Metastatic Renal Cell Carcinoma of Mandible: A Case Report with Review of Literature
Article Information
Prabhanshu S*, Suchitra G, Dipak G, Nivedita K, Maya N
Department of Oral Pathology, Government Dental College & Hospital, Nagpur, India
*Corresponding Author: Prabhanshu Shrivastava, Department of Oral Pathology, Government Dental College & Hospital, Nagpur, India
Received: 09 December 2023; Accepted 18 December 2023; Published: 21 December 2023
Citation: Prabhanshu Shrivastava, Suchitra Gosavi, Dipak Ghatage, Nivedita Kaorey, Maya Nishad. Metastatic Renal Cell Carcinoma of Mandible: A Case Report with Review of Literature. Dental Research and Oral Health. 6 (2023): 95-98.
View / Download Pdf Share at FacebookAbstract
Metastatic lesions to the jaw bones are seldom found, but when observed pose a great diagnostic dilemma to the pathologists. In such cases, immunohistochemical markers and imaging modalities play a significant role in enabling the diagnosis. This case report discusses one such perplexing case of a metastasized renal cell carcinoma to the mandible with undiagnosed primary neoplasm. Discussed here is a case of a 66-year-old male who presented with a proliferative mass involving the left alveolobuccal complex.
Keywords
<p>Metastasis, Renal Cell Carcinoma, Jaw bones</p>
Metastasis articles Metastasis Research articles Metastasis review articles Metastasis PubMed articles Metastasis PubMed Central articles Metastasis 2023 articles Metastasis 2024 articles Metastasis Scopus articles Metastasis impact factor journals Metastasis Scopus journals Metastasis PubMed journals Metastasis medical journals Metastasis free journals Metastasis best journals Metastasis top journals Metastasis free medical journals Metastasis famous journals Metastasis Google Scholar indexed journals Renal Cell Carcinoma articles Renal Cell Carcinoma Research articles Renal Cell Carcinoma review articles Renal Cell Carcinoma PubMed articles Renal Cell Carcinoma PubMed Central articles Renal Cell Carcinoma 2023 articles Renal Cell Carcinoma 2024 articles Renal Cell Carcinoma Scopus articles Renal Cell Carcinoma impact factor journals Renal Cell Carcinoma Scopus journals Renal Cell Carcinoma PubMed journals Renal Cell Carcinoma medical journals Renal Cell Carcinoma free journals Renal Cell Carcinoma best journals Renal Cell Carcinoma top journals Renal Cell Carcinoma free medical journals Renal Cell Carcinoma famous journals Renal Cell Carcinoma Google Scholar indexed journals Jaw bones articles Jaw bones Research articles Jaw bones review articles Jaw bones PubMed articles Jaw bones PubMed Central articles Jaw bones 2023 articles Jaw bones 2024 articles Jaw bones Scopus articles Jaw bones impact factor journals Jaw bones Scopus journals Jaw bones PubMed journals Jaw bones medical journals Jaw bones free journals Jaw bones best journals Jaw bones top journals Jaw bones free medical journals Jaw bones famous journals Jaw bones Google Scholar indexed journals mandible articles mandible Research articles mandible review articles mandible PubMed articles mandible PubMed Central articles mandible 2023 articles mandible 2024 articles mandible Scopus articles mandible impact factor journals mandible Scopus journals mandible PubMed journals mandible medical journals mandible free journals mandible best journals mandible top journals mandible free medical journals mandible famous journals mandible Google Scholar indexed journals Oral Surgery articles Oral Surgery Research articles Oral Surgery review articles Oral Surgery PubMed articles Oral Surgery PubMed Central articles Oral Surgery 2023 articles Oral Surgery 2024 articles Oral Surgery Scopus articles Oral Surgery impact factor journals Oral Surgery Scopus journals Oral Surgery PubMed journals Oral Surgery medical journals Oral Surgery free journals Oral Surgery best journals Oral Surgery top journals Oral Surgery free medical journals Oral Surgery famous journals Oral Surgery Google Scholar indexed journals mandibular canal articles mandibular canal Research articles mandibular canal review articles mandibular canal PubMed articles mandibular canal PubMed Central articles mandibular canal 2023 articles mandibular canal 2024 articles mandibular canal Scopus articles mandibular canal impact factor journals mandibular canal Scopus journals mandibular canal PubMed journals mandibular canal medical journals mandibular canal free journals mandibular canal best journals mandibular canal top journals mandibular canal free medical journals mandibular canal famous journals mandibular canal Google Scholar indexed journals alveolobuccal complex articles alveolobuccal complex Research articles alveolobuccal complex review articles alveolobuccal complex PubMed articles alveolobuccal complex PubMed Central articles alveolobuccal complex 2023 articles alveolobuccal complex 2024 articles alveolobuccal complex Scopus articles alveolobuccal complex impact factor journals alveolobuccal complex Scopus journals alveolobuccal complex PubMed journals alveolobuccal complex medical journals alveolobuccal complex free journals alveolobuccal complex best journals alveolobuccal complex top journals alveolobuccal complex free medical journals alveolobuccal complex famous journals alveolobuccal complex Google Scholar indexed journals
Article Details
Introduction
Metastasis is a highly complex biological process that begins as the tumor cells detach from the primary tumor, spread into the distant tissues and/or organs, and invade through the lymphovascular structures followed by their survival in the circulation and subsequent proliferation in competent organs leading to colonization [1]. Metastatic tumors in the oral cavity are very rare, accounting for only 1%-3% of all malignant oral neoplasms [2]. The most common sites of primary malignancies are Breast, lung and kidney. In the orofacial region, the mandible is the most frequently affected site for metastases, particularly in the molar region. The peculiarity of these metastatic lesions is that they often show non aggressive clinical findings that frequently favours a reactive or benign lesions or odontogenic lesion [3]. The diagnosis of a metastatic lesion in the oral region is challenging, both to the clinician and to the pathologist. The clinician must recognize the possibility that a lesion may represent a metastasis, and the pathologist must determine the site of tumor origin [4]. Diagnosis of these metastatic tumours often require adjuvant investigations and advanced diagnostic procedures. This article discusses one such case with a perplexing clinical and histological picture which was diagnosed with the aid of immunohistochemical markers and advanced radiographic modalities.
Case Report
A 66 years old male patient reported to our OPD with chief complaint of pain and swelling in the lower left back region of the jaw since last 6 months. There was no history of bleeding or pus discharge from the site. He reported a bidi smoking habit 5 to 6 times a day for 20 years. Extra oral Examination revealed pain and swelling present on the left side of the mandible.
On palpation, a solitary, fixed and tender submandibular lymph node was noted. Intra orally, an ulceroproliferative growth was visible involving the Left alveolobuccal complex. Antero-posteriorly, the lesion extended from the mesial aspect of the mandibular left canine to the third molar on the same side. Vertical extension was from the crest of the alveolar bone and involving the buccal vestibule. The size of the lesion was approximately 4cm x 1cm. On inspection, the growth was pale pink in colour, had everted margins and indurated base. The patient had lost all his teeth on the affected side (Figure 1). Radiography revealed a large lytic lesion on left mandible presenting as an ill-defined bone resorption extending from symphyseal region till third molar and supero-inferiorly, from crest of alveolus till mandibular canal (Figure 2). CECT findings included a 25 × 15 × 10 mm sized heterogeneously enhancing lesion involving left lower alveolus causing expansion and lytic changes in bone. Medially the lesion was abutting floor of mouth and extension in the left sub lingual space. Laterally the lesion was involving lower GBS along its anterior aspect. Left mandibular canal appeared to be involved by the lesion. Based on the history and the clinical findings, a provisional diagnosis of squamous cell carcinoma of left alveolobuccal complex (ABC) was given and the patient was referred to the department of Oral Surgery for biopsy.
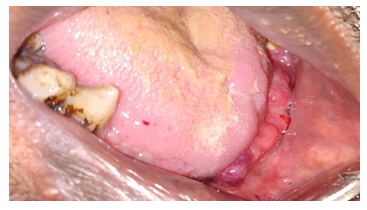
Figure 1: Clinical image of the patient showing ulceroproliferative growth on left side of mandible.
Histopathology revealed multiple bits of tissue devoid of surface epithelium. Lesional tissue showed extensive proliferation of tumor cell population in the form of clear cells. These tumor cells were large and arranged in the form of small islands, cords and dissociated cells with hyperchromatic nuclei varying from round to pleomorphic in shape located central or eccentrically. Intervening connective tissue stroma was scanty and comprised of haphazardly arranged collagen fibers and chronic inflammatory cells (Figure 3). Part of the section showed numerous proliferative strands of tumor cells with abundant cytoplasm and large round to oval pleomorphic hyperchromatic nuclei in a stroma of hyalinized collagen fibers, fibroblasts and inflammatory cells. A differential diagnosis of Oral squamous cell carcinoma with clear cell variant, mucoepidermoid carcinoma, Clear cell odontogenic lesion and metastatic lesion was made. Immunohistochemical assay comprising of CK 14,19 and 20, EMA and vimentin were performed on the tissue sections to rule out tumours of salivary and odontogenic origin. Tumor cells stained positive for EMA and Vimentin (Figures 4 and 5) both of which are strong indicators of metastasis. The patient was further advised to undergo Chest X-ray (CXR) and Ultrasonography (USG)of the abdomen. CXR revealed no active pleuro-parenchymal lesion. USG abdomen reported well-defined round to oval heterogeneously isoechoic to hyperechoic lesion of size 3.4 X 2.8 cm in relation to upper pole of right kidney. Correlating the findings and the diagnostic results, we arrived at a final diagnosis of metastatic renal cell carcinoma. The patient provided the informed consent for publication of this case.
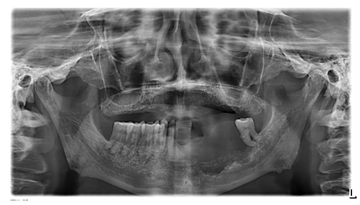
Figure 2: Radiograph of the patient showing large lytic lesion on left mandible presenting as an ill-defined bone resorption
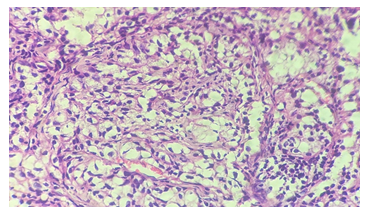
Figure 3: H & E-stained section (10X) showing proliferation of tumor cell population in the form of clear cells
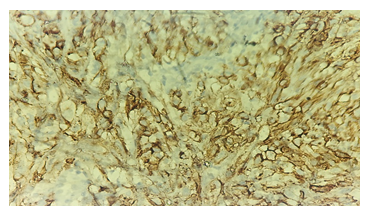
Figure 4: Immunohistochemistry showing EMA positivity
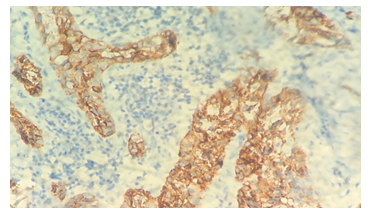
Figure 5: Immunohistochemistry showing Vimentin positivity
Discussion and Review of literature
Renal cell carcinoma (RCC) accounts for 3% of adult malignancies and 90% to 95% of kidney tumors [4]. The rate of metastasis in RCC is reported to be around 30% [5,6]. RCC has been known metastasize to distant organs, including bilateral adrenal glands, skin, pancreas and spleen, gastric or duodenal systems, skeleton, pancreas and bladder. However, reports of metastases to the head and neck region have been extremely rare [7]. The incidence of oral metastases and their origins may vary according to the study population. Boles R and Cerny J, reported the incidence of metastasis from RCC to head and neck to be 15% [8]. According to Hirshberg et al. the kidney is the third most common primary site for oral metastases [9]. As was seen in our case, jaw lesions were the first reported manifestation of an undiagnosed primary malignancy. The common clinical features of this oral metastatic disease include submucosal or jaw mass, jaw or oral pain, lip numbness, gingival swelling, or even an extensive bilobed mass. Bleeding, tooth mobility, trismus, paraesthesia, and paralysis may be accompanying findings in some cases. Radiography may reveal extensive lytic lesions of the jaw bones with ill defined, irregular margins. Further imaging modalities like USG abdomen and PET scan etc. facilitates the diagnosis of the underlying primary or further metastasized masses [10]. According to the literature review by Naoto Nishii et al, a distinct male predominance was observed with the male to female ratio being 3:1 with mean age of 61.4 years. The tongue was involved in renal metastases in most patients (39%) followed by mandible(19.1%). Out of the 153 cases reviewed, they found that 58 cases were diagnosed before the diagnosis of primary RCC and 93 after the primary malignancy was diagnosed [11]. Our patient was a 66 year old male with mandibular involvement wherein the oral lesion was the first clinical manifestation of the primary neoplasm. Clinically the lesion mimicked a squamous cell carcinoma. Histopathology of clear cell renal cell carcinoma depicts significant vascularisation, cells with pleomorphic and hyperchromatic nuclei and cells with a clear cytoplasm [12,13]. As was observed in our case, if the primary neoplasia was undiagnosed immunohistochemical assay was mandatory to establish the diagnosis. The main markers in the panel include PAX8, presenting positivity in approximately 90% of cases, and CD10 with positivity in 98%. Another classic marker is AE1/AE2, positive in 88% of the cases. The reported case was negative for this marker, which can be explained by its poorly differentiated cells [14]. In addition to these markers, another that can be used is CK7, an important marker for epithelial tumours and with high expression in renal tumors [15]. Management primarily includes radiotherapy and chemotherapy- pazopanib, sunitinib and sorafenib. These drugs are angiogenic inhibitors which target VEGF receptors resulting in decreased tumour volume [16,17]. Surgical management is opted after the tumor size is reduced. Literature review of similar cases revealed the longest survival time to be 11 months and smaller survival time to be around six months [18,19].
Conclusion
Owing to their rarity, diagnosis of oral metastatic diseases is a challenge pathologists encounter. In these cases, stress lies upon the judicious and optimal utilisation of the advanced diagnostic techniques along with histopathology to arrive at an accurate diagnosis.
References
- Franziska VZ, Georg K, Wolfgang M. Initial steps of metastasis: Cell invasion and endothelial transmigration.Mutat Res 728 (2011): 23-34.
- Kumar GS, Manjunatha BS. Metastatic tumors to the jaws and oral cavity. J Oral Maxillofac Pathol 17 (2013): 71-75.
- Nishanth G, Rajesh E, Aravindha BN, et al. Metastatic tumors of jaw - A Review. European Journal of Molecular & Clinical Medicine 7 (2020): 708-711.
- Hafez KS, Fergany AF, Novick AC. Nephron sparing surgery for localized renal cell carcinoma: Impact of tumor size on patient survival, tumor recurrence and TNM staging. J Urol 162 (1999): 1930-1933.
- Ather MH, Masood N, Siddiqui T. Current management of advanced and metastatic renal cell carcinoma. Urol J 7 (2010): 1-9.
- Dimanovski J, Popovic A. Metastatic renal cell carcinoma. Acta Clin Croat 39 (2000): 171-174.
- Louqiang Z, Hongbin Y, Xuebin Z. Metastatic renal cell carcinoma to the jaws: report of cases. World Journal of Surgical Oncology 12 (2014): 204-208.
- Boles R, Cerny J. Head and neck metastases from renal carcinomas. Mich Med 70 (1971): 616-618.
- Hirshberg A, Shnaiderman-Shapiro A, Kaplan I, et al. Metastatic tumours to the oral cavity - pathogenesis and analysis of 673 cases. Oral Oncology 44 (2008): 743-752.
- Rafael Netto. Metastasis of Renal Cell Carcinoma Causing Significant Facial Asymmetry. Case Reports in Surgery 5 (2019): 6840873.
- Naoto N. Renal cell carcinoma metastasis to the maxillary bone successfully treated with surgery after vascular embolization: a case report.Journal of Medical Case Reports 14 (.2020): 193.
- Jatti D, Puri G, Aravinda K, et al. An atypical metastasis of renal clear cell carcinoma to the upper lip: a case report. J Oral Maxillofac Surg 73 (2015): 371.e1-e6.
- Lee C, Park JW, Suh J, et al. Histologic variations and immunohistochemical features of metastatic clear cell renal cell carcinoma. Korean J Pathol 47 (2013): 426-432.
- Ma F, Dai L, Wang Z, et al. Correction: evaluating prognosis by CK7 differentiating renal cell carcinomas from oncocytomas can be used as a promising tool for optimizing diagnosis strategies. Oncotarget 9 (2018): 29285.
- Altuntas O, Petekkaya I, Süslü N, et al. Renal cell carcinoma metastatic to the tongue: a case report and review of the literature. J Oral Maxillofac Surg 73 (2015): 1227-1230.
- Costantino C, Thomas GV, Ryan C, et al. Metastatic renal cell carcinoma without evidence of a renal primary. Int Urol Nephrol 48 (2015): 73-77.
- Rini BI, Plimack ER, Takagi T, et al. A phase II study of pazopanib in patients with localized renal cell carcinoma to optimize preservation of renal parenchyma. J Urol 194 (2015): 297-303.
- Derakhshan S, Rahrotaban S, Mahdavi N, et al. Metastatic renal cell carcinoma presenting as maxillary lesion: report of two rare cases. J Oral Maxillofac Pathol 22 (2018): S39-43.
- Costantino C, Thomas GV, Ryan C, et al. Metastatic renal cell carcinoma without evidence of a renal primary. Curr Oncol 21 (2014): e521-e524.
