On Similarities and Differences of HIV and SARS Cov-2; Open Questions Relevant for COVID-19 Disease
Article Information
Eva Zerovnik*
Department of Biochemistry and Molecular and Structural Biology, Jo?ef Stefan Institute, Ljubljana, Slovenia
*Corresponding Author: Eva ?erovnik, Department of Biochemistry and Molecular and Structural Biology, Jo?ef Stefan Institute, Jamova 39, 1000 Ljubljana, Slovenia
Received: 23 October 2021; Accepted: 05 November 2021; Published: 10 November 2021
Citation:
Eva Zerovnik. On Similarities and Differences of HIV and SARS Cov-2; Open Questions Relevant for COVID-19 Disease. Journal of Biotechnology and Biomedicine 4 (2021): 147-158.
View / Download Pdf Share at FacebookAbstract
This paper is written from an educated bio-scientist’ perspective who follows studies about COVID-19 in order to better understand some relevant questions about the disease and vaccination. I first pose myself a question about similarities and differences of SARS CoV-2 and HIV viruses. After the available literature search this idea was soon confirmed. Several papers have appeared where authors discuss similarities and differences between HIV and SARS CoV-2. I later come across some open questions on SARS CoV-2, which do or do not have clear answers. I discuss these questions; among them are some about the current vaccines. In my opinion, the hot questions should be addressed as getting clear answers will bring more understanding of the matter and could help in future endeavors.
Keywords
HIV; SARS CoV-2
viruses articles viruses Research articles viruses review articles viruses PubMed articles viruses PubMed Central articles viruses 2023 articles viruses 2024 articles viruses Scopus articles viruses impact factor journals viruses Scopus journals viruses PubMed journals viruses medical journals viruses free journals viruses best journals viruses top journals viruses free medical journals viruses famous journals viruses Google Scholar indexed journals HIV articles HIV Research articles HIV review articles HIV PubMed articles HIV PubMed Central articles HIV 2023 articles HIV 2024 articles HIV Scopus articles HIV impact factor journals HIV Scopus journals HIV PubMed journals HIV medical journals HIV free journals HIV best journals HIV top journals HIV free medical journals HIV famous journals HIV Google Scholar indexed journals SARS CoV-2 articles SARS CoV-2 Research articles SARS CoV-2 review articles SARS CoV-2 PubMed articles SARS CoV-2 PubMed Central articles SARS CoV-2 2023 articles SARS CoV-2 2024 articles SARS CoV-2 Scopus articles SARS CoV-2 impact factor journals SARS CoV-2 Scopus journals SARS CoV-2 PubMed journals SARS CoV-2 medical journals SARS CoV-2 free journals SARS CoV-2 best journals SARS CoV-2 top journals SARS CoV-2 free medical journals SARS CoV-2 famous journals SARS CoV-2 Google Scholar indexed journals co-infection articles co-infection Research articles co-infection review articles co-infection PubMed articles co-infection PubMed Central articles co-infection 2023 articles co-infection 2024 articles co-infection Scopus articles co-infection impact factor journals co-infection Scopus journals co-infection PubMed journals co-infection medical journals co-infection free journals co-infection best journals co-infection top journals co-infection free medical journals co-infection famous journals co-infection Google Scholar indexed journals heart-intestine axis articles heart-intestine axis Research articles heart-intestine axis review articles heart-intestine axis PubMed articles heart-intestine axis PubMed Central articles heart-intestine axis 2023 articles heart-intestine axis 2024 articles heart-intestine axis Scopus articles heart-intestine axis impact factor journals heart-intestine axis Scopus journals heart-intestine axis PubMed journals heart-intestine axis medical journals heart-intestine axis free journals heart-intestine axis best journals heart-intestine axis top journals heart-intestine axis free medical journals heart-intestine axis famous journals heart-intestine axis Google Scholar indexed journals proteins articles proteins Research articles proteins review articles proteins PubMed articles proteins PubMed Central articles proteins 2023 articles proteins 2024 articles proteins Scopus articles proteins impact factor journals proteins Scopus journals proteins PubMed journals proteins medical journals proteins free journals proteins best journals proteins top journals proteins free medical journals proteins famous journals proteins Google Scholar indexed journals glycoproteins articles glycoproteins Research articles glycoproteins review articles glycoproteins PubMed articles glycoproteins PubMed Central articles glycoproteins 2023 articles glycoproteins 2024 articles glycoproteins Scopus articles glycoproteins impact factor journals glycoproteins Scopus journals glycoproteins PubMed journals glycoproteins medical journals glycoproteins free journals glycoproteins best journals glycoproteins top journals glycoproteins free medical journals glycoproteins famous journals glycoproteins Google Scholar indexed journals symptoms articles symptoms Research articles symptoms review articles symptoms PubMed articles symptoms PubMed Central articles symptoms 2023 articles symptoms 2024 articles symptoms Scopus articles symptoms impact factor journals symptoms Scopus journals symptoms PubMed journals symptoms medical journals symptoms free journals symptoms best journals symptoms top journals symptoms free medical journals symptoms famous journals symptoms Google Scholar indexed journals antiretroviral therapies articles antiretroviral therapies Research articles antiretroviral therapies review articles antiretroviral therapies PubMed articles antiretroviral therapies PubMed Central articles antiretroviral therapies 2023 articles antiretroviral therapies 2024 articles antiretroviral therapies Scopus articles antiretroviral therapies impact factor journals antiretroviral therapies Scopus journals antiretroviral therapies PubMed journals antiretroviral therapies medical journals antiretroviral therapies free journals antiretroviral therapies best journals antiretroviral therapies top journals antiretroviral therapies free medical journals antiretroviral therapies famous journals antiretroviral therapies Google Scholar indexed journals neurodegeneration articles neurodegeneration Research articles neurodegeneration review articles neurodegeneration PubMed articles neurodegeneration PubMed Central articles neurodegeneration 2023 articles neurodegeneration 2024 articles neurodegeneration Scopus articles neurodegeneration impact factor journals neurodegeneration Scopus journals neurodegeneration PubMed journals neurodegeneration medical journals neurodegeneration free journals neurodegeneration best journals neurodegeneration top journals neurodegeneration free medical journals neurodegeneration famous journals neurodegeneration Google Scholar indexed journals
Article Details
1. Similarities between the Two Viruses
Several papers comparing these two viruses and the effect of possible co-infection have appeared in the literature [1]. At first glance one would conclude that these two viruses do not cause similar disease and that they are not similar by origin and sequence, however, they both are RNA viruses and some symptoms of the disease caused by them are shared [2] – as will be described later. A common presentation at least in the more severe cases of COVID-19 is the so called cytokine storm, the increased production of proinflammatory cytokines [3]. It is well known that HIV infection also leads to steady increase in cytokine release that generates chronic inflammation. In COVID-19 disease this results in systemic inflammation (cardiovascular system, heart, lungs, intestine, pancreas) and in HIV infected patients it also leads to cardiovascular disease, cancer, etc. Borges et al., [4]. Changes of microbiota to the worse are a common denominator, too. This happens in COVID-19 patients with cardiac complication, due to higher levels of intestinal permeability and activation of inflammasomes, suggesting a heart-intestine axis. Similarly, HIV infection produces unfavorable effects on microbiota (increase in pro-inflammatory bacteria and a reduction in beneficial bacteria), which also affects the immune system Tincati et al., [5].
Another similarity is NET (neutrophil extracellular traps) where NETs capture bacteria, fungi and viruses Papayannopoulos et al., [6]. The mechanism of NETosis stems from neutrophil death in which neutrophils release chromatin fibres containing histones, microbicidal peptides and some enzymes into the extracellular space. Acute NETosis is an efficient defense mechanism that prevents collateral tissue damage. However, a chronic and aberrant NETosis contributes to the pathology Soehnlein et al., 2017 [7]. As predicted by Mozzini and Girelli [8] NETs in COVID-19 patients may lead to thrombotic complications [9], similarly as in HIV patients. Common is also the psychological effect of the fear of unknown and bad outcome [2]. The fear increases stress and anxiety, which suppress the immune system. In addition, the panic is even increased by the Internet news, spreading some unfounded rumors, both, on the disease and vaccines.
2. Cleavage by the Host Protease Furin
Next similarity is the cleavage of the viral envelop proteins by the host protease furin. Spike glycoproteins of many enveloped viruses are proteolytically cleaved at the carboxytermini of sequences containing the basic motif R-X-K/R-R. SARS CoV-2 similarly uses the host protease furin to process the spike protein before entry. It has been reported recently in a Science paper by Daly et al., 2020 that neurophilin-1 acts as a host factor interacting with the cleaved part of the spike protein [10]. Of interest HIV also uses the subtilisin-like endoprotease furin to process the envelop glycoproteins as shown by Garten et al., already in 1994 [11]. The furin active site is thus surprisingly shared with HIV, which raises some doubts whereas it might not be inserted [12]. However, other coronaviruses of natural origin also developed the furin active site [13].
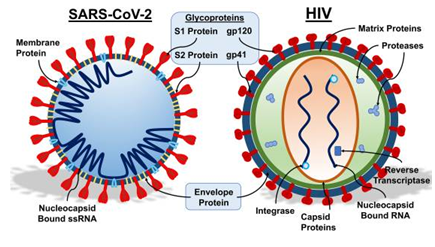
Figure 1: Copy right, Image credit to Nicholas J. Evans. HIV AIDS (Auckl). 2021; 13: 361–375.
Published online 2021 Mar 31. doi: 10.2147/HIV.S300055.
2.1 HIV symptoms
During the early acute phase HIV infected patients present with mild symptoms of fever, headache, muscle pain, sore throat, skin rashes and mouth sores, as well as swollen lymph nodes. In the later chronic phase, the virus continues to spread and destroy immune cells and to suppress immune response. If not treated, HIV turns into AIDS in about 10 years. As the immune system is compromised, opportunistic infections, neurodegenerative diseases and cancers appear more frequently.
2.2 SARS COV-2 symptoms
Most common symptoms include fever, cough, and dyspnea. Diarrhea and abdominal pain are also frequent. Although most cases have mild symptoms, in the more severe cases, the infection can cause pneumonia, severe difficulty breathing, kidney failure, and even death, just as SARS-CoV-1 does, Lake 2020 [14]. It has been observed that COVID-19 disease also affects the brain and increases risk for dementia, Wang et al. [15].
2.3 Therapies against HIV
There is no cure for HIV infection, however, with the antiretroviral therapies (ART) [16] this disease has become well manageable, as any other chronic disease. New drugs, such as integrase inhibitors and a cocktail of protease inhibitors have contributed to better outlook. Still, HIV persists in the body in the viral reservoirs, which allows competent viruses to replicate. Vaccination has not been found for HIV, even though the researchers are looking at mRNA-based vaccine technology to apply the concept to HIV.
2.4 Therapies against SARS CoV-2 virus
COVID-19 was initially treated by known drugs used to treat HIV, such as lopinavir and ritonavir with not much success, Cao et al., [17]. Nowadays there are more drugs in development, among them anti-protease inhibitors [18]. Plant based inhibitors of the main 3CL protease are also considered as starting drug candidates [19]. Other plant derived antivirals have been considered at least as preventive measure against initial viral spread [20]. Numerous trials are on-going to selectively stop the SARS CoV-2 by small drugs, for example molnupiravir [21]. As applied in Mayo Clinic (Rochester, MN) https://www.mayoclinic.org/diseases-conditions/coronavirus/expert-answers/coronavirus-drugs/faq-20485627 in practical terms drugs combinations: tocilizumab or baricitinib (rheumatoid arthritis drug) with dexamethasone (this latter is cortico-steroid) and remdesivir with dexamethasone are used in hospitalized COVID-19 patients who are on mechanical ventilation or need supplemental oxygen. Ivermectin and hydroxychloroquine are not recommended and can cause serious adverse reactions.
There also is an over activity of the immune system observed leading to inflammation in the severe COVID-19 disease. Dr Ivan Marazzi says: “host inflammatory response, rather than viral activity is driving disease severity during severe COVID-19, accordingly, COVID-19 can be regarded an immune disease. Therefore, there is definitely a need to re-align our focus to host, immune-modulatory treatments for this disease, as opposed to antivirals”. The study about topotecan (TPT), an anti-cancer drug, inhibiting inflammation via topoisomerase I complex was published in Cell [22]. Long COVID is believed to result from autoantibodies against ACE-2 receptor as recent report implicates [23]. Unfortunately, similar albeit very rare adverse reactions have been observed upon vector based vaccines.
3. How is the Brain Involved?
Patients after more severe COVID-19 and long COVID often experience neurological symptoms, such as memory loss, difficulty in concentration, fatigue, and loss of smell, taste, and muscle weakness. Researchers suggest at least three possible pathways, how SARS CoV-2 could affect the brain. Either via infecting the astrocytes, by reduced blood flow also leading to high pressure or by autoantibodies. The immune system over-reaction could contribute to neurological symptoms by autoantibodies as described by Prüss in a recent review published in Nature Reviews [24]. There is evidence that these autoantibodies can pass through the blood–brain barrier (BBB), which reflects in neurological disorders such as memory loss and even psychosis [24]. As cited from Farhadian et al. [25]: “Neuroimmune perturbations and vascular inflammation observed in people with COVID-19 warrant investigation of immune-modulating interventions to ameliorate neurological complications associated with acute SARS-CoV-2 infection”. Such therapies may also be applied to treat the so called long COVID-19 [25]. A preprint of magnetic resonance brain imaging study by Douaud et al. [26] shows that grey matter in several brain areas, significantly so in the lateral orbitofrontal cortex and parahippocampal gyrus, gets reduced in the infected with SARS CoV-2 as compared to controls. Whereas brain atrophy reflected by cognitive decline observed with some survivors of the COVID-19 is a temporal effect of the infection remains to be followed [26].
As for the neurology of HIV infection [27] there have been many studies done over years. Somewhat similar but also different to SARS CoV-2 there are at least two pathways how HIV infects the brain, i.e., by chronic immune activation and by compromised BBB integrity [28]. In both of these the infected monocytes/macro-phages play an important role [28]. Cognitive decline in HIV infected patients known as HIV-associated neurocognitive disorders (HAND) can be classified as asymptomatic neurocognitive impairment (ANI), mild neurocognitive disorder (MND), and HIV associated dementia (HAD) [28]. The effect of a latent infection hidden in the brain remains a worry when looking for a potential HIV cure [28].
3.1 Amyloidogenecy and possible neurodegeneration
Human amyloid proteins can act as reducing or promoting agents for viral infectivity. The mechanisms are not well understood yet several hypothesis are being put forward [29]. For example, amyloid proteins aggregate can be enhanced by the virus particles surface or specific regions and on the other side, amyloid aggregates can help reduce viral burden, likely by inducing clearance by autophagy. SARS CoV-2 proteins were predicted to possess aggregation prone regions and hot spots for aggregation [30]. Specifically and importantly, as an entry protein interacting with ACE-2 receptor, spike protein S1 peptide was shown to bind heparin and attract other amyloidogenic proteins from Aβ, α-syn, prion to TDP-43 and to enhance their aggregation [31]. That this could happen also in vivo remains to be shown, how-ever, widespread inflammation is characteristic for COVID-19 and activation of the NLRP3 inflammasome, could lead to downstream tau aggregation and neuro-degeneration [32]. HIV infection also affects the brain, however, HAND did not cause widespread amyloid accumulation [33].
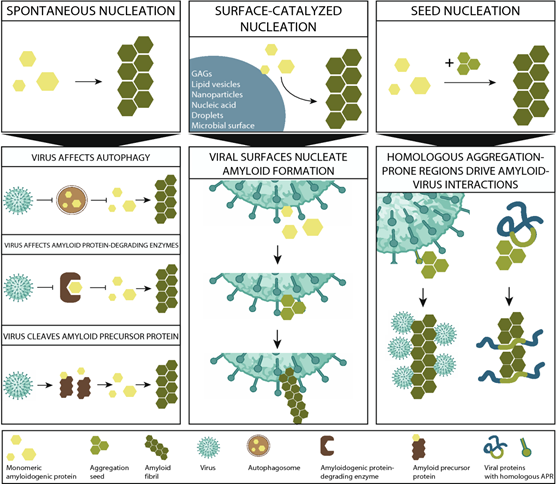
Figure 2: A schematic overview of the mechanisms underlying amyloid–virus interactions. The three upper panels represent three possible different mechanisms: spontaneous nucleation, surface-catalyzed nucleation (SCN), and seed nucleation (SN), which are non-mutually exclusive [29].
4. Differences and Some Burning Questions
The two viruses use different means to enter the host cell. Zhou et al. [34] confirmed that SARS-CoV-2 uses the angiotensin-converting enzyme 2 (ACE-2) to enter cells as SARS-CoV-1 did [35]. Spike protein is cleaved and one-part (S1) binds to the ACE-2 receptor while another (S2) fuses with the plasma membrane. In likely a distinct process, the HIV protein envelope (Env) protein binds to the primary cellular receptor CD4 and then to a cellular co-receptor [36]. There are several steps for HIV Env to achieve membrane fusion and perforation, through which the viral contents entry the cell [36]. However, there might be some structural similarities in SARS CoV S2 protein and HIV glycoprotein gp41, which share the same two α helices, suggesting similar membrane fusion mechanism. Ligand-binding measurements showed that two inhibitors GGL and D-peptide from HIV-1 gp41 may equally bind to S2 of SARS-CoV and prevent its entry [37].
Next difference is that HIV is a double-stranded RNA virus – retrovirus while SARS CoV-2 is a positive-sense single-stranded RNA virus. Therefore, HIV can transform into a latent state by integrating itself into the host DNA and can be reactivated upon various stimulations such as inflammation by co-infecting pathogens. In contrast and consistent with its single stranded RNA nature, SARS-CoV-2 does not integrate in the host DNA. The generation of antibodies 15 days after infection is 100%. Therefore, SARS COV-2 would be expected to dye off gradually. However, the phenomenon that symptoms persist months after the infection suggests possible presence of viral reservoirs on mucus membranes. A question is still debated whereas SARS CoV-2 could reside in a latent form as HIV does. A latent form appears when a virus remains dormant and does not replicate, however, under certain conditions it can be re-activated. The suspects arise from observations that in some cases COVID-19 infection appears again after a few weeks or months, where the second infection usually occurs with milder symptoms. It has been argued that this is not re-infection but rather re-activation of the virus. Examples of latent viruses comprise: Herpes simplex viruses type 1 and 2, varicella-zoster virus, HIV, Epstein-Barr virus (human herpesvirus 4), and cytomegalovirus [38]. Indeed, a study by Zhang et al. [39] has shown that SARS-CoV-2 RNAs can be reversely transcribed and integrated into the human genome by different reverse transcriptase’s, such as activated human LINE-1 or co-infected retrovirus (HIV) transcriptase’s. As cited from Zhang et al. “From an evolutionarily perspective, retrointegration of viral RNA could be an adaptive response by the host to provide sustaining antigen expression possibly enhancing protective immunity.” On the other hand, retrointegration of viral RNAs could be detrimental due to overwhelming immune response resulting in the so called “cytokine storm” or auto-immunity. Using reverse transcription in both cases could maybe explain why HIV infected people are at more risk to get a more severe COVID-19.
4.1 Questions about mRNA vaccines
Some viruses, such as dengue and respiratory syncytial virus themselves or vaccines producing non-neutralizing antibodies against the virus in question, can lead to AER (antibody enhancement reaction) also known as ADE (antibody disease enhancement), which results in worsening of the disease. By avoiding AER, by choosing careful target epitopes and T-cell enhancers, the new mRNA based vaccines seem avoid of such reactions and allow high protection. Concern is raised, even though at the moment purely theoretically, about the ratio of neutralizing against non-neutralizing, disease promoting antibodies in the case of delta variant [40].
An apparently naïve question is posed by some if the mRNAs get somehow transcribed back into genome by reverse transcription? The answer by experts is definite NO. This is not possible as all proteins information goes from DNA (gene), which is promoted to get expressed and is translated into mRNA, then transcribed into the protein on the ribosome. This is a dogma DNA – to RNA to protein. Well, dogmas have been challenged. And indeed, there exists a phenomenon of »retrotranscription« as described above and some viruses use it, such as HIV and lately they claim it also for SARS COV-2 [38]. As well retroposons are a common phenomenon. The answer about mRNA pieces retrotranscription is by majority of the specialists: NO. Scientists assure that mRNA injected to the adipose tissue is fast degraded (in about 2 days) and does not reach the nucleus of mast cells. However, the mRNA used for vaccines is not a simple one. It is prepared to last longer to enable higher load of proteins and corresponding antibodies. Questions go on, does the full-length spike protein (the result of mRNA translation, which is present in the system for about 2 – 3 weeks and also able to circulate in the blood and lymph as well as crossing the BBB) exert its function? Is it not dangerous that it would act as antagonist of ACE-2 receptors as we know it binds strongly to the active site and such receptors are vital in many tissues (lung, intestine, heart, the brain). In the heart and vascular system this protein might produce signs of inflammation (myocarditis is a rare side effect reported for adolescent boys). Spike protein may promote transformation of other proteins into prion or amyloid form [41] and disrupts lipid bilayers [42] (which might not be beneficial when it gets into the brain).
However, all these effects are temporary, whereas viral infection can stay for months. The concentrations of the spike protein after translation are not that low, higher than the shedded protein from the virus, only, that virus multiplies and the spike protein does not, it gets eventually degraded by phagocytosis. Therefore, after the first dose of mRNA vaccine spike protein is found in the blood plasma, whereas after the 2nd, it is not detectable, it gets captured by neutralizing antibodies.
Rare adverse effects of mRNA based vaccines boosting spike protein and corresponding antibodies comprise myocarditis or pericarditis (in adolescent and young boys). Whereas, with vector DNA vaccines (Astra Zeneca and Johnson-Johnson) thrombocytopenia and potentially fatal or long-lasting consequences of blood clots, even in young women was documented. The reasons behind are being explored and indicate certain autoimmune reactions [43]. For COVID-19 Vaccine Janssen there are reports of a very rare but extremely unpleasant and dangerous condition Guillain-Barré syndrome (GBS). Personally, I would feel much more at ease if peptide vaccines would be made. Results of phase 3 clinical study of a recombinant spike protein vaccine (Novavax) have been published [44]. Whole spike recombinant protein base vaccines would not involve the cellular transcription and translation machineries; however, the spike protein physiological and/or pathological functions would still be a concern, see above. Similarly peptide epitopes based T cells vaccine [45] (Epivax) are being tested. Peptide based vaccines are already used to treat cancer by a personal medicine approach and are already underway for COVID-19. In this approach antigenic peptides are introduced to cells enwrapped into nanoparticles (instead of current vaccines mRNA). The antigen producing sites (epitopes) can be obtained from the serum of convalescent COVID-19 patients screened for neutralizing antibodies. Likely these antibodies would be efficient to new variants if they would exclude the very binding site, which has changed [46]. Consortium just reported on the many epitopes of antibodies raised in recovalescents to other parts of spike than ACE-2 binding region, which could be used for further design of useful vaccines [47]. Somewhat reassuring is a recent report that the polyclonal antibodies elicited against SARS-CoV-2 may be resilient to substantial future SARS-CoV-2 variants. The authors have designed a polymutant of the spike protein and computationally showed that polyclonal neutralizing antibodies would neutralize the current delta and possible new variants [48].
4.2 What about innate immunity response?
Innate immunity boosters are another general strategy to be considered. Among them are live-attenuated vaccines (LAV) targeting tuberculosis, measles, and polio [49], which is a safe procedure to otherwise healthy people. Busting innate immunity can also be achieved by plant substances, such as Echinacea purpurea extract [50]. Of interest, a study found a range of memory T cells that can react to both the SARS COV-2 virus and the four previously known common cold coronaviruses and thus may augment innate immune defense [51]. However, another study opposes any significant cross-reactivity, even though the antibodies against common cold viruses are raised upon SARS CoV-2 infection [52]. A new vaccine is being studied as yet in mice, which is based on the common parts of proteins causing cross-reactivity to coronaviruses as recently published in Science [53]. Some immunologists believe that SARS CoV-2 will lose its strength in some years and will become one of the common coronaviruses. One would still encounter it every 2 – 3 years and each time get more or less mildly infected. On the contrary, others think more dangerous variants will survive while those neutralized by vaccines will dye.
4.3. Added along preparation of the review - as the field changes very fast:
Repurposing of approved drugs such as Ivermectin could be worthy of attention. An evidence-based review article discusses the mechanism of action of ivermectin against SARS-CoV-2 [54].
As mentioned, spike protein S can be cleaved by furin at the S1/S2 site and by the transmembrane serine protease 2 (TMPRSS2) at the S2' site. It was demonstrated that TMPRSS2 is essential for activation of SARS-CoV-2 S in human airway epithelial cells. Two serine protease inhibitors, camostat mesylate and nafamostat mesylate were tested human airway epithelial cells as well as mice models and proved effective in reducing viral load and also weight loss and mortality of mice [55]. Furthermore, combining various TMPRSS2 inhibitors with furin inhibitor MI-1851 produced more potent antiviral activity against SARS-CoV-2 than an equimolar amount of any single serine protease inhibitor [56].
Acknowledgement
I am part of the programme Proteolysis and its regulation (led by B. Turk). I am thankful for the continuous support.
Declarations
Funding
The author is financed by the programme P1- 140 funded by ARRS (Slovenian research agency).
Conflicts of interest/Competing interests
There is no conflict of interest
Availability of data and material
n.a.
Code availability
n.a.
Authors' contributions
The author composed and wrote the paper herself, after some informal discussion with a colleague, who prefers not to be mentioned.
References
- Evans N, Martinez E, Petrosillo N, et al. (SARS-CoV-2 and Human Immunodeficiency Virus: Pathogen Pincer Attack, HIV AIDS (Auckl) 13 (2021): 361-375.
- Illanes-Álvarez F, Márquez-Ruiz D, Márquez-Coello M, et al. Similarities and differences between HIV and SARS-CoV-2. Int J Med Sci 18 (2021): 846-851.
- Kanimozhi G, Pradhapsingh B, Singh Pawar C, et al. SARS-CoV-2: Pathogenesis, Molecular Targets and Experimental Models, Frontiers in Pharmacology 12 (2021).
- Borges Á H, O'Connor J L, Phillips A N, et al. Interleukin 6 Is a Stronger Predictor of Clinical Events Than High-Sensitivity C-Reactive Protein or D-Dimer During HIV Infection, The Journal of infectious diseases 214 (2016): 408-416.
- Tincati C, Douek D C, Marchetti G. Gut barrier structure, mucosal immunity and intestinal microbiota in the pathogenesis and treatment of HIV infection, AIDS research and therapy 13 (2016): 19.
- Papayannopoulos V. Neutrophil extracellular traps in immunity and disease, Nature reviews. Immunology 18 (2018): 134-147.
- Soehnlein O, Steffens S, Hidalgo A, et al. Neutrophils as protagonists and targets in chronic inflammation, Nature reviews. Immunology 17 (2017): 248-261.
- Mozzini C, Girelli D. The role of Neutrophil Extracellular Traps in Covid-19: Only an hypothesis or a potential new field of research?. Thromb Res 191 (2020): 26-27.
- Ouwendijk W J D, Raadsen M P, van Kampen J J A, et al. High Levels of Neutrophil Extracellular Traps Persist in the Lower Respiratory Tract of Critically Ill Patients With Coronavirus Disease 2019, The Journal of Infectious Diseases 223 (2021): 1512-1521.
- Daly J L, Simonetti B, Klein K, et al. Neuropilin-1 is a host factor for SARS-CoV-2 infection, Science 370 (2020): 861.
- Garten W, Hallenberger S, Ortmann D, et al. Processing of viral glycoproteins by the subtilisin-like endoprotease furin and its inhibition by specific peptidylchloroalkylketones, Biochimie 76 (1994): 217-225.
- Segreto R, Deigin Y, McCairn K, et al. Should we discount the laboratory origin of COVID-19?. Environmental Chemistry Letters (2021).
- Wu Y, Zhao S. Furin cleavage sites naturally occur in coronaviruses, Stem Cell Research 50 (2021): 102115.
- Lake M A. What we know so far: COVID-19 current clinical knowledge and research. Clinical medicine (London, England) 20 (2020): 124-127.
- Wang Q, Davis P B, Gurney M E, et al. COVID-19 and dementia: Analyses of risk, disparity, and outcomes from electronic health records in the US, Alzheimer's & dementia : the journal of the Alzheimer's Association (2021).
- Borges Á H, Neuhaus J, Babiker A G, et al. Immediate Antiretroviral Therapy Reduces Risk of Infection-Related Cancer During Early HIV Infection, Clin Infect Dis 63 (2016): 1668-1676.
- Cao B, Wang Y, Wen D, et al. A Trial of Lopinavir-Ritonavir in Adults Hospitalized with Severe Covid-19, N Engl J Med 382 (2020): 1787-1799.
- Mengist H M, Dilnessa T, Jin, T. Structural Basis of Potential Inhibitors Targeting SARS-CoV-2 Main Protease, Frontiers in Chemistry 9 (2021).
- Mandal A, Jha A K, Hazra B. Plant Products as Inhibitors of Coronavirus 3CL Protease. Frontiers in Pharmacology 12 (2021).
- Shin J A, Oh S, Jeong J-M. The potential of BEN815 as an anti-inflammatory, antiviral and antioxidant agent for the treatment of COVID-19, Phytomedicine Plus 1 (2021): 100058.
- Painter G R, Natchus M G, Cohen O, et al. Developing a direct acting, orally available antiviral agent in a pandemic: the evolution of molnupiravir as a potential treatment for COVID-19, Current opinion in virology 50 (2021): 17-22.
- Ho J S Y, Mok B W, Campisi L, et al. TOP1 inhibition therapy protects against SARS-CoV-2-induced lethal inflammation. Cell 184 (2021): 2618-2632.e2617.
- Arthur J M, Forrest J C, Boehme K W, et al. Development of ACE2 autoantibodies after SARS-CoV-2 infection. Plos One 16 (2021): e0257016.
- Prüss H. Autoantibodies in neurological disease, Nature reviews. Immunology (2021): 1-16.
- Farhadian S F, Seilhean D, Spudich S. Neuropathogenesis of acute coronavirus disease 2019, Current Opinion in Neurology 34 (2021).
- Douaud G, Lee S, Alfaro-Almagro F, et al. Brain imaging before and after COVID-19 in UK Biobank. medRxiv (2021).
- Manji H, Miller R. The Neurology of Hiv Infection. Journal of Neurology, Neurosurgery & Psychiatry 75 (2004): i29.
- Marban C, Forouzanfar F, Ait-Ammar A, et al. Targeting the Brain Reservoirs: Toward an HIV Cure 7 (2016).
- Michiels E, Rousseau F, Schymkowitz J. Mechanisms and therapeutic potential of interactions between human amyloids and viruses, Cellular and Molecular Life Sciences 78 (2021): 2485-2501.
- Gour S, Yadav J K. Aggregation hot spots in the SARS-CoV-2 proteome may constitute potential therapeutic targets for the suppression of the viral replication and multiplication, Journal of proteins and proteomics (2021): 1-13.
- Idrees D, Kumar V. SARS-CoV-2 spike protein interactions with amyloidogenic proteins: Potential clues to neurodegeneration, Bioche-mical and biophysical research communications 554 (2021): 94-98.
- Pratt J, Lester E, Parker R. Could SARS-CoV-2 cause tauopathy?. The Lancet Neurology 20 (2021): 506.
- Howdle G C, Quidé Y, Kassem M S, et al. Brain amyloid in virally suppressed HIV-associated neurocognitive disorder, Neurology - Neuroimmunology Neuroinflammation 7 (2020): e739.
- Zhou P, Yang X-L, Wang X-G, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin, Nature 579 (2020): 270-273.
- Hamming I, Timens W, Bulthuis M L, et al. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis, The Journal of pathology 203 (2004): 631-637.
- Wilen C B, Tilton J C, Doms R W. HIV: cell binding and entry, Cold Spring Harb Perspect Med 2 (2012): a006866.
- Wu Zhang X, Leng Yap Y. Structural similarity between HIV-1 gp41 and SARS-CoV S2 proteins suggests an analogous membrane fusion mechanism, Journal of Molecular Structure: THEOCHEM 677 (2004): 73-76.
- Sheleg S, Vasilevsky A. Could COVID-19 be a latent viral infection? Glob J Infect Dis Clin Res 6 (2020): 029-030.
- Zhang L, Richards A, Barrasa M I, et al. Reverse-transcribed SARS-CoV-2 RNA can integrate into the genome of cultured human cells and can be expressed in patient-derived tissues, Proceedings of the National Academy of Sciences 118 (2021): e2105968118.
- Yahi N, Chahinian H, Fantini J. Infection-enhancing anti-SARS-CoV-2 antibodies recognize both the original Wuhan/D614G strain and Delta variants. A potential risk for mass vaccination?. J Infect (2021).
- Idrees D, Kumar V. SARS-CoV-2 spike protein interactions with amyloidogenic proteins: Potential clues to neurodegeneration, Bioche-mical and biophysical research communications 554 (2021): 94-98.
- Luchini A, Micciulla S, Corucci G, et al. Lipid bilayer degradation induced by SARS-CoV-2 spike protein as revealed by neutron reflec-tometry, Scientific reports 11 (2021): 14867.
- Elrashdy F, Tambuwala M M, Hassan S S, et al. Autoimmunity roots of the thrombotic events after COVID-19 vaccination, Autoimmunity Reviews (2021): 102941.
- Heath P T, Galiza E P, Baxter D N, et al. Safety and Efficacy of NVX-CoV2373 Covid-19 Vaccine. The New England journal of medicine (2021).
- Meyers L M, Gutiérrez A H, Boyle C M, et al. Highly conserved, non-human-like, and cross-reactive SARS-CoV-2 T cell epitopes for COVID-19 vaccine design and validation, npj Vaccines 6 (2021): 71.
- He L, Lin X, Wang Y, et al. Single-component, self-assembling, protein nanoparticles presenting the receptor binding domain and stabilized spike as SARS-CoV-2 vaccine candidates, Sci Adv 7 (2021): eabf1591.
- Voss W N, Hou Y J, Johnson N V, et al. Prevalent, protective, and convergent IgG recognition of SARS-CoV-2 non-RBD spike epitopes, Science 372 (2021): 1108-1112.
- Schmidt F, Weisblum Y, Rutkowska M, et al. High genetic barrier to SARS-CoV-2 polyclonal neutralizing antibody escape. Nature (2021).
- Alavi Darazam, I, Shokouhi S, Pourhoseingholi MA, et al. Role of interferon therapy in severe COVID-19: the COVIFERON randomized controlled trial. Sci Rep 11, 8059 (2021).
- Zhai Z, Liu Y, Wu L, et al. Enhancement of innate and adaptive immune functions by multiple Echinacea species, Journal of medicinal food 10 (2007): 423-434.
- Sagar M, Reifler K, Rossi M, et al. Recent endemic coronavirus infection is associated with less-severe COVID-19, The Journal of Clinical Investigation 131 (2021).
- Anderson E M, Goodwin E C, Verma A, et al. Seasonal human coronavirus antibodies are boosted upon SARS-CoV-2 infection but not associated with protection. Cell 184 (2021): 1858-1864.e1810.
- Cohen A A, Gnanapragasam P N P, Lee Y E. Mosaic nanoparticles elicit cross-reactive immune responses to zoonotic coronaviruses in mice 371 (2021): 735-741.
- Zaidi AK, and Dehgani-Mobaraki P. RETRACTED ARTICLE: The mechanisms of action of Ivermectin against SARS-CoV-2: An evidence-based clinical review article, The Journal of Antibiotics 2021.
- Li K, Meyerholz DK, Bartlett JA and McCray P. B Jr. The TMPRSS2 Inhibitor Nafamostat Reduces SARS-CoV-2 Pulmonary Infection in Mouse Models of COVID-19 mBio 12 (2021): e0097021-e0097021.
- Bestle D, Heindl MR, Limburg H, Van Lam van T, Pilgram O, et al. TMPRSS2 and furin are both essential for proteolytic activation of SARS-CoV-2 in human airway cells, Life science alliance 3 (2020).
