Nutritional and Functional Properties of Four Traditional Mucilaginous Vegetables used by Rural Populations in Benin Republic
Article Information
Léandre Gandji1, 2*, C Evariste S Mitchikpe2, Julien G Djego1
1Laboratory of Applied Ecology, University of Abomey-Calavi, Cotonou, Benin
2Human Nutrition Laboratory, University of Abomey-Calavi, Cotonou, Benin
*Corresponding Author: Léandre Gandji, Laboratory of Applied Ecology, Faculty of Agricultural Sciences, University of Abomey-Calavi, BP 1724 Abomey-Calavi, Benin
Received: 18 April 2019; Accepted: 30 April 2019; Published: 15 May 2019
Citation: Léandre Gandji, C Evariste S Mitchikpe, Julien G Djego. Nutritional and Functional Properties of Four Traditional Mucilaginous Vegetables used by Rural Populations in Benin Republic. Journal of Food Science and Nutrition Research 2 (2019): 076-086.
View / Download Pdf Share at FacebookAbstract
Child malnutrition is one of the public health problems. Unfortunately, some of our vegetables such as Adansonia digitata, Bombax brevicuspe, Ceiba pentandra and Grewia venusta, with important nutritional value, are neglected. To assess the nutritional value of these vegetables, their protein, lipid, fibre and mineral content as well as polyphenols have been determined using reference methods as per AOAC (Association of Official Analytical Chemists). The results showed that the leaves of A. digitata, B. brevicuspe, C. pentandra and the flower buds of G. venusta have a protein content from 17.19 g/100g; 19.84 g/100g; 9.26 g/100g; 13.01 g/100g respectively. Their lipid content is respectively 1.03 g/100g; 1.71 g/100g; 2.75 g/100g; 0.12 g/100g and their fibre content varies from 9.42 to 24.95 g/100g. These plants are also rich in iron, calcium, magnesium and vitamin C. In addition, the polyphenol content of these plants shows that they are important in the prevention of certain diseases. Given the different nutritional potential of each of these plants, it would be useful to popularize them in order to contribute to improving the quality of the food consumed by the population.
Keywords
Malnutrition, Nutritional potential, Mucilaginous vegetables, Polyphénol
Malnutrition articles Malnutrition Research articles Malnutrition review articles Malnutrition PubMed articles Malnutrition PubMed Central articles Malnutrition 2023 articles Malnutrition 2024 articles Malnutrition Scopus articles Malnutrition impact factor journals Malnutrition Scopus journals Malnutrition PubMed journals Malnutrition medical journals Malnutrition free journals Malnutrition best journals Malnutrition top journals Malnutrition free medical journals Malnutrition famous journals Malnutrition Google Scholar indexed journals Nutritional potential articles Nutritional potential Research articles Nutritional potential review articles Nutritional potential PubMed articles Nutritional potential PubMed Central articles Nutritional potential 2023 articles Nutritional potential 2024 articles Nutritional potential Scopus articles Nutritional potential impact factor journals Nutritional potential Scopus journals Nutritional potential PubMed journals Nutritional potential medical journals Nutritional potential free journals Nutritional potential best journals Nutritional potential top journals Nutritional potential free medical journals Nutritional potential famous journals Nutritional potential Google Scholar indexed journals Mucilaginous vegetables articles Mucilaginous vegetables Research articles Mucilaginous vegetables review articles Mucilaginous vegetables PubMed articles Mucilaginous vegetables PubMed Central articles Mucilaginous vegetables 2023 articles Mucilaginous vegetables 2024 articles Mucilaginous vegetables Scopus articles Mucilaginous vegetables impact factor journals Mucilaginous vegetables Scopus journals Mucilaginous vegetables PubMed journals Mucilaginous vegetables medical journals Mucilaginous vegetables free journals Mucilaginous vegetables best journals Mucilaginous vegetables top journals Mucilaginous vegetables free medical journals Mucilaginous vegetables famous journals Mucilaginous vegetables Google Scholar indexed journals Polyphénol articles Polyphénol Research articles Polyphénol review articles Polyphénol PubMed articles Polyphénol PubMed Central articles Polyphénol 2023 articles Polyphénol 2024 articles Polyphénol Scopus articles Polyphénol impact factor journals Polyphénol Scopus journals Polyphénol PubMed journals Polyphénol medical journals Polyphénol free journals Polyphénol best journals Polyphénol top journals Polyphénol free medical journals Polyphénol famous journals Polyphénol Google Scholar indexed journals quality of the food articles quality of the food Research articles quality of the food review articles quality of the food PubMed articles quality of the food PubMed Central articles quality of the food 2023 articles quality of the food 2024 articles quality of the food Scopus articles quality of the food impact factor journals quality of the food Scopus journals quality of the food PubMed journals quality of the food medical journals quality of the food free journals quality of the food best journals quality of the food top journals quality of the food free medical journals quality of the food famous journals quality of the food Google Scholar indexed journals Child malnutrition articles Child malnutrition Research articles Child malnutrition review articles Child malnutrition PubMed articles Child malnutrition PubMed Central articles Child malnutrition 2023 articles Child malnutrition 2024 articles Child malnutrition Scopus articles Child malnutrition impact factor journals Child malnutrition Scopus journals Child malnutrition PubMed journals Child malnutrition medical journals Child malnutrition free journals Child malnutrition best journals Child malnutrition top journals Child malnutrition free medical journals Child malnutrition famous journals Child malnutrition Google Scholar indexed journals nutritional value articles nutritional value Research articles nutritional value review articles nutritional value PubMed articles nutritional value PubMed Central articles nutritional value 2023 articles nutritional value 2024 articles nutritional value Scopus articles nutritional value impact factor journals nutritional value Scopus journals nutritional value PubMed journals nutritional value medical journals nutritional value free journals nutritional value best journals nutritional value top journals nutritional value free medical journals nutritional value famous journals nutritional value Google Scholar indexed journals Food and nutrition articles Food and nutrition Research articles Food and nutrition review articles Food and nutrition PubMed articles Food and nutrition PubMed Central articles Food and nutrition 2023 articles Food and nutrition 2024 articles Food and nutrition Scopus articles Food and nutrition impact factor journals Food and nutrition Scopus journals Food and nutrition PubMed journals Food and nutrition medical journals Food and nutrition free journals Food and nutrition best journals Food and nutrition top journals Food and nutrition free medical journals Food and nutrition famous journals Food and nutrition Google Scholar indexed journals nutritional quality articles nutritional quality Research articles nutritional quality review articles nutritional quality PubMed articles nutritional quality PubMed Central articles nutritional quality 2023 articles nutritional quality 2024 articles nutritional quality Scopus articles nutritional quality impact factor journals nutritional quality Scopus journals nutritional quality PubMed journals nutritional quality medical journals nutritional quality free journals nutritional quality best journals nutritional quality top journals nutritional quality free medical journals nutritional quality famous journals nutritional quality Google Scholar indexed journals
Article Details
1. Introduction
Food and nutrition insecurity is a recurrent problem in traditional systems in sub-Saharan Africa and particularly in Benin. The origins of child malnutrition in Benin were an incorrect timely complementary feeding practices and cereal porridges and extracts from family diets were the two categories of complementary foods provided to children [1]. Complementary feeding was mostly initiated using locally prepared maize porridges in Benin. West African flora is known for its diversity of species that can serve humans well, but most of them have not been studied in depth [2].
The young leaves or the shoots are normally used for soup (sausage) [3]. A powder prepared from the dried leaves is used to prepare sauce during the dry season [3, 4]. This is the case of Adansonia digitata, Bombax brevicuspe, Ceiba pentandra and Grewia venusta all from the Malvaceae family (APG II) whose leaves and/or flowers are consumed in rural Benin [5-9]. In Nigeria, the flowers and young shoots are sometimes used as a soup or sauce vegetable [10]. Some findings demonstrated that the mucilage obtained from the stem bark can serve as a good binder in paracetamol formulations [10]. The mucilage of this plant could be useful as a functional ingredient on account of its physical/functional properties and phytochemical composition [11]. Baobab leaves are a pool of essential and protective compounds and minerals [12]. G. venusta mucilage appeared to affect the physical, chemical and organoleptic properties of akara prepared from bambara groundnuts in Nigeria [13]. Chemically, G. venusta had high levels of minerals such potassium (K), iron (Fe) and phosphorus (P) [14]. Fruits of Grewia spp. are a good source of phytic acid (antioxidant) which lowers blood cholesterol [14]. As for C. pentandra, constituents confer their protective, preventive and therapeutic properties on these leaves and these improve the benefits that can be derived from the consumption of these leaves [3]. In Côte d’Ivoire, Konan et al. [15] showed that Bombax costatum contains, on the one hand, phytophenols (phenol acids, coumarins, flavonoids, tannins), sterols, terpenes and on the other hand, has demonstrated its antioxidant potential. In Nigeria, different parts of Bombax costatum are employed for various purposes. The immature fruits are prepared as an emollient; decoction of young leaves is used as a warm bath for febrile children [16]. Bombax costatum is a useful plant resource that could help alleviate poverty. It has many benefits for local populations and a great potential for food and medicine [8]. Moreover, a real interest for consumers is emerging since epidemiological studies have linked the dietary habits and the prevalence of certain diseases [17-19]. Many of the local vegetable materials are underexploited because of inadequate scientific knowledge of their nutritional potentials [20, 21]. It is why this study was conducted with the objective of assessing the nutritional and functional values of traditional perennial mucilaginous vegetables.
The purpose of this study is to evaluate the nutritional quality of four neglected vegetables (A. digitata, B. brevicuspe, C. pentandra and G. venusta) through their content of mineral ions, vitamins and secondary metabolite content.
2. Materials and Methods
2.1 Materials
The leaves of A. digitata and B. brevicuspe were harvested in the village of Ouèdèmè, those of C. pentandra were harvested in Djallouma and the flower buds of G. venusta in Aglamidjodji in the department of Collines in central Benin during the dry season. The samples were cleaned and were subsequently separated into 2 portions. The two portions were treated differently. One portion was dried at room temperature. The vegetables in this dried batch were ground into powder and used for the determination of macronutrients and micronutrients. The second fresh portion, which was extracted in water, was used for the determination of total polyphenols and pH.
2.2 Methods
2.2.1 Physico-chemical analyses of dried vegetable powders:
2.2.1.1 Determination of mineral and vitamin C contents of raw vegetable powders: Minerals such as calcium, iron and magnesium were determined using the spectrophotometric method [22]. Thus, 2 g of each of the treated samples were weighed and incinerated in a crucible at 550°C in a muffle furnace. The resultant ash was dissolved in 5 ml of HNO3/HCl/H2O and heated gently on a hot plate until brown fumes disappeared. To Then 5 ml of deionized water was added to each crucible and the mixture was heated until a colorless solution was obtained. The mineral solution in each crucible was transferred into a 100 ml volumetric flask by filtration through a whatman N° 42 filter paper and the volume was made to the mark with deionized water. This solution was used for elemental analysis by atomic absorption spectrophotometer. A 10 cm-long cell was used and the concentration of each element in the sample was calculated on percentage of dry matter.
2.2.1.2 Dosage of vitamin C: Ascorbic acid or vitamin C was extracted in the presence of a solution of metaphosphoric acid/acetic acid and measured by 2,6-dichlorophenol indophenol calibrated by the standard vitamin C of known concentration [23]. An amount (10 mL) of acid (metaphosphoric acid / acetic acid) was added to 1g of powder of the extract and the mixture was stirred for 30 minutes and filtered on whatman paper N°1. 3 mL of the filtrate was titrated with 2,6-dichlorophenol indophenol until a persistent champagne rose coloration appeared. The volume of 2,6-dichlorophenol indophenol (Ve) added was noted. A solution of metaphosphoric acid-acetic acid was also titrated with 2,6-dichlorophenol indophenol. The volume added (Vo) was noted. A 1 mg/mL vitamin C solution prepared just before the assay was also titrated with 2,6-dichlorophenol indophenol under the same conditions. The volume paid (Vs) was noted. Each assay was performed in triplicate. The pH of the samples was measured according to the AACC method (02-31-01) with a pH meter (pHep®) previously calibrated with buffer solutions at pH 4.0 and pH 9.0.
2.2.2 Determination of lipid, protein and dietary fibre content of raw vegetable powders:
2.2.2.1 Crude lipid: Crude lipid was determined by using the method described by Osborne and Voogt [24]. 2 g of each sample were placed into separate extraction thimbles and then covered with cotton wool. The extraction thimbles containing the samples were placed in the extraction jacket. Clean dried 500 ml round bottom flasks containing few anti-bumping granules were weighed (W1) and 300 ml of petroleum ether was poured into each flask fitted with sohxlet extraction units. The round bottom flasks and the condenser were connected to the sohxlet extractor and cold-water circulation was put on. The heating mantle was switched on; the heating rate was adjusted until the solvents were refluxing at a steady rate. Extraction was carried out for 6 h. The solvents were recovered and the oil was dried in the oven at 70°C for 1 h. The round bottom flask and oil were cooled and then weighed (W2). The lipid content was calculated.
2.2.2.2 Crude protein: Micro-Kjeldahl method as described by AOAC [22] was used. Briefly, 0.5 g of each sample were weighed and placed on each nitrogen free filter paper, then folded and dropped into a Kjeldahl digestion tubes. 3.0 g of digesting mixed catalyst (CuSO4 + Na2SO4) and 25 ml of Conc. Na2SO4 were added to each sample in the digestion tubes. The mixtures in the digestion tubes were transferred to the Kjeldahl digestion apparatus; the heater was regulated at a temperature below the boiling point of the acid until frothing ceased. The mixtures boil vigorously as the temperature was increased, until clear (light) green color was obtained. The digests were allowed to cool and then transferred into separate 100 cm3 volumetric flasks and diluted with distilled water to make up 100 cm3. 10 ml aliquot of each digest was introduced into the distillation jacket of the micro-steam distillation apparatus that was connected to the main, as the water in the distiller flask boils. 20 ml of 40% NaOH was added to each digest in the distillation jacket. 50 ml of 40% boric acid was measured into separate conical flasks, four (4) drops of methyl red indicator was added to each. The conical flasks containing the mixture were placed onto the distillation apparatus with the outlet tubes inserted into each conical flask and NH3 was collected through the condenser. The distillation continued until 25 ml of the distillate were trapped into the boric acid solution and colour changes from red to yellow. The distillates were then titrated with 0.02 M HCL and the titre values were recorded. Percentage nitrogen was first calculated and crude protein was determined by multiplying the percentage nitrogen with a factor of 5.3 for vegetables [25].
2.2.2.3 Crude fibre: Crude fibre was analyzed following the procedure of AOAC [22]. 2.0 g of each sample were weighed into separate 500 ml round bottom flasks. 100 ml of 0.25 M sulphuric acid solutions was added to each sample in the flask, and the mixtures were boiled under reflux for 30 min. The hot solutions were quickly filtered under suction. The residues were thoroughly washed with hot water until acid free. Each residue was transferred into the round bottom flasks and 100 ml of hot 0.3 M sodium hydroxide solutions was added and the mixtures were boiled again under reflux for 30 min and filtered quickly under suction. Each insoluble residue was washed with hot water until it was base free. They were dried to a constant weight in an oven at 100°C for 2 hours, cooled in desiccators and weighed (C1). The weighed samples were then incinerated, and reweighed (C2). Percentage crude fibre content was calculated [25].
2.2.3 Determination of secondary metabolite (total polyphenols) contents of raw vegetable extracts:
2.2.3.1 Sample extraction: The leaves of A. digitata, B. brevicuspe, C. pentandra and the flower buds of G. venusta were crushed. Water was used as an extraction solvent. The extracts were concentrated in a vacuum.
2.2.4 Total polyphenols: The polyphenol dosage method chosen uses the Folin-Ciocalteu reagent. The Folin-Ciocalteu reagent is a mixture of phosphotungstic acid (H3PW12O40) and phosphomolybdic acid (H3PMo12O40). In basic solution, polyphenols reduce the yellow-coloured Folin-Ciocalteu reagent to blue-coloured tungsten and molybdenum oxide. The intensity of this blue color indicates the total polyphenol content in the mixture. The latter has an absorption maximum at 760 nm, the intensity of which is proportional to the amount of polyphenols present in the sample [26]. To determine the total polyphenols in our samples, we used the method of Singleton et al. [27] with some modifications using 96-well microplates with a transparent bottom. To 20 µL of each sample contained in a well, we add 100 µL of Folin-Ciocalteu reagent and then shake it for 30 seconds before incubating for 5 minutes. An addition of 80 µL of Na2CO3 to 75 mg.mL-1 was made in each well to stop the reaction. The absorbance readings of the wells were taken at 760 nm. The polyphenols content was obtained using a calibration curve of gallic acid (0.2 mg/mL) as standard.
2.2.5 Statistical analysis of results: Calculations of nutrient and secondary metabolite content from vegetables were performed using Excel software. The estimates reported for nutrient and secondary metabolite contents for vegetables are averages ± standard deviation. The values were calculated using the ANOVA software.
3. Results and Discussions
3.1 The pH of the leaves of A. digitata, B. brevicuspe, C. pentandra and the flowers of G. venusta
From the results (Figure 1), it appears that the linear trend curve of vegetable pH ranges from 6.75 to 7.00 for A. digitata, B. brevicuspe and G. venusta. Therefore, it appears that they are classified as neutral leafy vegetables. These results are consistent with those of Tchiégang and Aissatou [28] in Cameroon, who report that mucilaginous vegetables in general and A. digitata, Corchorus olitorius, Ceratotheca sesamoïdes and Abelmoschus esculentus in particular are neutral. These results are close to those found (6.16 and 6.51) in Côte d'Ivoire by Assi et al. [29] in the mucilages of Irvingia gabonensis and Abelmoschus esculentus respectively. This organoleptic property (taste neither acid nor alkaline) is another criteria of choice in addition to the smell, colour and gelatinous texture. Moreover, research by Dansi et al. [5] revealed that taste is the first organoleptic criteria of preference in the choice of leafy vegetables in the centre and south of Benin. And with regard to the importance of mucilaginous vegetables, Chadare et al. [9] reported that the leaves of A. digitata and C. pentandra are real traditional food ingredients for complementary food formulations to combat infant malnutrition in Benin. The use of these neutral vegetables would reduce the prevalence of digestive tract diseases (diarrhoea and infections) caused by the aggression of the “young intestinal flora” of young children after consumption of the family meal, whose physico-chemical properties are very poorly controlled.
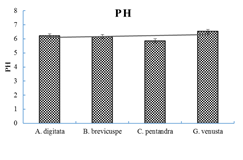
Figure 1: pH of mucilaginous vegetables.
3.2 The protein, lipid and fibre content of fresh and raw vegetables
In Table 1, the average values of the protein, lipid and fibre contents of the leaves of A. digitata, B. brevicuspe, C. pentandra and the flowers of G. venusta were presented. The protein content of raw vegetables ranges from 9.26 to 19.84 g/100g. These values are higher than 6.21 g/100g reported for Corchorus olitorius leaves [25] but similar to 14.47 g/100g reported for Hibiscus sabdariffa leaves [30]. The high protein content of these vegetables can reduce or even eliminate deficiencies in children, pregnant women and the poor in rural areas. Therefore, it appears that these vegetables can be used for building and repairing of body tissue, regulation of body process and formation of enzyme and hormones [31].
|
Species |
Protein |
Lipid |
Fibre |
|
Adansonia digitata |
17.19 ± 0.77 |
1.03 ± 0.34 |
9.42 ± 1.00 |
|
Bombax brevicuspe |
19.84 ± 1.18 |
1.71 ± 0.33 |
11.29 ± 1.17 |
|
Ceiba pentandra |
9.26 ± 1.98 |
2.75 ± 0.43 |
24.95 ± 0.60 |
|
Grewia venusta |
13.01 ± 1.54 |
0.12 ± 0.04 |
13.01 ± 1.74 |
Table 1: Protein, fat and fibre content of fresh and raw vegetables in g/100g of fresh material.
Lipid contents of raw vegetables vary from one species to another and range from 0.12 g/100g DM for C. pentandra to 2.75 g/100g DM for B. brevicuspe. These values are similar to 2.10 g/100g reported for A. digitata 1.39 g/100g reported for C. pentandra [30], but lower than 6.64 g/100g in Chorchorus olitorius [32]. The very low values obtained for lipid levels corroborate the findings of studies that indicate that vegetables contain little lipid [17]. Despite their low lipid content, vegetables provide the body with an essential amount of lipid [33]. Lipids are essential fats that play a very important role in the human body. Lipids help with brain function, joint mobilization and even energy production, they also help the body to absorb fat-soluble vitamins such as vitamins A and E [25]. Regarding the fibre content of raw vegetables, it is between 9.42 and 24.95 g/100g and is in the same order of importance as the fibre content 24.01 g/100g for C. pentandra [19]. Fibre intake has many health benefits. Soluble and insoluble fibres have a beneficial effect on the intestinal mucosa [17] and also contribute to reducing the risk of cardiovascular disease and cancer [19, 34].
3.3 The content of iron, calcium, magnesium, vitamin C and polyphenols in fresh and raw vegetables
Iron is required for blood formation and is said to be an important element in the diet of pregnant women, nursing mothers, infants convulsing patients and elderly to prevent anaemia and other related diseases. In Table 2, the average values of the mineral, vitamin C and polyphenol contents of the four plants were presented. The iron content was highest in raw leaf powders of A. digitata (25.11 mg/100g DM) and lowest in B. brevicuspe (6.70 mg/100g DM). These levels are lower than 54-56 mg/100g in C. olitorius and 89-92 mg/100g in H. sabdariffa [35].
|
Species |
Iron |
Calcium |
Magnesium |
Vitamin C |
Polyphenols |
|
Adansonia digitata |
25.11 ± 0.52 |
357.09 ± 38.71 |
278.00 ± 36.93 |
51.31 ± 7.06 |
11.68 ± 0.14 |
|
Bombax brevicuspe |
6.70 ± 0.43 |
107.34 ± 3.36 |
518.05 ± 46.01 |
38.05 ± 2.62 |
21.31 ± 1.36 |
|
Ceiba pentandra |
18.03 ± 0.57 |
475.56 ± 7.56 |
667.55 ± 46.96 |
35.00 ± 5.36 |
10.72 ± 0.61 |
|
Grewia venusta |
20.54 ± 1.49 |
228.70 ± 26.82 |
478.23 ± 18.62 |
22.10 ± 2.50 |
36.28 ± 1.21 |
Table 2: Content of iron, calcium, magnesium, vitamin C and polyphenols (mg GAE/100g) in fresh and raw vegetables.
Traditional vegetables contribute significantly to calcium intake in the traditional diet. Calcium is the most abundant mineral in humans existing as hydroxyapatite and very important to humans for it role in blood clotting, muscle contraction, neurological function, bone and teeth formation [36]. Calcium content was highest in the leaves of C. pentandra (475.56 mg/100g MS) and lowest in those of B. brevicuspe (107.34 mg/100g MS). These results are similar to those obtained 110.16 mg/100g for H. sabdariffa [37] and 402.21 mg/100g [30] but higher than those obtained 12.41 mg/100g for C. olitorius [38] in Ghana. Vegetables also contain a good magnesium content. The magnesium content of these mucilaginous plants ranges from 278.00 mg/100g to 667.55 mg/100g. These values are also similar to the results obtained by Sahoré et al. [35] in Côte d'Ivoire 405-422 mg/100g for C. olitorius and H. sabdariffa leaves. Magnesium is a cofactor in over 300 enzymatic reactions that regulate various biochemical reactions in the body including protein synthesis, muscle function, blood glucose, blood pressure and heart rate regulation [39]. Considering the recommended dietary allowance (RDA) for minerals, calcium (1000 mg/day); magnesium (400 mg/day), iron (8 mg/day) and zinc (6 mg/day) these leafy vegetables could cover RDA and contribute substantially for improving human diet [40]. Traditional leafy vegetables are also some of the best sources of vitamin C in African traditional diets [41]. The vegetables studied contain vitamin C whose concentrations vary from species to species and range from 22.1 mg/100g DM for G. venusta to 51.31 mg/100g DM for A. digitata. Vitamin C is a powerful antioxidant essential for healthy formation of bone and teeth [31]. Lastly, polyphenol contents vary by 10.72 to 36.28 mg GAE/100g and are of the same range as the phenolic compound content of olive oil, 16.72 mg GAE/100g [42]. These results show that vegetables contain phytophenols on the one hand, and on the other hand, highlight their obvious antioxidant potential. This virtue opens a promising avenue that will turn these plant matrices into a source of natural antioxidants. It seems clear, moreover, that the antioxidant activity exhibited by B. costatum and therefore by B. brevicuspe could explain its recurrent use in the traditional care of people with mental disorders [15]. Moreover, the presence of these different metabolites in plants is very useful because they may protect individuals from the damaging effects of free radicals, precursors to diseases such as Alzheimer’s disease, cardiovascular disease and cancer [43]. This property of B. brevicuspe has often been cited in Kétou and Pouignan in Benin by traditional therapists.
It should be noted that some of the differences in the nutritional values compared to others may be due to factors such as climate, leaf age, and nature of soil, growing conditions and the period of analysis. It should also be noted that there is a synergistic effect between the nutritional elements of vegetable when used in the body. The iron present in plants is in non-hematic form (Fe3+) and therefore generally consists of a set of inorganic ferric salts. During digestion, this inorganic iron is partially reduced to its more absorbable iron form (Fe2+). This conversion is remarkably helped by the presence of vitamin C, hence the preservation of the vitamin C contained in the vegetables. In addition to the organoleptic characteristics recognized (gelatinous texture, taste neither alkaline nor acidic), the vegetables studied contain primary metabolites (proteins, lipids, fibres, iron, Ca, Vit C and Mg) and secondary metabolites (antioxidants; polyphenols) that give them nutritional and functional properties. In a context of a high prevalence of child malnutrition supported by climate change, it is imperative to popularize the use of perennial mucilaginous vegetables because they are available when others (Ceratotheca sesamoides, Sesamum radiatum, Hibiscus sabdariffa, Abelmoschus esculentus and Corchorus olitorius) disappear and are also little known and therefore inexpensive to buy. However, the influence of traditional processing methods on the nutritional value of vegetables must be investigated.
4. Conclusion
The results obtained show that the leaves of A. digitata, B. brevicuspe, C. pentandra and the floral buds of G. venusta are rich in vitamins C, as well as in iron (Fe), calcium (Ca) and magnesium (Mg). Most of them play a very important role in immuno-stimulation. The richness of these leaves and flowers in phenolic compound, particularly phenols and flavonoids, gives them a strong antimicrobial activity (antibacterial and antifungal). These results therefore show the enormous potential of these vegetables in the management of malnutrition and numerous microbial infections. They would therefore be well indicated for undernourished children as well as for pregnant women, convalescents and the elderly. The valorization of the leaves of A. digitata, B. brevicuspe, C. pentandra and the flowers of G. venusta, four mucilaginous perennial plants, would effectively contribute to the health of populations. In addition, it would help to combat poverty, as leaf collection is an income-generating activity. However, traditional methods of preservation and preparation merit investigation in order to preserve the nutritional and functional values of these traditional vegetables.
Conflict of Interests
The authors did not declare any conflict of interest.
References
- Mitchodigni IM, Hounkpatin WA, Ntandou-Bouzitou G, et al. Complementary Feeding Practices of Children Aged 6-23 Months in Rural Area, Southern-Benin: Challenges and Opportunities. IJTDH 24 (2017): 1-12.
- Arbonnier M. Trees, shrubs and lianas in the dry zones of West Africa. (2nd), CIRAD-MNHN Paris (2002): 573.
- Raimi MM, Oyekanmi AM, Farombi AG. Proximate and phytochemical composition of leaves of Ceiba pentandra, Manihot esculentus and Abelmoschus esculentus in Southwestern Nigeria. Scientific Research Journal (SCIRJ) 2 (2014): 30-34.
- Friday ET, Omale J, Olupinyo D, et al. Investigations on the nutritional and medicinal potentials of Ceiba pentandra leaf: A common vegetable in Nigeria. International Journal of Plant Physiology and Biochemistry 3 (2011): 95-101.
- Dansi A, Adjatin A, Adoukonou-Sagbadja H, et al. Traditional leafy vegetables and their use in the Benin Republic. Genet Resour Crop Ev 55 (2008): 1239-1256.
- Djego J, Djego-Djossou S, Cakpo Y, et al. Evaluation du potentiel ethnobotanique des populations rurales au Sud et au centre du Bénin. Int J Biol Chem Sci 5 (2011): 1432-1447.
- Assogbadjo AE, Glele Kakai R, Idohou R, et al. Building capacity for sustainable baobab leaves production in Benin. RUFORUM working document 14 (2016): 349-353.
- Assogba GA, Fandohan AB, Salako VK, et al. Usages de Bombax costatum (Malvaceae) dans les terroirs riverains de la réserve de biosphère de la Pendjari, République du Bénin. Bois et Forets des Tropiques 333 (2017): 17-29.
- Chadare FJ, Madode YE, Fanou-Fogny N, et al. Indigenous food ingredients forcomplementary food formulations to combat infant malnutrition in Benin: a review (2017): 8568.
- Al-Youssef HM, Musarat A and El-shafae AM. Biological evaluation of constituents from Grewia mollis. Journal of Chemical and Pharmaceutical Research 4 (2012): 508-518.
- Alobo AP and Arueya GL. Physical, functional and chemical properties of Grewia venusta (ururu) mucilage extract. International Food Research Journal 24 (2017): 2107-2115.
- Ogbaga CC, Nuruddeen FA, Alonge OO, et al. Phytochemical, Elemental and Proximate Analyses of Stored, Sun-Dried and Shade-Dried Baobab (Adansonia digitata) Leaves 1 (2017): 11.0148
- Alobo Arubi P and Arueya Gibson L. Effect of Grewia venusta (‘Ururu’) mucilageon the physicochemical and sensory properties of fried cake (‘akara’) prepared from bambara groundnut seed flour. Afr J Food Sci Technol 6 (2014): 12-17.
- Sati NME, Ahmed FAM. Botanical Overview and Chemical Composition of some Grewia spp. “Gudeim Plant” in Sudan. Open Science Journal 3 (2018): 1-12
- Konan KV, Mamyrbékova-Békro JA, Guiffrey P, et al. Composition organique et activité anti-radicalaire de Bombax brevicuspe de Côte d’Ivoire. Revue CAMES-Série Pharm Méd Trad Afr 18 (2017): 21-27
- Abubakar UD and Barminas JT. Evaluation of Bombax costatum calyx hydrogel as a potential insulin delivery system. BJAST 20 (2017): 1-9
- Vodouhè S, Dovoedo A, Anihouvi VB, et al. Influence du mode de cuisson sur la valeur nutritionnelle de Solanum macrocarpum, Amaranthus hybridus et Ocimum gratissimum, trois légumes feuilles traditionnels acclimatés au Bénin. Int J Biol Chem Sci 5 (2012): 1926-1937
- Li M, Fan Y, Zhang X, et al. Fruit and vegetable intake and risk of type 2 diabetes mellitus: meta-analysis of prospective cohort studies. BMJ Open 4 (2014): e00597.
- Ehilé SE, Kouassi NK, N’Dri DY, et al. Proximate composition of five varieties of spontaneous leafy vegetables regularly consumed in Côte d’Ivoire Areas. Int J Curr Microbiol App Sci 6 (2017): 3536-3542.
- Awobajo FO, Olatunji- Bello II, Obilade TT, et al. Knowledge of the Nutritional and medicinal use of some vegetables among a cross section of market women in two major food markets in Lagos State, South West Nigeria. Pakistan Journal of Nutrition 9 (2010): 216-221.
- Dansi A, Vodouhè R, Azokpota P, et al. Diversity of the neglected and underutilized crop species of importance in Benin. Scientific World Journal (2012): 932947.
- Official Method of Analysis. 15th ed. Association of Official Analytical Chemist, Washington, DC (1990).
- Pongracz G, Weiser H, Matzinger D. Tocopherols-Antioxydant. Fat Science Technology 97 (1971): 90-104.
- Osborne DR, Voogt P. The Analysis of Nut rients in Foods London Academic Press (6th) (1978): 239-245.
- Adeniyi SA, Ehiagbonare JE, Nwangwu SCO. Nutritional evaluation of some staple leafy vegetables in Southern Nigeria. International Journal of Agricultural and Food Science 2 (2012): 37-43.
- Khadhri A, El Mokni R, Smiti S. Composés phénoliques et activités antioxydantes de deux extraits de chardon à glu: Atractylis gummifera. Revue Soc Sci Nat de Tunisie 39 (2013): 44-52.
- Singleton VL, Orthofer R, Lamuela-Raventós RM. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Methods in Enzymology 299 (1999): 152-178.
- Tchiégang C, Aissatou K. Données ethnonutritionnelles et caractéristiques physico-chimiques des légumes feuilles consommés dans la savane de l’Adamaoua (Cameroun). Tropicultura 22 (2004): 11-18.
- Assi OY, Sidibé D, Kouakou P, et al. Characterization of the Mucilages of Four Food Plants, Abelmoschus esculentus, Beilschmiedia mannii, Corchorus olitorius, and Irvingia gabonensis, from Côte d’lvoire. Biotechnology Journal International 19 (2017): 1-10.
- Oulai PD, Zoué LT, Niamké SL. Evaluation of nutritive and antioxidant properties of blanched leafy vegetables consumed in Northern Côte d’Ivoire. Pol J Food Nutr Sci 65 (2015): 31-38.
- Misra S and Misra MK. Nutritional evaluation of some leafy vegetable used by the tribal and rural people of south Odisha, India. J Nat Prod Plant Resour 4 (2014): 23-28.
- Traoré K, Parkouda C, Savadogo A, et al. Effect of processing methods on the nutritional content of three traditional vegetables leaves: Amaranth, black nightshade and jute mallow. Food Sci Nutr 25 (2017): 1139-1144.
- Badau MH, Abba HZ, Agbara GI, et al. Proximate composition, mineral content and acceptability of granulated maize dumpling (Dambu Masara) with varying proportions of ingredients. Global Advanced Research Journal of Agricultural Science 2 (2013): 320-329.
- Madhu C, Krishna KM, Reddy KR, et al. Estimation of Crude Fibre Content from Natural Food Stuffs and its Laxative Activity Induced in Rats. Int J Pharma Res Health Sci 5 (2017): 1703-06.
- Sahore AD, Abouattier L, Gbogouri FG. Assessment of some mineral elements (Ca, Na, K, Mg, Fe, Mn Cu and Zn) and their nutritional intake of two traditional leafy vegetables: Leaves of Corchorus olitotorius (Tiliaceae) and Hibiscus sabdariffa (Malvaceae). International Journal of Agriculture Innovations and Research 3 (2014): 786-791.
- Senga Kitumbe P, Opota Onya D, Tamba Vemba A, et al. Chemical composition and nutritive value study of the seed oil of Adenanthera pavonina L. (Fabaceae) growing in Democratic Republic of Congo. International journal of Pharmtech Research 5 (2013): 205-216.
- Asaolu SS, Adefemi OS, Oyakilome IG, et al. Proximate and Mineral Composition of Nigerian Leafy Vegetables. Journal of Food Research 1 (2012): 214-218.
- Amagloh FK, Atuna R.A, McBride R, et al. Nutrient and Total Polyphenol Contents of Dark Green Leafy Vegetables, and Estimation of Their Iron Bioaccessibility Using the In Vitro Digestion/Caco-2 Cell Model. Food 6 (2017): 1-12.
- Rude RK, Singer FR, and Gruber HE, et al. Skeletal and hormonal effects of magnesium deficiency. J Am Coll Nutr 28 (2009): 131-141.
- Institute of Medicine. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, FattyAcids, Cholesterol, Protein, and AminoAcids. Washington, DC: The National Academies Press (2005): 10490.
- Aworh OC. Promoting food security and enhancing Nigeria’s small farmers’ income through value-added processing of lesser-known and under-utilized indigenous fruits and vegetables. Food Research International 76 (2015): 986-991.
- Merouane A, Noui A, Medjahed H, et al. Activite antioxydante des composés phénoliques d’huile d’olive extraite par méthode traditionnelle. Int J Biol Chem Sci 8 (2014): 1865-1870
- Oboh G, Akinyemi AJ, Ademiluyi AO, et al. Inhibitory effect of some tropical green leafy vegetables on key enzymes linked to Alzheimer’s disease and some pro-oxidant induced lipid peroxidation in rats’ brain. Journal of Food Science and Technology 51 (2011): 884-891.
