Peracute Onset Pediatric Myelin Oligodendrocyte Glycoprotein Antibody Associated Focal Cortical Encephalitis: A Case Report
Article Information
Johanna Sophie Pohl1, Frank Leypoldt2,3, Naomi Larsen4, Andreas van Baalen1, Klaus-Peter Wandinger2, Kevin Rostasy5, Hiltrud Muhle1*
1Department of Neuropediatrics, University Center Schleswig-Holstein, Kiel University (CAU), Kiel, Germany
2Institute of Clinical Chemistry, University Hospital Schleswig-Holstein, Kiel/Luebeck, Germany
3Department of Neurology, University Hospital Schleswig-Holstein, Campus Kiel, Germany
4Institute of Radiology and Neuroradiology, University Hospital Schleswig-Holstein, Campus Kiel, Germany
5Pediatric Neurology, University of Witten/Herdecke, Children’s Hospital Datteln, Datteln, Germany
*Corresponding Author: Hiltrud Muhle, Department of Neuropediatrics, University Center Schleswig-Holstein, Kiel University (CAU), Kiel, Germany
Received: 22 June 2022; Accepted: 20 July 2022; Published: 08 August 2022
Citation: Johanna Sophie Pohl, Frank Leypoldt, Naomi Larsen, Andreas van Baalen, Klaus-Peter Wandinger, Kevin Rostasy, Hiltrud Muhle. Peracute Onset Pediatric Myelin Oligodendrocyte Glycoprotein Antibody Associated Focal Cortical Encephalitis: A Case Report. Archives of Clinical and Medical Case Reports 6 (2022): 586-590.
View / Download Pdf Share at FacebookAbstract
Recently, several case series have described pediatric patients presenting with new onset focal seizures together with relapsing fever and cortical T2/FLAIR hyperintensities in association with myelin oligodendrocyte glycoprotein (MOG) antibodies, in adults known as unilateral cortical fluid-attenuated inversion recovery (FLAIR) hyperintense lesions in anti- MOG-associated encephalitis with seizures (FLAMES).
A previously healthy 12-year-old girl first arrived to the emergency department with stroke-like symptoms, including hemiparesis, facial palsy, and severe headache. Heparin therapy was initiated; seizures were successfully treated with valproate. Due to persistent cortical edema in brain magnetic resonance imaging (MRI), anti-inflammatory treatment with high dose dexamethasone was initiated and led to prompt recovery.
One month later, the patient displayed fever and headache of unknown origin. Brain MRI showed cortical FLAIR hyperintensities and leptomeningeal contrast enhancement. Cerebrospinal fluid (CSF) analyses including autoimmune diagnostics revealed a lymphocytic pleocytosis, elevated protein, and positive oligoclonal bands. MOG antibodies in serum and CSF were positive. Intravenous methylprednisolone (IVMP), followed by oral tapering, led to complete recovery, yet one relapse occurred and intravenous immunoglobulins (IVIG) were added, given monthly to date. Our patient remained relapse-free for eight months when moderate but persisting headache and paraesthesia reoccurred. Methylprednisolone therapy was given, but outstanding MOG antibodies in serum and CSF were negative. Therefore, maintenance therapy was not extended. Single focal seizures led to adjustment of antiseizure medication.
In summary, this is the first case report of a pediatric patient with MOG encephalitis, first presenting stroke-like symptoms, rem
Keywords
<p>FLAMES; Anti-MOG-antibodies; Encephalitis; Stroke; Seizure; Children</p>
FLAMES articles; Anti-MOG-antibodies articles; Encephalitis articles; Stroke articles; Seizure articles; Children articles
FLAMES articles FLAMES Research articles FLAMES review articles FLAMES PubMed articles FLAMES PubMed Central articles FLAMES 2023 articles FLAMES 2024 articles FLAMES Scopus articles FLAMES impact factor journals FLAMES Scopus journals FLAMES PubMed journals FLAMES medical journals FLAMES free journals FLAMES best journals FLAMES top journals FLAMES free medical journals FLAMES famous journals FLAMES Google Scholar indexed journals COVID-19 articles COVID-19 Research articles COVID-19 review articles COVID-19 PubMed articles COVID-19 PubMed Central articles COVID-19 2023 articles COVID-19 2024 articles COVID-19 Scopus articles COVID-19 impact factor journals COVID-19 Scopus journals COVID-19 PubMed journals COVID-19 medical journals COVID-19 free journals COVID-19 best journals COVID-19 top journals COVID-19 free medical journals COVID-19 famous journals COVID-19 Google Scholar indexed journals Anti-MOG-antibodies articles Anti-MOG-antibodies Research articles Anti-MOG-antibodies review articles Anti-MOG-antibodies PubMed articles Anti-MOG-antibodies PubMed Central articles Anti-MOG-antibodies 2023 articles Anti-MOG-antibodies 2024 articles Anti-MOG-antibodies Scopus articles Anti-MOG-antibodies impact factor journals Anti-MOG-antibodies Scopus journals Anti-MOG-antibodies PubMed journals Anti-MOG-antibodies medical journals Anti-MOG-antibodies free journals Anti-MOG-antibodies best journals Anti-MOG-antibodies top journals Anti-MOG-antibodies free medical journals Anti-MOG-antibodies famous journals Anti-MOG-antibodies Google Scholar indexed journals Encephalitis articles Encephalitis Research articles Encephalitis review articles Encephalitis PubMed articles Encephalitis PubMed Central articles Encephalitis 2023 articles Encephalitis 2024 articles Encephalitis Scopus articles Encephalitis impact factor journals Encephalitis Scopus journals Encephalitis PubMed journals Encephalitis medical journals Encephalitis free journals Encephalitis best journals Encephalitis top journals Encephalitis free medical journals Encephalitis famous journals Encephalitis Google Scholar indexed journals Chemotherapy articles Chemotherapy Research articles Chemotherapy review articles Chemotherapy PubMed articles Chemotherapy PubMed Central articles Chemotherapy 2023 articles Chemotherapy 2024 articles Chemotherapy Scopus articles Chemotherapy impact factor journals Chemotherapy Scopus journals Chemotherapy PubMed journals Chemotherapy medical journals Chemotherapy free journals Chemotherapy best journals Chemotherapy top journals Chemotherapy free medical journals Chemotherapy famous journals Chemotherapy Google Scholar indexed journals treatment articles treatment Research articles treatment review articles treatment PubMed articles treatment PubMed Central articles treatment 2023 articles treatment 2024 articles treatment Scopus articles treatment impact factor journals treatment Scopus journals treatment PubMed journals treatment medical journals treatment free journals treatment best journals treatment top journals treatment free medical journals treatment famous journals treatment Google Scholar indexed journals CT articles CT Research articles CT review articles CT PubMed articles CT PubMed Central articles CT 2023 articles CT 2024 articles CT Scopus articles CT impact factor journals CT Scopus journals CT PubMed journals CT medical journals CT free journals CT best journals CT top journals CT free medical journals CT famous journals CT Google Scholar indexed journals Stroke articles Stroke Research articles Stroke review articles Stroke PubMed articles Stroke PubMed Central articles Stroke 2023 articles Stroke 2024 articles Stroke Scopus articles Stroke impact factor journals Stroke Scopus journals Stroke PubMed journals Stroke medical journals Stroke free journals Stroke best journals Stroke top journals Stroke free medical journals Stroke famous journals Stroke Google Scholar indexed journals hemolytic anemia articles hemolytic anemia Research articles hemolytic anemia review articles hemolytic anemia PubMed articles hemolytic anemia PubMed Central articles hemolytic anemia 2023 articles hemolytic anemia 2024 articles hemolytic anemia Scopus articles hemolytic anemia impact factor journals hemolytic anemia Scopus journals hemolytic anemia PubMed journals hemolytic anemia medical journals hemolytic anemia free journals hemolytic anemia best journals hemolytic anemia top journals hemolytic anemia free medical journals hemolytic anemia famous journals hemolytic anemia Google Scholar indexed journals acute kidney injury articles acute kidney injury Research articles acute kidney injury review articles acute kidney injury PubMed articles acute kidney injury PubMed Central articles acute kidney injury 2023 articles acute kidney injury 2024 articles acute kidney injury Scopus articles acute kidney injury impact factor journals acute kidney injury Scopus journals acute kidney injury PubMed journals acute kidney injury medical journals acute kidney injury free journals acute kidney injury best journals acute kidney injury top journals acute kidney injury free medical journals acute kidney injury famous journals acute kidney injury Google Scholar indexed journals
Article Details
Abbreviations:
ADEM: acute disseminated encephalomyelitis; ANA: antinuclear antibody; ANCA: anti-neutrophil cytoplasmic antibodies; CASPR2: contactin-associated protein 2; CMV: Cytomegalovirus; CNS: central nervous system; CRP: c-reactive protein; CSF: cerebrospinal fluid; DPPX: dipeptidyl-peptidase-like protein; DWI: diffusion weighted imaging; EBV: Epstein-Barr Virus; EEG: electroencephalography; FLAIR: fluid-attenuated inversion recovery; FLAMES: unilateral cortical FLAIR hyperintense lesions in Anti-MOG-associated encephalitis with seizures; GABA-B: gamma-aminobutyric acid transporters; GLI-1: glioma-associated oncogene-1; HIV: Human Immunodeficiency Virus; HSV: Herpes Simplex Virus; IVMP: intravenous methylprednisolone; IVIG: intravenous immunoglobulins; MDEM: multiphasic disseminated encephalomyelitis; MOG: myelin oligodendrocyte glycoprotein; MOGAD: MOG-associated disease; MRI: magnetic resonance imaging; NMDAR: N-methyl-D-aspartate-receptor; NSAID: nonsteroidal anti-inflammatory drug; ON: opticus neuritis; OXC: Oxcarbazepine; VPA : Valproate; VZV: Varicella-Zoster Virus
1. Introduction
Myelin oligodendrocyte glycoprotein (MOG) is found exclusively in the central nervous system (CNS) [1]. MOG antibody disease (MOGAD) is an inflammatory demyelinating condition that can present as monophasic or relapsing forms [2]. Clinical phenotypes of MOGAD are variable, occur more often in children, and range from isolated opticus neuritis (ON) to acute disseminated encephalomyelitis (ADEM) [2-4].
A recently described phenotype primarily in children is MOG encephalitis characterized by fever, headache, somnolence, seizures, a typical imaging pattern, and good response to steroids [5]. Here we describe a girl with MOG encephalitis reminiscent of FLAMES presenting stroke-like symptoms, which has not been described in this context before. As acute treatment with intravenous methylprednisolone (IVMP) and slow steroid tapering leads to a favourable outcome [6], a timely diagnosis and treatment is important to prevent relapse or long-term sequela [5]. This case report expands the spectrum of clinical symptoms of MOG encephalitis and aims to extend the warning signs that should lead to autoimmune diagnostics.
In adults, unilateral cortical fluid-attenuated inversion recovery (FLAIR) - hyperintense lesions in anti-MOG-associated encephalitis with seizures (FLAMES) is a new clinico-radiography syndrome with few cases and variable symptoms [7]. It was first reported by Ogawa et al. in 2017 [8], who described four MOG antibody positive patients presenting with encephalopathy and epileptic seizures, showing unilateral cortical hyperintensities best seen in magnetic resonance imaging (MRI) FLAIR-sequence. Combined, FLAMES can be described as a triad of MOG antibody encephalopathy, typical MRI-findings, and seizures.
2. Case Report
A previously healthy 12-year-old girl was admitted to the hospital with slurred language, hemiparesis with facial palsy on the left side, and headache. Brain MRI showed a faint hyperintensity on diffusion weighted imaging (DWI) in the right thalamus with a discreet corresponding hyperintense lesion on FLAIR images and equivocal DWI-signal in the right parietal cortex and left thalamus (Figure 1a and 1b). Laboratory findings revealed only a mild leucocytosis. Four hours after symptom onset, left focal seizures with twitching of the left face developed. Treatment with clonazepam led to behavior abnormalities. A second brain MRI performed the very next day showed restricted diffusion on DWI and cortical hyperintensities on FLAIR images in the right parietal and right frontal cortex (Figure 2a and 2b). Because of ongoing focal seizures and headaches, our patient was treated with levetiracetam. An electroencephalogram (EEG) showed profound right hemispheric slowing in keeping with a lesion or a post-ictal state.
Combining symptoms of one sided-hemiparesis, profound headache, impaired consciousness, nausea, and vomiting the differential diagnosis of hemiplegic migraine was considered. Slurred speech, hemiparesis of the leg, and facial palsy disappeared within hours of admission, whereas hemiparesis of the left arm and headache improved less rapidly. Treatment with valproate (VPA) stopped the seizures promptly. In a follow-up MRI on day 6, cortical edema in the right occipital cortex was clearly seen on T2-FLAIR-images (Figure 3a and 3b). Because of persistent vasogenic edema, dexamethasone was given orally for five days leading to significant clinical improvement of arm paresis. The girl was discharged home in good condition and she remained seizure-free with valproate therapy. Symptoms disappeared completely within three weeks.
One month later, the girl suffered from headache and fever episodes lasting for several days leading to a second hospitalisation. Viral infection was suspected and symptoms were treated with nonsteroidal anti-inflammatory drugs (NSAID). Again, blood analyses revealed solely leucocytosis. A brain MRI showed leptomeningeal enhancement in the right hemisphere (Figure 4a and 4b). Lumbar puncture revealed pleocytosis (231/µl), elevated protein (782 mg/l), and positive oligoclonal bands. Autoimmune antibodies were outstanding when treatment with antibiotic and antiviral therapy was introduced. Serum and liquor samples showed negative results for extensive testing for infection. Clinically, headache and fever episodes did not improve for another week despite normalization of leucocytes.
Eventually, MOG antibodies were detected in CSF (live cell-based assay [9], titer 1:4) and serum (titer 1:160). Testing of other antibodies revealed GluN1 NMDAR antibodies in CSF (cell-based assay, titer 1:2, confirmed by tissue-based testing using rat-brain immunohistochemistry) but not in serum. Altogether clinical presentation with seizures, fever, and behavior abnormalities, accompanied by focal MRI abnormalities and MOG antibodies, led to the diagnosis of MOG encephalitis and coexisting NMDAR antibodies. A treatment with IVMP (20 mg/kg/d) for three days was initiated. Headache and fever improved within hours. Steroid tapering for another 6 weeks with reduction of treatment to 2 mg/kg/d every second day to a final maintenance therapy of 10 mg prednisolone every alternate day was initiated. A routine follow-up brain MRI, in a symptom-free interval, 9 weeks later, showed regression of leptomeningeal enhancement. Blood samples revealed negative results for NMDAR antibodies and MOG antibody titers of only 1:80.
Three weeks after the end of steroid tapering, the girl again presented sudden onset of headache and loss of sensitivity in the right leg. Brain MRI revealed hyperintensities in the left parietal cortex on DWI with corresponding hyperintensities on T2-FLAIR-imaging (Figure 5a and 5b). Overall, the findings were consistent with a relapse. Acute therapy with IVMP (20 mg/kg/d) and additionally intravenous immunoglobulins (IVIG) (1 mg/kg/d) led to prompt improvement of clinical symptoms. Steroid tapering was performed for another 6 weeks. Since then, the girl receives IVIG-therapy monthly.
Under this maintenance therapy the patient remained relapse-free for eight months. Serum samples in the course of disease showed negative results for MOG- and NMDAR-antibodies. However, reoccurring focal epileptic seizures led to change of antiseizure medication from VPA to Oxcarbazepine (OXC) monotherapy.
13 months after her first presentation to hospital, our patient suffered from ongoing headache for 3 weeks, non-responsive to NSAIDS, once again. Due to hyponatremia (129 mmol/l) and high level of OXC in serum (38 μg/mL), a reduction of antiseizure medication was initiated. CSF showed mild pleocytosis (12/µl) and mildly elevated protein (482 mg/l). A few days later, paraesthesias and focal seizures appeared and our patient was treated with IVMP. Antiseizure medication was extended with low dose VPA, leading to prompt recovery. MOG antibodies in serum and CSF were negative in retrospect for that broadening of maintenance therapy was not initiated to date.
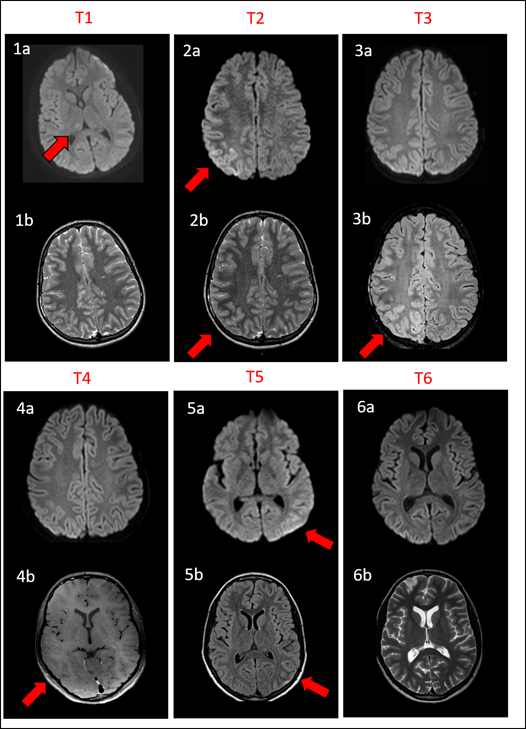
Figure 1: Brain magnetic resonance imaging (MRI) of our patient with MOG encephalitis. On the first day of admission (T1) a focal hyperintensity in the right thalamus is demonstrated on axial diffusion-weighted-images (DWI) (1a). There is no cortical edema visible on axial T2-weighted images (1b). On the second day of admission (T2), increased signal in the right parietal and occipital cortex on axial DWI, (2a) with only very slight vasogenic edema on T2-weighted images (2b) is seen. One week after the first MRI (T3) on axial DWI the hyperintensities have resolved (3a). Cortical swelling in the right occipital cortex is more clearly discernible on T2-FLAIR-images (3b). On the second admission to the hospital (T4), no pathologies were seen DWI (4a), but leptomeningeal enhancement throughout the right hemisphere was seen on contrast-enhanced T1-weighted images (4b). As part of a follow up two months after the second admission to the hospital, no pathologies were seen on axial DWI or contrast-enhanced T1-weighted images (not shown). On the third admission to the hospital (T5), hyperintensities in the left parietal cortex were seen on DWI (5a), with corresponding hyperintensities on axial T2-FLAIR-imaging (5b). During another follow up one month after the last admission to the hospital (T6), no pathologies were seen on DWI (6a) and T2-images (6b).
3. Discussion
This case report illustrates the variability of symptoms of autoimmune encephalitis associated with MOG antibodies. Our 12-year-old patient first arrived with stroke-like symptoms and headache. Only in combination with positive MOG antibodies the diagnosis of autoimmune encephalitis associated with MOG antibodies was made, in adults described as FLAMES. Until today, case series for FLAMES are rare. The first description was given by Ogawa and his team in 2017 [8], who identified four male adult patients with unilateral hyperintensities in T2-FLAIR-MRI and positivity for MOG antibodies. All four patients showed abnormal behavior, suffered from epileptic seizures, and recovered quickly after therapy with high dose IVMP. Budhram et al. were responsible for the original naming of FLAMES in 2019 [7]. They reviewed literature from 2009 until 2019 and identified 20 patients (age ranging from 11 – 46 years, mean age 29 years) with predominantly unilateral cortical hyperintensities in T2-FLAIR-MRI and positive MOG antibodies in serum or CSF. 85 % suffered from epileptic seizures, 70 % reported headache, 65 % reported fever, 55 % had neurological symptoms referable to MRI-findings, and 95 % reported at least two of these four findings.
Additionally to initial MRI description by Ogawa et al., contralateral cortical signal abnormalities, adjacent sulcal T2-FLAIR hyperintensities, and leptomeningeal enhancement were reported by Budhram and his team7. Hence, MRI-findings are not solely pathognomic for FLAMES, making differential diagnosis of CNS infection difficult. Like in our patient, only the combination of clinical symptoms, MRI-findings, and autoimmunity diagnostics in liquor leads to diagnosis of MOG encephalitis.
After diagnosis is made, the question of maintenance immunosuppressive therapy to prevent relapses arises. In October 2020, Bruijstens, Rostasy et al [6] suggested a consensus-based treatment recommendation for pediatric MOGAD (see Table 1). Patients treated with IVMP during an acute phase following slow steroid tapering up to three months showed a favourable outcome. IVIG and plasma exchange present alternative therapies in case of insufficiency or incompatibility of IVMP. After the first relapse (new clinical episode compatible with radiologic evidence at least one month after last attack) maintenance therapy should be initiated (see Table 1).
|
Course of MOG antibody disease (MOGAD) |
|||||
|
First symptoms |
1st relapse |
2nd relapse |
3rd relapse |
4th relapse |
|
|
Acute treatment |
IVMP (20-30 mg/kg/d for 3-5 d) + Steroid tapering (over 3 m, max. 60 mg/d) |
IVMP + steroid tapering |
IVMP + steroid tapering |
IVMP + steroid tapering |
IVMP + steroid tapering |
|
Alternative 1: IVIG* Alternative 2: PLEX |
Alternative: IVIG*/PLEX |
Alternative: IVIG*/PLEX |
Alternative: IVIG*/PLEX |
Alternative: IVIG*/PLEX |
|
|
Maintenance therapy |
Start IVIG*, monthly Or Rituximab Every 6 month |
Switch to other maintenance treatment e.g. Rituximab |
Combine maintenance treatments e.g. Rituximab + IVIG |
Combine maintenance treatments e.g. Rituximab + IVIG |
|
|
Alternative: AZA (daily) or MMF (daily) |
Prednisone Option A: daily Option B: monthly |
||||
Table 1: mg = milligram, kg = kilogram, d = days, w = week, m = months, IVMP = intravenous methylprednisolone, IVIG = intravenous immunoglobulins, *1-2 g/kg for 1-5 d and max. 1 g/kg/d, PLEX = plasma exchange, RTX = Rituximab, AZA = Azathioprine, MMF = Mycophenolate Mofetil, Adapted from Bruijstens et al., 2020 [6].
As recommended, our patient was treated with high dose IVMP (20 mg/kg/d over three days) [6]. This led to prompt recovery. IVMP is also recommended as a first step when a patient rearrives with relapse [10]. However, the effectiveness of acute IVMP therapy in relapse prevention remains uncertain [6]. Studies describing oral steroid tapering, comparing prednisolone doses and duration, are limited but show vulnerability to relapse on steroid reduction and cessation [11].
After the first relapse, our patient was treated with IVIG (1 g/kg/d monthly over two days) and did not experience further relapses for eight months until headache and seizures reoccurred. With outstanding anti MOG antibodies in CSF and negative anti MOG antibodies in serum, a second relapse could not be proven. Monthly IVIG was continued. To date, she remains relapse-free.
In our patient, autoimmune diagnostics showed positive results for NMDAR and MOG antibodies. Antibodies against MOG- and NMDA-receptors can coexist in children with autoimmune encephalitis [5, 12, 13]. Patients with both, positive MOG and positive NMDAR antibodies, showed increased risk of relapse and a high percentage of preceding infection [14]. In general, high and persisting titers are associated with a relapsing course. Wegener-Panzer et al. demonstrated in 2020 that five out of ten children with MOG encephalitis had relapses and that initial MOG serum titers in pediatric patients were higher in patients with relapses (median 1:640) than in patients without relapses (median 1:60) [5]. In addition, persistent MOG antibodies over time increase risk for relapse, although conversion to MOG antibody serum-negativity over time does not preclude relapse [15]. All in all, our patient was at high risk for relapses.
In our patient, interictal EEGs showed profound hemispheric slowing without epileptic discharges during acute symptoms twice, whereas EEGs in symptom-free intervals were normal in accordance to MRI-improvement. This finding is consistent with previous studies [5].
In conclusion, our case report aims at increasing awareness for autoimmune encephalitis associated with MOG antibodies which can occur in children. Any focal, mostly cortical T2-FLAIR hyperintensity detected in the context of new onset focal seizures should trigger a suspicion of MOGAD and serum testing for MOG antibodies. Importantly, different test systems with variable sensitivity and specificity exist [16] and care must be taken to correctly interpret serological findings. Relapsing fever and peracute onset of motor symptoms can be an additional valuable clinical hint and should lead to autoimmune diagnostics.
Declarations of Interest
None
Acknowledgments
We thank our patient and her family for participating in this case report.
Disclosures
None of the authors has any conflict of interest to disclose. We confirm that we have read the Journals position on issues involved in ethical publication and affirm that this report is consistent with those guidelines.
References
- Schluesener H, Sobel R, Linington C, et al. A monoclonal antibody against a myelin oligodendrocyte glycoprotein induces relapses and demyelination in central nervous system autoimmune disease. J Immunol 12 (1987): 4016-4021.
- Lechner C, Baumann M, Hennes E-M, et al. Antibodies to MOG and AQP4 in children with neuromyelitis optica and limited forms of the disease. J Neurol Neurosurg Psychiatry 8 (2016): 897-905.
- Reindl M, Di Pauli F, Rostásy K, et al. The spectrum of MOG autoantibody-associated demyelinating diseases. Nat Rev Neurol 8 (2013): 455-461.
- Pröbstel AK, Dornmair K, Bittner R, et al. Antibodies to MOG are transient in childhood acute disseminated encephalomyelitis. Neurology 6 (2011): 580-588.
- Wegener-Panzer A, Cleaveland R, Wendel E-M, et al. Clinical and imaging features of children with autoimmune encephalitis and MOG antibodies. Neurol Neuroimmunol Neuroinflamm (2020).
- Bruijstens AL, Wendel E, Lechner C, et al. European Journal of Paediatric Neurology E .U. paediatric MOG consortium consensus?: Part 5 e Treatment of paediatric myelin oligodendrocyte glycoprotein antibody-associated disorders Ronny Wickstr o. Eur J Paediatr Neurol 29 (2020): 41-53.
- Budhram A, Mirian A, Le C, et al. Unilateral cortical FLAIR ? hyperintense Lesions in Anti ? MOG ? associated Encephalitis with Seizures (FLAMES): characterization of a distinct clinico ? radiographic syndrome. J Neurol (2019): 2481-2487.
- Ogawa R, Sato DK, Nakshima I, et al. MOG antibody – positive, benign, unilateral, cerebral cortical encephalitis with epilepsy. Neurol Neuroimmunol Neuroinflamm (2017): 1-10.
- Höftberger R, Sepulveda M, Armangue T, et al. Antibodies to MOG and AQP4 in adults with neuromyelitis optica and suspected limited forms of the disease. Mult Scler 21 (2015): 866-874.
- Ambrosius W, Michalak S, Kozubski W, et al. Myelin Oligodendrocyte Glycoprotein Antibody-Associated Disease?: Current Insights into the Disease Pathophysiology, Diagnosis and Management. Int J Mol Sci 22 (2021): 100.
- Ramanathan S, Mohammad S, Tantsis E, et al. Clinical course , therapeutic responses and outcomes in relapsing MOG antibody-associated demyelination. J Neurol Neurosurg Psychiatr 89 (2018): 127-137.
- Hou C, Wu W, Tian Y, et al. Clinical analysis of anti-NMDAR encephalitis combined with MOG antibody in children. Mult Scler Relat Disord. (2020).
- Titulaer MJ, Höftberger R, Iizuka T, et al. Overlapping demyelinating syndromes and anti-NMDA receptor encephalitis. Ann Neurol 75 (2015): 411-428.
- Zhang J, Ji T, Chen Q, et al. Pediatric Autoimmune Encephalitis?: Case Series From Two Chinese Tertiary Pediatric Neurology Centers. Front Neurol 10 (2019): 1-9.
- Waters P, Fadda G, Woodhall M, et al. Serial Anti–Myelin Oligodendrocyte Glycoprotein Antibody Analyses and Outcomes in Children With Demyelinating Syndromes. Jama Neurol 19104 (2020): 82-93.
- Gastaldi M, Scaranzin S, Jarius S, et al. Cell-based assays for the detection of MOG antibodies: a comparative study. J Neurol 12 (2020): 3555-3564.
