Nutritional Assessment In Hemodialysis Patients : Influence of Method and Sex
Article Information
Niakhaleen Keita1, Maria Faye2,3, Moustapha Faye2,3, Bacary Ba2, Seynabou Diagne4, Mansour Mbengue1,3, Ahmed Tall Lemrabott2,3, Sidy Mouhamed Seck4,6, Abdou Niang1,3, El Hadji Fary Ka2,3
1Nephrology Department, Dalal Jamm National Hospital Center, Dakar, Senegal.
2Nephrology and Dialysis Department, Aristides le Dantec University Hospital Center.
3Cheikh Anta Diop University, Dakar, Senegal.
4Nephrology and Dialysis Department, Pikine National Hospital Center, Dakar, Senegal.
5 Nephrology and Dialysis Department, Ouakam Military Hospital, Dakar, Senegal.
6Gaston Berger University, Saint-Louis, Senegal.
*Corresponding Author: Niakhaleen Keita, Nephrology Department, Dalal Jamm National Hospital Center
Received: 23 March 2023; Accepted: 03 April 2023; Published: 05 April 2023
Citation: Niakhaleen Keita, Maria Faye, Moustapha Faye, Bacary Ba, Seynabou Diagne, Mansour Mbengue, Ahmed Tall Lemrabott, Sidy Mouhamed Seck, Abdou Niang, El Hadji Fary Ka. Nutritional Assessment In Hemodialysis : Influence of Method and Sex. Archives of Nephrology and Urology. 6 (2023): 31-36.
View / Download Pdf Share at FacebookAbstract
Introduction: Several nutritional assessment methods are available. The objective of this study was to assess the nutritional status of chronic hemodialysis patients based on anthropometry, bioimpedance and albuminemia.
Patients and Method: A cross-sectional study was conducted in two hemodialysis units, having included patients regularly on dialysis, with no history of hospitalization during the last month and consenting. The nutritional evaluation was based on the collection of anthropometric markers, the determination of body composition by bioelectrical impedance analysis and the determination of albuminemia.
Results: 68 hemodialysis patients (46.7 years old, 51.5% women) analyzed. The prevalence of undernutrition was 47.1% (95% CI [34.8 – 59.6]) according to BMI, 25% (95% CI [15.3 – 37.0]) according to brachial circumference, by 10.3% (95% CI [4.2 – 20.1]) according to the percentage of ideal weight, by 11.8% (95% CI [5.2 – 21.9]) according to the FM, by 55.9% (95% CI [43.3 – 67.9]) according to the LM index and by 10.3% (95% CI [4.2 – 20.1]) according to serum albumin . Similarly, the prevalence of obesity varied from 8.8% (95% CI [-1.8 – 22.4]) according to BMI and from 36.8% (95% CI [24.7 – 48 ,9]) according to the FM. According to bioimpedance, women on hemodialysis were more exposed to malnutrition than men. Impedance nutritional parameters were thus strongly correlated and agreement with anthropometric parameters in the diagnosis of undernutrition and obesity.
Conclusion: Due to its availability and simplicity, bioelectrical impedance analysis (BIA) has considerable potential as a complement to conventional anthropometric techniques for assessing the nutritional status of hemodialysis patients.
Keywords
<p>Undernutrition; Obesity; Hemodialysis patients; Nutrition; Sex</p>
Undernutrition articles; Obesity articles; Hemodialysis patients articles; Nutrition articles; Sex articles
Undernutrition articles Undernutrition Research articles Undernutrition review articles Undernutrition PubMed articles Undernutrition PubMed Central articles Undernutrition 2023 articles Undernutrition 2024 articles Undernutrition Scopus articles Undernutrition impact factor journals Undernutrition Scopus journals Undernutrition PubMed journals Undernutrition medical journals Undernutrition free journals Undernutrition best journals Undernutrition top journals Undernutrition free medical journals Undernutrition famous journals Undernutrition Google Scholar indexed journals Obesity articles Obesity Research articles Obesity review articles Obesity PubMed articles Obesity PubMed Central articles Obesity 2023 articles Obesity 2024 articles Obesity Scopus articles Obesity impact factor journals Obesity Scopus journals Obesity PubMed journals Obesity medical journals Obesity free journals Obesity best journals Obesity top journals Obesity free medical journals Obesity famous journals Obesity Google Scholar indexed journals Hemodialysis patients articles Hemodialysis patients Research articles Hemodialysis patients review articles Hemodialysis patients PubMed articles Hemodialysis patients PubMed Central articles Hemodialysis patients 2023 articles Hemodialysis patients 2024 articles Hemodialysis patients Scopus articles Hemodialysis patients impact factor journals Hemodialysis patients Scopus journals Hemodialysis patients PubMed journals Hemodialysis patients medical journals Hemodialysis patients free journals Hemodialysis patients best journals Hemodialysis patients top journals Hemodialysis patients free medical journals Hemodialysis patients famous journals Hemodialysis patients Google Scholar indexed journals Nutrition articles Nutrition Research articles Nutrition review articles Nutrition PubMed articles Nutrition PubMed Central articles Nutrition 2023 articles Nutrition 2024 articles Nutrition Scopus articles Nutrition impact factor journals Nutrition Scopus journals Nutrition PubMed journals Nutrition medical journals Nutrition free journals Nutrition best journals Nutrition top journals Nutrition free medical journals Nutrition famous journals Nutrition Google Scholar indexed journals Thermobalancing therapy articles Thermobalancing therapy Research articles Thermobalancing therapy review articles Thermobalancing therapy PubMed articles Thermobalancing therapy PubMed Central articles Thermobalancing therapy 2023 articles Thermobalancing therapy 2024 articles Thermobalancing therapy Scopus articles Thermobalancing therapy impact factor journals Thermobalancing therapy Scopus journals Thermobalancing therapy PubMed journals Thermobalancing therapy medical journals Thermobalancing therapy free journals Thermobalancing therapy best journals Thermobalancing therapy top journals Thermobalancing therapy free medical journals Thermobalancing therapy famous journals Thermobalancing therapy Google Scholar indexed journals Sex articles Sex Research articles Sex review articles Sex PubMed articles Sex PubMed Central articles Sex 2023 articles Sex 2024 articles Sex Scopus articles Sex impact factor journals Sex Scopus journals Sex PubMed journals Sex medical journals Sex free journals Sex best journals Sex top journals Sex free medical journals Sex famous journals Sex Google Scholar indexed journals dual-photon X-ray absorptiometry articles dual-photon X-ray absorptiometry Research articles dual-photon X-ray absorptiometry review articles dual-photon X-ray absorptiometry PubMed articles dual-photon X-ray absorptiometry PubMed Central articles dual-photon X-ray absorptiometry 2023 articles dual-photon X-ray absorptiometry 2024 articles dual-photon X-ray absorptiometry Scopus articles dual-photon X-ray absorptiometry impact factor journals dual-photon X-ray absorptiometry Scopus journals dual-photon X-ray absorptiometry PubMed journals dual-photon X-ray absorptiometry medical journals dual-photon X-ray absorptiometry free journals dual-photon X-ray absorptiometry best journals dual-photon X-ray absorptiometry top journals dual-photon X-ray absorptiometry free medical journals dual-photon X-ray absorptiometry famous journals dual-photon X-ray absorptiometry Google Scholar indexed journals computed tomography articles computed tomography Research articles computed tomography review articles computed tomography PubMed articles computed tomography PubMed Central articles computed tomography 2023 articles computed tomography 2024 articles computed tomography Scopus articles computed tomography impact factor journals computed tomography Scopus journals computed tomography PubMed journals computed tomography medical journals computed tomography free journals computed tomography best journals computed tomography top journals computed tomography free medical journals computed tomography famous journals computed tomography Google Scholar indexed journals Waist circumference articles Waist circumference Research articles Waist circumference review articles Waist circumference PubMed articles Waist circumference PubMed Central articles Waist circumference 2023 articles Waist circumference 2024 articles Waist circumference Scopus articles Waist circumference impact factor journals Waist circumference Scopus journals Waist circumference PubMed journals Waist circumference medical journals Waist circumference free journals Waist circumference best journals Waist circumference top journals Waist circumference free medical journals Waist circumference famous journals Waist circumference Google Scholar indexed journals
Article Details
1. Introduction
Chronic hemodialysis (CHD)patients have high risk for malnutrition due to anorexia, dietary restrictions, physical inactivity, chronic inflammation, comorbidities and metabolic disorders [1]. An assessment of undernutrition and obesity is necessary for future nutritional advice given by nephrologists and dieticians to CHD patients [2]. The prevalence of protein-energy malnutrition varies from 20 to 75%, partly depending on the characteristics of the population studied, but also on differences in methodology and diagnostic criteria. Obesity is also common [3, 4]. A few studies have simultaneously assessed the prevalence of undernutrition and obesity in CHD patients [5, 6]. They reported an overlap between malnourished (protein-energy wasting) and obese (excess energy) patients, indicating a likely problem in nutritional assessment methodologies [2]. Several of these methods have, however, been reported useful in HDC patients such as subjective global assessment, comparison of anthropometric and serum biochemical parameters, as well as changes in body composition assessed by dual-photon X-ray absorptiometry (DEXA), a computed tomography or bioimpedance [7 – 13]. In this study, we sought to assess the nutritional status of a CHD patients from anthropometry, bioimpedance and albuminemia.
2. Methods
A cross-sectional study was conducted from July 18 to 28, 2018, in two (2) hemodialysis units of the nephrology department of the University Hospital Center Aristide Le Dantec. All patients were targeted. We included, patients on hemodialysis for more than 3 months, on a regular basis (2 or 3 sessions per week), aged at least 18 years. Patients hospitalized within the last month, those with a medical implant (pacemaker, orthopedic nail, total hip replacement) and those who could not stand on the impedance scale, were not included. We excluded also, patients who had decided to withdraw from the study. Oral consent was obtained from all patients after a clear explanation of the protocol in the local language. The study received the approval of the Research Ethics Committee of the Cheikh Anta Diop University of Dakar (CER/UCAD).
The nutritional assessment was based on anthropometric markers, body composition determination by bioelectrical impedance analysis (BIA) and serum albumin. The ideal weight was calculated using the Lorentz formula [14] modified and taking age into account: ideal weight (kg) = 50 + [(height (cm) – 150) / 4] + [(age (years) ) – 20) / 4]. The ideal weight percentage was calculated from the ideal weight to weight ratio. Body mass index (BMI in kg/m2) was calculated by dividing weight (kilograms) by height squared (meters). Waist circumference (WC) and mid-upper arm circumference (MUAC) measurements were taken with a tape measure and are expressed in cm. A single-frequency foot-to-foot bioelectrical impedance analyzer (SF-BIA, TANITA® model BC-730, 50 kHz, Japan) was used. The measurement is made after the end of the mid-week session, according to the recommendations issued by the European Society of Clinical Nutrition and Metabolism (ESPEN) [15]. The fat mass index (FMI, in kg/m2) and the lean mass index (LMI, in kg/m2) were calculated by dividing the fat mass (FM) and the lean mass (LM) respectively, by height squared [16]. The serum biochemical parameters had been carried out in the month of the weighing.
Various parameters of undernutrition studied : BMI (value less than 20 kg/m2 [15]), percentage of ideal weight (value less than 80% [17]), MUAC (value less than 22 cm [15 ]), FM (value less than 10% of body weight [15]), LMI (value less than 15 kg/m2 for women and 17 kg/m2 for men [15]) and serum albumin (value less than 35 g/L [18]). Parameters of obesity studied were: BMI (value greater than 30 kg/m2 [19]) and FM (value greater than 30%).
Statistical analysis: data were entered using EXCEL® software version 2013 (Microsoft®, Albuquerque, USA) and analyzed using SPSS® software version 21.0 (IBM®, Endicott, USA). The anthropometric and impedance measurements were compared to the reference standards of the impedance data and to the reference standards established for the general population. Mann-Whitney, Kruskal Wallis, chi-square, Fisher and Pearson tests were used. The results were considered significant for an alpha risk threshold of 5% (p < 0.05). In order to determine the agreement between the methods for estimating the prevalence of obesity and undernutrition, we divided the patients into two groups (obese/non-obese, undernourished/normal). Cohen's kappa (κ) measures the level of agreement between two raters or judges who each classify items into mutually exclusive categories: agreement, here, meaning that a patient diagnosed undernourished (or with normal nutrition) or obese (or not obese) using a method also has been diagnosed undernourished (or normally nourished) or obese (or non-obese) using another method. The strength of agreement was interpreted as follows [20]: almost perfect agreement (κ = 0.81–1.00), substantial (κ = 0.61–0.80), moderate (κ = 0.41–0.60), fair (κ = 0.21–0.40), slight (κ = 0.00–0.20), and poor (κ < 0.00).
3. Results
Sixty eight (68) CHD patients (mean age of 46.7 years, 51.5% women, mean duration on dialysis of 83.04 ± 48.1 months). The main causal nephropathy was hypertensive nephropathy in 29/68 patients. The baseline characteristics are summarized in Table I.
The mean BMI was 21.7 ± 5.1 kg/m2. According to the WHO classification, 33/68 had a normal BMI, 20/68 patients were lean including 2/68 cachectic patients, 7/68 patients were overweight and 6/68 patients were obese. Thirty-two patients had a BMI of less than 20 kg/m2 (see Figure 1). Mean MUAC was 25.5 ± 5.6 cm. The average weight was 64.9 ± 16.1 kg or 104.0 ± 22.0% of ideal weight. The mean FM was 17.6 ± 12 kg or 25.9 ± 13.9% of body weight. The mean LM was 45.4 ± 14.3 kg with an mean LMI of 15.4 ± 4.4 kg/m2. Albuminemia was measured in 32 patients with an mean of 38.9 ± 5.1 g/l.
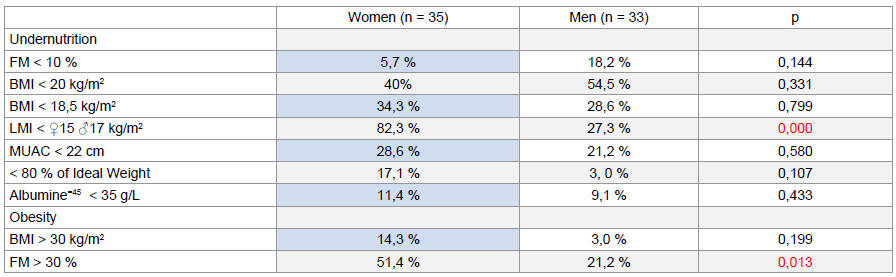
The linear correlation between anthropometric nutritional markers, impedance markers and serum albumin was studied (see Table II). Figure 2 shows the variation in the prevalence of undernutrition in the population studied according to the nutritional marker (anthropometry, BIA, serum albumin) used. It was 47.1% (95% CI [34.8 – 59.6]) based on BMI (threshold of 20 kg/m2), 25% (95% CI [15.3 – 37 .0]) according to MUAC, 10.3% (95% CI [4.2 – 20.1]) according to ideal weight, 11.8% (95% CI [5.2 – 21. 9]) according to FM, 55.9% (95% CI [43.3 – 67.9]) according to LMI and 10.3% (95% CI [4.2 – 20.1]) according to serum albumin. Table III illustrates the prevalence of undernutrition and obesity in patients by sex and by different assessment methods.
Agreement in diagnosis of undernutrition by FM was fair with BMI (< 20 kg/m2 [κ = 0.261 and p = 0.001]), BMI (< 18.5 kg/m2 [κ = 0.275 and p = 0.006]), MUAC (κ = 0.381 and p = 0.001) and slight with serum albumine (κ = 0.075 and p = 0.025). Diagnosis of undernutrition by LMI had a slight agreement with ideal weight (κ = 0.166 and p = 0.013), BMI (< 18.5 kg/m2 [κ = 0.301 and p = 0.005]), and a fair agreement with MUAC (κ = 0.288 and p = 0.003). The diagnosis of malnutrition by serum albumin was also slight agreement with ideal weight (κ = 0.092 and p = 0.004), BMI (< 18.5 kg/m2 [κ = 0.107 and p = 0.018]) and BMI (< 20 kg/m2 [κ = 0.142 and p = 0.002]).
The prevalence of obesity was 8.8% (95% CI [-1.8 – 22.4]) according to WHO criteria and 36.8% (95% CI [24.7 – 48 ,9]) according to FM. The diagnosis of obesity by FM (> 30%) had a fair agreement with WHO criteria (κ = 0.210 and p = 0.013) in the diagnosis of obesity.
|
Parameters |
Women (n = 35) |
Men (n = 33) |
p |
|
|
General |
||||
|
Age (years) |
48 ± 14 |
45 ± 14 |
0,297 |
|
|
Dialysis duration (months) |
87 ± 50 |
79 ± 46 |
0,432 |
|
|
Nephropathies |
0,468 |
|||
|
Vascular (%) |
40,0 |
48,5 |
||
|
Glomerular (%) |
22,9 |
24,2 |
||
|
CIN (%) |
11,4 |
18,2 |
||
|
No indeterminate (%) |
20,0 |
6,1 |
||
|
PKD (%) |
5,7 |
3,0 |
||
|
Vascular access |
0,480 |
|||
|
Catheter (%) |
20,0 |
27,3 |
||
|
AV Fistula (%) |
80,0 |
72,7 |
||
|
Dry weight (kg) |
62,1 ± 17,3 |
66,1 ± 14,5 |
0,075 |
|
|
Kt/V=45 |
1,32 ± 0,18 |
1,30 ± 0,17 |
0,836 |
|
|
Anthropometric |
||||
|
Ideal weight (kg) |
61,3 ± 3,8 |
62,9 ± 4,5 |
0,072 |
|
|
BMI (kg/m2) |
22,2 ± 5,8 |
21,1 ± 4,2 |
0,628 |
|
|
MUAC (cm) |
25,9 ± 6,5 |
24,9 ± 4,2 |
0,882 |
|
|
BIA |
||||
|
Fat mass (%) |
29,8 ± 11,3 |
21,7 ± 15,3 |
0,002 |
|
|
Lean mass index (kg/m2) |
15,1 ± 3,3 |
15,7 ± 5,3 |
0,005 |
|
|
Biologic |
||||
|
Hemoglobin (g/dl) |
8,9 ± 1,9 |
9,4 ± 1,4 |
0,146 |
|
|
CRP (mg/l) |
14,8 ± 26,5 |
12,0 ± 17,9 |
0,947 |
|
|
Kalemia (mEq/l) |
4,9 ± 1,1 |
4,7 ± 1,0 |
0,710 |
|
|
Phosphataemia (mg/l) |
29,6 ± 12,0 |
30,5 ± 10,4 |
0,540 |
|
|
Albumine=32 (g/l) |
37,5 ± 3,6 |
40,4 ± 6,3 |
0,105 |
|
Albuminie=45 : made at 32 patients. Kt/V=45 : made at 45 patients. AV fistulas : arteriovenous fistula. BMI : body mass index. CNI : chronic interstitial nephropathy. MUAC : mid-upper arm circumference. PKD : polycystic kidney disease.
Table 1: Baseline characteristics of chronic hemodialysis patients
|
Fat Mass |
Lean Mass Index |
|||
|
r |
p |
r |
p |
|
|
Ideal weight (%) |
0,939 |
0,000 |
0,625 |
0,000 |
|
BMI |
0,514 |
0,000 |
0,488 |
0,000 |
|
MUAC |
0,611 |
0,000 |
0,470 |
0,000 |
|
Albumine |
-0,008 |
0,966 |
0,241 |
0,184 |
Same as Table I.
Table 2: Correlation between anthropometric and bioelectrical impedance nutritional parameters
Same as Table I
Table 3: Prevalence of malnutrition according to different methods and sex
4. Discussion
Assessing nutritional state of CHD patients is important and challenging. Indeed, they present an increased risk of malnutrition due to many factors, nutritional or non-nutritional, but changes in their body composition are frequent [21]. Also, there is no single criterion for identifying malnutrition, which sometimes delays the diagnosis [22]. It has been suggested to assess the nutritional state of CHD patients from several markers such serum biochemical parameters and body composition analysis (weight, anthropometry, bioimpedance, total body nitrogen and DEXA) [7]. In this study, we evaluated the prevalence of undernutrition and obesity in our patients by comparing different evaluation methods. Malnutrition is a significant cause of morbidity and mortality in CHD. Its prevalence would be between 25% and 80% in different studies [7, 22, 23] and this variability would be due to the different criteria used to diagnose the nutritional state and could also be attributable to the variation in demographic and cultural characteristics and the traditional eating habits specific to each country and each ethnic group. Oliveira et al. [22] reported a prevalence between 12.1 and 94.8% using different methods. In our study, it was also assessed differently and ranged from 10.3 to 55.9%. The lowest value was found during the evaluation by albuminemia and the percentage of ideal weight and the highest during the evaluation by IMM. The prevalence of obesity also varied according to the diagnostic criteria with the highest value during the evaluation by FM in all patients and in men and women. BIA is thus a fast, non-invasive, cheap, safe method and can be done anywhere using a small portable device. It is therefore suitable for large-scale studies [24]. BIA data have been reported for white, African American, and Hispanic populations [25–28]. From a nutritional point of view, a high risk of malnutrition in CHD patients has been revealed in numerous studies that have used BIA [29]. In a study of 118 hemodialysis patients, SF-BIA was consistent with the gold standard, DEXA [30]. However, it can be affected by water status [12, 28]. In our study, BIA nutritional parameters were strongly and positively correlated with anthropometric markers. In addition, the agreement in the diagnosis of undernutrition and obesity was good according to a statistically significant kappa coefficient. Due to its availability and simplicity, BIA has considerable potential as a complement to conventional anthropometric techniques for assessing the nutritional status of hemodialysis patients. Our results show that the prevalence of malnutrition varied according to the evaluation method and sex in our patients. It was significantly higher in women when evaluated undernutrition by LMI and obesity by FM. Women are thus more exposed to undernutrition than men at the expense of a low LM in them. This relationship between a low LMI and female sex had already been highlighted by Rosenberger et al. in 748 patients [31]. Similarly, they are also more exposed to obesity in our cohort in relation to a high FM. This suggests the possible coexistence of protein malnutrition (reduced LM) and high adiposity (high MG). As shown in Figure 3, 31.4% of women are affected.
The limitations of our study were observational type, small sample size. But our results suggest that BIA has considerable potential in the nutritional assessment of our patients, due to its availability, simplicity and reliability.
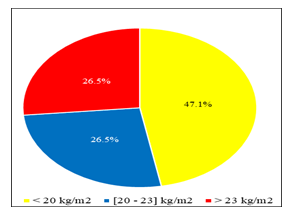
Figure 1: Distribution of patients according to their body mass index (BMI).
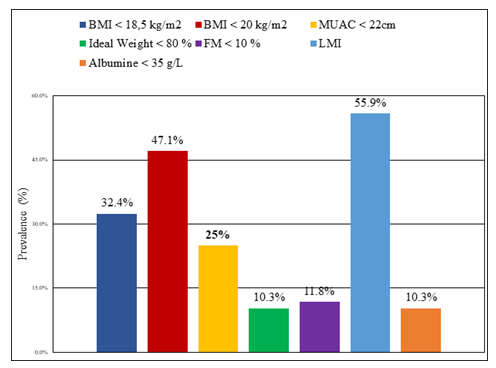
Figure 2: Prevalence of undernutrition according to nutritional markers.
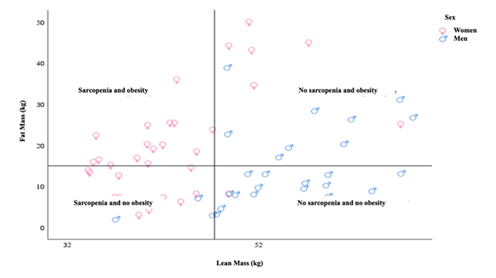
Figure 3: Non-linear plot of fat mass and lean mass of chronic hemodialysis patients. The horizontal and vertical axes indicate the medians of fat and lean mass in the population.
5. Conclusion
Different methods of nutritional assessment lead to very different estimates of undernutrition and obesity. Our results indicate that the prevalence of malnutrition varied in CHD patients depending on the method of nutritional assessment but also on sex. Our results also indicate an average prevalence of undernutrition of 26.7% in CHD patients, while more than half had a low lean mass and 36.8% of them had a high fat mass. According to the BIA, women on hemodialysis were at greater risk of malnutrition than men and they had an overlap of reduced lean mass and high fat mass. In order to clearly identify the latter, we suggest simple measurements of lean mass and fat mass indices, available from bioelectrical impedance analysis, in order to establish better diagnostic strategies and nutritional care in CHD patients.
Conflicts of interest
The authors declare no conflict of interest
Author contributions
Study design: Niakhaleen Keita, Maria Faye, Sidy Mouhamed Seck. Bibliography: Niakhaleen Keita, Maria Faye, Sidy Mouhamed Seck. Definition of the methodology: Niakhaleen Keita, Maria Faye, Moustapha Faye, Sidy Mouhamed Seck. Writing, co-writing or validation of the protocol: Niakhaleen Keita, Maria Faye, Sidy Mouhamed Seck. Steering: Niakhaleen Keita, Bacary Ba, Seynabou Diagne, Mansour Mbengue. Data analysis and validation: Niakhaleen Keita, Maria Faye, Moustapha Faye, Ahmed Tall Lemrabott, Sidy Mouhamed Seck. Manuscript writing: Niakhaleen Keita, Maria Faye, Moustapha Faye, Sidy Mouhamed Seck. Proofreading and validation of the manuscript: Niakhaleen Keita, Maria Faye, Moustapha Faye, Bacary Ba, Seynabou Diagne, Mansour Mbengue, Sidy Mouhamed Seck, Abdou NIANG, El Hadji Fary Ka.
Acknowledgments
Not Applicable
References
- Ikizler TA, Cano NJ, Franch H, et al. Prevention and treatment of protein energy wasting in chronic kidney disease patients: a consensus statement by the International Society of Renal Nutrition and Metabolism. Kidney Int 84(6) (2013):1096-107.
- Koefoed M, Kromann CB, Juliussen SR, et al. Nutritional Status of Maintenance Dialysis Patients: Low Lean Body Mass Index and Obesity Are Common, Protein-Energy Wasting Is Uncommon. PLoS One 11(2) (2016): e0150012.
- Kramer HJ, Saranathan A, Luke A, et al. Increasing body mass index and obesity in the incident ESRD population. J Am Soc Nephrol 17(5) (2006): 1453-9.
- van Biesen W, Claes K, Covic A, et al. A multicentric, international matched pair analysis of body composition in peritoneal dialysis versus haemodialysis patients. Nephrol Dial Transplant 28(10) (2013): 2620-8.
- Honda H, Qureshi AR, Axelsson J, et al. Obese sarcopenia in patients with end-stage renal disease is associated with inflammation and increased mortality. Am J Clin Nutr 86(3) (2007): 633-8.
- Cordeiro AC, Qureshi AR, Stenvinkel P, et al. Abdominal fat deposition is associated with increased inflammation, protein-energy wasting and worse outcome in patients undergoing haemodialysis. Nephrol Dial Transplant 25(2) (2010): 562-8.
- Ikizler TA, Hakim RM. Nutrition in end-stage renal disease. Kidney Int 50(2) (1996):343-57.
- Kaysen GA, Stevenson FT, Depner TA. Determinants of albumin concentration in hemodialysis patients. Am J Kidney Dis 29(5) (1997): 658-68
- Marcén R, Teruel JL, de la Cal MA, et al. The impact of malnutrition in morbidity and mortality in stable haemodialysis patients. Spanish Cooperative Study of Nutrition in Hemodialysis. Nephrol Dial Transplant. 12(11) (1997):2324-31.
- Zabetakis PM. Nutritional monitoring techniques in dialysis. Can routine monitoring impact outcome? ASAIO J 43(3) (1997):243-5.
- Qureshi AR, Alvestrand A, Danielsson A. et al. Factors predicting malnutrition in hemodialysis patients: a cross-sectional study. Kidney Int 53(3) (1998): 773-82.
- Ikizler TA, Wingard RL, Harvell J, et al. Association of morbidity with markers of nutrition and inflammation in chronic hemodialysis patients: a prospective study. Kidney Int 55(5) (1999): 1945-51.
- Kopple JD, Zhu X, Lew NL, et al. Body weight-for-height relationships predict mortality in maintenance hemodialysis patients. Kidney Int 56(3) (1999): 1136-48.
- Lorentz FH. Der konstitutionsindex der frau. Klin Wochenshr 8(16) (1929): 734-6.
- Fouque D, Vennegoor M, ter Wee P, et al. EBPG guideline on nutrition. Nephrol Dial Transplant 2(2007): ii45-87.
- VanItallie TB, Yang MU, Heymsfield SB, et al. Height-normalized indices of the body’s fat-free mass and fat mass: potentially useful indicators of nutritional status. Am J Clin Nutr 52(6) (1999): 953-9.
- Clinical practice guidelines for nutrition in chronic renal failure. K/DOQI, National Kidney Foundation. Am J Kidney Dis 35(2000):S17-104.
- Clinical practice guidelines for nutrition in chronic renal failure. K/DOQI, National Kidney Foundation. Am J Kidney Dis 38(4) (2001): 917.
- 2003 annual report: ESRD clinical performance measures project. Am J Kidney Dis 44(2004): S1-92.
- Physical status: the use of and interpretation of anthropometry, report of a WHO expert committee [Internet] 29(2022).
- Altman DG. Measuring agreement. Chapter 14: Some common problems in medical research. In: Practical statistics for medical research. New York: Chapman and Hall 10 (1991): p404.
- Kaysen GA, Zhu F, Sarkar S, al. Estimation of total-body and limb muscle mass in hemodialysis patients by using multifrequency bioimpedance spectroscopy. Am J Clin Nutr 82(5) (2005): 988-95.
- Oliveira CMC de, Kubrusly M, Mota RS, et al. [Malnutrition in chronic kidney failure: what is the best diagnostic method to assess?]. J Bras Nefrol 32(1) (2010): 55-68.
- Lowrie EG, Lew NL. Death risk in hemodialysis patients: the predictive value of commonly measured variables and an evaluation of death rate differences between facilities. Am J Kidney Dis 15(5) (1990): 458-82.
- Dumler F, Kilates C. Body composition analysis by bioelectrical impedance in chronic maintenance dialysis patients: comparisons to the National Health and Nutrition Examination Survey III. Ren Nutr 13(2) (2003): 166-72.
- Piccoli A, Nigrelli S, Caberlotto A, et al. Bivariate normal values of the bioelectrical impedance vector in adult and elderly populations. Am J Clin Nutr 61(2) (1995): 269-70.
- Piccoli A, Pillon L, Dumler F. Impedance vector distribution by sex, race, body mass index, and age in the United States: standard reference intervals as bivariate Z scores. Nutrition 18(2) (2002):153-67.
- Piccoli A, Brunani A, Savia G, et al. Discriminating between body fat and fluid changes in the obese adult using bioimpedance vector analysis. Int J Obes Relat Metab Disord 22(2) (1998): 97-104.
- Piccoli A, Italian CAPD-BIA Study Group. Bioelectric impedance vector distribution in peritoneal dialysis patients with different hydration status. Kidney Int 65(3) (2004): 1050-63.
- Johansen KL, Kaysen GA, Young BS, et al. Longitudinal study of nutritional status, body composition, and physical function in hemodialysis patients. Am J Clin Nutr 77(4) (2003): 842-6.
- Bross R, Chandramohan G, Kovesdy CP, et al. Comparing body composition assessment tests in long-term hemodialysis patients. Am J Kidney Dis 55(5) (2010): 885-96.
- Rosenberger J, Kissova V, Majernikova M, et al. Body composition monitor assessing malnutrition in the hemodialysis population independently predicts mortality. J Ren Nutr 24(3) (2014): 172-6.
