Neonate with Severe Tricuspid Regurgitation and Flail Anterior Leaflet of the Tricuspid Valve
Article Information
Matthew F Pizzuto*, Chris S Iskander, J. Ryan Shea
Department of Pediatrics, University of North Carolina at Chapel Hill, North Carolina Children’s Hospital
*Corresponding Author: Matthew F Pizzuto, Campus Box 7221, 417 MacNider Hall, Chapel Hill, North Carolina.
Received: 30 August 2023; Accepted: 13 September 2023; Published: 23 October 2023
Citation: Matthew F Pizzuto, Chris S Iskander, J. Ryan Shea. Neonate with Severe Tricuspid Regurgitation and Flail Anterior Leaflet of the Tricuspid Valve. Archives of Clinical and Medical Case Reports. 7 (2023): 374-376.
View / Download Pdf Share at FacebookAbstract
Flail anterior tricuspid valve leaflet is a rare source of primary tricuspid regurgitation. It can be challenging to manage medically in neonates and often requires surgical repair. This case describes the use of medical management of a patient with flail tricuspid valve leaflet with severe tricuspid regurgitation to avoid neonatal cardiac surgery
Keywords
Flail tricuspid valve; Tricuspid regurgitation; Neonate; Congenital Heart Disease
Flail tricuspid valve articles; Tricuspid regurgitation articles; Neonate articles; Congenital Heart Disease articles
COVID-19 articles COVID-19 Research articles COVID-19 review articles COVID-19 PubMed articles COVID-19 PubMed Central articles COVID-19 2023 articles COVID-19 2024 articles COVID-19 Scopus articles COVID-19 impact factor journals COVID-19 Scopus journals COVID-19 PubMed journals COVID-19 medical journals COVID-19 free journals COVID-19 best journals COVID-19 top journals COVID-19 free medical journals COVID-19 famous journals COVID-19 Google Scholar indexed journals Flail tricuspid valve articles Flail tricuspid valve Research articles Flail tricuspid valve review articles Flail tricuspid valve PubMed articles Flail tricuspid valve PubMed Central articles Flail tricuspid valve 2023 articles Flail tricuspid valve 2024 articles Flail tricuspid valve Scopus articles Flail tricuspid valve impact factor journals Flail tricuspid valve Scopus journals Flail tricuspid valve PubMed journals Flail tricuspid valve medical journals Flail tricuspid valve free journals Flail tricuspid valve best journals Flail tricuspid valve top journals Flail tricuspid valve free medical journals Flail tricuspid valve famous journals Flail tricuspid valve Google Scholar indexed journals Tricuspid regurgitation articles Tricuspid regurgitation Research articles Tricuspid regurgitation review articles Tricuspid regurgitation PubMed articles Tricuspid regurgitation PubMed Central articles Tricuspid regurgitation 2023 articles Tricuspid regurgitation 2024 articles Tricuspid regurgitation Scopus articles Tricuspid regurgitation impact factor journals Tricuspid regurgitation Scopus journals Tricuspid regurgitation PubMed journals Tricuspid regurgitation medical journals Tricuspid regurgitation free journals Tricuspid regurgitation best journals Tricuspid regurgitation top journals Tricuspid regurgitation free medical journals Tricuspid regurgitation famous journals Tricuspid regurgitation Google Scholar indexed journals Neonate articles Neonate Research articles Neonate review articles Neonate PubMed articles Neonate PubMed Central articles Neonate 2023 articles Neonate 2024 articles Neonate Scopus articles Neonate impact factor journals Neonate Scopus journals Neonate PubMed journals Neonate medical journals Neonate free journals Neonate best journals Neonate top journals Neonate free medical journals Neonate famous journals Neonate Google Scholar indexed journals Ultrasound articles Ultrasound Research articles Ultrasound review articles Ultrasound PubMed articles Ultrasound PubMed Central articles Ultrasound 2023 articles Ultrasound 2024 articles Ultrasound Scopus articles Ultrasound impact factor journals Ultrasound Scopus journals Ultrasound PubMed journals Ultrasound medical journals Ultrasound free journals Ultrasound best journals Ultrasound top journals Ultrasound free medical journals Ultrasound famous journals Ultrasound Google Scholar indexed journals treatment articles treatment Research articles treatment review articles treatment PubMed articles treatment PubMed Central articles treatment 2023 articles treatment 2024 articles treatment Scopus articles treatment impact factor journals treatment Scopus journals treatment PubMed journals treatment medical journals treatment free journals treatment best journals treatment top journals treatment free medical journals treatment famous journals treatment Google Scholar indexed journals CT articles CT Research articles CT review articles CT PubMed articles CT PubMed Central articles CT 2023 articles CT 2024 articles CT Scopus articles CT impact factor journals CT Scopus journals CT PubMed journals CT medical journals CT free journals CT best journals CT top journals CT free medical journals CT famous journals CT Google Scholar indexed journals Congenital Heart Disease articles Congenital Heart Disease Research articles Congenital Heart Disease review articles Congenital Heart Disease PubMed articles Congenital Heart Disease PubMed Central articles Congenital Heart Disease 2023 articles Congenital Heart Disease 2024 articles Congenital Heart Disease Scopus articles Congenital Heart Disease impact factor journals Congenital Heart Disease Scopus journals Congenital Heart Disease PubMed journals Congenital Heart Disease medical journals Congenital Heart Disease free journals Congenital Heart Disease best journals Congenital Heart Disease top journals Congenital Heart Disease free medical journals Congenital Heart Disease famous journals Congenital Heart Disease Google Scholar indexed journals Neural Crest articles Neural Crest Research articles Neural Crest review articles Neural Crest PubMed articles Neural Crest PubMed Central articles Neural Crest 2023 articles Neural Crest 2024 articles Neural Crest Scopus articles Neural Crest impact factor journals Neural Crest Scopus journals Neural Crest PubMed journals Neural Crest medical journals Neural Crest free journals Neural Crest best journals Neural Crest top journals Neural Crest free medical journals Neural Crest famous journals Neural Crest Google Scholar indexed journals SARS-Cov-2 articles SARS-Cov-2 Research articles SARS-Cov-2 review articles SARS-Cov-2 PubMed articles SARS-Cov-2 PubMed Central articles SARS-Cov-2 2023 articles SARS-Cov-2 2024 articles SARS-Cov-2 Scopus articles SARS-Cov-2 impact factor journals SARS-Cov-2 Scopus journals SARS-Cov-2 PubMed journals SARS-Cov-2 medical journals SARS-Cov-2 free journals SARS-Cov-2 best journals SARS-Cov-2 top journals SARS-Cov-2 free medical journals SARS-Cov-2 famous journals SARS-Cov-2 Google Scholar indexed journals
Article Details
1. Introduction
Primary tricuspid regurgitation occurs due to a structural abnormality of the tricuspid valve. This abnormality causes inappropriate coaptation of one or multiple leaflets of the valve relative to the other(s). Flail anterior tricuspid valve leaflet is a rare source of primary tricuspid regurgitation typically caused by damage to the papillary muscle.
There are multiple potential causes of papillary muscle injury in the neonate including prenatal and perinatal factors, genetic factors, and concomitant congenital heart disease lesions [1].
2. Case Presentation
A 2.5kg male infant was born at 36 weeks gestation to 38-year-old G6P4 mother whose pregnancy was complicated by gestational diabetes, pre-eclampsia, bipolar disorder, chronic kidney disease, and maternal drug use. Prenatal ultrasounds were unremarkable. APGARs were 8 and 9 at 1 and 5 minutes, respectively. The patient became cyanotic within the first hour of life requiring tracheal intubation. Pulse oximetry saturations remained approximately 70% despite 100% FiO2. A murmur was noted and given the concern for congenital heart disease, prostaglandin E1 (PGE) was started. Echocardiography revealed severe tricuspid valve regurgitation, prompting transfer to a tertiary care center.
Repeat echocardiography on arrival was notable for severe tricuspid regurgitation with prolapse of the anterior leaflet of the tricuspid valve (Figure 1). The septal leaflet was minimally tethered. Right ventricular size and function were normal for age. Apical displacement of the tricuspid valve was only 5.9 mm/m2 (An index of ≥8mm/m2 is supportive of the diagnosis of Ebstein’s anomaly) [2].
There was a small patent ductus arteriosus (PDA) with left to right shunting and a moderate patent foramen ovale with right to left shunting. Initially on day of life 1 there was functional pulmonary atresia presumably due to elevated pulmonary vascular resistance (PVR) as well as left to right ductal flow.
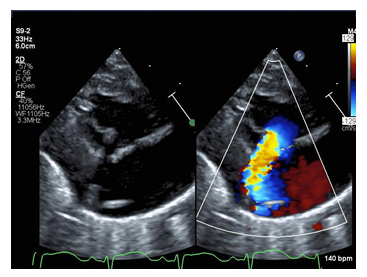
Figure 1: Echocardiogram still image of the parasternal long axis view with color compare of the patient demonstrating flail tricuspid valve leaflet (arrow) and corresponding severe tricuspid regurgitation jet with color.
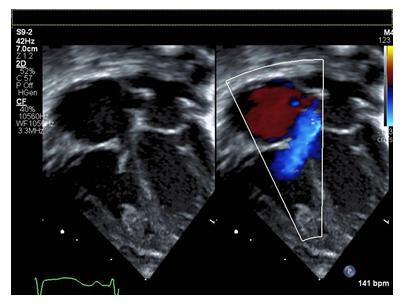
Figure 2: Apical four chamber view of flail tricuspid valve with color compare in systole demonstrating failure of coaptation and wide based regurgitant jet.
Considering the increased PVR and functional pulmonary atresia, inhaled nitric oxide (iNO) was started, mechanical ventilation was titrated, and PGE was continued. This led to a clinical improvement in oxygen saturations to 87%. The management strategy was to give time to allow for the PVR to decrease as normally occurs in all newborns, leading to adequate antegrade flow across the pulmonary valve. At day of life 4, the patient was initially trialed off PGE. He developed prohibitive hypoxemia requiring reinstitution of PGE and the decision was made to give more time for the PVR to decrease further. His second trial off PGE was successful and by 13 days of life the patient was weaned off iNO, PGE, and the ventilator. The decision was made to delay surgical intervention given continued clinical improvement. At 4 weeks of age, he was discharged home in good condition, feeding by mouth, and gaining weight.
He initially did well but at 23 months of age he developed symptoms of fatigue and periorbital edema. He then underwent surgical repair of his abnormal tricuspid valve. Intraoperative inspection showed a thickened anterior leaflet with prolapse. The subvalvar apparatus consisted of a minor chordal attachment but no obvious papillary muscle present. His surgery consisted of a repair of tricuspid valve with a modified annuloplasty, PDA ligation, and closure of patent foramen ovale. There were no complications. Echocardiography showed that his pre-operative tricuspid regurgitation was now mild and without significant tricuspid stenosis; there was normal biventricular systolic function and mild-moderate right ventricular dilation. He was discharged home on post-operative day six.
3. Discussion
Flail anterior tricuspid valve leaflet is a rare, but life-threatening diagnosis made based on imaging in the context of a clinical picture consisting of murmur, cyanosis, and respiratory distress failing to improve despite optimized ventilation and oxygenation. Though typically provoked by a ruptured papillary muscle, this pathology should remain on the differential diagnosis even with intact papillary muscles.
Medical management of a neonate with flail tricuspid valve is challenging. Optimizing multiple sources of pulmonary blood flow is key. Maneuvers to achieve this goal include intubation with supplemental oxygen and ventilator titration to minimize PVR, iNO, milrinone, and a tincture of time. PGE is also useful to keep the PDA open for additional pulmonary blood flow and act as an additional pulmonary vasodilator. Many of the management strategies are physiologically similar to that of neonatal Ebstein’s anomaly [1, 3].
It should be noted, however, that in certain scenarios, PGE could be detrimental and may prevent antegrade flow across the pulmonary valve causing functional pulmonary atresia [4]. When to start and stop PGE can be a difficult decision for the clinician at the bedside and must be individualized to the patient.
4. Conclusions
Understanding this rare anatomy and complex physiology of flail tricuspid valve as well as ways to optimize pulmonary blood flow were key to successful medical management of this case. Importantly, this led to avoidance of surgical intervention in the neonatal period.
Acknowledgements
The authors acknowledge the support from the Department of Pediatrics at University of North Carolina at Chapel Hill.
Financial Support
This research received no specific grant from any funding agency, commercial or not-for-profit sectors.
Conflict of Interest
None.
References
- Loftus PD, Arrington CB, Kaza AK. Neonatal flail tricuspid valve: diagnosis and management. Ann Thorac Surg. 2014;98(3):1098-1101.
- Attenhofer Jost CH, Connolly HM, Dearani JA, Edwards WD, Danielson GK. Ebstein's anomaly. Circulation. 2007;115(2):277-285.
- Baek JS, Yu JJ, Im YM, Yun TJ. Outcomes of neonatal Ebstein's anomaly without right ventricular forward flow [published correction appears in J Thorac Cardiovasc Surg. 2016 Dec;152(6):1644]. J Thorac Cardiovasc Surg. 2016;152(2):516-521.
- Wald RM, Adatia I, Van Arsdell GS, Hornberger LK. Relation of limiting ductal patency to survival in neonatal Ebstein's anomaly. Am J Cardiol. 2005;96(6):851-856.
