Placental Fatty Acid Metabolism and Transport in a Rat Model of Gestational Diabetes Mellitus
Article Information
Jay S Mishra1, Sathish Kumar1, 2, 3*
1Department of Comparative Biosciences, School of Veterinary Medicine, University of Wisconsin, Madison, Wisconsin, United States of America
2Department of Obstetrics and Gynecology, School of Medicine and Public Health, University of Wisconsin, Madison, Wisconsin, United States of America
3Endocrinology-Reproductive Physiology Program, University of Wisconsin, Madison, Wisconsin, United States of America
*Corresponding author: Sathish Kumar, Department of Comparative Biosciences, School of Veterinary Medicine, University of Wisconsin, Madison, Wisconsin, United States of America.
Received: 30 March 2023; Accepted: 07 April 2023; Published: 04 May 2023
Citation: Jay S Mishra, Sathish Kumar. Placental Fatty Acid Metabolism and Transport in a Rat Model of Gestational Diabetes Mellitus. Journal of Women’s Health and Development 6 (2023): 56-67.
View / Download Pdf Share at FacebookAbstract
Gestational Diabetes Mellitus (GDM) is a form of heightened insulin resistance triggered during gestation. This study examines how insulin resistance alters placental Long-Chain Polyunsaturated Fatty Acid (LCPUFA) transport and metabolism in a rat model of lean GDM. Pregnant Sprague Dawley rats were administered with S961, an insulin receptor antagonist (30 nmol/kg s.c. daily), or vehicle from Gestational Day (GD) 7 to 20. Daily maternal body weight, food, and water intake were measured. Blood pressure assessment and glucose tolerance test were done on GD20. Fetal plasma and placenta were collected on GD20 and processed for fatty acid measurement using LC-mass spectrometry. The expression of fatty acid metabolism-related genes in the placenta was assessed using RT2 Profiler PCR arrays. The results were validated by qRT-PCR. Blockade of insulin receptors with S961 in pregnant rats resulted in glucose intolerance with increased fasting glucose and insulin levels. Maternal body weight gain, food, and water intake were not affected; however, S961 significantly increased maternal blood pressure and heart rate. The placenta n3 and n6 LCPUFA concentrations were significantly decreased by 8% and 11%, respectively, but their levels in the fetal plasma were increased by 15% and 4%. RT2 profiler arrays revealed that placental expressions of 10 genes related to fatty acid β-oxidation (Acaa1a, Acadm, Acot2, Acox2, Acsbg1, Acsl4, Acsm5, Cpt1b, Eci2, Ehhadh) and 3 genes related to fatty acid transport pathway (Fabp2, Fabp3, Slc27a3) were significantly upregulated. In summary, lack of insulin action increased the expression of genes related to placental fatty acid β-oxidation and transport with an increased transfer of LCPUFA to the fetus. The increased lipid levels routed toward the fetus may lead to fat adiposity and later-life metabolic dysfunction.
Keywords
Fatty Acid; Gestational Diabetes; Insulin Resistance; Pregnancy; Placenta
Fatty Acid articles Fatty Acid Research articles Fatty Acid review articles Fatty Acid PubMed articles Fatty Acid PubMed Central articles Fatty Acid 2023 articles Fatty Acid 2024 articles Fatty Acid Scopus articles Fatty Acid impact factor journals Fatty Acid Scopus journals Fatty Acid PubMed journals Fatty Acid medical journals Fatty Acid free journals Fatty Acid best journals Fatty Acid top journals Fatty Acid free medical journals Fatty Acid famous journals Fatty Acid Google Scholar indexed journals Gestational Diabetes articles Gestational Diabetes Research articles Gestational Diabetes review articles Gestational Diabetes PubMed articles Gestational Diabetes PubMed Central articles Gestational Diabetes 2023 articles Gestational Diabetes 2024 articles Gestational Diabetes Scopus articles Gestational Diabetes impact factor journals Gestational Diabetes Scopus journals Gestational Diabetes PubMed journals Gestational Diabetes medical journals Gestational Diabetes free journals Gestational Diabetes best journals Gestational Diabetes top journals Gestational Diabetes free medical journals Gestational Diabetes famous journals Gestational Diabetes Google Scholar indexed journals Insulin Resistance articles Insulin Resistance Research articles Insulin Resistance review articles Insulin Resistance PubMed articles Insulin Resistance PubMed Central articles Insulin Resistance 2023 articles Insulin Resistance 2024 articles Insulin Resistance Scopus articles Insulin Resistance impact factor journals Insulin Resistance Scopus journals Insulin Resistance PubMed journals Insulin Resistance medical journals Insulin Resistance free journals Insulin Resistance best journals Insulin Resistance top journals Insulin Resistance free medical journals Insulin Resistance famous journals Insulin Resistance Google Scholar indexed journals Pregnancy articles Pregnancy Research articles Pregnancy review articles Pregnancy PubMed articles Pregnancy PubMed Central articles Pregnancy 2023 articles Pregnancy 2024 articles Pregnancy Scopus articles Pregnancy impact factor journals Pregnancy Scopus journals Pregnancy PubMed journals Pregnancy medical journals Pregnancy free journals Pregnancy best journals Pregnancy top journals Pregnancy free medical journals Pregnancy famous journals Pregnancy Google Scholar indexed journals Placenta articles Placenta Research articles Placenta review articles Placenta PubMed articles Placenta PubMed Central articles Placenta 2023 articles Placenta 2024 articles Placenta Scopus articles Placenta impact factor journals Placenta Scopus journals Placenta PubMed journals Placenta medical journals Placenta free journals Placenta best journals Placenta top journals Placenta free medical journals Placenta famous journals Placenta Google Scholar indexed journals Gestational diabetes mellitus articles Gestational diabetes mellitus Research articles Gestational diabetes mellitus review articles Gestational diabetes mellitus PubMed articles Gestational diabetes mellitus PubMed Central articles Gestational diabetes mellitus 2023 articles Gestational diabetes mellitus 2024 articles Gestational diabetes mellitus Scopus articles Gestational diabetes mellitus impact factor journals Gestational diabetes mellitus Scopus journals Gestational diabetes mellitus PubMed journals Gestational diabetes mellitus medical journals Gestational diabetes mellitus free journals Gestational diabetes mellitus best journals Gestational diabetes mellitus top journals Gestational diabetes mellitus free medical journals Gestational diabetes mellitus famous journals Gestational diabetes mellitus Google Scholar indexed journals pathogenesis articles pathogenesis Research articles pathogenesis review articles pathogenesis PubMed articles pathogenesis PubMed Central articles pathogenesis 2023 articles pathogenesis 2024 articles pathogenesis Scopus articles pathogenesis impact factor journals pathogenesis Scopus journals pathogenesis PubMed journals pathogenesis medical journals pathogenesis free journals pathogenesis best journals pathogenesis top journals pathogenesis free medical journals pathogenesis famous journals pathogenesis Google Scholar indexed journals hypertension articles hypertension Research articles hypertension review articles hypertension PubMed articles hypertension PubMed Central articles hypertension 2023 articles hypertension 2024 articles hypertension Scopus articles hypertension impact factor journals hypertension Scopus journals hypertension PubMed journals hypertension medical journals hypertension free journals hypertension best journals hypertension top journals hypertension free medical journals hypertension famous journals hypertension Google Scholar indexed journals infections articles infections Research articles infections review articles infections PubMed articles infections PubMed Central articles infections 2023 articles infections 2024 articles infections Scopus articles infections impact factor journals infections Scopus journals infections PubMed journals infections medical journals infections free journals infections best journals infections top journals infections free medical journals infections famous journals infections Google Scholar indexed journals cesarean section articles cesarean section Research articles cesarean section review articles cesarean section PubMed articles cesarean section PubMed Central articles cesarean section 2023 articles cesarean section 2024 articles cesarean section Scopus articles cesarean section impact factor journals cesarean section Scopus journals cesarean section PubMed journals cesarean section medical journals cesarean section free journals cesarean section best journals cesarean section top journals cesarean section free medical journals cesarean section famous journals cesarean section Google Scholar indexed journals
Article Details
Introduction
Gestational diabetes mellitus (GDM) is a growing health problem worldwide, and the number of affected women is constantly increasing [1]. GDM is marked by elevated glucose levels in the bloodstream. The pathogenesis of GDM involves heightened pathological insulin resistance, which is characterized by a loss and/or insensitivity of insulin receptors in body cells. As a result, the cells become less responsive to the effects of insulin, ultimately leading to elevated blood glucose levels [2]. GDM prevalence varies depending on ethnicity and geographic location and is between 1% and 30% of all pregnancies [3]. GDM women are more prone to pregnancy complications such as hypertension, infections, cesarean section, and long-term adverse effects, such as type 2 diabetes and cardiovascular diseases [4]. Additionally, children born to mothers with GDM are at increased risk of obesity, metabolic syndrome, and diabetes later in life [5]. These adverse effects of GDM have profound public health implications and highlight the urgency of understanding the underlying mechanisms. The placenta plays a crucial role in regulating the exchange of nutrients between the mother and fetus [6]. The placenta progressively adapts its structure and function to meet the growing fetal demands and also responds to changes in maternal nutrient availability [7]. GDM is known to affect the growth and function of the placenta. For example, studies show glucose flux across the placenta to the fetus, and the expressions of glucose transporters and glycolytic genes are increased in GDM [8-10]. In addition to glucose, the placenta also transports n-3 and n-6 long-chain polyunsaturated fatty acids (LCPUFAs), which play a vital role in fetal growth and development; however, there is no consensus on how GDM affects placental LCPUFA metabolism and transport. Some studies show that the expression of placental fatty acid transporters and the level of essential fatty acids in cord blood are reduced in GDM [11, 12]. However, no such difference was observed in other studies [13]. In contrast, other studies show that placental fatty acid transporters were upregulated in diabetic women [14, 15]. The reasons for such inconsistencies are undetermined. One possible explanation could be that studies on GDM are conducted inconsistently in overweight and obese women. Animal studies also use high-fat diets to model GDM features [16]. Obesity and high-fat consumption, independent of GDM, are known to influence placental lipid metabolism [17-19]. Also, GDM modeled with diabetogenic drugs like streptozotocin have pertinent drawbacks of causing non-specific free radical damage [20, 21]. Hence, dissecting the direct effect of insulin resistance, the key feature of GDM, on placental lipid metabolism in lean subjects is necessary for evaluating intrinsic abnormalities. In this study, we used an animal model to establish insulin resistance and mimic GDM pathophysiology by administering a specific insulin receptor blocker, S961, to pregnant rats. This model has been shown to induce hyperglycemia, hyperinsulinemia, and glucose intolerance in rodents without causing obesity or changes in feed intake [22-24]. Using this model, we examined the impact of insulin resistance on placental lipid transport and metabolism. Experiments were designed to investigate (i) whether body weight, glucose tolerance, plasma insulin levels, and blood pressure are altered in S961-treated compared with control dams; (ii) whether the placental and fetal plasma n-3 and n-6 LCPUFA concentrations are altered in S961-treated compared with control dams; and (iii) whether the expression of genes related to fatty acid transport and fatty acid β-oxidation are altered in the placenta of S961-treated compared with control dams.
Materials and Methods
Animals and Treatment
All experiments were approved by Animal Care and Use Committee at the University of Wisconsin-Madison and conducted in accordance with the NIH guidelines (NIH Publication No. 85–23, revised 1996). Timed-pregnant Sprague-Dawley rats at gestational day (GD) 5 (day of positive plug = GD 1) were obtained from Envigo (Indianapolis, IN) and housed under a temperature- and light-controlled room (25°C, 12:12-h light/dark cycle) with water and food provided ad libitum. After acclimatization, the dams were randomly divided into two groups and injected subcutaneously with either saline (i.e., the control group) or insulin receptor antagonist S961 (30 nmol/kg/day; MedChem Express, Monmouth Junction, NJ, USA) from GD7 to GD20. The dosing regimen of S961 administration was chosen based on previous studies [24-26] and our initial pilot studies. Maternal body weight, food, and water intake were recorded throughout the study period. Intraperitoneal glucose tolerance test and blood pressure measurement were done at GD18 and GD20, respectively. After collecting fetal blood under isoflurane anesthesia, dams were sacrificed on GD20 to collect placenta for fatty acid analysis.
Glucose and Insulin Measurement
An automatic glucometer (StatStrip; Nova Biomedical, Waltham, MA) was used to measure blood glucose levels. Insulin levels were measured using a rat insulin ELISA kit (Mercodia, Winston Salem, NC) following the manufacturer's instructions. Briefly, heparinized blood was centrifuged for 15 min at 3000 rpm and 4°C for plasma isolation, and 10 μl of plasma or calibrators and 100 μl of enzyme conjugate were mixed and held at room temperature for 2 h with constant shaking. After washing, 200 μl of 3,3',5,5'-tetramethylbenzidine substrate was added and incubated for 15 min for color development. The reaction was stopped with the addition of 50 μl of stop solution. Absorbance was measured at 450 nm, and the results were calculated with cubic spline regression fit using data analysis software (SpectraMax i3x plate reader, Molecular Devices; San Jose, CA). The sensitivity of the insulin assay is 0.07 g/L, with an inter-and intraassay precision of 3.3 and 1.8, respectively.
Intraperitoneal Glucose Tolerance Test
The intraperitoneal glucose tolerance test was performed after fasting overnight (12 h). The tip of the rat tail was snipped for blood collection. The first drop of blood was discarded, and the second was used to determine glucose levels (time 0) using a glucometer as described before [27]. After that, glucose (2 g/kg body weight) was administered intraperitoneally, and blood samples were collected from the tail tip at 15, 30, 60, and 120 min for blood glucose measurements. The data were graphed, and the area-under-glucose-curve (AUC) was calculated.
Systemic Blood Pressure and Heart Rate Measurement
Mean blood pressure was recorded using a computerized noninvasive CODA system (Kent Scientific, Litchfield, CT, USA) as in our previous studies [28, 29]. Rats were acclimatized in a pre-warmed Plexiglas restrainer for 15 min for 2 consecutive days, and on the third day, blood pressure was measured. An occlusion cuff and a volume pressure-recording cuff were applied to the base of the tail. The cuff was programmed to inflate and deflate automatically within 90 s. The data from the last 5 cycles were taken as the mean blood pressure for that rat. All recordings and data analyses were done using Kent Scientific Software.
Fetal Plasma and Placenta Collection
Rats were anesthetized using isoflurane, and maternal laparotomy was done to expose individual fetal units. Fetal blood was collected by nicking the left ventricle of the heart using a 26G BD precision slide needle and 1 ml syringe. Collected samples from six to eight fetuses of individual dams were pooled in lightly heparinized tubes for plasma separation. After blood collection, the dams were perfused with heparinized saline. Then placentas were collected, rinsed thoroughly in ice-chilled saline, blotted with Whatman filter paper, snap-frozen in liquid nitrogen, and stored at −80 °C until further processing.
Measurement of LCPUFA in Placenta and Fetal Plasma
Total fatty acid extraction from placental tissues was performed according to Brown et al. [30]. In brief, 40 mg of frozen placental tissue was homogenized in 400 μl methanol containing 50 μg/ml of butylated hydroxytoluene (BHT) using a bead homogenizer. The homogenate was vortexed for 2 min and left on ice for 2 min (repeated thrice). Insoluble material was cleared by centrifugation at 16000g for 5 min at 4 °C. The supernatant (25 μl) was added to 250 μl MTBE and vortexed overnight at 4 °C. Ammonium acetate (150 μl of 100 mM) was added for phase separation. The upper organic layer was transferred to a new vial, dried in a speed vac, and resuspended in 125 μl of methanol: dichloromethane (1:1 v/v) containing 2 mM ammonium acetate before mass spec analysis. All samples were kept at concentrations below 20 μM to minimize ion suppression effects [31]. For fetal plasma, fatty acid extraction was performed using chloroform: methanol 2:1 (v/v) containing BHT to prevent oxidation during sample preparation [32]. Five microliters of plasma were mixed with 1.5 ml of chloroform: methanol (2: 1 v/v). To this mixture, 300 μl of water was added to facilitate phase separation. The bottom phase was removed, mixed with 500 μl of chloroform:methanol: water (3/48/47; v/v/v), vortexed, and centrifuged at 16000g for 5 min at 4 °C. The lower phase containing lipids was dried in a speed vac and reconstituted with 200 μl of methanol: dichloromethane (1:1 v/v) containing 2 mM ammonium acetate before mass spec analysis. Mass spectra were acquired using an ABSciex TripleTOF 5600 (Sciex, Foster City, CA) equipped with an electrospray interface, 25 μm iD capillary, and coupled with Eksigent μUHPLC (Eksigent, Redwood City, CA) as per Tran et al. [33]. Data processing and acquisition were made with Analyst TF 1.7 software. Data in both positive and negative ionization modes were analyzed with LipidView (ABSciex) software version 1.2, including smoothing, identification, removal of isotope contribution from lower mass species, and correction for isotope distribution. Methanol: dichloromethane (1:1 v/v) containing 2 mM ammonium acetate was used as mobile phase.
RT2 Profiler PCR Array
Total RNA was extracted using RNeasy plus Universal Kit (Qiagen, Valencia, CA), and RNA concentration and integrity were determined by Nanodrop spectrophotometer (ThermoFisher Scientific, Newark, DE). Two micrograms of RNA from each sample were reverse transcribed to complementary DNA (cDNA) using RT2 First Strand Kit (Qiagen). RT2 Profiler PCR Array (PARN-007Z) for 84 fatty acid metabolism pathway genes was performed in 96-well plates in CFX Connect™ Real-Time Detection System (Bio-Rad, USA). The relative gene expression was calculated using Qiagen web-based PCR Array Data Analysis Software and normalized with the average of 5 housekeeping genes contained in the array. The results from three experiments are expressed as fold changes in the GDM placenta compared to controls.
Quantitative Real-Time PCR Validation
RNA extraction and cDNA synthesis was done as described for RT2 profiler PCR array. After dilution, cDNA corresponding to 20 ng of RNA was amplified using SsoAdvanced Universal SYBR Green Supermix (Bio-Rad, Hercules, CA) and CFX Connect real-time thermal cycler (Bio-Rad). Gene-specific primer sequences are shown in Table 2. The 2−??CT method was used for calculation, and the results were expressed as a fold change of the gene of interest in GDM versus control placenta. All reactions were performed in duplicate, and β-Actin was used as an internal control.
|
Gene |
Forward |
Reverse |
|
Aca1a |
ACAGGAAAACCATCACCGTGTCTC |
TGTCTATGTCATTCACAGTCAGCCC |
|
Acadm |
GGCATATGGGTGTACAGGGG |
ACGCAGTAGGCACACATCAT |
|
Acot2 |
CCTGGTGGGTGCTAACATCA |
AGGGGGCACCTCCTAACTC |
|
Acox2 |
CACAGCTCCTACAGTGCTATTC |
GCCTTCATACCAGTGCTCAA |
|
Cpt1b |
CAGCCATGCCACCAAGATC |
AAGGGCCGCACAGAATCC |
|
ECi2 |
GTCACCCTTCTGGGACTATTTG |
GAGGTGGCTGAATGGAGTATG |
|
Fabp2 |
AAGGAATAGGCCAGCTTCTTG |
CAGTGAGTGAGCCTGCATTAT |
|
Fabp3 |
CGGTACCTGGAAGCTAGTGG |
TCATCTGCTGTGACCTCGTC |
|
Cd36 |
AGCAAGATCAAGGTTAAACAGAGAGGTCC |
CATTGGGTTGTACAAAAGAGACAGTGCT |
|
Fatp6 |
CACGTCACCTCAAGCCTTAAGTCTACTT |
CTTCAACACTTGGAACTGGCTAATCACAG |
|
Fatp1 |
CTGGCGCTTTCTGCGTATCGTCTGCA |
TCTCCTGCTCGCCGGTGTCGTCGTA |
|
Fatp3 |
GCCTGTACATCTTCACCTCTGGCACTA |
GACTCCACAAAGCTGGTAGAATCCCTG |
|
Fatp4 |
ACGAGCCCTCATCTTTGGCAGCGAGAT |
AGGAACCAGAGCAGAAGAGGGTGAGTG |
Table 1: Rat-specific quantitative real-time PCR primer sequence.
Statistical Analysis
Data were analyzed with GraphPad Prism (GraphPad Software, San Diego, CA) and presented as mean ± SE. Significant differences were assessed by unpaired Student's t-test or one-way ANOVA followed by Newman–Keuls tests. The normality and homogeneity of the variances were analyzed with the one-sample Kolmogorov–Smirnov test and Anderson-Darling test, respectively. When the data were not normally distributed, a non-parametric Kruskal–Wallis test, with Dunn’s multiple comparisons, was used. Differences with a P-value of less than 0.05 were considered statistically significant.
Results
S961 altered Fasting Glucose and Insulin Levels
The fasting plasma glucose (7.23 ± 0.37 vs. 3.96 ± 0.31 mmol/L, n = 6, P < 0.05, Fig 1A) and insulin levels (1106.20 ± 29.11 vs. 115.07 ± 28.06 pmol/L, n = 6, P < 0.05, Fig 1B) were significantly higher in S961 treated pregnant rats than in controls. S961 treatment did not affect maternal weight gain as the daily changes in body weight and the total gestational weight gain were similar to controls (Figs 1C and D). Similarly, placental, and fetal weights were unchanged between groups. In addition, daily food and water intake in S961-treated pregnant rats were similar to controls throughout gestation (Figures 1E and F).
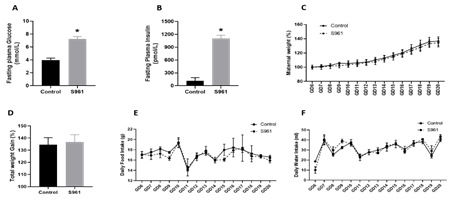
Figure 1: Changes in (A) fasting glucose, (B) fasting insulin, (C) maternal weight, (D) total maternal weight gain, (E) daily food intake, and (F) daily water intake in pregnant rats treated from GD7 to GD20 with S961 or vehicle. All data are expressed as means ± SE of six dams in each group; ∗P < 0 .05 vs. control.
S961 Dysregulates Glucose Tolerance and Insulin Responses
An intraperitoneal glucose tolerance test was performed to characterize the effects of S961 treatment on glucose handling and insulin responses. After 2 g/kg glucose administration, plasma glucose levels were significantly higher in S961-treated pregnant rats compared to controls at all-time points; 15 min (20.59 ± 0.56 vs. 15.41 ± 0.35 mmol/L, P < 0.05), 30 min (20.05 ± 1.98 vs. 14.39 ± 0.80 mmol/L, P < 0.05), 60 min (16.69 ± 1.39 vs. 7.11 ± 0.42 mmol/L, P < 0.05), 90 min (14.17 ± 1.28 vs. 6.41 ± 0.31 mmol/L, P < 0.05), and 120 min (12.18 ± 0.75 vs. 5.73 ± 0.13 mmol/L, P < 0.05); n = 6 in each group (Fig 2A, top panel). Area under the curve (AUC) analysis (mmol/L × 120 min) of glucose responses (Figure 2A, bottom panel) revealed significant glucose intolerance in S961-treated pregnant rats (1923.0 ± 110.1, n = 6, P < 0.05) compared with controls (1077.0 ± 38.87, n = 6). In response to an intraperitoneal glucose challenge, S961-treated pregnant rats exhibited a significant increase in plasma insulin levels compared to controls at 15 min (1099.96 ± 24.35 vs. 633.07 ± 162.85 pmol/L, P < 0.05), 30 min (1068.73 ± 48.70 vs. 593.14 ± 160.98 pmol/L, P < 0.05), 60 min (936.16 ± 68.55 vs. 601.64 ± 165.11 pmol/L, P < 0.05), 90 min (812.21 ± 53.13 vs. 434.48 ± 155.43 pmol/L, P < 0.05), and 120 min (840.26 ± 39.03 vs. 274.02 ± 50.82 pmol/L, P < 0.05); n = 6 in each group (Fig 2B, top panel). The overall plasma insulin responses, expressed as insulin AUC after intraperitoneal glucose administration, were greater in S961-treated pregnant rats (114.12 ± 6.64 nmol/L, n = 6, P < 0.05) compared with controls (58.64 ± 15.46 nmol/L, n = 6) (Figure 2B, bottom panel).
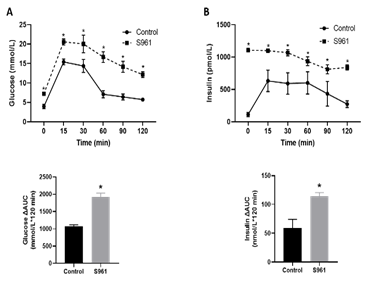
Figure 2: Blood glucose levels and insulin responses following intraperitoneal glucose tolerance test in control and S961-treated rats. Pregnant rats were treated with S961 or vehicle from GD7 to GD20. Following overnight fasting, intraperitoneal glucose tolerance tests were performed on GD19. Blood samples were collected at 0, 30, 60, 90, and 120 min following intraperitoneal glucose (2 g/kg) administration for measurement of (A) plasma glucose and (B) insulin levels. The overall plasma glucose levels and insulin responses are expressed as the AUC calculated by the trapezoidal method (bottom panel). Results are expressed as means ± SE of six dams in each group; ∗P < 0 .05 vs. control.
S961 affected Maternal Blood Pressure and Heart Rate
Studies show that insulin resistance during pregnancy has persistent effects on maternal cardiovascular function [34]. Thus, we assessed the impact of S961 treatment on mean blood pressure and heart rate in pregnant rats. As shown in Figure 3, mean arterial blood pressure and heart rate were significantly higher by 13.93 mmHg and 99 beats/min, respectively, in S961-treated pregnant rats than in controls (n=6 dams; P<0.05).
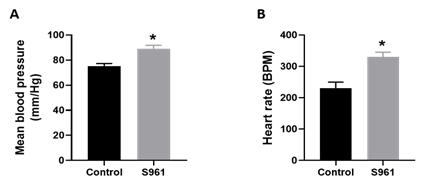
Figure 3: Changes in (A) maternal blood pressure and (B) heart rate in pregnant rats treated from GD7 to GD20 with S961 or vehicle. Mean arterial blood pressure and heart rate were measured using a noninvasive CODA system on GD20. All data are expressed as means ± SE of six dams in each group; ∗P < 0 .05 vs. control.
3.4 S961 has Differential Effects on Placental and Fetal Levels of n-3 and n-6 Fatty Acids
S961-treated pregnant rats (n = 6) had significantly lower placental concentrations of total n-3 (↓8%) and n-6 (↓11%) fatty acids than in controls (n=6 dams; P<0.05). In specific, the placental concentrations of n-3 fatty acids like eicosapentaenoic acid (↓32%) and docosatrienoic acid (↓19%), and n-6 fatty acid like linoleic acid (↓17%), and eicosadienoic acid (↓16) were lower in the placenta of S961-treated pregnant rats than in controls (Table 2).
|
Fatty acid |
Chain |
Control |
S961 |
%Change |
|
Omega-3 fatty acids (n-3 LCPUFAs) |
||||
|
Stearidonic acid |
18:04 |
0.44 ± 0.06 |
0.39 ± 0.04* |
-11.36 |
|
a-Linolenic acid |
18:03 |
3.59 ± 0.39 |
3.08 ± 0.33* |
-14.21 |
|
Eicosapentaenoic acid |
20:05 |
23.89 ± 2.33 |
16.26 ± 0.80* |
-31.94 |
|
Docosahexaenoic acid |
22:06 |
874.10 ± 81.83 |
811.65 ± 38.25* |
-7.14 |
|
Docosapentaenoic acid |
22:05 |
6.03 ± 0.66 |
7.18 ± 0.13* |
19.07 |
|
Docosatrienoic acid |
22:04 |
9.93 ± 0.80 |
7.94 ± 0.52* |
-20.04 |
|
Total |
917.97 ± 87.07 |
846.50 ± 40.07 |
-7.79 |
|
|
Omega-6 fatty acids (n-6 LCPUFAs) |
||||
|
g-Linolenic acid |
18:03 |
3.59 ± 0.39 |
3.08±0.33* |
-14.21 |
|
Linoleic acid |
18:02 |
1487.06 ± 116.34 |
1231.98±58.84* |
-17.15 |
|
Arachidonic acid |
20:04 |
1807.55 ± 151.40 |
1544.02±74.34* |
-14.58 |
|
Dihomo-g-linolenic acid |
20:03 |
11.01 ± 0.94 |
9.38±0.43* |
-14.8 |
|
Eicosadienoic acid |
20:02 |
133.12 ± 13.19 |
110.92±6.98* |
-16.68 |
|
Docosapentaenoic acid |
22:05 |
539.21 ± 84.81 |
659.32±35.05* |
22.28 |
|
Docosatetraenoic acid |
22:04 |
159.83 ± 17.17 |
144.67±7.11* |
-9.49 |
|
Docosadienoic acid |
22:02 |
20.78 ± 2.27 |
18.66±1.77* |
-10.2 |
|
Total |
4162.16 ± 386.51 |
3722.03 ± 184.26* |
-10.57 |
|
|
Values are expressed as mean ± SEM. n = 6 in each group. *P < 0.05 vs control group. |
||||
Table 2: n3- and n6-LCPUFA levels in placenta (µg/g).
In contrast, significantly higher plasma n-3 (↑15%) and n-6 (↑4%) fatty acid concentrations were observed in the plasma of fetuses of S961-treated pregnant rats (n = 6) than in control fetuses (Table 2). In specific, the plasma concentrations of n-3 fatty acids like docosapentaenoic acid (↑30%) and docosahexaenoic acid (↑15%), and n-6 fatty acids like linoleic acid (↑21%) and docosapentaenoic acid (↑8) were higher in the fetuses from S961-treated pregnant rats than in controls (Table 3).
|
Fatty acid |
Chain |
Control |
S961 |
% Change |
|
Omega-3 fatty acids (n-3 LCPUFAs) |
||||
|
Stearidonic acid |
18:4 |
0.19 ± 0.02 |
0.2 ± 0.03 |
7.34 |
|
a-Linolenic acid |
18:3 |
1.14 ± 0.24 |
0.77 ± 0.08* |
-31.82 |
|
Eicosapentaenoic acid |
20:5 |
6.04 ± 0.50 |
6.04 ± 0.91 |
0.05 |
|
Docosahexaenoic acid |
22:6 |
147.72 ± 14.71 |
170.87 ± 12.45* |
15.67 |
|
Docosapentaenoic acid |
22:5 |
1.53 ± 0.14 |
2 ± 0.13* |
30.36 |
|
Docosatrienoic acid |
22:4 |
1.19 ± 0.30 |
1.08 ± 0.30* |
-9.56 |
|
Total |
157.82 ± 15.92 |
180.96 ± 13.91* |
14.66 |
|
|
Omega-6 fatty acids (n-6 LCPUFAs) |
||||
|
g- Linolenic acid |
18:3 |
1.14 ± 0.24 |
0.77 ± 0.08* |
-31.82 |
|
Linoleic acid |
18:2 |
163.37 ± 14.48 |
198.44 ± 12.07* |
21.47 |
|
Arachidonic acid |
20:4 |
161.02 ± 28.15 |
140.11 ± 2.82* |
-12.99 |
|
Dihomo-g-linolenic acid |
20:3 |
0.95 ± 0.20 |
0.84 ± 0.05* |
-12.46 |
|
Eicosadienoic acid |
20:2 |
9.7 ± 2.13 |
10.07 ± 1.86* |
3.86 |
|
Docosapentaenoic acid |
22:5 |
36.76 ± 4.25 |
39.92 ± 3.97* |
8.58 |
|
Docosatetraenoic acid |
22:4 |
3.11 ± 0.53 |
2.66 ± 0.10* |
-14.49 |
|
Docosadienoic acid |
22:2 |
1.64 ± 0.38 |
1.7 ± 0.45* |
4 |
|
Total |
377.68 ± 50.36 |
394.51 ± 21.41* |
4.46 |
|
|
Values are expressed as mean ± SEM. n = 6 in each group. *P < 0.05 vs. control group. |
||||
Table 3: n3- and n6-LCPUFA levels in fetal plasma (µg/ml).
S961 Treatment affected the Expression of Genes related to Fatty Acid β-Oxidation and Fatty Acid Transport in the Placenta
The Fatty Acid RT2 Profiler PCR Array allowed us to assess the expression of 84 genes involved in fatty acid biosynthesis, metabolism, and transport. Among the 26 genes involved in fatty acid β-oxidation, S961 treatment significantly increased the expression of 10 genes, including Acaa1a, Acadm, Acot2, Acox2, Acsbg1, Acsl4, Acsm5, Cpt1b, Eci2, and Ehhadh (Table 4). Similarly, out of the 19 genes related to fatty acid transport, S961 treatment significantly increased the expression of 3 genes, including Fabp2, Fabp3, and Slc27a3 (Table 4).
|
Gene Symbol |
Gene Name |
Fold Up regulation |
Group |
|
Acaa1a |
Acetyl-CoA Acyltransferase 1 |
1.52 |
FAM |
|
Acadm |
Acyl-CoA Dehydrogenase Medium Chain |
1.96 |
FAM |
|
Acot2 |
Acyl-CoA Thioesterase 2 |
1.58 |
FAM |
|
Acox2 |
Acyl-CoA Oxidase 2 |
1.64 |
FAM |
|
Acsbg1 |
Acyl-CoA Synthetase Bubblegum Family Member 1 |
1.88 |
FAM |
|
Acsl4 |
Acyl-CoA Synthetase Long-Chain Family Member 4 |
1.66 |
FAM |
|
Acsm5 |
Acyl-CoA Synthetase Medium-Chain Family Member 5 |
1.53 |
FAM |
|
Cpt1b |
Carnitine Palmitoyltransferase 1B |
1.82 |
FAM |
|
Eci2 |
Enoyl-CoA Delta Isomerase 2 |
1.75 |
FAM |
|
Ehhadh |
Enoyl-CoA Hydratase And 3-Hydroxyacyl CoA Dehydrogenase |
2.29 |
FAM |
|
Fabp2 |
Fatty Acid Binding Protein 2 |
1.63 |
FAT |
|
Fabp3 |
Fatty Acid Binding Protein 3 |
1.59 |
FAT |
|
Slc27a3 |
Solute Carrier Family 27 Member 3 |
1.65 |
FAT |
|
FAM - Fatty Acid Metabolism; FAT - Fatty Acid Transport |
|||
Table 4: S961-upregulated fatty acid β-oxidation and transport genes in placenta
qRT-PCR validation confirmed that S961 treatment upregulated the expression of genes related to fatty acid β-oxidation and transport (Figure 4). Further examination of additional genes involved in placental fatty acid transport revealed that S961 did not affect the mRNA expression of fat/Cd36, Fatp1, Fatp3, and Fatp4 compared to controls (Figure 5).
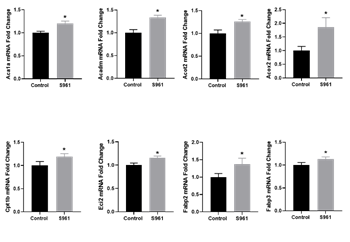
Figure 4: qPCR validation of mRNA expression of genes related to fatty acid β-oxidation and fatty acid transport in the placenta of control and S961 treated dams. mRNA expressions were determined by quantitative RT-PCR, and results were normalized with β-actin. Values are expressed as means ± SE of six dams in each group; ∗P < 0 .05 vs. control.
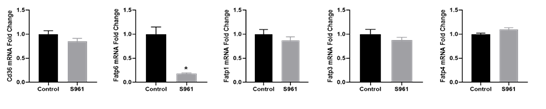
Figure 5: Changes in mRNA levels of genes related to fatty acid transport in the placenta exposed to vehicle and S961. mRNA expressions were determined by quantitative RT-PCR, and results were normalized with β-actin. Values are expressed as means ± SE of six dams in each group; ∗P < 0 .05 vs. control.
Discussion
The present work shows that S961 treatment in pregnant rats (i) induces key features of GDM, including hyperglycemia, hyperinsulinemia, and glucose intolerance without increases in maternal weight gain, (ii) increases maternal blood pressure, (iii) decreases placental n-3 and n-6 LCPUFA content, while increasing their concentrations in the fetal plasma, and (iv) increases the placental mRNA expression of genes involved in fatty acid β-oxidation and transport. Our work is the first to study placental LCPUFA levels and expression of key genes involved in LCPUFA trafficking and handling in lean GDM pregnancies, which provides new data to a poorly studied field. Treatment of pregnant rats with S961 resulted in glucose intolerance with higher fasting blood glucose, exhibiting the key features of GDM in humans. A compensatory rise in insulin levels generally accompanies insulin resistance. A significant increase in plasma insulin levels demonstrates the activation of homeostatic mechanisms in response to the S961 treatment. The hyperglycemia and glucose intolerance in S961-treated pregnant rats was not associated with increased maternal weight as the body weight gain in S961-treated pregnant rats was comparable to controls throughout the gestation. In addition, S961 did not affect daily food intake, consistent with a lack of effect on maternal weight gain. This indicates that S961-treatment induces features of the lean model of GDM in contrast to most animal models, which recapitulates insulin resistance due to obesity and high-fat feeding [35]. This is consistent with human data that over 25% of all cases of GDM occur in lean women [36]. The blood pressure in S961-treated pregnant rats was higher than controls indicating that disruption of insulin signaling could lead to the development of cardiovascular dysfunction [34]. Insulin is an important endothelium-dependent vasodilator, and enhanced endothelium-mediated vasodilation is critical during pregnancy [37]. Thus, the blockade of insulin-mediated endothelium-dependent vasodilation could contribute to increased blood pressure. This aligns with previous studies where S961 treatment caused decreased endothelium-dependent vasodilation in the aorta of male C57BL/6Jrj mice [38]. The significant increase in heart rate in S961-treated pregnant rats suggests enhanced adrenergic activity, which may also contribute to the hypertensive effects. In addition, hyperglycemia in S961-treated pregnant rats could promote the production of free radicals and inflammation and contribute to vascular dysfunction and hypertension [39, 40]. Whether high blood pressure in S961-treated pregnant rats results from specific or combined effects of endothelial dysfunction, enhanced adrenergic activity, or hyperglycemia remains to be determined. The concentrations of n-3 and n-6 fatty acids in the placenta were reduced while their levels were increased in the fetal plasma in S961-treated pregnant rats, indicating increased utilization/metabolism and/or transplacental transport of LCPUFA. This is in contrast with human studies, which show that n-3 and n-6 fatty acids were higher in the placenta and lower in the fetus of pregnant women with GDM [41-44]. However, these clinical studies were done in women with higher BMI (>25, categorized as overweight). Thus, the presence of overweight along with GDM could have confounded the results since obesity (in the absence of GDM) is known to cause placental LCPUFA accumulation and reduced fetal levels [41-44]. In support of this notion, animal studies with high-fat diets fed to pregnant rats show that LCPUFA was increased in the placenta and reduced in the fetus [45, 46]. Together, this data suggests that lean conditions of GDM could decrease placental LCPUFA levels but increase their fetal concentrations. One human study in lean pregnant women observed reduced placental LCPUFA levels in the placenta [47], similar to that observed in this study, but the fetal levels of LCPUFA were not measured in this study. Thus, clinical studies focusing on measuring placental and fetal levels of LCPUFA in lean GDM mothers would be necessary to validate our findings. Many fatty acid uptake transporters (FATPs) and fatty acid translocase (FAT/CD36) were expressed in the rat placenta. The sheer number of transport proteins at the maternal-placenta interface suggests that this site is critical for maintaining high rates of fatty acid uptake into the placenta. Once inside the trophoblast, fatty acids must bind with fatty acid-binding proteins (FABPs) to be transferred through intra-cellular aqueous cytoplasm to either mitochondria for oxidation, organelles for esterification and storage or to fetal-facing trophoblast membrane for delivery to fetal circulation [48-50]. At the transcriptional level, no differences in placental fatty acid uptake gene expression were observed between control and S961-treated pregnant rats. Other reports also suggest that fatty acid transporters in the placenta may not be insulin sensitive [51-53]. However, major intracellular fatty acid carriers, Fabp2, Fabp3, and solute carrier family 27, member 3 (Slc27a3), were upregulated in the placentas of S961-treated pregnant rats. Consistently, Scifres et al. reported that placental FABP3 mRNA expression is higher in diabetic women compared to controls [54]. These findings suggest that placental uptake is unaffected, but intracellular trafficking of LCPUFA is increased in the placenta of S961-treated pregnant rats. Additionally, mRNA expression of genes related to fatty acid β-oxidation was upregulated in the placenta of S961-treated pregnant rats. The upregulated genes include carnitine palmitoyltransferase 1b (Cpt1b), the rate-limiting step that delivers the long-chain fatty acids from the cytoplasm into mitochondria. Further, acetyl-CoA synthesis (acetyl-CoA synthetase), an important regulation site for intracellular long-chain fatty acid metabolism and other target genes essential for a series of β-oxidation reactions such as dehydrogenation (acyl-CoA dehydrogenase), hydration (enoyl hydratase also called crotonase) and dehydrogenation (β-hydroxy acyl-CoA dehydrogenase) were upregulated in the placentas of S961 treated pregnant rats. Studies show that in vitro treatment of primary trophoblasts with insulin downregulated multiple lipid oxidation genes [55]; hence it is logical that blockade of insulin action with S961 would increase the expression of fatty acid β-oxidation genes. Thus, these gene expression changes in the placenta of S961-treated rats suggest that insulin resistance increases placental lipid processing and transport of LCPUFA to the fetus. However, further studies that analyze these target proteins and activity levels and what cell population is affected could enhance our understanding of how insulin resistance could regulate placental lipid handling in a non-obese environment. Figure 6 summarizes the possible mechanisms involved in altered placental lipid processing and LCPUFA transfer to the fetus in lean GDM conditions. In conclusion, our study shows that lack of insulin action during pregnancy leads to GDM with no apparent weight gain and placental adaptations resulting in increased placental fatty acid β-oxidation and fetal lipid transport. This increased routing of lipids to the fetus might result in adiposity and contribute to the fetal programming of obesity and metabolic syndrome.
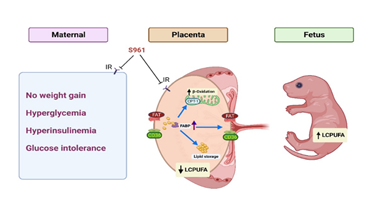
Figure 6: Possible mechanisms for altered placental lipid processing and LCPUFA transfer in lean GDM condition.
Acknowledgement
This study was supported by National Institutes of Health (NIH) grants (R01 HL134779 and R01 ES033345 to SK).
Conflicts of Interest
None.
References
- Sheiner E, Kapur A, Retnakaran R, et al. FIGO (International Federation of Gynecology and Obstetrics) Postpregnancy Initiative: Long-term Maternal Implications of Pregnancy Complications-Follow-up Considerations. Int J Gynaecol Obstet 147 (2019): 1-31.
- American Diabetes Association Professional Practice C. Summary of Revisions: Standards of Medical Care in Diabetes-2022. Diabetes Care 45 (2022): S4-S7.
- McIntyre HD, Catalano P, Zhang C, et al. Gestational diabetes mellitus. Nat Rev Dis Primers 5 (2019): 47.
- Group HSCR, Metzger BE, Lowe LP, et al. Hyperglycemia and adverse pregnancy outcomes. N Engl J Med 358 (2008): 1991-2002.
- Schneider S, Hoeft B, Freerksen N, et al. Neonatal complications and risk factors among women with gestational diabetes mellitus. Acta Obstet Gynecol Scand 90 (2011): 231-237.
- Sferruzzi-Perri AN, Camm EJ. The Programming Power of the Placenta. Front Physiol 7 (2016): 33.
- Sferruzzi-Perri AN, Higgins JS, Vaughan OR, et al. Placental mitochondria adapt developmentally and in response to hypoxia to support fetal growth. Proc Natl Acad Sci U S A 116 (2019): 1621-1626.
- Jayabalan N, Lai A, Ormazabal V, et al. Adipose Tissue Exosomal Proteomic Profile Reveals a Role on Placenta Glucose Metabolism in Gestational Diabetes Mellitus. J Clin Endocrinol Metab 104 (2019): 1735-1752.
- Jansson T, Ekstrand Y, Wennergren M, et al. Placental glucose transport in gestational diabetes mellitus. Am J Obstet Gynecol 184 (2001): 111-116.
- Gaither K, Quraishi AN, Illsley NP. Diabetes alters the expression and activity of the human placental GLUT1 glucose transporter. J Clin Endocrinol Metab 84 (1999): 695-701.
- Prieto-Sanchez MT, Ruiz-Palacios M, Blanco-Carnero JE, et al. Placental MFSD2a transporter is related to decreased DHA in cord blood of women with treated gestational diabetes. Clin Nutr 36 (2017): 513-521.
- Zhao JP, Levy E, Fraser WD, et al. Circulating docosahexaenoic acid levels are associated with fetal insulin sensitivity. PLoS One 9 (2014): e85054.
- Zhu Y, Li M, Rahman ML, et al. Plasma phospholipid n-3 and n-6 polyunsaturated fatty acids in relation to cardiometabolic markers and gestational diabetes: A longitudinal study within the prospective NICHD Fetal Growth Studies. PLoS Med 16 (2019): e1002910.
- Castillo-Castrejon M, Yamaguchi K, Rodel RL, et al. Effect of type 2 diabetes mellitus on placental expression and activity of nutrient transporters and their association with birth weight and neonatal adiposity. Mol Cell Endocrinol 532 (2021): 111319.
- Castillo-Castrejon M, Powell TL. Placental Nutrient Transport in Gestational Diabetic Pregnancies. Front Endocrinol (Lausanne) 8 (2017): 306.
- Dahlhoff M, Pfister S, Blutke A, et al. Peri-conceptional obesogenic exposure induces sex-specific programming of disease susceptibilities in adult mouse offspring. Biochim Biophys Acta 1842 (2014): 304-317.
- Rasool A, Mahmoud T, Mathyk B, et al. Obesity downregulates lipid metabolism genes in first trimester placenta. Sci Rep 12 (2022): 19368.
- Bidne KL, Uhlson C, Palmer C, et al. Human placental lipid content and lipid metabolic enzyme abundance in obesity and across gestation. Clin Sci (Lond) 136 (2022): 1389-1404.
- Calabuig-Navarro V, Haghiac M, Minium J, et al. Effect of Maternal Obesity on Placental Lipid Metabolism. Endocrinology 158 (2017): 2543-2555.
- Gutierrez JC, Bahamonde J, Prater MR, et al. Production of a type 2 maternal diabetes rodent model using the combination of high-fat diet and moderate dose of streptozocin. Endocr Res 35 (2010): 59-70.
- Serradas P, Giroix MH, Portha B. Evaluation of the pancreatic B-cell function in the rat after prenatal exposure to streptozotocin or N-nitrosomethylurea. Diabete Metab 15 (1989): 30-37.
- Kleiner S, Gomez D, Megra B, et al. Mice harboring the human SLC30A8 R138X loss-of-function mutation have increased insulin secretory capacity. Proc Natl Acad Sci U S A 115 (2018): E7642-E9.
- Cieniewicz AM, Kirchner T, Hinke SA, et al. Novel Monoclonal Antibody Is an Allosteric Insulin Receptor Antagonist That Induces Insulin Resistance. Diabetes 66 (2017): 206-217.
- Vikram A, Jena G. S961, an insulin receptor antagonist causes hyperinsulinemia, insulin-resistance and depletion of energy stores in rats. Biochem Biophys Res Commun 398 (2010): 260-265.
- de Sousa RAL, de Lima EV, da Silva TP, et al. Late Cognitive Consequences of Gestational Diabetes to the Offspring, in a New Mouse Model. Mol Neurobiol 56 (2019): 7754-7764.
- Schaffer L, Brand CL, Hansen BF, et al. A novel high-affinity peptide antagonist to the insulin receptor. Biochem Biophys Res Commun 376 (2008): 380-383.
- Mishra JS, More AS, Kumar S. Elevated androgen levels induce hyperinsulinemia through increase in Ins1 transcription in pancreatic beta cells in female rats. Biol Reprod 98 (2018): 520-531.
- Dangudubiyyam SV, Mishra JS, Song R, et al. Maternal perfluorooctane sulfonic acid exposure during rat pregnancy causes hypersensitivity to angiotensin II and attenuation of endothelium-dependent vasodilation in the uterine arteries dagger. Biol Reprod 107 (2022): 1072-1083.
- Mishra JS, Kumar S. Activation of angiotensin type 2 receptor attenuates testosterone-induced hypertension and uterine vascular resistance in pregnant ratsdagger. Biol Reprod 105 (2021): 192-203.
- Brown SH, Eather SR, Freeman DJ, et al. A Lipidomic Analysis of Placenta in Preeclampsia: Evidence for Lipid Storage. PLoS One 11 (2016): e0163972.
- Stahlman M, Ejsing CS, Tarasov K, et al. High-throughput shotgun lipidomics by quadrupole time-of-flight mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci 877 (2009): 2664-2672.
- Ulmer CZ, Patterson RE, Koelmel JP, et al. A Robust Lipidomics Workflow for Mammalian Cells, Plasma, and Tissue Using Liquid-Chromatography High-Resolution Tandem Mass Spectrometry. Methods Mol Biol 1609 (2017): 91-106.
- Tran PN, Brown SH, Mitchell TW, et al. A female gametocyte-specific ABC transporter plays a role in lipid metabolism in the malaria parasite. Nat Commun 5 (2014): 4773.
- Muniyappa R, Sowers JR. Role of insulin resistance in endothelial dysfunction. Rev Endocr Metab Disord 14 (2013): 5-12.
- Pasek RC, Gannon M. Advancements and challenges in generating accurate animal models of gestational diabetes mellitus. Am J Physiol Endocrinol Metab 305 (2013): E1327-38.
- Kim SY, England L, Wilson HG, et al. Percentage of gestational diabetes mellitus attributable to overweight and obesity. Am J Public Health 100 (2010): 1047-1052.
- Lopes van Balen VA, van Gansewinkel TAG, de Haas S, et al. Physiological adaptation of endothelial function to pregnancy: systematic review and meta-analysis. Ultrasound Obstet Gynecol 50 (2017): 697-708.
- Proniewski B, Bar A, Kieronska-Rudek A, et al. Systemic Administration of Insulin Receptor Antagonist Results in Endothelial and Perivascular Adipose Tissue Dysfunction in Mice. Cells 10 (2021).
- Steven S, Frenis K, Oelze M, et al. Vascular Inflammation and Oxidative Stress: Major Triggers for Cardiovascular Disease. Oxid Med Cell Longev 2019 (2019): 7092151.
- Dinh QN, Drummond GR, Sobey CG, et al. Roles of inflammation, oxidative stress, and vascular dysfunction in hypertension. Biomed Res Int 2014 (2014): 406960.
- Dijck-Brouwer DA, Hadders-Algra M, Bouwstra H, et al. Impaired maternal glucose homeostasis during pregnancy is associated with low status of long-chain polyunsaturated fatty acids (LCP) and essential fatty acids (EFA) in the fetus. Prostaglandins Leukot Essent Fatty Acids 73 (2005): 85-87.
- Min Y, Lowy C, Ghebremeskel K, et al. Fetal erythrocyte membrane lipids modification: preliminary observation of an early sign of compromised insulin sensitivity in offspring of gestational diabetic women. Diabet Med 22 (2005): 914-920.
- Thomas BA, Ghebremeskel K, Lowy C, et al. Plasma fatty acids of neonates born to mothers with and without gestational diabetes. Prostaglandins Leukot Essent Fatty Acids 72 (2005): 335-341.
- Wijendran V, Bendel RB, Couch SC, et al. Fetal erythrocyte phospholipid polyunsaturated fatty acids are altered in pregnancy complicated with gestational diabetes mellitus. Lipids 35 (2000): 927-931.
- Louwagie EJ, Larsen TD, Wachal AL, et al. Placental lipid processing in response to a maternal high-fat diet and diabetes in rats. Pediatr Res 83 (2018): 712-722.
- Grant WF, Gillingham MB, Batra AK, et al. Maternal high fat diet is associated with decreased plasma n-3 fatty acids and fetal hepatic apoptosis in nonhuman primates. PLoS One 6 (2011): e17261.
- Segura MT, Demmelmair H, Krauss-Etschmann S, et al. Maternal BMI and gestational diabetes alter placental lipid transporters and fatty acid composition. Placenta 57 (2017): 144-151.
- Islam A, Kagawa Y, Sharifi K, et al. Fatty Acid Binding Protein 3 Is Involved in n-3 and n-6 PUFA transport in mouse trophoblasts. J Nutr 144 (2014): 1509-1516.
- Makkar A, Mishima T, Chang G, et al. Fatty acid binding protein-4 is expressed in the mouse placental labyrinth, yet is dispensable for placental triglyceride accumulation and fetal growth. Placenta 35 (2014): 802-807.
- Haggarty P. Placental regulation of fatty acid delivery and its effect on fetal growth--a review. Placenta 23 (2002): S28-38.
- Anam AK, Cooke KM, Dratver MB, et al. Insulin increases placental triglyceride as a potential mechanism for fetal adiposity in maternal obesity. Mol Metab 64 (2022): 101574.
- Hulme CH, Nicolaou A, Murphy SA, et al. The effect of high glucose on lipid metabolism in the human placenta. Sci Rep 9 (2019): 14114.
- Desoye G, Hofmann HH, Weiss PA. Insulin binding to trophoblast plasma membranes and placental glycogen content in well-controlled gestational diabetic women treated with diet or insulin, in well-controlled overt diabetic patients and in healthy control subjects. Diabetologia 35 (1992): 45-55.
- Scifres CM, Chen B, Nelson DM, et al. Fatty acid binding protein 4 regulates intracellular lipid accumulation in human trophoblasts. J Clin Endocrinol Metab 96 (2011): E1083-91.
- Lassance L, Haghiac M, Leahy P, et al. Identification of early transcriptome signatures in placenta exposed to insulin and obesity. Am J Obstet Gynecol 212 (2015): 647 e1-11.
