues demonstrated that tissue oxygen saturation (StO2) measured by NIRS of the thenar muscle was an indicator of delayed lactate clearance after cardiac surgery with higher sensitivity and specificity than cardiac index, central venous saturation and mean arterial pressure in 40 patients undergoing cardiac surgery [6]. They identified a threshold of StO2 <75% of baseline to be predictive of delayed lactate clearance on ICU. A prospective clinical trial to use NIRS for diagnosis of low cardiac output status (LCOS) in neonates and infants after cardiac surgery is currently under way and has completed enrollment of 35 neonates (NCT01849120). This study correlates NIRS measurements in neonates after cardiac surgery with venous lactate levels and blood oxygen saturation levels.
However, to date no prospective study investigates the use of NIRS on ICU after adult cardiac surgery with simultaneous continuous cardiac index monitoring to observe, if frequent hemodynamic complications, such as pericardial tamponade or other low output states, may be visible through a decline in NIRS values. There is evidence that LCOS may occur in 20% of chronic heart failure patients after coronary artery bypass grafting (CABG) [7] and diagnosis is often delayed until clinical signs become obvious. No data exist how NIRS correlates with cardiac index in such clinical scenarios and no threshold values for pathological changes in LCOS have been defined, while the general assumption is that any fall below 50% is pathologic. If cerebral desaturation is an early indicator of pericardial tamponade as suggested by Woodford and van Roest, this could be true for any form of LCOS after cardiac surgery.
The aim of this study is to investigate the diagnostic accuracy of NIRS for LCOS and the time between cerebral desaturation until clinical recognition of LCOS on ICU after cardiac surgery. The results of this observational trial may influence recommendations on hemodynamic monitoring of ICU post cardiac surgery. We anticipate showing that cerebral desaturation is an early and valid indicator of LCOS in these patients. This study may also generate hypotheses for future studies, investigating NIRS as an early indicator for more specific post-operative complications, such as pericardial tamponade or myocardial infarction. Also, data obtained from this study may serve as foundation for a randomized trial comparing continuous cardiac index monitoring with NIRS in cardiac surgical patients with reduced ejection fraction. The hypothesis of our study is, that in CABG patients with pre-existing reduced LVEF NIRS detects LCOS earlier than routine continuous cardiac index monitoring. Thus, the objective of this study is first to show that there is a close correlation between LCOS after cardiac surgery and cerebral desaturation measured via NIRS and additionally, to investigate sensitivity and specificity of the diagnostic test for this complication. As reference standard, routine Swan-Ganz catheter measurements, blood gas analysis and standard hemodynamic parameters will be documented. Only patients with formal indication to receive Swan-Ganz catheter will be included (according to official guideline LVEF<30% prior to surgery). Findings of our research could impact guideline recommendations on routine hemodynamic monitoring on cardiac ICU.
Methods/Design
General methods
The target condition to be tested is low cardiac output syndrome (LCOS), with cerebral NIRS as the index test and Swan Ganz catheter measurements with continuous cardiac index monitoring as reference standard. For cerebral NIRS measurement, INVOS™ 5100C produced by Medtronic Inc. (Minneapolis, MN, USA) will be used, allowing for data transfer of continuous measurements on a USB portable device and data analysis on a dedicated software (INVOS™ Monitoring System Analytics Tool). Informed consent will be obtained before initiation of cardiac surgery if patients meet the inclusion criteria. Also, formal consent to receive Swan-Ganz catheter will be obtained. A Swan-Ganz catheter is per guideline recommendation indicated in these patients with severely reduced ejection fraction since risk for postoperative low-cardiac output syndrome is as high as 20%.
Study visits
At induction of anesthesia, baseline values for NIRS, Swan-Ganz catheter measurements, blood gas analysis and standard hemodynamic measurements will be obtained (see table 1). Upon admission to ICU, screening is repeated, and patients may be excluded according to inclusion/exclusion criteria and thereafter handled as screening failures were further data is not collected or included as per protocol (see figure 1). If the patient still fulfills all inclusion criteria, measurements will be performed according to protocol for up to 48 hours. If Swan-Ganz catheter is no longer clinically indicated (i.e. the patient is extubated an without inotropes) before the 48hr post-operative period is completed, it may be removed. The study visits with documentation of hemodynamic values are completed by 48 hours post procedure. Patients will be contacted 30-days post surgery to identify 30-day survival.
|
Study Period |
||||
|
Enrollment |
Baseline |
Follow-up |
||
|
Timepoint |
0 |
0 |
t1: Admission to ICU |
t2-t26: 1h-48h post-procedure |
|
Screening |
X |
X |
X |
|
|
Informed Consent |
X |
|||
|
Cerebral saturation (NIRS) |
||||
|
Swan Ganz Catheter measurements (PCWP, CI, PVRI, SVRI) |
X |
X |
1-24h, 24-48h if further indicated |
|
|
Central Venous Pressure (mmHg) |
||||
|
Serum Hemoglobin mmol/L |
X |
X |
X |
X |
|
FiO2 |
X |
X |
X |
|
|
Partial CO2 Pressure (kPa) |
X |
X |
X |
|
|
Mean Arterial Pressure mmHg |
||||
|
Heart rate (min-1) |
||||
|
Body Temperature (°C) |
X |
X |
X |
|
|
Arterial SO2 (%) |
||||
|
Arterial pH |
X |
X |
X |
|
|
Base deficit |
X |
X |
X |
|
|
Central Venous SO2 (%) |
X |
X |
X |
|
|
Mixed Venous SO2 (%) |
X |
X |
As per hospital standard |
|
|
Arterial Lactate (mmol/L) |
X |
X |
X |
|
|
Dosage of Inotropes |
||||
|
Urine output (ml/h) |
X |
X |
||
|
Drain output (ml/h) |
X |
X |
||
--> Indicates continuous measurements of values
X Indicates discontinuous measurement of values at set time points
Abbreviations: NIRS = Near infrared spectroscopy; PCWP = Pulmonary capillary wedge pressure; CI = Cardiac index; PVRI = Pulmonary vascular resistance index; SVRI = Systemic vascular resistance index; FiO2 = Fractional concentration of oxygen during inspiration; ICU = Intensive care unit.
Baseline: Induction of general anesthesia
Table 1: Study visits
Primary Endpoint
The primary endpoint is LCOS and its timepoint of onset. During the 48 hours post-operative period, exact time of diagnosis of LCOS – if the endpoint is reached – is documented. Diagnosis of LCOS is based on the definition given by the study protocol and not the NIRS measurements itself.
LCOS is defined as the occurrence of the following conditions:
1) Two consecutive measurements of low cardiac output (defined as a cardiac output of ≤ 2.2 L/min/m2, without associated relative hypovolaemia) after calibration OR
2) One measurement of low cardiac output ≤ 2.2 L/min/m2 plus the use of two or more inotropes OR
3) If baseline cardiac index was ≤ 2.2 L/min/m2, then any measurement showing a reduction of >20% of its pre-operative value will be counted as LCOS.
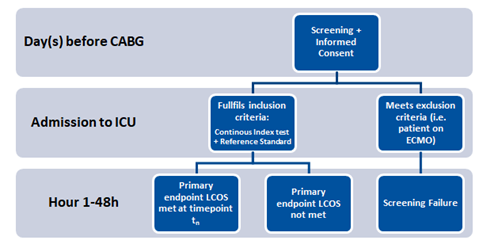
Figure 1: Screening process and endpoint assessment.
Proposed sample size and statistical analysis
The incidence of LCOS in the study population is considered to be 20%. Assuming that 68 individuals are enrolled in the study and a significant change of NIRS is defined at 12% decline (62% average to 50%), difference in NIRS in LCOS and non-LCOS can be shown with a standard deviation of 10, a power of 95% and alpha of 0.05. In this calculation, drop outs are already considered. Minimum number of 60 patients would be necessary without drop outs. Therefore, we plan the inclusion of at least 68 individuals. After inclusion of 68 individuals, number of screening failures and incidence of LCOS will be analysed and more patients may be included if necessary for exploratory statistical analysis (maximum 100 patients).
NIRS is not only influenced by cardiac output – NIRS values are driven by various factors, which in our study act as confounders. They will be collected during the process of the study and adjusted for statistically. The most important covariates with influence on NIRS are (but not exclusively), hemoglobin value, carbon dioxide partial pressure, arterial oxygen saturation, systemic vascular resistance and blood pressure. Variations in these values may affect NIRS measurements and lead to a drop not related to any form of LCOS. Some of these values, in an intensive care setting, are somewhat controlled for – hemoglobin values are maintained at levels above 8g/dL through patient blood management and mean blood pressure is maintained at a minimum value of 60mmHg to ensure adequate organ perfusion and arterial oxygen saturation is maintained above 96% through mechanical ventilation or other form of oxygen supply. However, these variables may show acute significant variations, even in a controlled intensive care setting. Therefore, it is imperative, to not only collect those variables during the study, but also to adjust for them statistically during analyses. As blood pressure is a direct effect of cardiac output, we may not adjust for that since overadjustment can lead to inconclusive results. The amount of variables one can adjust for is limited by the amount of events that occurred. With an expected sample size of 68 patients and an expected incidence of LCOS in 14 cases, statistically one could adjust for one or two variables. Consequently, we plan to adjust for hemoglobin values and systemic vascular resistance via logistic regression analysis. Other variables, such as carbon dioxide partial pressure levels, arterial oxygen saturation and temperature, may also influence NIRS measurements and will have to be taken into account when looking at the results of our study. NIRS measurements will be analyzed using Receiver-Operating Curve (ROC) Analysis for Sensitivity and Specificity for detecting LCOS in our patients.
Data management
Baseline and peri-operative data as well as hemodynamic measurements will be collected via RedCap. NIRS Data will be obtained on a portable USB device and analyzed on a dedicated software (INVOS™ Monitoring System Analytics Tool) and ultimately exported to Microsoft Excel. Data will be published in accordance with the STARD criteria [8].
Feasibility of recruitment / Trial sites
The study will be monocentric. We perform >600 Coronary artery bypass grafting surgeries (CABG) at our center annually. Not all CABG patients will be eligible for participation since severely reduced ejection fraction <35% is only present in a share of patients. All CABG patients with severely reduced ejection fraction may be included in the trial, therefore recruitement of 68 patients over a period of 24 months seems feasible.
Inclusion Criteria
Subjects will only be included in the study if they meet all of the following criteria:
- Patients ≥18 years undergoing on-pump or of-pump coronary artery bypass grafting. The procedure may be combined with other cardiac procedures (e.g. valve surgery).
- The surgery is elective or urgent (either planned or within one hospital admission between diagnosis and treatment), can be for ST-Elevation or non-ST-Elevation myocardial infarction (STEMI/NSTEMI) or any form of acute coronary syndrome (ACS).
- Pre-operative left ventricular function is <35% as measured by left ventricular angiography or transthoracic echocardiography by simpson biplane method.
- The patient gives written informed consent to take part in the study.
- The patient is hemodynamically stable upon admission to ICU (max. dose epinephrine ≤0.05μg/kg/min, dobutamine ≤3μg/kg/min, enoximone/milrinone ≤2μg/kg/min, Noradrenaline ≤0.2μg/kg/min).
- No metabolic acidosis is present on admission to ICU (Base excess ≥ -10, pH >7.30).
- Upon admission to ICU, Cardiac index is ≥80% of the pre-operative value.
Exclusion Criteria
Subjects will not be included in the study if any of the following criteria applies:
- Manifest LCOS (see definition in primary endpoint section) upon admission to ICU.
- Cardiac arrest with external cardiopulmonal resuscitation prior to admission to ICU.
- Patient is on extracorporal life-support or has an intra-aortic ballon pump (IABP) upon admission to ICU.
- Procedure was performed as salvage procedure (i.e. with cardiopulmonary rescucitation prior to initiating cardiopulmonary bypass, patient was unstable upon transfer to operating theatre “sliding on inotropes”).
- Subject without legal capacity who is unable to understand the nature, scope, significance and consequences of this clinical trial.
Discussion
We present a study for further evaluation of the role of cerebral venous oxygenation during LCOS. Patients undergoing cardiac surgery present a valuable cohort for analysis of this clinical scenario, since those with pre-operative reduced left ventricular ejection fraction <30% are at specific risk to develop some form of LCOS in the early postoperative course. Current guideline recommendations favor invasive hemodynamic monitoring in those patients on cardiac ICUs. This study may gather evidence, if continuous NIRS monitoring may be advantageous as a non-invasive technique detecting LCOS during the early post-operative course. To date, this is the only study to simultaneously correlate continuous cardiac index and continuous NIRS through a 48hr postoperative period in adult cardiac surgical patients. Three publications already hinted at the significance of NIRS monitoring in LCOS through case reports and case series [4-6]. This study may provide sufficient data to either evaluate NIRS monitoring in other clinical scenarios or to initiate a randomized controlled trial for comparison of these diagnostic tools in LCOS patients. Since NIRS measurements are not blinded, the values themselves may lead to further diagnostics and clinical decision-making. Since this is only a pilot study to correlate NIRS values with continuous cardiac index, this will not interfere with our research hypothesis. Swan-Ganz continuous cardiac index is an invasive but valuable diagnostic tool in situations like LCOS, but may lead to complications such arrhythmias and perforation. For this reason, it has been used less during the last decade [2]. Another form of hemodynamic monitoring, arterial pulse contour analysis, is increasingly used and may replace Swan-Ganz catheter measurements in some clinical scenarios. However, Swan-Ganz still acts as the reference method to evaluate cardiac function and vascular resistance. Moreover, it offers estimation of left-ventricular pre-load and evaluation of pulmonary hypertension, which seems advantageous in cardiac surgical patients. Moreover, methods like arterial pulse contour analysis or repeated transeosophageal echocardiograms to assess cardiac functions still are invasive. Therefore, it seems justified to evaluate a non-invasive, readily available alternative monitoring device for such a clinical scenario as LCOS.
Trial status
We were able to enroll 79 patients in this study so far. However, due to necessity for mechanical circulatory support, a significant share of patients counts as screening failure. Also, rate of LCOS seems lower as expected so far, therefore enrollment is ongoing. Completion is expected for July 2023.
List of abbreviations
ACS Acute coronary syndrome
CABG Coronary Artery Bypass Grafting
CI Cardiac index
FiO2 Fractional concentration of oxygen during inspiration
ICU Intensive Care Unit
LCOS Low Cardiac Output
NIRS Near infrared spectroscopy
NSTEMI Non-ST-Elevation myocardial infarction
PCWP Pulmonary capillary wedge pressure
PVRI Pulmonary vascular resistance index
ROC Receiver Operating Curve
STEMI ST-Elevation myocardial infarction
SVRI Systemic vascular resistance index
Declarations
Ethics approval
The study was approved by the ethics committee of the University Hospital Bonn (No.: 119/19).
Availability of data and materials
Not applicable.
Competing interests
MS received support from Medtronic through a research support agreement, providing equipment needed for the study as items of loan. All other authors declare that they have no competing interests with regard to this study.
Funding
The study is funded through a research grant of the Committee for clinical studies at the University Hospital Bonn. Additionally, the study is supported through a research support agreement with Medtronic. The INVOS 5100 monitoring devices are provided as items of loan for the duration of the study by Medtronic Inc. (Minneapolis, MN, USA). None of the funding parties have a role in study design, data collection, analysis or interpretation of data and also not in writing the final manuscript after data analysis.
Author´s contributions
MS is the main responsible person for design, enrollment, data collection and analysis. MV, MR, MT, JK, CP, MH, DR, FB all participate in the conductance of the clinical trial, patient enrollment and monitoring and they all read and approved the final version of the manuscript.
Acknowledgments
None.
References
- Schön JHM, Berger KU, Groesdonk V, et al. Relationship between mixed venous oxygen saturation and regional cerebral oxygenation in awake, spontaneously breathing cardiac surgery patients. Minerva Anestesiol. 77 (2011): 952-958.
- Habicher M, Zajonz T, Heringlake M, et al. S3 guidelines on intensive medical care of cardiac surgery patients : Hemodynamic monitoring and cardiovascular system-an update. Anaesthesist 67 (2018): 375-379.
- Moerman A, Vandenplas G, Bové T, et al. Relation between mixed venous oxygen saturation and cerebral oxygen saturation measured by absolute and relative near-infrared spectroscopy during off-pump coronary artery bypass grafting. Br J Anaesth. 2013;110/2: 258-265.
- Van Roest MH, Vercauteren M, Vrints C, et al. Early recognition of cardiac tamponade using cerebral oximetry during ventricular tachycardia ablation. Eur J Anaesthesiol 32 (2015): 581-582.
- Woodford S. 277: NIRS as a tool in the cardiac ICU defining the niche for a new ICU technology. Critical Care Medicine 41 (2013): A64.
- Kopp R, Dommann K, Rossaint R, et al. Tissue oxygen saturation as an early indicator of delayed lactate clearance after cardiac surgery: a prospective observational study. BMC anesthesiology 15 (2015): 158.
- Algarni KD, Maganti M, Yau TM. Predictors of low cardiac output syndrome after isolated coronary artery bypass surgery: trends over 20 years. Ann Thorac Surg 92 (2011): 1678-1684.
- Bossuyt PM, Reitsma JB, Bruns DE, et al. STARD 2015: an updated list of essential items for reporting diagnostic accuracy studies. BMJ (Clinical research ed.) 351 (2015): 5527.
