Nutritional Genomics and Microbiota to Control Postprandial Dysmetabolism
Article Information
Giuseppe Merra1*, Annunziata Capacci2, Giuseppe Cenname3, Ernesto Esposito4, Maria Dri5, Laura Di Renzo1, Marco Marchetti1
1Section of Clinical Nutrition and Nutrigenomic, Department of Biomedicine and Prevention, University of Rome Tor Vergata, 00133 Rome, Italy
2Department of Medical and Surgical Sciences, Agostino Gemelli General Hospital Foundation-IRCCS, Rome, Italy
3Comando Generale Arma Carabinieri, Direzione di Sanità, 00197 Rome, Italy
4Department of Human Policies [General Directorate] of Basilicata Region, 85100 Potenza, Italy
5Department of Surgical Sciences, School of Applied Medical-Surgical Sciences, University of Rome Tor Vergata, 00133 Rome, Italy
*Corresponding Author: Giuseppe Merra, Section of Clinical Nutrition and Nutrigenomic, Department of Biomedicine and Prevention, University of Rome Tor Vergata, 00133 Rome, Italy
Received: 11 Macrh 2022; Accepted: 18 March 2022; Published: 22 March 2022
Citation: Giuseppe Merra, Annunziata Capacci, Giuseppe Cenname, Ernesto Esposito, Maria Dri, Laura Di Renzo, Marco Marchetti. Nutritional Genomics and Microbiota to Control Postprandial Dysmetabolism. Journal of Food Science and Nutrition Research 5 (2022): 398-429.
View / Download Pdf Share at FacebookAbstract
There is a causal relationship between cardiovascular risk and plasma levels of triglycerides, lipoproteins rich in triglycerides and remnants triglycerides. The importance of postprandial hyperlipidemia in the development of atherosclerosis was suggested by several studies. The mechanisms through which postprandial lipoproteins exert the atherogenic effect can be manifold. It has been hypothesized that the postprandial phase corresponds to an inflammatory condition involving leukocytes and potentially contributing to endothelial dysfunction. In the pathogenesis of postprandial lipemia alterations, insulin resistance undoubtedly plays a leading role that is expressed at various levels. Obesity is associated with hypertriglyceridemia and low fasting HDL cholesterol levels. These abnormalities could explain the high incidence of cardiovascular disease in the obese population. In recent years there has been a dramatic increase in diseases related to the alteration of metabolism, causing conditions such as obesity, type 2 diabetes (TD2), cardiovascular disease and metabolic syndrome (MetS). In addition to the numerous factors to which this increase has been attributed, such as diet, physical activity and inflammation, numerous studies provide evidence of a correlation between intestinal microbiota and metabolic diseases. The aim of this umbrella review is clarify the interactions between diet and lifestyle, microbes and health and therefore the relationship between gut microbiota and metabolic diseases so that one day we may perhaps know exactly how to direct our diet in order to be able to "shape" our microbiota to im-prove our health.
Keywords
Metabolism, Obesity, Microbiota
Metabolism articles; Obesity articles; Microbiota articles
Metabolism articles Metabolism Research articles Metabolism review articles Metabolism PubMed articles Metabolism PubMed Central articles Metabolism 2023 articles Metabolism 2024 articles Metabolism Scopus articles Metabolism impact factor journals Metabolism Scopus journals Metabolism PubMed journals Metabolism medical journals Metabolism free journals Metabolism best journals Metabolism top journals Metabolism free medical journals Metabolism famous journals Metabolism Google Scholar indexed journals Obesity articles Obesity Research articles Obesity review articles Obesity PubMed articles Obesity PubMed Central articles Obesity 2023 articles Obesity 2024 articles Obesity Scopus articles Obesity impact factor journals Obesity Scopus journals Obesity PubMed journals Obesity medical journals Obesity free journals Obesity best journals Obesity top journals Obesity free medical journals Obesity famous journals Obesity Google Scholar indexed journals Microbiota articles Microbiota Research articles Microbiota review articles Microbiota PubMed articles Microbiota PubMed Central articles Microbiota 2023 articles Microbiota 2024 articles Microbiota Scopus articles Microbiota impact factor journals Microbiota Scopus journals Microbiota PubMed journals Microbiota medical journals Microbiota free journals Microbiota best journals Microbiota top journals Microbiota free medical journals Microbiota famous journals Microbiota Google Scholar indexed journals triglycerides articles triglycerides Research articles triglycerides review articles triglycerides PubMed articles triglycerides PubMed Central articles triglycerides 2023 articles triglycerides 2024 articles triglycerides Scopus articles triglycerides impact factor journals triglycerides Scopus journals triglycerides PubMed journals triglycerides medical journals triglycerides free journals triglycerides best journals triglycerides top journals triglycerides free medical journals triglycerides famous journals triglycerides Google Scholar indexed journals type 2 diabetes articles type 2 diabetes Research articles type 2 diabetes review articles type 2 diabetes PubMed articles type 2 diabetes PubMed Central articles type 2 diabetes 2023 articles type 2 diabetes 2024 articles type 2 diabetes Scopus articles type 2 diabetes impact factor journals type 2 diabetes Scopus journals type 2 diabetes PubMed journals type 2 diabetes medical journals type 2 diabetes free journals type 2 diabetes best journals type 2 diabetes top journals type 2 diabetes free medical journals type 2 diabetes famous journals type 2 diabetes Google Scholar indexed journals cardiovascular risk articles cardiovascular risk Research articles cardiovascular risk review articles cardiovascular risk PubMed articles cardiovascular risk PubMed Central articles cardiovascular risk 2023 articles cardiovascular risk 2024 articles cardiovascular risk Scopus articles cardiovascular risk impact factor journals cardiovascular risk Scopus journals cardiovascular risk PubMed journals cardiovascular risk medical journals cardiovascular risk free journals cardiovascular risk best journals cardiovascular risk top journals cardiovascular risk free medical journals cardiovascular risk famous journals cardiovascular risk Google Scholar indexed journals postprandial hyperlipidemia articles postprandial hyperlipidemia Research articles postprandial hyperlipidemia review articles postprandial hyperlipidemia PubMed articles postprandial hyperlipidemia PubMed Central articles postprandial hyperlipidemia 2023 articles postprandial hyperlipidemia 2024 articles postprandial hyperlipidemia Scopus articles postprandial hyperlipidemia impact factor journals postprandial hyperlipidemia Scopus journals postprandial hyperlipidemia PubMed journals postprandial hyperlipidemia medical journals postprandial hyperlipidemia free journals postprandial hyperlipidemia best journals postprandial hyperlipidemia top journals postprandial hyperlipidemia free medical journals postprandial hyperlipidemia famous journals postprandial hyperlipidemia Google Scholar indexed journals diet articles diet Research articles diet review articles diet PubMed articles diet PubMed Central articles diet 2023 articles diet 2024 articles diet Scopus articles diet impact factor journals diet Scopus journals diet PubMed journals diet medical journals diet free journals diet best journals diet top journals diet free medical journals diet famous journals diet Google Scholar indexed journals lipoproteins articles lipoproteins Research articles lipoproteins review articles lipoproteins PubMed articles lipoproteins PubMed Central articles lipoproteins 2023 articles lipoproteins 2024 articles lipoproteins Scopus articles lipoproteins impact factor journals lipoproteins Scopus journals lipoproteins PubMed journals lipoproteins medical journals lipoproteins free journals lipoproteins best journals lipoproteins top journals lipoproteins free medical journals lipoproteins famous journals lipoproteins Google Scholar indexed journals
Article Details
1. Introduction
There is a causal relationship between cardiovascular risk and plasma levels of tri-glycerides, lipoproteins rich in triglycerides and remnants triglycerides. Lipoproteins rich in triglycerides and remnants accumulate in the plasma at TG values above 100 mg/dL and cardiovascular risk increases significantly for triglyceridemia greater than 150 mg/dL [1]. So the pathogenetic relationship between hypertriglyceridemia and cardio-vascular disease has been enough clarified thanks to the large amount of research carried out in recent decades but it has yet to be fully explained. One reason behind these diffi-culties is, first of all, the fact that the lipoproteins that transport triglycerides in the plasma (chylomicrons, very low density lipoproteins - VLDL - and their remnants, i.e. the particles that are formed by their catabolism) constitute a population very heterogeneous in origin, structure and mode of interaction with cell receptors, so it is likely that they also have a different degree of atherogenicity [1]. But an even more important reason is certainly the fact that the plasma levels of lipids used to assess cardiovascular risk are measured in the fasting state and not in the postprandial phase, a condition in which, also considering today's conditions of well-being, we practically spend the 'all day. It should be considered that "postprandial lipemia" is a physiological process in which plasma lipoproteins and their subfractions undergo specific changes in concentration and composition following the intake of a meal, and in particular, a meal rich in fat. By "postprandial hyperlipidemia", on the other hand, we mean the quantitative, or even qualitative, alterations of this process. The clinical relevance of these lipoprotein alterations [2] lies in the important role they appear to play in the development of cardiovascular diseases. The aim of this umbrella review is clarify the interactions between diet and lifestyle, microbes and health and therefore the relationship between gut microbiota and metabolic diseases so that one day we may perhaps know exactly how to direct our diet in order to be able to "shape" our microbiota to improve our health.
2. Postprandial metabolism of lipoproteins
Most dietary fats are represented by triglycerides, which under normal conditions are almost completely absorbed in the small intestine, where pancreatic lipase splits the ester bond between fatty acids and glycerol. The intestinal lipid content is emulsified by bile salts and phospholipids and collected in water-soluble micelles which penetrate the enterocytes by diffusion. Glycerol and short-chain fatty acids (with less than 10-12 carbon atoms) then enter the bloodstream and are transported directly to the liver via the portal vein while long-chain fatty acids are reesterified to triglycerides and introduced into the chylomicrons, which instead enter the bloodstream through the lymphatic one. The subsequent metabolic fate of triglycerides is determined by the protein component of the chylomicrons. Apolipoprotein (apo) B-48, synthesized in the intestine, is its structural component. Apo C-II, activator of lipoprotein lipase (LPL) and apo E, ligand of the receptor for chylomicron remnants, are transferred to them by plasma HDL (high density lipoprotein). LPL linked to the vascular endothelium of adipose tissue and muscle tissue mediates the hydrolysis of most of the triglycerides contained in the chylomicron core. In conjunction with this lipolysis, phospholipids and other surface molecules are transferred to the HDL, which donate cholesterol esters in exchange for triglycerides. The remnants of the chylomicrons are then cleared from the circulation by receptor-mediated hepatic uptake. VLDL of hepatic origin contribute to the pool of postprandial plasma lipoproteins. Like chylomicrons, they undergo a reduction in their triglyceride content by the action of LPL and are then removed from the circulation by endocytosis mediated by the receptor that recognizes apo B-100 and apo E. A certain amount of VLDL, however, it is directly converted into intermediate (IDL) and low (LDL) density lipoproteins. Therefore, in the postprandial phase the particles of intestinal origin (chylomicrons and their remnants) and those of hepatic origin (different subfractions of VLDL) compete for the same enzymatic and receptor systems responsible for their catabolism [3].
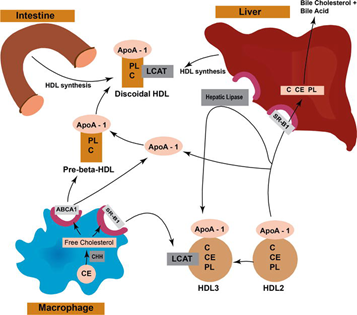
Figure 1: Lipoprotein metabolism (by Zhyvotovska, Yusupov, McFarlane, Introductory Chapter: Overview of Lipoprotein Metabolism, DOI: 10.5772/intechopen.85094).
2.1 Role of the apo E genotype on postprandial lipemia
There are different isoforms of apo E (E2, E3 and E4), which seem to have different binding properties and therefore affect the efficiency of the receptor-mediated uptake of the remnants of the chylomicrons. The genotype E2/E2, which is present only in 1% of the general population, but in 90% of patients with hyperlipidemia of type III, is associated with prolonged retention of chylomicron remnants compared to the more common E3/E33 genotype. However, it is still controversial whether the heterozygous E2 geno-type is associated with a delay in the clearance of chylomicron remnants in normolipidemic subjects. Indeed, a reduced clearance in subjects with the E2/E3 genotype was observed in some studies in both healthy subjects [5-7] and in patients with type 2 diabetes mellitus [7] but not in other studies in healthy subjects [4,9-11] and in obese women [12]. Also for the E4 allele, which is associated with increased fasting total and LDL cholesterol levels and an increased cardiovascular risk [13,14], the data are conflicting regarding the independent effect on the postprandial lipemic response [5-6,10,15-16]. A preferential association of the E4 allele with lipoproteins rich in triglycerides (TRL) was found compared to HDL [17-18]. This different conformation of the apo E4 on the TRLs could produce a reduction in receptor binding compared to the apo E3 and therefore a lower uptake of VLDL with a consequent greater conversion of these lipoproteins into LDL [19]. It can be concluded that the apo E genotype is most likely an important factor in determining the postprandial lipemic response, but further studies are needed to clarify the relative weight of the different isoforms regardless of the basal values of plasma lipids and in different populations.
2.2 Methods of evaluation of postprandial lipemia
There are several methods for studying postprandial hyperlipemia [20]. The meth-odological objective is the precise characterization and quantification of lipoproteins rich in triglycerides that are formed in the postprandial period. The determination of plasma triglycerides after a meal is the simplest method, but it does not give information on the distribution in the different lipoproteins and does not allow to distinguish the exogenous particles from those of hepatic origin. Ultracentrifugation (UCF) allows lipoproteins to be separated based on their density and therefore based on their sedimentation constant (Sf). The different UCF techniques (analytical, preparative in solvent density and in density gradient, zonal etc.) allow the isolation, among the various classes of lipoproteins, of different subfractions in physico-chemical characteristics, but also in origin and fate metabolic. Many researchers have used the metabolic pathway of vitamin A on the basis that it is extensively incorporated into chylomicrons in the form of retinyl palmitate [21]. In human plasma, only a small amount of retinyl palmitate is exchanged with other lip-oproteins, while, for the most part, it remains part of the chylomicrons until their hepatic uptake. Furthermore, vitamin A is excreted by the liver bound exclusively to reti-nol-binding proteins present in chylomicrons and is not incorporated into VLDL or LDL. Retinyl palmitate is therefore a specific marker for chylomicrons and their remnants, at least in the first part of the postprandial phase. The direct quantification of chylomicrons can be obtained through the determination of apo B-48 using an electrophoresis on so-dium polyacrylamide dodecyl-sulfate gel which also allows its separation from particles containing apo B-100 by exploiting their different molecular weight [22]. It is a method subject to wide biological variability, but it is able to give information on the number of circulating particles, as there is one apo B-48 particle for each chylomicronemic particle. Very recently, an enzyme immunoassay (ELISA) method has been proposed for the de-termination of apo B-48 which uses monoclonal antibodies directed against apo B-48 [23]. Data on its reliability are still missing. There are also immunological methods for the separation of remnants-like particles (RLP) from VLDL, which involve the use of im-munoaffinity chromatography with monoclonal antibodies directed against an apo B-100 epitope, which is not present in the apo B-48, and which in addition to apo B-48 do not recognize a population of particles rich in triglycerides containing apo B-100 and rich in cholesterol esters and apoE [24]. In conclusion, the detailed assessment of postprandial lipemia, that is, the exact differentiation of the particles with regard to their lipid com-position and their hepatic or intestinal origin, is difficult to perform as the methods cur-rently available are complex and expensive. On the other hand, the simple determination of postprandial triglyceridemia, possible through the use, even at home, of portable re-flectometers, can provide useful information for the identification and correction of the condition of postprandial hyperlipidemia [25-26].
2.3 Standardization of the test meal
Currently, very different meals are used for the study of dietary lipemia and this naturally makes it difficult to compare the results of different studies and makes it im-possible to establish criteria for the normality of the lipemic response [27,28]. For a de-sirable standardization of the meal, several variables must be considered including mainly the type of nutrients to be included in the meal (fats/carbohydrates), the chemical composition of fats (saturated, monounsaturated, polyunsaturated), the caloric content of the meal and the time of its intake, in addition to the environmental variables that are able to influence postprandial lipemia, such as smoking, exercise and alcohol intake [19,28]. A key element is the inclusion or not of nutrients other than lipids. In fact, meals composed only of fats or containing a variable proportion of carbohydrates were used, ranging from a load of cream to a mixed liquid meal or, more adherent to real life, solid. Of course, a meal made up of 100% fat, using for example cream as the only component, has the advantage of being simple to make and more reproducible and comparable in different situations and geographical areas than a mixed meal. On the other hand, it is too far from the usual diet as a "normal" meal completely devoid of carbohydrates is difficult to achieve. It is therefore impossible to build a standard meal that does not interfere with carbohydrate metabolism at all. In this regard, some studies suggest that sucrose, ingested with fat, increases postprandial triglyceridemia [29-31], unlike glucose which could even increase the clearance of chylomicrons probably due to the stimulation of insulin secretion. A high quantity of complex carbohydrates, on the other hand, has a fairly low acute lipogenic effect [32]. As for the protein content of the meal, it does not appear to influence postprandial triglyceridemia [33]. As regards the type of fats eaten with the meal, it must first of all be considered that dairy products contain a significant amount of short and medium chain fatty acids, which are absorbed as such and do not form chylomicrons, in addition to probably having an accelerated postprandial clearance [34-35]. Some studies [36,37], but not others [38,39], have found the formation of smaller chylo-microns with the intake of saturated fats than unsaturated ones. This could be due to methodological problems, as at 4°C (temperature at which plasma is generally treated) the chylomicrons rich in saturated fatty acids tend to flocculate more easily since the core components move from the liquid to the crystalline phase [40]. This does not happen when the determination is carried out at 27°C. Vegetable oils, on the other hand, have a higher melting point and are therefore preferable from an analytical-methodological point of view. There is evidence that n-3 fatty acids, contained in fish oil, produce a lower triglyceride response than vegetable fatty acids and even more so in saturated ones [41-43]. In a study in healthy young people, a meal containing butter induced a higher response than one containing olive oil, with an increase in triglycerides concerning the fraction rich in chylomicrons and a greater and prolonged response of retinyl palmitate in this fraction [44]. However, it was reported in rats that clearance of chylomicrons containing a saturated fatty acid, palmitic, was faster than chylomicrons containing oleic or linoleic acid [41]. Overall, the fatty acid composition of the acute fat load does not appear to have a relevant effect on the lipemic response. With regard to the size of the meal, it must be taken into account that this should be such as to stimulate the metabolism without compromising absorption and gastric emptying times and, in any case, palatability. The optimal amount of fat in a meal should be between 50 and 80g [45,46]. A necessary condition for the execution of the meal is that this occurs fasting since the plasma levels of triglycerides before a fat load are a predictor of the postprandial lipemic response [47]. It would also be advisable to prohibit the intake of alcohol the day before the test as well as the execution of strenuous physical activity as it is able to modify the ability to remove circulating triglycerides [48]. In women, then, it is necessary to consider the possible intake of estrogen-based contraceptives as the latter seem to influence the metabolism of chylomicron remnants [49]. In conclusion, there is currently no consensus on the type of meal to be used to stimulate the lipemic response. However, the clinical-diagnostic need which would require a less physiological meal, but more reproducible at an interpersonal and population level, must be distinguished from the clinical-prognostic need which in-stead can benefit from the detection of the lipemic response to the usual diet of a single individual.
2.4 Postprandial hyperlipidemia and cardiovascular diseases
The importance of postprandial hyperlipidemia in the development of atheroscle-rosis was suggested by the studies of Zilversmit, in which it was shown in animal models how the esterified cholesterol of the remnant of the chylomicrons accumulated in the anterior part of the aorta [50]. The evidence supporting the atherogenic effect of post-prandial plasma lipoproteins is numerous. These are mainly studies that have compared the postprandial lipemic response of patients with different clinical forms of atheroscle-rosis with that of control subjects without vasculopathy, by evaluating the degree of coronary obstruction [51-53] and the presence of peripheral arterial disease [54-56], but also of early indicators of vascular involvement such as carotid mid-intimal thickness [57-59]. Already in the 1950s it was reported that coronary heart disease patients showed increased levels of plasma triglycerides for a prolongation of the postprandial condition [60]. A relationship with ischemic heart disease was then noted by Simons et al. [61] with an increased apo B-48/apo B-100 ratio in the postprandial phase and by Simpson et al. [62] with prolonged postprandial lipemia evaluated with retinyl palmitate. Patients with hypertriglyceridemic ischemic heart disease have a slowed removal of chylomicrons from plasma [3]. Furthermore, the progression of the disease over a period of 5 years was related to the level of lipoprotein containing apo B-48 [63]. In a group of patients with severe coronary artery stenosis, age, postprandial plasma levels of triglyc-erides and fasting apo B were the best indicators of coronary risk [64]. Ginsberg et al. [65] also reported that myocardial ischaemia was associated with high postprandial plasma triglyceride levels regardless of other risk factors such as fasting LDL and HDL concen-tration. More recently it has been confirmed that the removal of chylomicron remnants from the circulation is slowed in both male [52] and female patients with normolipidemic ischemic heart disease [66,67]. It should be remembered that post-menopausal women, with or without ischemic heart disease, experienced a slowed removal of chylomicron remnants, so it is possible that estrogen deficiency is associated with impaired postprandial metabolism [68]. In conclu-sion, these studies indicate quite clearly that slow removal of chylomicrons from the remnant circulation is an independent risk factor for the development of atherosclerosis. At present, there are no prospective studies that have assessed the incidence of cardio-vascular events in relation to the presence or absence of postprandial lipemia alterations. There are also no intervention studies on the effect of correcting postprandial lipemia on cardiovascular disease.
2.5 Possible atherogenic mechanisms
The mechanisms through which postprandial lipoproteins exert the atherogenic ef-fect can be manifold. The remnants of lipoproteins rich in triglycerides could act directly in an atherogenic sense. In fact, they have been detected in human atherosclerotic plaques [69] and can promote an inflammatory response in the arterial wall [70]. Rem-nants could also act by intervening in the coagulation processes, as it has been observed that they promote the activation of factor VII, an enzyme important in the production of thrombin, which in turn converts fibrinogen to fibrin in clot formation [71,72]. The acti-vation of platelets and monocytes is a process potentially involved in the development of cardiovascular disease which appears to be associated with postprandial lipemia. In fact, after a moderately high-fat meal (40%) there was a significant increase in platelets ex-pressing P-selectin on the surface and increased activation of the GP IIIb-IIa receptor, as well as an increase in platelet-monocyte aggregates and percentage of monocytes ex-pressing TN-α and IL-1 beta [73].
2.6 Endothelial dysfunction
It has been hypothesized that the postprandial phase corresponds to an inflamma-tory condition involving leukocytes and potentially contributing to endothelial dysfunc-tion. In fact, an increase in the blood of neutrophils, IL-8 and hydroperoxides in associa-tion with a reduction in flow-mediated vasodilation was observed in a group of healthy males during postprandial lipemia [74]. It also appears that a substantial increase in postprandial hyperlipidemia is associated with a reduction in endothelium-mediated vasodilation in the brachial artery [75-77]. This functional and structural marker of early atherosclerosis correlates proportionally to the degree of postprandial hyperlipidemia as it is related to the reduction of HDL, with antioxidant potential, and the increase of small and dense LDL which, on the contrary, oxidize easily [78-81]. Evans et al. [82] also ob-served that postprandial deterioration of endothelial function correlated directly with triglyceride levels in VLDL and LDL and inversely with HDL cholesterol. In this study, in the postprandial phase, the triglyceride enrichment of VLDL was the only parameter correlated with oxidative stress evaluated as the level of free radicals. In conclusion, there is evidence that postprandial lipemia alters the function of the endothelium predisposing it to vasoconstriction and causing a proinflammatory and procoagulant state in various physiological and pathophysiological conditions. It would be interesting to link these activities with the development of atherosclerosis and/ or the triggering of acute coronary syndromes, but the evidence is still contradictory [83].
2.7 Postprandial hyperlipidemia and insulin resistance
In the pathogenesis of postprandial lipemia alterations, insulin resistance un-doubtedly plays a leading role that is expressed at various levels. First of all, the altered sensitivity of the adipose and hepatic tissue to the action of insulin causes the normal suppression of the release of free fatty acids in the first and the production of VLDL in the second is altered. This implies that in the postprandial state the chylomicrons of intestinal origin and the hepatic VLDLs produced in excess compete for the common disposal mechanism that involves both the action of the LPL and the receptor-mediated uptake. In this regard, there are data that show, in the course of insulin resistance, a reduction in the expression of lipoprotein lipase in adipose tissue and an increase in the concentration of hepatic lipase [84]. Despite such evidence, the role of lipolytic enzymes in the pathogenesis of postprandial lipemia remains controversial [85]. Since insulin resistance is the common denominator of the various metabolic abnormalities that make up the metabolic syndrome, it contributes significantly to the genesis of postprandial lipid alterations present in these conditions [86].
2.8 Type 2 diabetes mellitus
Postprandial hyperlipidemia, understood as the high persistence in the circulation of chylomicrons, VLDLs and their remnants, is an element characterizing the dyslipidemia of patients with type 2 diabetes [86-88]. The postprandial triglyceride peak is delayed (4-6 hours) and this means that, due to the succession of meals and the progressive accumulation, diabetic patients have high concentrations of triglycerides throughout the day, as has been found in studies in which the profile daily triglyceridemia was determined in patients with type 2 diabetes using a portable device [24]. The abnormalities of postprandial metabolism observed in type 2 diabetes consist of a prolonged stay in the circulation of both intestinal and hepatic origin particles, especially the remnants of these particles. These alterations have been found in diabetic patients with fasting hypertriglyceridemia. even if only moderate, and / or unsatisfactory glycometabolic compensation, while the observations are more disagree with regard to diabetic patients without fasting hypertriglyceridemia and in excellent glycometabolic compen-sation [89-92]. However, Rivellese et al.[93] have shown that even subjects with type 2 diabetes, fasting normotriglyceridemics and with optimal glycemic compensation have alterations in postprandial lipoproteins affecting both exogenous and endogenous lipo-proteins. There are numerous evidences supporting the relationship between insulin re-sistance, which is a fundamental element in the pathogenesis of type 2 diabetes, and the alterations of postprandial lipid metabolism. Jeppesen et al. [94] have, in fact, observed that in healthy non-diabetic individuals, insulin-mediated resistance to the use of glucose was an independent factor associated with lipid alterations in the postprandial phase. Axelsen et al. [95] also reported an increase in the postprandial incremental area of triglycerides after a standard mixed meal in first-degree relatives of type 2 diabetic pa-tients, who presented insulin resistance, but were normoglycemic and normotriglycer-idemic. Annuzzi et al. [96] experimentally demonstrated for the first time the specific role of insulin resistance in type 2 diabetic patients in the absence of the confounding factors represented by hyperglycemia and hyperinsulinemia, which are characteristic of these patients. In fact, their lipemic response to a high-fat meal during a hyperinsulinemic glycemic clamp, compared with that of non-diabetic control subjects, was altered due to a postprandial increase in lipoproteins rich in triglycerides of both exogenous and endog-enous origin. The fact that this has been observed in patients with excellent glycemic control and optimal fasting triglyceride values is clinically relevant, as it suggests that these postprandial changes are even more evident in patients with glycemic metabolic decompensation.
2.9 Obesity
Obesity is associated with hypertriglyceridemia and low fasting HDL cholesterol levels. These abnormalities could explain the high incidence of cardiovascular disease in the obese population. However, obesity appears to be an independent risk factor for coronary heart disease even in subjects with normal fasting plasma lipid levels and glu-cose tolerance. Android-type obesity patients with normal fasting triglyceride levels and no different from those of healthy controls show an abnormal postprandial response of plasma lipids, indicated by an increase in triglycerides in non-chylomicronemic subfrac-tions, significantly higher than in healthy controls [96]. These alterations could be due, in particular, to the modifications of the endogenous lipoproteins consequent to the insulin resistance. In particular, a study by Couillard et al. [97] investigated the relationship be-tween postprandial lipemia, fasting triglyceridemia, and visceral accumulation of adipose tissue. The results of this study suggested that the increase in lipolysis at the level of visceral adipocytes, due to the state of insulin resistance related to obesity, determines an increase in plasma levels of triglycerides and VLDL for the hepatic reesterification of high levels of non-esterified fatty acids released into the portal circulation.
2.10 Hypertension
A study by Kolovou et al.[98] examined the relationship between hypertension and postprandial hyperlipidemia by comparing the response to a high-fat meal of a group of 25 hypertensive patients with that of 25 healthy subjects. The results of this study indi-cated that hypertension, regardless of other conditions of the metabolic syndrome, is associated with an excessive postprandial peak and delayed clearance of plasma triglyc-erides.
2.11 Other conditions associated with insulin resistance
Recent data have highlighted the existence of a relationship between insulin re-sistance syndrome and cigarette smoking[99]. It has been shown that some aspects of the insulin resistance syndrome such as hyperinsulinemia, high levels of hypertriglycer-idemia, reduced HDL-cholesterol levels and dysfibrinolysis are related to the daily con-sumption of nicotine. Axelsen et al[100] observed that smokers also seem to have an impaired elimination of triglycerides after the intake of a high-fat meal regardless of fasting triglyceride values.
2.12 Treatment of postprandial hyperlipidemia
2.12.1 Diet: Studies in humans and animals have shown that the fatty acid composition of the usual diet can influence the extent and duration of postprandial lipemia. First of all, as already discussed, fatty acids a medium and short chain are not incorporated in the chylomicrons so there is less postprandial increase triglyceridemia when more medium- and short-chain fatty acids are used than those of the chain long. It has been observed that polyunsaturated fatty acids of the ω3 type are able to reduce the concentration of chylomicrons and their remnants, probably as a result of a reduced production of VLDL and therefore less competition for the lipolysis of these particles in the postprandial phase [101,102]. On the contrary, there is evidence of a higher and prolonged postprandial lipemic response in the presence of an increased dietary intake of saturated fatty acids compared to polyunsaturated ones and this seems to be due to an increased secretion of VLDL [103,104]. As regards the influence of the dietary intake of cholesterol, it was reported that no differences were observed in postprandial lipemia if the diet contained 0 to 4 eggs per day [105]. In this study, however, healthy young subjects were evaluated for which the effect may be different in patients with cardiovascular risk factors and, more-over, it must be remembered that the effects of dietary cholesterol are dependent on the fatty acid composition of the diet. It has been shown that the cholesterol esters contained in chylomicrons (and probably also free cholesterol) derive from endogenous sources and that only a limited quantity is of food origin [106], even if 50% of the cholesterol taken from the diet is still absorbed and can not only be transported by the large chylomicrons that are secreted by the intestine immediately after a meal, but also by small chylomicrons produced continuously in the post-absorbing state [107]. With regard to the other components of the diet, it should be remembered that carbohydrates, if chronically con-sumed in excessive quantities (especially if it is sucrose and fructose), can induce an ex-cessive formation of VLDL by the liver and therefore hypertriglyceridemia [108]. In these conditions, the postprandial lipemic response is increased [109], particularly in patients with diabetes mellitus type 2 [110]. Dietary fibers, with a well-documented cholester-ol-lowering effect, which has been attributed to intraluminal sequestration of cholesterol, appear to have a limited effect on postprandial triglycerides [111-113]. Acute and chronic alcohol intake is associated with a slowdown in the removal of postprandial triglycer-ides [114] and the effect seems to be more evident if the meal contains saturated fatty ac-ids [115].
2.12.2 Physical activity: Several studies indicate that a training period reduces postprandial lipemia [116-119]. There is evidence that even a single session of exercise is able to improve the postprandial lipemic response [120] and that the effects are only observed up to three days after the end of a training period [121] and could therefore be the result of the last training session [48]. This therefore implies that, in order to maintain the benefits in terms of reducing the presence of postprandial atherogenic particles in the circulation, physical exercise, in line with the recommendations proposed for improving cardiovascular risk, must be conducted regularly and frequently.
2.12.3 Pharmacological therapy: Lipid-lowering drugs. The drugs commonly used to treat hyperlipidemias seem to be able to reduce the concentration of lipoproteins rich in triglycerides and their remnants present in the postprandial phase. Several studies have evaluated the effects of fibrates. In a study in type 2 diabetic patients with moderate hypertriglyceridemia, gemfibrozil reduced the concentration of the largest postprandial lipoproteins (Sf> 400), of almost exclusively intestinal origin [122]. In this study, the largest VLDLs (Sf 60-400) also decreased with a reduction in retinyl palmitate in this fraction, demonstrating the effect of chylomicrons on remnants. Similar results were obtained with gemfibrozil in hyper-lipidemic patients [123], while a reduction in the triglyceride concentration in the post-prandial fraction d <1006 was observed with fenofibrate, but not in the levels of retinyl palmitate [62]. Attia et al. [124] observed a reduction in triglycerides in fractions with Sf> 400, 12-400 and <12 both fasting and after a predominantly lipid meal in moderately hy-perlipidemic diabetic patients treated for four weeks with bezafibrate. The hypothesized mechanism underlying these results would be the ability of the fibrates to reduce the levels of triglycerides of endogenous origin with a consequent lower competition between particles of endogenous and exogenous origin at the level of receptor-mediated hepatic removal. Treatment with bezafibrate also appears to be able to restore normal cholesterol efflux levels from both fasting and postprandial cells by improving the reverse transport of cholesterol, the latter mechanism which in insulin-resistant patients is altered due to slowing postprandial lipolysis of lipoproteins rich in triglycerides [125]. Other mechanisms on which fibrates could act are increased oxidative stress and endothelial dysfunction, phenomena which, as already discussed, are observed during the postprandial phase. In fact, in a controlled study in 20 diabetic patients, treatment for three months with ciprofibrate resulted in the reduction of fasting plasma triglycerides and their incremental postprandial area, as well as an increase in HDL cholesterol levels. And these effects were closely correlated with the improved endothelial function and reduction of oxidative stress observed in the postprandial phase [126].Several studies have evaluated the effects of statins on postprandial lipemia. In patients with type 2 diabetes in stable glycemic control and combined hyperlipidaemia, treatment with simvastatin induced not only a reduction in total and LDL cholesterol levels and an increase in fasting HDL cholesterol, but also a reduction in postprandial triglycerides [127]. Atorvastatin was also effective compared to placebo in reducing postprandial triglyceridaemia in a group of patients with impaired fasting glucose [128]. In a study in patients with mixed hyperlipidaemia, the beneficial effects of atorvastatin on postprandial lipemic profile and cardiovascular risk were enhanced by the addition of a low dose of omega-3 fatty acids, which produced a further significant increase in cholesterol levels. HDL and decrease in small and dense LDL concentration and postprandial triglyceridemia [129]. The association atorvastatin-omega-3 fatty acids was also found to be able to reduce the activation of coagulation in the course of postprandial lipemia, reducing the concentration of activated factor VII, its coagulant activity and the FVII antigen [129]. Some studies have compared the postprandial effects of statins with those of fibrates. In a study that compared gemfibrozil and lovastatin in patients with hypoalphalipoproteinemia and mild/moderate hypertriglyceridemia, the reduction in fasting triglyceridemia was related to the improvement in postprandial lipemia, probably thanks to the increase in LPL mediated lipolysis [130]. Gemfibrozil was more effective than lovastatin in reducing postprandial lipemia[131]. Wilmink et al. [132] compared the effects of cerivastatin and gemfibrozil on changes in endothelial function induced by a high-fat meal. Cerivastatin but not gemfibrozil was able to significantly improve postprandial endothelial dysfunction in direct proportion to the effect on lipoproteins, suggesting that lowering remnant cholesterol rather than lowering total triglyceride levels may contribute to the prevention of endothelial dysfunction. postprandial. Statins are the treatment of choice for lowering LDL cholesterol because they have proven efficacy in reducing cardiovascular morbidity and mortality. Statins inhibit hydroxymethylglutaryl-CoA reductase, a key enzyme in the synthesis of cholesterol, leading to positive regulation of LDL receptors and increased LDL clearance. They reduce LDL cholesterol by up to 60% and cause small increases in HDL and a modest reduction in triglycerides. Additionally, statins appear to reduce intra-arterial inflammation and systemic inflammation, or both, by stimulating endothelial nitric oxide production and may have other beneficial effects. Other classes of lipid-lowering drugs are not the first choice, because they have not demonstrated equivalent efficacy for reducing atherosclerotic cardiovascular disease [134]. Statin treatment is classified as high, moderate or low intensity and is given based on treatment group and age. Statin choice may depend on patient comorbidities, other medications, risk factors for adverse events, statin intolerance, cost and patient preference. Adverse effects with statins are rare but include elevated liver enzymes and myositis or rhabdomyolysis. Elevations in liver enzymes are rare, and severe liver toxicity is extremely rare. Muscle problems occur in 10% of statin-treated patients and can be dose-dependent. Muscle symptoms can occur without enzyme elevation. Adverse effects are more frequent in elderly patients, in patients with several comorbidities and in those taking numerous drug therapies. In some patients, changing the statin type or reducing the dose (after temporarily stopping the drug) solves the problem. Muscle toxicity appears to be more common if some of the statins are used in combination with drugs that inhibit cytochrome P3A4 (eg, macrolide antibiotics, azole antifungals, cyclosporine) and with fibrates, especially gemfibrozil. Statins are contraindicated during pregnancy and breastfeeding. In patients with atherosclerotic cardiovascular disease, the greater the reduction in LDL cholesterol achieved by statin therapy, the greater the reduction in risk. Thus, the initial treatment is a statin at the maximum tolerated dose to reduce LDL cholesterol> 50% (high intensity therapy). Despite maximal statin therapy, patients with very high-risk athero-sclerotic cardiovascular disease (eg, those with recent myocardial infarction or unstable angina, or with high-risk comorbidities such as diabetes) and LDL cholesterol values > 70 mg / dL (> 1.2 mmol / L) should be added to therapy with ezetimibe or a PCSK9 inhibitor (eg, evolocumab, alirocumab). With the advent of combination therapy using ezetimibe and/or proprotein convertase subtilisin/kexin type 9 inhibitors in addition to statins, the routine attainment of extremely low LDL-C levels in the clinic has become a reality [133]. In large clinical trials [134,135], these therapies in combination with statin therapy have been shown to reduce major cardiovascular adverse events. Bile acid sequestering drugs block intestinal reabsorption of bile acids, resulting in the positive regulation of hepatic LDL receptors, in order to find circulating cholesterol for the synthesis of bile. They are promising to reducing cardiovascular mortality. Bile acid sequestrants are usually used in conjunction with statins or nicotinic acid to enhance the LDL cholesterol-lowering effect and are the drugs of choice for pregnant or maternity-planning women. Bile acid sequestrants are safe, but their use is limited by the adverse effects of bloating, nausea, cramping pain and constipation. They can also increase triglyceride levels and therefore their use is contraindicated in patients with hypertriglyceridemia. Cholestyramine, colestipol and colesevelam (but to a lesser extent) interfere with the absorption of other drugs, especially thiazides, beta-blockers, warfarin, digoxin, and thyroxine, an effect that can be minimized by administration at least 4 h before or 1 h after other drugs. Drugs that sequester bile acids must be given with meals to increase their effectiveness. Cholesterol absorption inhibitors, such as ezetimibe, inhibit the intestinal absorption of cholesterol and phytosterols. Usually ezetimibe lowers LDL cholesterol by 15-20% and causes a small increase in HDL and a slight decrease in triglycerides. Ezetimibe can be used as monotherapy in statin intolerant patients or added to statins in patients with persistently elevated LDL cholesterol despite taking a maximal statin dosage. Adverse effects are rare. PCSK9 monoclonal antibodies are available as subcutaneous injections administered once or twice a month. These drugs prevent PCSK9 from binding to LDL receptors, resulting in improved function of these receptors. LDL cholesterol is lowered by 40 to 70%. A cardiovascular outcome test with evolocumab and alirocumab showed a reduction in cardiovascular events in patients at high risk for atherosclerotic cardiovascular disease [134]. Dietary supplements that lower LDL cholesterol levels include fiber supplements and commercially available margarines and other products that contain plant sterols (sitosterol, campesterol) or stanols. Fiber-based supplements reduce cho-lesterol levels in multiple ways, including reduced absorption and increased excretion. Oat fiber supplements can reduce total cholesterol by up to 18%. Plant sterols and stanols decrease cholesterol absorption by replacing cholesterol from intestinal micelles and can reduce LDL cholesterol by up to 10% without affecting HDL or triglycerides. Medicines for homozygous familial hypercholesterolaemia include mipomersen and lomitapide. Mipomersen is an apo B antisense oligonucleotide that decreases the synthesis of apo B in liver cells and decreases the levels of LDL, apo B, and Lp(a). It is given by subcutaneous injection and can cause injection site reactions, flu-like symptoms, increased liver fat and elevated liver enzymes. Lomitapide is a microsomal triglyceride transfer protein inhibitor that interferes with the secretion of triglyceride-rich lipoproteins in the liver and intestines. The dose was started at a low dose and gradually titrated up about every 2 weeks. Patients should be on a diet with less than 20% calories from fat. Lomitapide can cause gastrointestinal adverse effects (eg, diarrhea, increased hepatic steatosis, elevated liver enzymes). Procedural approaches are reserved for patients with severe hyperlipidemia (LDL cholesterol> 300 mg / dL [> 7.74 mmol / L]) and no vascular disease. LDL apheresis can be performed on patients with LDL cholesterol> 200 mg / dL (> 5.16 mmol / L) and vascular disease refractory to conventional therapy, as occurs in familial hyper-cholesterolemia. Options are extracorporeal LDL apheresis (in which LDL is removed by plasmapheresis [plasma exchanges]) and, rarely, ileal bypass (to block the reabsorption of bile acids) and liver transplantation (which also transplants the receptors for LDL). LDL apheresis is the procedure of choice in most cases when maximum tolerated therapy does not adequately reduce LDL. Apheresis is also the usual therapy in patients with the homozygous form of familial hypercholesterolemia, which has little or no response to drug therapy. Fibrates reduce triglycerides by about 50%. They appear to stimulate en-dothelial lipoprotein lipase, leading to increased oxidation of fatty acids in the liver and muscles and reduced hepatic synthesis of VLDL. They also increase HDL by up to 20%. Fibrates can cause adverse effects on the gastrointestinal tract, such as dyspepsia, ab-dominal pain and elevated liver enzymes. They rarely cause cholelithiasis. Fibrates can potentiate muscle toxicity when used in combination with statins and enhance the effect of warfarin. Statins can be used in patients with triglycerides <500 mg / dL (<5.65 mmol / L) if elevated LDL cholesterol levels are also present; statins can reduce both LDL cho-lesterol and triglycerides by reducing VLDL. If only triglycerides are elevated, fibrates are the drugs of choice.High-dose omega-3 fatty acids (1-6 g / day of eicosapentaenoic acid and docosahexaenoic acid) can be effective in lowering triglycerides. The omega-3 fatty acids eicosapentaenoic acid and docosahexaenoic acid are the active ingredients in fish oil or omega-3 capsules. Adverse effects include belching and diarrhea. These effects can be reduced by giving the fish oil capsules during meals in divided doses (eg, 2 or 3 times / day). Omega-3 fatty acids can be helpful in addition to other therapies. Prescription omega-3 fatty acids are indicated for triglyceride levels> 500 mg / dL (> 5.65 mmol / L). The Apo CIII inhibitor (an antisense inhibitor of apo CIII), volanesorsen, is currently available in some countries. Lowers triglyceride levels in patients with severely elevated triglyceride levels, including patients with lipoproteinlipase deficiency. It is given as a weekly injection[136]. Hypoglycemic drugs. Metformin by virtue of its insulin-sensitizing properties in the different types of tissues proved effective in improving postprandial lipemia both in patients with type 2 diabetes in poor glycemic compensation only with sulfonylureas [136], and in non-diabetic subjects but with impaired glucose tolerance [136]. In both studies, treatment with metformin, in addition to reducing the area under the postprandial glucose and insulin curve, reduced the postprandial area of retinyl-palmitate in chylomicrons (56%) and in the non-chylomicronemic fraction (32%). As for insulinotropic drugs, however, a study in type 2 diabetic patients in good glycemic control did not show any effect on postprandial lipemia of either glibenclamide or nateglinide [137-139]. Sulfonylureas are insulin secretagogues. They lower blood glucose by promoting insulin secretion by pancreatic beta cells and may secondarily improve peripheral and hepatic sensitivity to insulin, reducing glucose toxicity. First generation drugs (see table Some Combinations of Oral Hypoglycemic Drugs by Class) cause adverse effects more frequently and are rarely used. All sulfonylureas promote hyperinsulinemia and a weight gain of 2-5 kg which, over time, can enhance insulin resistance and limit their usefulness. All of them can also cause hypoglycemia. Risk factors include age> 65, use of long-acting drugs (especially chlorpropamide, glyburide, or glipizide), improperly planned diet and exercise, and kidney or liver failure. Hypoglycemia caused by long-acting drugs can last for days after stopping treatment, can sometimes cause per-manent neurological damage and can be fatal. For these reasons, some doctors prefer to hospitalize people with hypoglycemia, particularly the elderly. Chlorpropamide can also cause the syndrome of inappropriate antidiuretic hormone secretion. Ultimately, most patients on sulphonylurea alone require additional medications to maintain euglycemia, indicating that the sulfonylureas may deplete beta cell function. However, worsening of insulin secretion and insulin resistance is probably more a feature of diabetes mellitus itself than of the drugs used to treat it. Fast-acting insulin secretagogues (repaglinide, nateglinide) stimulate insulin secretion in a similar way to sulfonylureas. However, they have a faster action and can stimulate insulin secretion more during meals than at other times. They are therefore particularly effective in reducing postprandial hyperglycemia and appear to lead to a lower risk of hypoglycemia. There may be some weight gain, although evidently less than with sulfonylureas. Patients who have not responded to oral hypoglycemic agents of other classes (eg, sulphonylureas, metformin) are unlikely to respond to these drugs. Biguanides reduce blood sugar by lowering hepatic production of glucose (gluconeogenesis and glycogenolysis). They are considered peripheral insulin sensitizers, but their stimulation on peripheral glucose uptake may simply be the result of their hepatic effects. Biguanides also reduce lipid levels and can reduce the absorption of nutrients from the gastrointestinal tract, increase the sensitivity of beta cells to circulating glucose, and reduce the levels of plasminogen activator inhibitor 1, thus having an antithrombotic effect. Metformin is the only commercial biguanide in the United States. It is at least as effective as sulfonylureas in lowering blood sugar, rarely causes hypogly-cemia, and can be used safely in combination with other drugs and insulin. Additionally, metformin does not cause weight gain and may also promote weight loss by reducing appetite. However, the drug typically causes gastrointestinal adverse effects (eg, dys-pepsia, diarrhea) that, in most people, resolve over time. Less commonly, metformin causes vitamin B12 malabsorption, but clinically significant anemia is rare. The contribu-tion of metformin to the development of life-threatening lactic acidosis is very rare, but the drug is contraindicated in patients at risk of acidosis (including those with significant renal insufficiency, heart failure, hypoxia or severe respiratory disease, alcoholism, other forms of metabolic acidosis or dehydration). The drug should be discontinued during surgery, IV administration of contrast media and any serious illness. Many people treated with metformin alone eventually require the addition of an additional drug. Thiazolidinediones decrease peripheral insulin resistance (insulin sensitizers), but their specific mechanism of action is not fully understood. These drugs bind a nuclear receptor present mainly in fat cells (peroxisome-proliferator-activated receptor-γ [PPAR-gamma]), which is involved in the transcription of genes that regulate glucose and lipid metabolism. Furthermore, thiazolidinediones increase HDL levels, reduce triglycerides and can have anti-inflammatory and anti-atherosclerotic effects. Thiazolidinediones are as effective as sulfonylureas and metformin in reducing HbA1C. Thiazolidinediones may be useful in the treatment of nonalcoholic fatty liver disease (NAFLD). Although a thiazolidinedione (troglitazone) caused acute liver failure, currently marketed drugs have shown no hepatotoxicity. Nonetheless, periodic monitoring of liver function is recommended. Thiazolidinediones can cause peripheral edema, especially in patients taking insulin, and can worsen heart failure in predisposed patients. Weight gain, due to fluid retention and an increase in fat mass, is common and may be relevant (> 10 kg) in some patients. Rosiglitazone may increase the risk of heart failure, angina, myocardial infarction, stroke, and fractures. Pioglitazone may increase the risk of bladder cancer (although the data are conflicting), heart failure and fractures. Alpha-glucosidase inhibitors competitively inhibit intestinal enzymes that hydrolyze dietary carbohydrates; carbohydrates are digested and absorbed more slowly, thus reducing postprandial blood sugar. Alpha-glucosidase inhibitors are less effective than other oral drugs in lowering blood glucose and often cause dyspepsia, flatulence, and diarrhea, causing patients to discontinue therapy. Aside from that, these are otherwise safe drugs that can be used in combination with all other oral hypoglycemic agents and with insulin. Dipeptidyl peptidase-4 inhibitors (eg, alogliptin, linagliptin, saxagliptin, sitagliptin) prolong the action of endogenous glucagon-like peptide-1 (GLP-1) via inhibition of the implicated dipeptidyl peptidase-4 enzyme in the decomposition of GLP-1. There is a slightly increased risk of pancreatitis with dipeptidyl peptidase-4 inhibitors, but they are otherwise considered safe and well tolerated. The decrease in HbA1C is modest with dipeptidyl peptidase-4 inhibitors. Sodium glucose cotransporter type 2 inhibitors (canagliflozin, dapagliflozine, empagiflozine) inhibit the sodium glucose cotransporter type 2 in the proximal tubule of the kidney, which blocks glucose reabsorption causing glycosuria, and lowering plasma glucose. Sodium-glucose co-transporter type 2 inhibitors can also cause modest weight loss and lower blood pressure. Empagliflozin has been shown to reduce cardiovascular events in diabetic patients at high risk for cardiovascular disease. The most frequent side effects are genitourinary infections, in particular fungal infections. Orthostatic symptoms may also appear. There have been reports of diabetic ketoacidosis in patients with type 1 diabetes mellitus and type 2 diabetes mellitus. Bromocriptine is a dopamine agonist that lowers HbA1C by approximately 0.5% by an unknown mechanism. Although approved for type 2 diabetes mellitus, it is not commonly used due to the potential negative effects. Injectable hypoglycemic drugs other than insulin are glucagon-like peptide 1 receptor agonists [GLP-1] and the amylin analogue, pramlintide (see table Characteristics of non-insulin injectable hypoglycemic drugs). These drugs are used in combination with other hypoglycemic agents. GLP-1 agonists (exenatide [a hormone that stimulates secretion], liraglutide, dulaglutide, albiglutide) increase glucose-dependent insulin secretion and slow gastric emptying. GLP-1 agonists can also reduce appetite, promote weight loss, and stimulate beta cell proliferation. Formulations are available for dosing twice / day, once / day and weekly. The most common adverse effects of GLP-1 agonists are gastrointestinal, in particular nausea and vomiting. GLP-1 agonists also cause a slightly increased risk of pancreatitis. They are contraindicated in patients with a personal or family history of medullary thyroid carcinoma because an increased risk of this tumor has occurred in experiments with animal models. The amylin analog pramlintide mimics amylin, a pancreatic hormone produced by beta cells that helps regulate postprandial blood sugar. Pramlintide suppresses postprandial glucagon secretion, slows gastric emptying, and promotes satiety. It is given by injection and is used in combination with insulin given during meals. Patients with type 1 diabetes mellitus are given 30-60 mcg subcutaneously before meals, those with type 2 diabetes mellitus are given 120 mcg [137-139].
2.13 Relationship between intestinal microbiota and metabolic diseases
In recent years there has been a dramatic increase in diseases related to the alteration of metabolism, causing conditions such as obesity, type 2 diabetes (TD2), cardiovascular disease and metabolic syndrome (MetS). In addition to the numerous factors to which this increase has been attributed, such as diet, physical activity and inflammation, numerous studies provide evidence of a correlation between intestinal microbiota and metabolic diseases. Over the past decade, the body of published studies providing evidence on the relationship between gut microbiota and metabolic diseases has soared compared to twenty years ago. The ability to understand as microbiota be able to modulate it could represent the new frontier for the prevention and treatment of major metabolic diseases such as type 1 diabetes, type 2 diabetes, metabolic syndrome and obesity.
2.14 Microbiota and metabolism
The microbiota plays an important role in many metabolic functions, including the modulation of blood sugar and lipid homeostasis, the regulation of satiety, the production of energy and vitamins. It plays a role in the regulation of numerous biochemical and physiological mechanisms through the production of metabolites and substances. In ad-dition, the microbiota also carries out important anti-inflammatory actions. There is growing evidence that changes in the composition of the microbiota can lead to various diseases, including metabolic diseases, such as obesity and diabetes, and cardiovascular disease. It is interesting to underline how the current and future preventive and thera-peutic strategies to prevent these diseases definitely focus on adequate modulation of the microbiota [1].
2.15 The human microbiota
The microbiota is a complex ecosystem of microorganisms made up of bacteria, vi-ruses, protozoa, fungi and archea, which live in different areas of the human body, such as the gastrointestinal tract, skin, mouth, respiratory system and vagina. Over 70% of microbiota lives in the gastrointestinal tract in a mutually beneficial relationship with its host. The qualitative and quantitative composition of microorganisms varies in the dif-ferent sections of the digestive system. In particular at the gastric level it is possible to isolate a concentration equal to about 103 CFU/ml. This concentration progressively in-creases in the small intestine up to about 102-109 CFU/ml and in the large intestine up to about 104-1012 CFU/ml. These bacterial species mainly belong to the Firmicutes and Bac-teroidetes phyla, both deriving from a common precursor: Methanobrevibacter smithii. In an individual weighing 70kg, the human gut microbiota gathers more than 100 trillion microorganisms and weighs about 200g (equivalent to a medium-sized mango) [140-142]. It is possible to say that "the human intestinal microbiota has a metabolic activity equal to an organ in the organ" and through this activity it performs certain beneficial functions for the host, such as production of essential metabolites, formation of fecal mass and intestinal gas, immune modulation also including antibiotic activity, xenobiotic degradation, and metabolic function affecting the digestion and absorption of nutrients. Backhed et al. [143], in 2004, stated that the microbiota can be considered as a metabolic organ exquisitely converted to our physiology that performs functions that we are unable to perform otherwise. These functions include the ability to assimilate otherwise indi-gestible components of our diet, such as plant polysaccharides. The microbiota is influ-enced by nutrition, lifestyle, exercise, use of antibiotics and genetics. It is involved in the regulation of the production of short-chain fatty acids (SCFA), basic in the regulation of food absorption and insulin modulation in the generation of a low degree of inflamma-tion. Colonization of the intestine begins even before childbirth and the first three years of life seem essential for the development of the immune system. The imbalance of the intestinal microbiota leads to the so-called "intestinal dysbiosis" which seems to be in-volved in the development of the major immune-mediated diseases. The mode of delivery can also determine the microbiota of the unborn child. Babies born with spontaneous delivery take up part of the vaginal microbiota while babies born via caesarean section have a different imprinting with less microbiota diversification. Furthermore, premature babies have a microbiota characterized by poor diversity, instability and delayed matu-ration. Finally, breastfeeding also affects the development of the microbiota in a different way than breastfeeding with humanized milks. Human milk oligosaccharides (HMOs) are considered to be able to act as growth factors for beneficial intestinal bacteria in the infant. The intestinal microbiota has a great resilience and therefore tends to return to the starting conditions after the end of the disturbing event. Therefore, to modulate our mi-crobiota we have to think about very long interventions to avoid a rapid return to the in-itial dysbiosis. In other words, antibiotics can induce changes in the microbiota even in the long term. The main environmental factor for the modulation of the microbiota is perhaps nutrition [144]. Undigested or incompletely digested carbohydrates are important food substrates for the microbiota. For example, we find among these, complex starches and dietary fibers, such as cellulose and hemicellulose, which includes xylans, such as bran, glucomannans, and pectins. Lipids, on the other hand, cannot be used by intestinal anerobic bacteria, as the oxidation of long-chain fatty acids requires the presence of oxygen, which is poor in intestine.
Figure 2: The intestinal microbiota (by https://www.thesautonapproach.it/microbiota-intestinale-come-prendersene-cura/).
2.16 Microbiota and type 2 diabetes mellitus
Some studies have examined whether an altered intestinal microbiota can directly influence this disease and have shown that the intestinal microbiota of people with type 2 diabetes can be linked to a different metabolism of the aminoacid histidine, which derives mainly from the diet. This aminoacid, in turn, leads to the formation of imidaz-ol-propionic acid, a substance that alters the ability of cells to respond to insulin. Reduc-ing the amount of imidazole propionate produced by bacteria could therefore be a new way of treating patients with type 2 diabetes. This substance does not cause all types of type 2 diabetes, but a hypothesis is that there are subpopulations of type 2 diabetes pa-tients who may benefit from changing their diet or altering their gut microbiota to reduce imidazole propionate levels [143]. The researchers identified a high concentration of the substance imidazole propionate in patients with type 2 diabetes. Using fecal samples, it was also possible to show that the microbiota of people with type 2 diabetes produced imidazole propionate when histidine was added. This mechanism was not found in control subjects without diabetes. Gram negative bacteria, such as Bacteriodes, contain large amounts of lipopolysaccharide (LPS), a strong activator of TLR4 (Toll like receptor 4) receptors, expressed in many somatic and CNS cells as well as on macrophages. The link between LPS and TLR4 triggers an inflammatory response and the expression and secretion of cytokines. In obese subjects, the circulating levels of LPS are very high and this increase is directly related to the increase in intestinal permeability due to a decrease in intestinal "tight junctions". LPS are carried by chylomicrons into the circulation. The increase in LPS is one of the factors that induces inflammation, as already mentioned, as well as insulin resistance, glucose intolerance and obesity. The microbiota can also affect the absorption of monosaccharides. Some bacterial strains can induce a greater expression of SGLT-1 at the ileal level, while other strains can induce an increase in the expression of GLUT-2 at the level of the jejunum and ileum.
2.17 Microbiota and type 1 diabetes mellitus
The incidence of type 1 diabetes mellitus (DMT1) has been growing in all industri-alized countries since the end of the second world war. This increase cannot be explained by the genetic aspect alone. In fact, migrant children from a population with a low risk of developing DMT1 assume the same risk as high-risk populations. Viruses certainly ap-pear to be involved in the development of DMT1, but the cause and effect relationship between infection and disease has not yet been established. As already mentioned, the use of antibiotics also causes long-term changes and the western diet, high in fat, high in simple carbohydrates and low in fiber, can induce substantial functional changes in the intestinal microbiota. These data suggest that children without DMT1 may have had a more balanced and stable microbiota than children who developed DMT1[145].
2.18 Microbiota and obesity
A clear example of the relationship between intestinal microbiota and metabolic diseases is offered to us by obesity which is a multifactorial pathology deriving from the interaction of individual genetics with the environment with a prevalence in the world that is increasing more and more markedly starting from the 70s-80s. Especially the in-creasingly affordable food, by facile availability, high energy density and poor quality and excess nutrition are among the most relevant causes in determining a significant increase in obesity. The same factors also have a considerable importance in alteration of the microbiota. Although obesity and metabolic diseases have always been considered nutrition-related disorders, recent evidence shows that the intestinal microbiota plays an important role in the development of this metabolic disease. Recent studies have sug-gested that the microbiota of obese people may help the body increasing the energy ex-tracted from food, suggesting that certain components of the gut microbiota may increase the likelihood of becoming obese. Although much of the research is still at the beginning, however, studies have revealed that obese people have a different composition of gut bacteria compared to lean individuals. In 2006, one of the first studies confirmed the re-lationship between microbiota, weight gain and obesity, a relationship determined by the ability of the microbiota to make a greater amount of assimilable energy available to the host. The study showed that microbiota transplantation from genetically obese mice with the ob/ob genotype into germ-free mice caused weight gain; this increase was not evident when the microbiota of non-obese mice was transplanted, with the same food intake. Studies on homozygous twins discordant for weight have helped to determine the cor-relation between microbiota, weight and nutrition. Subjects with obese phenotype and with lower bacterial richness have a reduction in the bacteria that produce butyrate, a lower production of methane and hydrogen, an increase in oxidative stress, which re-sulted in a greater tendency to develop inflammatory pictures. The intestinal microbiota influences the host's metabolism through various mechanisms including: the extraction of energy from some nutrients, the modulation of lipid metabolism, the alteration of endocrine functions and the increase in the inflammatory level. Obesity has been associ-ated with an altered intestinal microbiota characterized by high levels of Firmicutes and low levels of Bacteroides. The Western diet (high content of saturated fats, low presence of fibers, etc.) induces a reduction of Bacteroidetes strains and favors the development of Firmicutes strains, involved in the metabolism of carbohydrates. The microbiota of obese subjects has a prevalence of Firmicutes, while the microbiota of lean subjects has a prev-alence of Bacteroidetes with a high Bacteroidetes/Firmicutes ratio. One of the most im-portant bacterial strains in the prevention of obesity is Akkermansia muciniphila[145], which belongs to the Verrucomicrobia group. Many studies describe an inverse correlation between Akkermansia muciniphila concentration and genetic and dietary obesity patterns. The administration of prebiotics, dietary fibers such as inulin and its derivatives, induces a marked increase in Akkermansia in genetically obese mice, with an im-provement in metabolic functions and glucose metabolism [146]. The microbiota influ-ences the fermentation of indigestible carbohydrates into short-chain fatty acids (SCFA: butyric acid, acetic acid, and propionic acid). Butyric and propionic acid have an anti-obesogenic effect while acetic acid has the opposite effect. In fact, the first two are mainly produced by Bacteroidetes while the third by Firmicutes. Butyric acid stimulates the re-lease of GLP-1 and GIP by L cells, improving insulin sensitivity. Propionic acid has posi-tive effects on the inhibition of the functioning of Acetyl-CoA synthetase and therefore an effect on the synthesis of cholesterol. Acetic acid, on the other hand, is a substrate for li-pogenesis and for the synthesis of cholesterol.
2.19 Microbiota and modulation strategies
Prebiotics are defined as fermentable carbohydrates but not digestible by humans capable of influencing the intestinal microbiota in order to induce a health benefit to the host. The demonstrated benefits include an improvement in the insulin signal and its production. Among the prebiotics we find inulin, resistant starches and oligosaccharides. The use of prebiotics is able to modulate the development of many bacterial strains and, in particular, of Bacteroidetes to the detriment of Firmicutes, thus favoring a relationship between bacterial strains such as that found in the microbiota of lean subjects. Further-more, prebiotics have been shown to improve many metabolic parameters such as fasting blood glucose levels, glucose tolerance, reduction of plasma triglyceride levels, lipid in-filtration of muscles, fat tissue mass, oxidative stress and to improve senleptin sensitivity. Prebiotics also modulate the secretion of GLP-1 and GLP-2, and increase the feeling of satiety, with effects on appetite and the glucose response after a meal. Probiotics are living microorganisms that can improve the health of the host by modifying the microbiota. Lactobacillus casei induces a decrease in glycemic levels in mice with type 1 diabetes and prevents the onset of type 2 diabetes in mouse models. Administration of probiotics has been shown to reduce β-cell destruction, reduce oxidative damage to the pancreas, exhibit anti-inflammatory properties, reduce bacterial translocation between the gut and host, and reduce the expression of pro-inflammatory cytokines [148-150].
3. Conclusions
Cross-sectional and prospective studies indicate that postprandial hyperlipidemia, even in the presence of normal levels fasting lipid, is associated with an increased car-diovascular risk. It is observed more frequently in the conditions characterizing the mul-timetabolic syndrome, that is, in the presence of fasting hypertriglyceridemia, type 2 di-abetes mellitus, obesity and arterial hypertension. Insulin resistance in the liver and ad-ipose tissue appears to be the common mechanism responsible for postprandial hyper-lipidemia in these conditions, all characterized by a high cardiovascular risk. Postpran-dial physical exercise is able to reduce the extent of postprandial lipemia. Lipid-lowering drugs, statins and in particular fibrates, are able to reduce postprandial lipemia. There is a lack of prospective studies on the relationship between postprandial lipemia and the incidence of cardiovascular events, as well as studies on the effect of correcting post-prandial hyperlipidemia on cardiovascular risk. Numerous researches on the human intestinal microbiota show that the diet can act as a modulator of the composition and function of this microbial community in humans and animals. In conclusion, the changes in the microbiota that we find in obesity, meta-bolic syndrome, type 1 and type 2 diabetes could represent a potential therapeutic target: the use of predominantly local-acting therapies, with systemic effects, could become a new perspective of intervention. positive on the correlation of intestinal microbiota and metabolic diseases. It is evident that the mechanisms through which the microbiota reg-ulates the host's metabolism are very complex and in the future, probably, new metabolic pathways will be identified. It is clear that if we feed our microbiota by optimizing the diet, reducing the use of antibiotics and recovering contact with the products that nature offers us, we will improve the well-being of this microbial community and therefore our health state. On this basis, the modification of the intestinal microecosystem can be taken as a key goal to achieve prophylactic or therapeutic effects in altered metabolic conditions and in inflammatory states. Using this knowledge to our advantage could help us stay healthy longer. In this way, fundamental ideas are also provided for the development of new foods and food models that aim at optimizing the microbial activities in our intestine for an adequate exploitation of this enormous resource that every human being possesses. Our nutrition message must therefore continue to be to increase the consumption of complex carbohydrates that have a beneficial effect on our microbiota. Considering that the diet of the modern world has negatively changed our microbiota, we should therefore really rethink our way of eating and living, putting the well-being of the intestine among our priorities. Certainly there are still many gaps in our understanding of the interactions between diet and lifestyle, microbes and health and therefore the relationship between gut microbiota and metabolic diseases so that one day we may perhaps know exactly how to direct our diet in order to be able to "shape" our microbiota to improve our health. Therefore, we can revise the old aphorism of the philosopher Fuerbach, arguing that we are not directly what we eat or what our mother eats, but we are what our microbiota eats and how the collective composition of our microbiota is modified by the diet. At present we haven’t understood everything about the relationship between intes-tinal microbiota and metabolic diseases but we are certainly aware that many human diseases can be treated through the microbiota. Therefore, the fascination of this great scientific challenge remains, which will certainly open up new scenarios in the near future and perhaps will be able to give great answers that will allow us, in all probability, to write important pages in the history of medicine in the coming years [15].
Funding
This research received no external funding
Data availability statement
Not applicable
Conflicts of interest
The authors declare no conflict of interest.
References
- Ginsberg HN, Packard CJ, Chapman MJ, et al. Triglyceride-rich lipoproteins and their remnants: metabolic insights, role in atherosclerotic cardiovascular disease, and emerging therapeutic strategies- a consensus statement from the European Atherosclerosis Society. Eur Heart J (2021).
- Annuzzi G, Bozzetto L. Iperlipemia postprandiale. GIDM Rassegna 25 (2005): 125-139.
- Karpe F, Steiner G, Olivecrona T, et al. Metabolism of triglyceride rich lipoproteins during alimentary lipemia. J Clin Invest 91 (1993): 748-758.
- Brenninkmeijer BJ, Stuyt PM, Demacher PN, et al. Catabolism of chylomicron remnants in normolipidemic subjects in relation to the apoprotein E phenotype. J Lipid Res 28 (1987): 361-370.
- Weintraub MS, Eisenberg S, Breslow JL. Dietary fat clearance in normal subjects is regulated by genetic variation in apolipoprotein E. J Clin Invest 80 (1987): 1571-1577.
- Boerwinkle E, Brown S, Sharrett AR, et al. Apolipoprotein E polymorphism influences post-prandial retinyl palmitate but not triglyceride concentrations. Am J Hum Genet 54 (1994): 341-360.
- Dallongeville J, Tiret L, Visvikis S, et al: Effect of apo E phenotype on plasma postprandial triglyceride levels in young male adults with and without a familial history of myocardial infarction. The EARS II study. European Atherosclerosis Research Study. Atherosclerosis 145 (1999): 381-388.
- Reznik Y, Morello R, Pousse P, et al. The effect of age, body mass index, and fasting tri-glyceride level on postprandial lipemia is dependent on apolipoprotein E polymorphism in subjects with noninsulin dependent diabetes mellitus. Metabolism 51 (2002): 1088-1092.
- Orth M, Wahl S, Hanisch M, et al. Clearance of postprandial lipoproteins in nor-molipemics: role of the apolipoprotein E phenotype. Biochim Biophys Acta 1303 (1996): 22-30.
- Brown AJ, Roberts DC. The effect of fasting triacylglyceride concentration and apolipoprotein E polymorphism on postprandial lipemia. Arterioscler Thromb 11 (1991): 1737-1744.
- Nikkila M, Solakivi T, Lehtimaki T, et al. Postprandial plasma lipoprotein changes in relation to apolipoprotein E phenotypes and low density lipoprotein size in men with and without coronary artery disease. Atherosclerosis 106 (1994): 149-157.
- Vansant G, Mertens A, Muls E. Determinants of postprandial lipemia in obese women. Int J Obes Relat Metab Disord 23 (1999): 14-21.
- Kumar P, Luthra K, Dwivedi M, et al. Apolipoprotein E gene polymorphisms in pa-tients with premature myocardial infarction: a casecontrolled study in Asian Indians in North India. Ann Clin Biochem 40 (2003): 382-387.
- Chen Q, Reis SE, Kammerer CM, et al. Women’s Ischemia Syndrome Evaluation (WISE) Study: APOE polymorphism and angiographic coronary artery disease severity in the Women’s Ischemia Syndrome Evaluation (WISE) study. Atherosclerosis 169 (2003): 159-167.
- Dart A, Sherrad B, Simpson H. Influence of apo E phenotype on postprandial triglyceride and glucose response in subjects with and without coronary heart disease. Atherosclerosis 130 (1997): 161-170.
- Bergeron N, Havel RJ. Prolonged postprandial responses of lipids and apolipoproteins in triglyceride-rich lipoproteins in individuals expressing an apolipoprotein epsilon 4 allele. J Clin Invest 97 (1996): 65-72.
- Gregg RE, Zech LA, Schaefer EJ, et al. Abnormal in vivo metabolism of apolipo-protein E4 in humans. J Clin Invest 78 (1986): 815-821.
- Saito H, Dhanasekaran P, Baldwin F, et al. Effects of polymorphism on the lipid interaction of human apolipoprotein E. J Biol Chem 278 (2003): 40723-40729.
- Bergeron N, Havel RJ. Assessment of postprandial lipemia: nutritional influences. Curr Opin Lipidol 8 (1997): 43-52.
- Patti L, Rubba F, Coppola S, et al. Lipemia postprandiale: implicazioni cliniche e problemi metodologici. Giorn Ital Diabetol 19 (1999): 169-176.
- Karpe F, Bell M, Bjorkegren J, et al. Quantification of postprandial triglyceride-rich lipoproteins in healthy men by retinyl ester labeling and simultaneous measurement of apolipoproteins B-48 and B-100. Arterioscler Thromb Vasc Biol 15 (1995): 199-207.
- Karpe F, Hamsten A: Determination of apolipoproteins B-48 and B-100 in triglyceride-rich lipoproteins by an-alytical SDS-PAGE. J Lipid Res 35 (1994): 1311-1317.
- Kinoshita M, Kojima M, Matsushima T, et al. Determination of apolipoprotein B-48 in serum by a sandwich ELISA. Clin Chim Acta 351 (2005): 115-120.
- Smith D, Proctor SD, Mamo JC. A highly sensitive assay for quantitation of apolipoprotein B48 using an anti-body to human apolipoprotein B and enhanced chemiluminescence. Ann Clin Biochem 34 (1997): 185-189.
- Iovine C, Vaccaro O, Gentile A, et al. Post-prandial triglyceride profile in a population-based sample of type 2 diabetic patients. Diabetologia 47 (2004): 19-22.
- Iovine C, Gentile A, Hattemer A, et al. Self-monitoring of plasma triglyceride levels to evaluate postprandial response to different nutrients. Metabolism 53(2004): 620-623.
- Heine RJ, Dekker JM. Beyond postprandial hyperglycaemia: metabolic factors associated with cardiovascular disease. Diabetologia 45 (2002): 461-475.
- Demacker PNM. Diets and postprandial lipoproteins. Curr Opin Lipidol 6 (1995): 43-47.
- Karpe F. Effects of diet on postprandial lipaemia: a suggestion for methodological standardization. Nutr Metab Cardiovasc Dis 7 (1997): 44-55.
- Mann JI, Truswell AS, Pimstone BL. The different effects of oral sucrose and glucose on alimentary lipaemia. Clin Sci 41 (1971): 123-129.
- Cohen JC, Schall R. Reassessing the effects of simple carbohydrates on the serum triglyceride responses to fat meals. Am J Clin Nutr 48 (1988): 1031-1034.
- Grant KI, Marais MP, Dhansay MA. Sucrose in a lipidrich meal amplifies the postprandial excursion of serum and lipoprotein triglyceride and cholesterol concentrations by decreasing triglyceride clearance. Am J Clin Nutr 59 (1994): 853-860.
- Pelikanova T, Krausova Z, Kohout M, et al. Glucose and fat utilization during intravenous admin-istration of glucose and lipid emulsion in non-insulindependent diabetic patient. Nutrition 9 (1993): 18-22.
- Cohen JC. Protein ingestion does not affect postprandial lipaemia or chylomicron-triglyceride clearance. Eur J Clin Nutr 43 (1989): 497-499.
- Barr SI, Kottke BA, Mao SJ. Postprandial distribution of apolipoproteins C-II and C-III in normal subjects and patients with mild hypertriglyceridemia: comparison of meals containing corn oil and medium-chain triglyc-eride oil. Metabolism 34 (1985): 983-992.
- Hultin M, Mullertz A, Zundel MA, et al. Metabolism of emulsions containing medium- and long-chain triglycerides or interesterified triglycerides. J Lipid Res 35 (1994): 1850-1860.
- Ockner RK, Jones AL. An electron microscopic and functional study of very low density lipoproteins in intes-tinal lymph. J Lipid Res 11 (1970): 284-292.
- Feldman EB, Russell BS, Chen R, et al. Dietary saturated fatty acid content affects lymph lipoproteins: studies in the rat. J Lipid Res 24 (1983): 967-976.
- Fraser R, Cliff WJ, Courtice FC. The effect of dietary fat load on the size and composition of chylomicrons in thoracic duct lymph. Q J Exp Physiol Cogn Med Sci 53 (1968): 390-398.
- Zilversmit DB, Sisco PH Jr, Yokoyama A. Size distribution of thoracic duct lymph chylomicrons from rats fed cream and corn oil. Biochim Biophys Acta 125 (1966): 129-135.
- Renner F, Samuelson A, Rogers M, et al. Effect of saturated and unsaturated lipid on the composition of mesenteric triglyceride-rich lipoproteins in the rat. J Lipid Res 27 (1966): 72-81.
- Zampelas A, Murphy M, Morgan LM, et al. Postprandial lipoprotein lipase, insulin and gastric in-hibitory polypeptide responses to test meals of different fatty acid composition: comparison of saturated, n-6 and n-3 polyunsaturated fatty acids. Eur J Clin Nutr 48 (1994): 849-858.
- Weintraub MS, Zechner R, Brown A, et al. Dietary polyunsaturated fats of the W-6 and W-3 series reduce postprandial lipoprotein levels, chronic and acute effects of fat saturation on postprandial lipo-protein metabolism. J Clin Invest 82 (1988): 1884-1893.
- Zampelas A, Peel AS, Gould BJ, et al. Polyunsaturated fatty acids of the n-6 and n-3 series: effects on postprandial lipid and apolipoprotein levels in healthy men. Eur J Clin Nutr 48 (1994): 842-848.
- Thomsen C, Rasmussen O, Lousen T, et al. Differential effects of saturated and monounsaturated fatty acids on postprandial lipemia and incretin responses in healthy subjects. Am J Clin Nutr 69 (1999): 1135-1143.
- Murphy MC, Isherwood SG, Sethi S, et al. Postprandial lipid and hormone responses to meals of varying fat contents: modulatory role of lipoprotein lipase? Eur J Clin Nutr 49 (1995): 578-588.
- Cohen JC. Chylomicron triglyceride clearance: comparison of three assessment methods. Am J Clin Nutr 49 (1989): 306-313.
- Annuzzi G, Holmquist L, Carlson LA. Concentrations of apolipoproteins B, C-I, C-II, C-III, E and lipids in serum and serum lipoproteins of normal subjects during alimentary lipemia. Scandinavian Journal of Clinical and Laboratory Investigation 49 (1989): 73-81.
- Annuzzi G, Jansson E, Kaijser L, et al. Increased removal rate of exogenous triglycerides after prolonged exercise: time course and effect of exercise duration. Metabolism 36 (1987): 438-443.
- Berr F, Eckel RH, Kern F. Contraceptive steroids increase hepatic uptake of chylomicron remnants in healthy young women. J Lipid Res 27 (1986): 645-651.
- Zilversmit DB. Atherogenesis: A postprandial phenomenon. Circulation 60 (1979): 473-485.
- Groot PHE, Van Stiphout WAHJ, Krauss XH, et al. Postprandial lipoprotein metabolism in normolipidemic men with and without coronary artery disease. Arterioscl Thromb 11 (1991): 653-662.
- Weintraub MS, Grosskopf I, Rassin T, et al. Clearance of chylomicron remnants in normolipidaemic patients with coronary heart disease: case control study over three years. BMJ 312 (1996): 936-939.
- Mero N, Malmström R, Steiner G, et al. Postprandial metabolism of apolipoprotein B 48- and B 100-containing particles in type 2 diabetes mellitus: relations to angiographically verified severity of coronary artery disease. Atherosclerosis 150 (2000): 167-177.
- Lupattelli G, Pasqualini L, Siepi D, et al. Increased postprandial lipemia in patients with normolipidemic peripheral arterial disease. Am Heart J 143 (2002): 733-738.
- Ryu JE, Howard G, Craven TE, et al. Postprandial triglyceridemia and carotid atherosclerosis in middle-aged subjects. Stroke 23 (1992): 823-828.
- Sharrett AR, Chambless LE, Heiss G, et al. Association of postprandial triglyceride and retinyl palmitate responses with asymptomatic carotid artery atherosclerosis in middle-aged men and women. Arterioscler Thromb Vasc Biol 15(1995): 2122-2129.
- Teno S, Uto Y, Nagashima H, et al. Association of postprandial hyper-triglyceridemia and carotid intima-media thickness in patients with type 2 diabetes. Diabetes Care 23 (2000): 1401-1406.
- Karpe F, De Faire U, Mercuri M, et al. Magnitude of alimentary lipemia is re-lated to intima-media thickness of the common carotid artery in middle-aged men. Atherosclerosis 141 (1998): 307-314.
- Boquist S, Ruotolo G, Tang R, et al. Alimentary lipemia, postprandial triglyceride-rich lipoproteins, and common carotid intima-media thickness in healthy, mid-dleaged men. Circulation 100 (1999): 723-728.
- Albrink MJ, Man EB. Serum triglycerides in coronary artery disease. Arch Int Med 103 (1959): 4-8.
- Simons LA, Dwyer T, Simons J, et al. Chylomicrons and chylomicron remnants in coronary artery disease: a case-control study. Atherosclerosis 65 (1987): 181-189.
- Simpson HS, Williamson CM, Olivecrona T, et al. Postprandial lipemia, fenofibrate and coronary artery disease. Atherosclerosis 85 (1990): 193-202.
- Karpe F, Steiner G, Uffelman K, et al. Postprandial lipoproteins and progression of cor-onary atherosclerosis. Atherosclerosis 106 (1994): 83-97.
- Patsch JR, Miesenböck G, Hopferwieser T, et al. Relation of triglyceride metabolism and coronary artery disease: studies in the postprandial state. Arterioscler Thromb 12 (1992): 1336-1345.
- Ginsberg HN, Jones J, Blaner WS, et al. Association of post-prandial triglyceride and retinyl palmitate responses with newly diagnosed exercise-induced myocardial is-chemia in middle-aged men and women. Arterioscler Thromb Vasc Biol 15(1995): 1829-1838.
- Meyer E, Westerveld HT, De Ruyter-Meijstek FC, et al. Abnormal postprandial apolipoprotein B48 and triglyceride responses in normolipidemic women with greater than 70% stenotic coronary artery disease: a case-control study. Atherosclerosis 124 (1996): 221-235.
- Weintraub MS, Grosskopf I, Charach G, et al. Hormone replacement therapy enhances postprandial lipid metabolism in postmenopausal women. Metabolism 48 (1999): 1193-1196.
- Van Beek AP, De Tuijter-Heijstek FC, Erkelens DW, et al. Menopause is associated with reduced pro-tection from postprandial lipemia. Arter Thromb Vasc Biol 19 (1999): 2737-2741.
- Rapp JH, Lespine A, Hamilton RL, et al. Triglyceride-rich lipoproteins isolated by selective-affinity anti-apolipoprotein B immunosorption from human atherosclerotic plaque. Arter Thromb 14 (1994): 1767-1774.
- Ross R. Atherosclerosis is an inflammatory disease. Am Heart J 138 (1999): S419-S420.
- Silveira A, Karpe F, Johnsson H, et al. In vivo demonstration in humans that large triglyc-eriderich lipoproteins activate coagulation factor VII through the intrinsic coagulation pathway. Arterioscler Thromb Vasc Biol 16 (1996): 1333-1339.
- Miller GJ, Martin JC, Mitropoulos KA, et al. Plasma factor VII is activated by postprandial triglycer-idemia, irrespective of dietary fat consumption. Atherosclerosis 86 (1991): 163-171.
- Hyson DA, Paglieroni TG, Wun T, et al. Postprandial lipemia is associated with platelet and monocyte activation and increased monocyte cytokine expression in normolipemic men. Clin Appl Thromb Hemost 8 (2002): 147-155.
- Van Oostrom AJ, Sijmonsma TP, Verseyden C, et al. Postprandial recruitment of neutrophils may contribute to endothelial dysfunction. J Lipid Res 44 (2003): 576-583.
- Vogel RA. Measurement of endothelial function by brachial artery flow-mediated vasodilation. Am J Cardiol 88 (2001): 31E-34E.
- Ong PJ, Dean TS, Hayward CS, et al. Effect of fat and carbohydrate con-sumption on endothelial function. Lancet 354 (1999): 21-34.
- Gaenzer H, Sturm W, Neumayr G, et al. Pro-nounced postprandial lipemia impairs endothelium-dependent dilation of the brachial artery in men. Cardiovasc Res 52 (2001): 509-516.
- Di Renzo L, Merra G, Botta R, et al. Post-prandial effects of hazelnut-enriched high fat meal on LDL oxidative status, oxidative and inflammatory gene expression of healthy subjects: a randomized trial. Eur Rev Med Pharmacol Sci 21 (2017): 1610-1626.
- De Lorenzo A, Bernardini S, Gualtieri P, et al. Mediterranean meal versus Western meal effects on postprandial ox-LDL, oxidative and inflammatory gene expression in healthy subjects: a randomized controlled trial for nutrigenomic approach in cardiometabolic risk. Acta Diabetol 54 (2017):141-149.
- Di Renzo L, Marsella LT, Carraro A, et al. Changes in LDL oxidative status and oxidative and inflammatory gene expression after red wine intake in healthy people: A randomized trial. Mediators Inflamm 11 (2015): 317-348.
- Perrone MA, Gualtieri P, Gratteri S, et al. Effects of postprandial hydroxytyrosol and derivates on oxidation of LDL, cardiometabolic state and gene expression: a nutrigenomic approach for cardiovascular prevention. J Cardiovasc Med (Hagerstown) 20 (2019):419-426.
- Evans M, Anderson RA, Graham J, et al. Ciprofibrate therapy improves endothelial function and reduces postprandial. Circulation 101 (2000): 1773-1779.
- Vigna GB, Delli Gatti C, Fellin R. Endothelial function and postprandial lipemia. Nutr Metab Cardiovasc Dis 14 (2004): 121-127.
- Eckel RH, Yost TJ, Jensen DR. Alterations in lipoprotein lipase in insulin resistance. Int J Obes Relat Metab Disord 19 (1995): S16-21.
- Taskinen MR. Insulin resistance and lipoprotein metabolism. Curr Opin Lipidol 6 (1995): 153-160.
- Syvänne M, Taskinen MR. Lipids and lipoproteins as coronary risk factors in non-insulin dependent diabetes mellitus. Lancet 350 (1997): S120-123.
- Taskinen MR. Pathogenesis of dyslipidemia in type 2 diabetes. Exp Clin Endocrinol Diabetes 109 (2001): S180-188.
- Ginsberg HN, Illingworth R. Postprandial dyslipidemia: an atherogenic disorder common in patients with diabetes mellitus. Am J Cardiol 88 (2001): H9-H15.
- Chen YD, Swami S, Skowronski R, et al. Differences in postprandial lipemia between pa-tients with normal glucose tolerance and noninsulindependent diabetes mellitus. J Clin Endocrinol Metab 76 (1993): 172-177.
- Reznik Y, Pousse P, Herrou M, et al. Postprandial lipoprotein metabolism in normotriglyceridemic non-insulindependent diabetic patients: influence of apolipoprotein E polymorphism. Metabolism 45 (1996): 63-71.
- Curtin A, Deegan P, Owens D, et al. Alteration in apolipoprotein B-48 in the postprandial state in NIDDM. Diabetologia 37 (1994): 1259-1264.
- Rivellese AA, De Natale C, Di Marino L, et al. Exogenous and endogenous postprandial lipid abnormalities in type 2 diabetic patients with optimal blood glucose control and optimal fasting triglyceride levels. J Clin Endocrinol Metab 89 (2004): 2153-2159.
- Jeppesen J, Hollenbeck CB, Zhou MY, et al. Relation between insulin resistance, hyperinsulinemia, postheparin plasma lipoprotein lipase activity, and postprandial lipemia. Arterioscler Thromb Vasc Biol 15 (1995): 320-324.
- Axelsen M, Smith U, Eriksson JW, et al. Postprandial hypertriglyceridemia and insulin resistance in normoglycemic first-degree relatives of patients with type 2 diabetes. Ann Intern Med 131 (1999): 27-31.
- Annuzzi G, De Natale C, Iovine C, et al. Insulin resistance is independently associated with postprandial alterations of triglyceride-rich lipoproteins in type 2 diabetes mellitus. Arterioscler Thromb Vasc Biol 24 (2004): 2397-2402.
- Roust LR, Jensen MD. Postprandial free fatty acid kinetics are abnormal in upper body obesity. Diabetes 42 (1993): 1567-1573.
- Couillard C, Bergeron N, Prud’homme D, et al. Postprandial triglyceride response in visceral obesity in men. Diabetes 47 (1998): 953-960.
- Kolovou GD, Daskalova DCh, Iraklianou SA, et al. Postprandial lipemia in hypertension. J Am Coll Nutr 22 (2003): 80-87.
- Facchini FS, Hollenbeck CB, Jeppesen J, et al. Insulin resistance and cigarette smoking. Lancet 339 (1992): 1128-1130.
- Axelsen M, Eliasson B, Joheim E, et al. Lipid intolerance in smokers. J Intern Med 237 (1995): 449-455.
- Nestel PJ, Connor WE, Reardon MF, et al. Suppression by diets rich in fish oil of very low density lipoprotein production in man. J Clin Invest 74 (1984): 82-89.
- Harris WS, Muzio F. Fish oil reduces postprandial triglyceride concentrations without accelerating lipid-emulsion removal rates. Am J Clin Nutr 58 (1993): 68-74.
- Demacker PN, Reijnen IG, Katan MB, et al. Increased removal of remnants of triglycer-ide-rich lipoproteins on a diet rich in polyunsaturated fatty acids. Eur J Clin Invest 21 (1991): 197-203.
- Bergeron N, Havel RJ. Influence of diets rich in saturated and omega-6 polyunsaturated fatty acids on the postprandial responses of apolipoproteins B-48, B-100, E, and lipids in triglyceride-rich lipoproteins. Arterioscler Thromb Vasc Biol 15 (1995): 2111-2121.
- Ginsberg HN, Karmally W, Siddiqui M, et al. A dose-response study of the effects of dietary cholesterol on fasting and postprandial lipid and lipoprotein metabolism in healthy young men. Arterioscler Thromb 14 (1994): 576-586.
- Dubois C, Armand M, Ferezou J, et al. Postprandial appearance of dietary deuterated cholesterol in the chylomicron fraction and whole plasma in healthy subjects. Am J Clin Nutr 64 (1996): 47-52.
- Beaumier-GG, Dubois C, Senft M, et al. Dietary cholesterol is secreted in intestinally derived chylomicrons during several subsequent postprandial phases in healthy hu-mans. Am J Clin Nutr 73 (2001): 870-877.
- Steiner G. Hypertriglyceridemia and carbohydrate intolerance: interrelations and therapeutic implications. Am J Cardiol 57 (1986): 27G-30G.
- Nestel PJ, Carroll KF, Havenstein N: Plasma triglyceride response to carbohydrates, fats and caloric intake. Metabolism 19 (1970): 1-18.
- Hollenbeck CB, Coulston AM, Reaven GM. Effects of sucrose on carbohydrate and lipid metabolism in NIDDM patients. Diabetes Care 12 (1989): 62-66.
- Dubois C, Armand M, Senft M, et al. Chronic oat bran intake alters postprandial lipemia and lipoproteins in healthy adults. Am J Clin Nutr 61 (1995): 325-333.
- Anderson JW, O’Neal DS, Riddell-Mason S, et al. Postprandial serum glucose, insulin, and lipoprotein responses to high- and lowfiber diets. Metabolism 44 (1995): 848-854.
- Mekki N, Dubois C, Charbonnier M, et al. Effects of lowering fat and increasing dietary fiber on fasting and postprandial plasma lipids in hypercholes-terolemic subjects consuming a mixed Mediterranean-Western diet. Am J Clin Nutr 66 (1997): 1443-1451.
- Superko HR. Effects of acute and chronic alcohol consumption on postprandial lipemia in healthy normotri-glyceridemic men. Am J Cardiol Mar 69 (1992): 701-704.
- Pownall HJ. Dietary ethanol is associated with reduced lipolysis of intestinally derived lipoproteins. J Lipid Res 35 (1994): 2105-2113.
- Hardman AE. The influence of exercise on postprandial triacylglycerol metabolism. Atherosclerosis 141 (1998): 93-100.
- Altekruse EB, Wilmore JH. Changes in blood chemistries following a controlled exercise program. J Occup Med 15 (1973): 10-13.
- Wirth A, Diehm C, Hanel W, et al. Traininginduced changes in serum lipids, fat tolerance, and adipose tissue metabolism in patients with hypertriglyceridemia. Atherosclerosis 54 (1985): 263-271.
- Weintraub MS, Rosen Y, Otto R, et al. Physical exercise conditioning in the absence of weight loss reduces fasting and postprandial triglyceride-rich lipoprotein levels. Circulation 79 (1989): 1007-1014.
- Gill JM, Mees GP, Frayn KN, et al. Moderate exercise, postprandial lipaemia and triacylglycerol clearance. Eur J Clin Invest 31 (2001): 201-207.
- Herd SL, Hardman AE, Boobis LH, et al. The effect of 13 weeks of running training followed by 9 d of detraining on postprandial lipaemia. Br J Nutr 80 (1998): 57-66.
- Syvanne M, Vuorinen-Markkola H, Hilden H, Taskinen MR: Gemfibrozil reduces postprandial lipemia in noninsulin-dependent diabetes mellitus. Arterioscler Thromb 13 (1993): 286-295.
- Weintraub MS, Charach G, Grosskopf Effects of fibric acid derivatives and metformin on postprandial li-pemia. Atherosclerosis 141 (1998): S71-75.
- Attia N, Durlach V, Cambilleau M, et al. Postprandial concentrations and distribution of apo C-III in type 2 diabetic patients. Effect of bezafibrate treatment. Atherosclerosis 149 (2000): 427-433.
- Autran D, Attia N, Dedecjus M, et al. Postprandial reverse cholesterol transport in type 2 diabetic patients: effect of a lipid lowering treatment. Atherosclerosis 153 (2000): 453-460.
- Sheu WH, Jeng CY, Lee WJ, et al. Simvastatin treatment on postprandial hypertriglycer-idemia in type 2 diabetes mellitus patients with combined hyperlipidemia. Metabolism 50 (2001): 355-359.
- Costa A, Casamitjana R, Casals E, et al. Effects of atorvastatin on glucose homeostasis, postprandial triglyceride response and C-reactive protein in subjects with impaired fasting glucose. Diabet Med 20 (2003): 743-745.
- Nordoy A, Hansen JB, Brox J, et al. Effects of atorvastatin and omega-3 fatty acids on LDL subfractions and postprandial hyperlipemia in patients with combined hyperlipemia. Nutr Metab Cardiovasc Dis 11 (2001): 7-16.
- Nordoy A, Svensson B, Hansen JB. Atorvastatin and omega-3 fatty acids protect against activation of the co-agulation system in patients with combined hyperlipemia. J Thromb Haemost 1 (2003): 690-697.
- Simo IE, Yakichuk JA, Ooi TC. Effect of gemfibrozil and lovastatin on postprandial lipoprotein clearance in the hypoalphalipoproteinemia and hypertriglyceridemia syndrome. Atherosclerosis 100 (1993): 55-64.
- Wilmink HW, Twickler MB, Banga JD, et al. Effect of statin versus fibrate on postprandial endothelial dysfunction: role of remnant-like particles. Cardiovasc Res 50 (2001): 577-582.
- Packard C, Chapman MJ, Sibartie M, et al. Intensive low-density lipoprotein cholesterol lowering in cardiovascular disease prevention: opportunities and challenges. Heart 107 (2021): 1369-1375.
- Grundy SM, Stone NJ, Bailey AL, et al. AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the management of blood cholesterol a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 139 (2018): e1082-e1143.
- Sabatine MS, Giugliano RP, Keech AC, et al: Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med 376 (2017):1713-1722.
- Schwartz GG, Steg PG, Szarek M, et al: Alirocumab and cardiovascular outcomes after acute coronary syn-drome. New Engl J Med 379 (2018): 2097-2107.
- Jeppesen J, Zhou MY, Chen YD, et al. Effect of metformin on postprandial lipemia in patients with fairly to poorly controlled NIDDM. Diabetes Care 17 (1994): 1093-1099.
- Grosskopf I, Ringel Y, Charach G, et al. Metformin enhances clearance of chylomicrons and chylomicron remnants in nondiabetic mildly overweight glucose-intolerant subjects. Diabetes Care 20 (1997): 1598-1602.
- Vakkilainen J, Mero N, Schweizer A, et al. Effects of nateglinide and glibenclamide on postprandial lipid and glucose metabolism in type 2 diabetes. Diabetes Metab Res 18 (2002): 484-490.
- Ron S, Shai F, Ron M. Revised Estimates for the Number of Human and Bacteria Cells in the Body. PLoS Biol 14 (2016): e1002533.
- Fox CS, Golden SH, Anderson C, et al. AHA/ADA Scientific statement: Update on prevention of cardiovascular disease in adults with type 2 diabetes mellitus in light of recent evidence. Circulation 132 (2015): 691-718.
- Karlsson CL, Onnerfält J, Xu J, et al. The microbiota of the gut in pre-school children with normal and excessive body weight. Obesity 20 (2012): 2257-2261.
- Bäckhed F, Ding H, Wang T, et al. The gut mi-crobiota as an environmental factor that regulates fat storage. Procedings of the National Academy of Sciences of the United States of America 101 (2004): 44.
- Eckburg PB, Bik EM, Bernstein CN, et al. Diversity of the human intestinal microbial flora. Science 308 (2005): 1635-1638.
- Everard A, Belzer C, Geurts L, et al. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet induced obesity. Proc Natl Acad Sci USA 110 (2013): 9066-9071.
- Flint HJ, Scott KP, Duncan SH, et al. Microbial degradation of complex carbohydrates in the gut. Gut Microbes 3 (2012): 289-306.
- Hara N, Alkanani AK, Ir D, et al. Prevention of virus induced type 1 diabetes with antibiotic therapy. J Immunol 189 (2012): 3805-3814.
- Tremaroli V, Bäckhed F. Functional interactions between the gut microbiota and host metabolism. Nature 489 (2012): 242-249.
- Turnbaugh PJ, Ley RE, Mahowald MA, et al. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 444 (2006): 1027-1031.
- Chindemi G. Rapporto tra microbiota intestinale e malattie metaboliche. Microbiologioa Italia 11 (2019): 10-20.
