Madelung Disease-a Challenge in Clinical Practice
Article Information
Micu Alexandru1,2*, Micu C Mihaela3
1Faculty of Medicine, "Iuliu Hatieganu" University of Medicine and Pharmacy Cluj-Napoca, Romania
2Medical Faculty Mannheim, Heidelberg University, Germany
3Rheumatology Division, Rehabilitation Clinical Hospital Cluj-Napoca, Romania
*Corresponding Author: Micu Alexandru, Faculty of Medicine, "Iuliu Hatieganu" University of Medicine and Pharmacy Cluj-Napoca, Romania
Received: 16 January 2022; Accepted: 27 January 2022; Published: 29 January 2022
Citation:
Micu Alexandru, Micu C Mihaela. Madelung Disease-a Challenge in Clinical Practice. Fortune Journal of Rheumatology 4 (2022): 01-11
View / Download Pdf Share at FacebookAbstract
Multiple Symmetric Lipomatosis (MSL) or Madelung’s disease is a rare disorder of the lipid metabolism characterized by symmetric growth of sometimes massive unencapsulated fatty deposits (lipomas) in anatomical regions ranging from typical sites such as neck, occipital area, upper arms, face, abdomen and back to the more unusual involvement of the scrotum and the tongue. In this article we present the diagnostic and therapeutic challenges in a patient developing an acute oligoarticular attack of gout generating important functional incapacity, alcoholic liver disease potentially evolving to liver cirrhosis, heart and kidney failure, bilateral parotid gland region swelling and Madelung disease. The recognition and screening for potential complications related to the Madelung disease is of great practical importance because treatment priorities should be stratified accordingly.
Keywords
<p>Gout; lipomatosis; Lipoma; Madelung disease; Multiple symmetric</p>
Gout articles; lipomatosis articles; Lipoma articles; Madelung disease articles; Multiple symmetric articles
Gout articles Gout Research articles Gout review articles Gout PubMed articles Gout PubMed Central articles Gout 2023 articles Gout 2024 articles Gout Scopus articles Gout impact factor journals Gout Scopus journals Gout PubMed journals Gout medical journals Gout free journals Gout best journals Gout top journals Gout free medical journals Gout famous journals Gout Google Scholar indexed journals lipomatosis articles lipomatosis Research articles lipomatosis review articles lipomatosis PubMed articles lipomatosis PubMed Central articles lipomatosis 2023 articles lipomatosis 2024 articles lipomatosis Scopus articles lipomatosis impact factor journals lipomatosis Scopus journals lipomatosis PubMed journals lipomatosis medical journals lipomatosis free journals lipomatosis best journals lipomatosis top journals lipomatosis free medical journals lipomatosis famous journals lipomatosis Google Scholar indexed journals Lipoma articles Lipoma Research articles Lipoma review articles Lipoma PubMed articles Lipoma PubMed Central articles Lipoma 2023 articles Lipoma 2024 articles Lipoma Scopus articles Lipoma impact factor journals Lipoma Scopus journals Lipoma PubMed journals Lipoma medical journals Lipoma free journals Lipoma best journals Lipoma top journals Lipoma free medical journals Lipoma famous journals Lipoma Google Scholar indexed journals Madelung disease articles Madelung disease Research articles Madelung disease review articles Madelung disease PubMed articles Madelung disease PubMed Central articles Madelung disease 2023 articles Madelung disease 2024 articles Madelung disease Scopus articles Madelung disease impact factor journals Madelung disease Scopus journals Madelung disease PubMed journals Madelung disease medical journals Madelung disease free journals Madelung disease best journals Madelung disease top journals Madelung disease free medical journals Madelung disease famous journals Madelung disease Google Scholar indexed journals Multiple symmetric articles Multiple symmetric Research articles Multiple symmetric review articles Multiple symmetric PubMed articles Multiple symmetric PubMed Central articles Multiple symmetric 2023 articles Multiple symmetric 2024 articles Multiple symmetric Scopus articles Multiple symmetric impact factor journals Multiple symmetric Scopus journals Multiple symmetric PubMed journals Multiple symmetric medical journals Multiple symmetric free journals Multiple symmetric best journals Multiple symmetric top journals Multiple symmetric free medical journals Multiple symmetric famous journals Multiple symmetric Google Scholar indexed journals epidemiology articles epidemiology Research articles epidemiology review articles epidemiology PubMed articles epidemiology PubMed Central articles epidemiology 2023 articles epidemiology 2024 articles epidemiology Scopus articles epidemiology impact factor journals epidemiology Scopus journals epidemiology PubMed journals epidemiology medical journals epidemiology free journals epidemiology best journals epidemiology top journals epidemiology free medical journals epidemiology famous journals epidemiology Google Scholar indexed journals oligoarticular attack articles oligoarticular attack Research articles oligoarticular attack review articles oligoarticular attack PubMed articles oligoarticular attack PubMed Central articles oligoarticular attack 2023 articles oligoarticular attack 2024 articles oligoarticular attack Scopus articles oligoarticular attack impact factor journals oligoarticular attack Scopus journals oligoarticular attack PubMed journals oligoarticular attack medical journals oligoarticular attack free journals oligoarticular attack best journals oligoarticular attack top journals oligoarticular attack free medical journals oligoarticular attack famous journals oligoarticular attack Google Scholar indexed journals therapeutic challenges articles therapeutic challenges Research articles therapeutic challenges review articles therapeutic challenges PubMed articles therapeutic challenges PubMed Central articles therapeutic challenges 2023 articles therapeutic challenges 2024 articles therapeutic challenges Scopus articles therapeutic challenges impact factor journals therapeutic challenges Scopus journals therapeutic challenges PubMed journals therapeutic challenges medical journals therapeutic challenges free journals therapeutic challenges best journals therapeutic challenges top journals therapeutic challenges free medical journals therapeutic challenges famous journals therapeutic challenges Google Scholar indexed journals mitochondrial DNA articles mitochondrial DNA Research articles mitochondrial DNA review articles mitochondrial DNA PubMed articles mitochondrial DNA PubMed Central articles mitochondrial DNA 2023 articles mitochondrial DNA 2024 articles mitochondrial DNA Scopus articles mitochondrial DNA impact factor journals mitochondrial DNA Scopus journals mitochondrial DNA PubMed journals mitochondrial DNA medical journals mitochondrial DNA free journals mitochondrial DNA best journals mitochondrial DNA top journals mitochondrial DNA free medical journals mitochondrial DNA famous journals mitochondrial DNA Google Scholar indexed journals bilateral knee articles bilateral knee Research articles bilateral knee review articles bilateral knee PubMed articles bilateral knee PubMed Central articles bilateral knee 2023 articles bilateral knee 2024 articles bilateral knee Scopus articles bilateral knee impact factor journals bilateral knee Scopus journals bilateral knee PubMed journals bilateral knee medical journals bilateral knee free journals bilateral knee best journals bilateral knee top journals bilateral knee free medical journals bilateral knee famous journals bilateral knee Google Scholar indexed journals
Article Details
1. Introduction
Multiple Symmetric Lipomatosis (MSL) or Made lung’s disease is a rare disorder of the lipid metabolism characterized by symmetric growth of sometimes massive unencapsulated fatty deposits (lipomas) in anatomical regions ranging from typical sites such as neck, occipital area, upper arms, face, abdomen and back to the more unusual involvement of the scrotum and the tongue [1, 2]. The epidemiology of the disease is linked to middle aged male patients of mostly of European Mediterranean origin. The etiology of the disease is still unknown, but possible causes point to mitochondrial DNA (mtDNA) mutations, abnormal adipose tissue and the patient’s positive history of chronic alcohol abuse [1, 3]. The consequences of the fatty accumulations are usually cosmetic, but dyspnea, dysphagia and difficulty in head movement have also been described in cases where lipomas of the neck region were more severe. An MSL classification has been proposed: Type 1 - involving symmetric, well circumscribed fat masses in the neck, proximal upper limbs and superior thorax; Type 2 - involving diffusely distributed subcutaneous fat layers with the appearance of simple obesity and Type 3 - fat deposits mainly located in the pelvic region resembling a gynecoid appearance [4, 5].
2. Case Report
A 77-year-old male patient presented with a history of bilateral knee and wrist joint pain, prolonged morning stiffness and walking incapacity, chronic lumbar pain, episodic constrictive chest pain correlated with moderate physical effort, fatigue and weight loss of 5 kg in the last 6 months. He identified himself as a nonsmoker but admitted a continuous moderate daily alcohol intake in the last 15 years. He denied experiencing xerophthalmia, xerostomia or dysphagia. On admission, clinical examination revealed a BMI of 30.6 kg/m2, facial venectasias and dyspnea on exertion. Painless bilateral and symmetric soft tissue tumors were present in the preauricular and nuchal region. Similar soft tissue tumors were identified at the levels of the chest and of the superior abdominal wall (Figure 1). Further physical examination revealed a respiratory frequency of 18/min, O2 saturation of 95%, persistent atrial fibrillation, NYHA stage III heart failure, arterial hypertension stage IIc, bilateral coarse crackles in the lower pulmonary lobes, swollen knees and wrists, both with limited flexion and extension movements, walking incapacity both due to the bilateral knee involvement and intermittent claudication (stage IIa Leriche Fontaine). Complementary lower limbs and heart ultrasound (US) evaluation identified lower extremity arterial disease and a low left ventricular ejection fraction <40%. US evaluation of the visible soft tissue tumors in the neck, upper limbs and trunk regions revealed the presence of unencapsulated lipomas with fibrous septa. No Doppler signal was detectable.
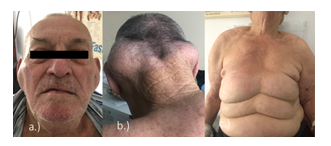
Figure 1abc: Bilateral, symmetric soft tissue masses in the preauricular and nuchal region, upper arms, chest and upper abdominal wall.
Complete blood count (CBC) and biochemistry was performed. The lab analyses identified the presence of mildly elevated CRP (0.8mg/dl), uric acid (8.7 mg/dl) and MEV (103.8 fL), no anemia, and decreased platelet number (131x103/µL) as well as creatinine clearance (71.38 ml/min). The rest of the CBC and biochemistry analysis were normal. Antinuclear antibodies panel, viral markers for B and C hepatitis were negative. The prostate-specific antigen, albumin, cholesterol and triglycerides were all in normal range. Musculoskeletal US evaluation of the hands identified mild bilateral radiocarpal synovitis and proximal as well as distal interphalangeal joint osteophytes. At knee level, bilateral joint effusion with crystal aggregates was identified, the double contour sign (highly specific for uric deposition) was present at the level of the hyaline femoral cartilage and hyperechoic cloudy aggregates were detected at the level of the patellar tendon bilaterally (Figure 2). Synovial fluid analysis identified the presence of needle-shaped intracellular and extracellular crystals, characteristic for monosodium urate crystals.
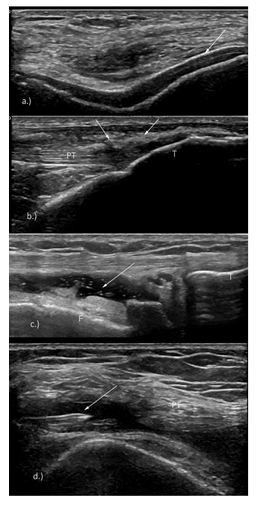
Figure 2a: Double contour sign at the level of the hyaline femoral cartilage; b) Arrows -Hyperechoic cloudy aggregates were detected at the level of the patellar tendon (PT) bilaterally, T – tibia cortical bone; c) Lateral parapatellar recess distended by a hypoechoic mass (arrow) containing hyperechoic dots, F – femoral bony cortex, T – tibia bony cortex; d) Transverse scanning of the suprapatellar bursa and ultrasound guided aspiration of the knee effusion (arrow – tip of the needle, PT – patellar tendon). The salivary glands US evaluation showed a normal volume and echotexture of the parotid, submandibular and sublingual glands. In the proximity of the superior pole of both parotid glands, unencapsulated subcutaneous fat tissue masses were detected (Figure 3).
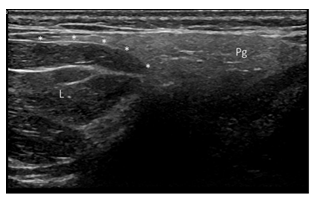
Figure 3: Normal Parotid Gland (Pg) and the unencapsulated subcutaneous fat tissue mass localized adjacent to the superior parotid pole (L).*- showing the interface between the fatty mass and parotid gland.
Apart from a diffuse intima thickening of the carotid arteries, no jugular or thyroid gland abnormalities were present at US evaluation. Thoraco-abdominal CT was performed in order to assess potential compressive intrathoracic and intraabdominal lipomatous masses or underlying neoplasias. In our patient, CT evaluation was negative for both. In addition, hepato-splenomegaly was detected. A more complex liver status evaluation was initiated in the gastroenterology department, given also the positive history of chronic alcohol consumption. Dual Energy X-ray Absorptiometry (DEXA) examination confirmed the diagnosis of vertebral and hip osteoporosis.
According to our evaluation, the patient’s diagnoses were the following: uric microcrystal arthropathy affecting both articular and periarticular structures, alcoholic liver disease potentially evolving to liver cirrhosis, heart and kidney failure and Madelung disease. The first phase treatment strategy was US guided corticosteroid knee joint injection with immediate recovery of the walking capacity, colchicine and allopurinol prescription for the long-term uric acid level control. The first step in the treatment approach had a major positive functional outcome and was possible without any additional risk due to prior exclusion of life threatening MSL complications (e.g. obstructive respiratory disorder caused by a combination of lipoma compression and/or pharyngeal adipose tissue infiltration). In addition, anticoagulants, digoxin, diuretics, betablockers and statins were prescribed for optimizing the cardiovascular and renal parameters and controlling the associated risk factors. Furthermore, the patient was transferred to the gastroenterology department for supplementary investigations.
2.1 Discussion and review of the literature
Herein we present the diagnostic and therapeutic challenges in a patient presenting with an acute attack of gout in the knees generating walking incapacity, alcoholic liver disease, bilateral parotid gland region swelling and Madelung disease. The recognition and screening for potential complications related to the Madelung disease is of great practical importance because treatment priorities should be stratified accordingly.
2.2 Epidemiology and classification
As of 2007, more than 300 cases of MSL have been reported worldwide, with a more consistent prevalence in Mediterranean countries such as Italy, Spain and France, but reports of patients range as far as far as southeast Asia [6-8]. Patients are usually male (male to female ratio 15:1) with a prior history of alcohol abuse [9]. Three main subgroups of MSL with distinct clinical appearances upon physical examination have been proposed, although interpolated physical aspects of the subtypes can occur. MSL Type 1 involves symmetric, well circumscribed fat masses in the neck, nuchal area of the head, shoulders, proximal part of the upper limbs and superior thorax, giving the appearance of a „Michelin Man“; MSL Type 2 presents as diffusely distributed subcutaneous fat layers that are mostly unelevated and extend on a greater surface of the body, resembling simple obesity; MSL type 3 was introduced at a later date and characterizes the gynecoid distribution of the fatty tissue in the pelvic region and gynecomastia [4, 5]. The presence of the lipomas in the neck region can resemble a collar-Madelung’s collar-a symmetrical structure with rather cosmetic consequences for the patient.
2.3 Etiology
Several theories have been postulated regarding the genesis of MSL. One of the particular findings in Madelung adipose tissue is the presence of brown fat. Brown fat is responsible for thermogenesis in babies without the involvement of chills. It becomes increasingly atrophic with age, to the point where only remnants are identifiable in healthy adults. The thermogenesis is physiologically mediated through the following mechanism: β3 receptors found in brown fat are highly responsive upon stimulation via Norepinephrine (NA), increasing the intracellular concentrations of Adenylate Cyclase (AC) via Gs proteins. Uncoupling protein 1 (UCP-1) uncouples the AC induced downhill cascade of Adenosine Triphosphate (ATP) production, favoring heat production instead [10, 11]. It is currently believed that a functionally defective brown fat tissue is present in MSL lipomas. NA responsiveness is decreased in MSL brown adipose tissue and NA fails to generate effective levels of Inducible Nitric Oxide Synthase (iNOS), responsible for anti-proliferative effects. The decreased adrenergic innervation of Madelung adipose tissue causes increased local storage of triglycerides, changes in the local lipid metabolism and hyperplasia of the adipose tissue [12, 13]. It should be noted that not all Madelung lipomas present brown fat components, so other possible mechanism of the disease should also be considered [14]. A secondary effect is that of alcohol, which not only decreases β receptor levels, further perturbing the adrenergic system, but also has a direct adipogenic effect. Several studies have linked the pathogenesis of the disease to mutations in mitochondrial DNA (mtDNA) (m.8344A>G and m.8363G>A). According to one study, up to 16% of the patients presented mtDNA mutations [15]. The m.8344A>G mutation is also present in MERRF syndrome, pointing to a possible common ground of the two disease entities [16, 17]. These mutations have been inconsistently reported in other studies and cases of MSL with wild type status of the mtDNA in MSL adipocytes have also been reported [14, 18].
2.4 Morphology and functionality of the adipocytes
Probe excision biopsies from patients with MSL have not only shown differences in the architecture of the fatty tissue but also in the individual cells. Compared to normal adipose tissue, the Madelung adipose tissue presents smaller adipocytes, more prominent septa and more atypical nuclei [18]. Further studies have identified the resemblance of the Madelung adipose tissue to that of the brown fat, through the presence of brown adipocyte markers such as UCP-1. It should be noted that UCP-1 and the brown fat associated with it is not present in all subjects and that it decreases with patient age and grade of obesity [14]. The growth kinetic of the Madelung adipocytes has shown changes in in vitro studies. Compared to normal adipocytes, these cells have demonstrated a higher growth potential, reaching a steady phase at higher cell concentrations. Interestingly, the secretome of the Madelung adipocytes can influence the phenotype of normal adjacent adipocytes and induce a switch towards an aberrant phenotype, thus decreasing the population doubling time and modifying the surface markers in previous healthy adipocytes. This points to an invasive character of the disease that also influences adjacent healthy tissue regions [18]. Surface phenotype of the Madelung adipocytes differs from the normal adipocytes’ one through reduced levels of CD90, CD9, CD73 and CD49f and increased levels of CD49a. This could lead to differences in adipogenic differentiation (CD90), proliferation and ATP metabolism (CD73). Lower CD9 levels are also linked to poorer cell adhesion and increased protection from cellular senescence [18, 19].
2.5 Diagnosis and clinical presentation
Diagnosis is usually established through physical examination. The hallmark of the disease is the presence of multiple sometimes symmetrical lipomas of sizes ranging from 1 to 20 cm in different body regions. Although the lipomas are easily spotted by physicians in regions with reduced physiological adipose tissue such as the nuchal area or the neck region, MSL type 2 or type 3 could be easily misinterpreted as simple obesity or gynecomastia [9, 20]. Patient history and temporal succession of events described by the patient are key in assessing the correct diagnosis. MSL usually has an abrupt progression at disease onset, afterwards becoming stationary for long periods of time and is correlated with chronic alcohol abuse and weight gain without apparent changes in patient diet [21, 22]. Disease progression is usually deemed slow and painless, patients usually complaining about difficulty upon actions requiring neck movement. Compression of adjacent neck structures, such as the carotid arteries and jugular veins, cervical plexus, recurrent laryngeal nerves, trachea and esophagus and infiltration further down into the mediastinum can lead to more severe symptoms of dysphagia, dyspnea, dysphonia, venous stasis and paresthesias. Several case reports have also highlighted the involvement of the tongue in MSL, depicting macroglossia as an additional property of the disease [23, 24]. Other more unusual particularities of this disease are the involvement of the scrotum or of the facial nerve with consequent paresthesias and paresis in the face region [1, 25]. Other disease clusters, such as hyperuricemia, hypertriglyceridemia, hypercholesterolemia, macrocytic anemia or different stages of hepatopathy that might be present are usually linked to chronic ethanol abuse and are not necessarily a consequence of the primary Madelung disease itself [21, 26].
The presence of polyneuropathy is currently believed to be of mixed genesis, both due to chronic alcohol toxicity and direct effects of the disease itself. CT and MRI are both excellent imaging techniques in helping establish the severity of lipoma infiltrations and possible compression of adjacent structures. CT and MRI examinations should be used in order to establish the possible presence and extent of mediastinal or abdominal lipomas that are inaccessible to US examination and as a preoperative management tool in symptomatic cases of MSL. US evaluation is useful in diagnosing superficial adipose masses, such as those in the face or neck region. US is a quick diagnostic method, efficient in time sensitive circumstances such as ambulatory patient presentation but lacks the accuracy of CT and MRI in diagnosing deeper located lipomas and their infiltration [27, 28].
2.6 Differential diagnosis
The pseudoathletic appearance of type 1 MSL could sometimes resemble cushingoid syndromes, whereas type 2 MSL is typically misdiagnosed as simple obesity [29].The presence of gynecomastia and/or gynecoid adipose tissue distribution requires further exclusion of alternative etiologies such as estrogen excess of tumoral or nontumoral cause, decreased testosterone or androgen resistance (primary causes - Klinefelter syndrome, secondary causes - Kallmann syndrome), decreased testosterone concentrations via systemic diseases (end stage renal disease, thyrotoxicosis, liver cirrhosis), drugs (spironolactone, efavirenz, ethanol, busulfan, vincristine, ketoconazole) [30]. The presence of lipomas in the neck region requires exclusion of other benign or malignant regional masses: cervical lymphadenopathy (reactive or malignant), goiter, carotid artery aneurysms, oropharyngeal squamous cell carcinoma, neurofibromas, salivary gland (infiltrations, neoplasms, cysts), congenital disorders such as thyroglossal duct cysts, and branchial cleft anomalies. Multiple lipomas and lipid dystrophies are also consequences of HAART therapy in HIV positive patients. These modifications are attributed to the effect of protease inhibitors [31-34]. In our case, salivary gland and neck region US examination performed immediately at bedside was extremely helpful for an accurate local evaluation and immediate exclusion of several differential diagnoses.
2.7 Associations with alcohol intake and other metabolic disorders
Alcohol consumption has been established to worsen the symptoms of MSL. The vast majority of the case reports published in the literature include patients with a positive history of alcohol abuse. The cessation of alcohol consumption does indeed slow the growth of lipomas but does not reduce the size of the already developed adipose masses [35]. The presence of MSL in patients denying alcohol consumption or even children speaks against alcohol as a solitary cause of the disease. Alcohol should rather be categorized as a catalyzer in the development of the lipomas. Other complications of chronic alcohol abuse in patients with MSL include liver disease, hyperuricemia, alcoholic pancreatitis and macrocytic anemia [36]. Polyneuropathy is present in several cases described in the literature. Axonal predominant neuropathy as well as autonomic dysfunctions are also common neurological complications of chronic alcohol consumption, so alcohol should be regarded as a cofactor in the disease pathogenesis. Evidence pleading for a mixed pathogenesis is the presence of neuropathy in children suffering from MSL and adults denying prior alcohol abuse. Histological features of MSL neuropathy also differ from those of ethanol induced neuropathy (progressive axonal atrophy in MSL vs. wallerian degeneration of the axon and reduced myelination), [37-39]. Other case reports have highlighted the presence of osteoporosis in patients suffering from MSL. The presence of osteoporosis in a male patient requires the exclusion of a possible Cushing syndrome resembling MSL or other osteolytic etiologies (glucocorticoid medication, hypogonadism, hyperparathyroidism, excessive alcohol consumption, gastrointestinal disease) [40].
3. Complications
Although not very frequent, if complications of MSL are present, they are usually linked to difficulties in neck movement and compression of adjacent neck or mediastinal structures. The most significantly impacted structures are the larger blood vessels (carotid arteries, brachyocephalic and jugular veins), nerves (cervical plexus, recurrent laryngeal nerves, facial nerve, glossopharyngeal nerve), trachea and the main bronchi, esophagus, regional lymph nodes and the thoracic duct. Rare cases of scrotal lipomatosis associated with dysuria and painful erections have also been reported [25]. The main concern is the presence of dyspnea, stridor or sleep apnea, all pointing to a significant obstructive respiratory disorder caused by a combination of lipoma compression, pharyngeal adipose tissue infiltration and macroglossia [41]. Very few cases of malignant transformation to liposarcoma or intramyxoid sarcoma have been described in the literature [42, 43].
4. Treatment
Nonsurgical treatments, such as intralipotherapy using phosphatidylcholine/ deoxycholate, only prevent further growth of the pathological adipose tissue, without reducing the actual volume. Eliminating alcohol consumption may also prevent further disease progression. Injection of enoxaparin and β2 agonists (salbutamol) has proven inconsistent curative effects [44, 45]. Surgical removal of lipomas is currently the main choice of treatment. The preferred methods are either through lipectomy or liposuction. Lipectomy allows for a better exposure of the desired adipose mass, while providing the surgeon with a superior view of adjacent structures., but the unencapsulated nature of the lipomas significantly impacts full excision capabilities. Being a more invasive procedure in comparison to liposuction, far more procedural complications have been reported [6, 45]. Liposuction is less invasive, but procedural success is significantly hindered if the adipose mass presents an important fibrous component. US assisted liposuction may be useful in differentiating tumor limits from critical structures, especially in the neck. The two surgical options are considered palliative. Recurrence is almost certain, due to incomplete lipoma resection through both methods [45, 46]. In patients presenting advanced stages of obstructive pulmonary disease, and MSL associated sleep apnea, CPAP is deemed the main conservative therapy [47, 48]. If proven unsuccessful, more invasive choices of either transoral or transcervical lipectomy or pharyngoplasty should be considered [49]. An idealistic approach in treating MSL would require the fulfillment of the following criteria: fully removing the already existent lipomas from the patient’s body, thus improving the cosmetic appearance, lowering the risk of developing severe complications (obstructive pulmonary disease, compression of vascular and nervous structures), and guaranteeing a very low risk of recurrence.
5. Conclusion
In our patient, we have dealt with a classical Type 1 Madelung disease. US evaluation on admission allowed for a rapid exclusion of parotid gland involvement and of other possible lymphadenopathies of the head and of the neck. CT scan of the thorax was essential in excluding possible life-threatening compressive phenomena in the neck, thorax (thoracic wall and mediastinum) and abdomen. Due to the lack of complications other than the modified cosmetic appearance, the patient did not opt for elective surgical excision of the lipomas. Nonsurgical therapy was also not a valid choice, due to the stationary status of the lipoma volume in the last years. The patient was encouraged to immediately cease alcohol consumption.
Conflict of interests
The authors declare none.
References
- da Costa JN, Gomes T, Matias J. Madelung Disease Affecting Scrotal Region. Ann Plast Surg 78 (2017): 73-77.
- Nikolic ZS, Jeremic JV, Drcic LJ, et al. Madelung disease: a rare case associated with gynaecomastia and scrotal involvement. J Plast Surg Hand Surg 47 (2013): 415-418.
- Hadjiev B, Stefanova P, Shipkov C, et al. Madelung disease: on the morphologic criteria for diagnosis and treatment. Ann Plast Surg 64 (2010): 807-808.
- Donhauser G, Vieluf D, Ruzicka T, et al. Benign symmetric Launois-Bensaude type III lipomatosis and Bureau-Barrière syndrome. Hautarzt 42 (1991): 311-314.
- Enzi G, Busetto L, Ceschin E, et al. Multiple symmetric lipomatosis: clinical aspects and outcome in a long-term longitudinal study. Int J Obes Relat Metab Disord 26 (2002): 253-261.
- Meningaud JP, Pitak-Arnnop P, Bertrand JC. Multiple symmetric lipomatosis: case report and review of the literature. J Oral Maxillofac Surg 65 (2007): 1365-1369.
- Zhang XY, Li NY, Xiao WL. Madelung disease: manifestations of CT and MR imaging. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 105 (2008): 57-64.
- Gao H, Xin ZY, Yin X, et al. Madelung disease: A case report. Medicine (Baltimore) 98 (2019): 14116.
- Liu Q, Lyu H, Xu B, et al. Madelung Disease Epidemiology and Clinical Characteristics: a Systemic Review. Aesthetic Plast Surg 45 (2021): 977-986.
- de Jong JMA, Wouters RTF, Boulet N, et al. The β (3)-adrenergic receptor is dispensable for browning of adipose tissues. Am J Physiol Endocrinol Metab 312 (2017): 508-518.
- Yang LK, Tao YX. Physiology and pathophysiology of the β (3)-adrenergic receptor. Prog Mol Biol Transl Sci 161 (2019): 91-112.
- Nisoli E, Regianini L, Briscini L, et al. Multiple symmetric lipomatosis may be the consequence of defective noradrenergic modulation of proliferation and differentiation of brown fat cells. J Pathol 198 (2002): 378-387.
- Kyaw H, Grillo M, Lin AN, et al. State of diagnostic quandary solved by modern technology: a rare case of Madelung's disease. BMJ Case Rep 2016 (2016): 213842.
- Moonen MPB, Nascimento EBM, van Kroonenburgh M, et al. Absence of (18) F-fluorodeoxyglucose uptake using Positron Emission Tomography/Computed Tomography in Madelung's disease: A case report. Clin Obes 9 (2019): 12302.
- Musumeci O, Barca E, Lamperti C, et al. Lipomatosis Incidence and Characteristics in an Italian Cohort of Mitochondrial Patients. Front Neurol 10 (2019): 160.
- Klopstock T, Naumann M, Schalke B, et al. Multiple symmetric lipomatosis: abnormalities in complex IV and multiple deletions in mitochondrial DNA. Neurology 44 (1994): 862-866.
- Klopstock T, Naumann M, Seibel P, et al. Mitochondrial DNA mutations in multiple symmetric lipomatosis. Mol Cell Biochem 174 (1997): 271-275.
- Caponnetto F, Manini I, Bulfoni M, et al. Human Adipose-Derived Stem Cells in Madelung's Disease: Morphological and Functional Characterization. Cells 10 (2020): 44.
- Cho JH, Kim EC, Son Y, et al. CD9 induces cellular senescence and aggravates atherosclerotic plaque formation. Cell Death Differ 27 (2020): 2681-2696.
- Lemaitre M, Chevalier B, Jannin A, et al. Multiple symmetric and multiple familial lipomatosis. Presse Med 50 (2021): 104077.
- Gao Y, Hu JL, Zhang XX, et al. Madelung's Disease: Is Insobriety the Chief Cause? Aesthetic Plast Surg 41 (2017): 1208-1216.
- Hoxha M, Cakoni R, Basho M. Rapidly progressive case of type I Madelung disease with bilateral parotid and minor salivary glands involvement. Br J Biomed Sci 77 (2020): 48-52.
- Vargas-Díez E, Daudén E, Jones-Caballero M, et al. Madelung's disease involving the tongue. J Am Acad Dermatol 42 (2000): 511-513.
- Calvo Hernadez LM, Riol López E, Peña Ferrera L, et al. Macroglossia in Madelung's disease. Med Clin (Barc) 147 (2016): 59.
- Penna V, Iblher N, Al Jamali J, et al. Rare locations of multiple symmetric lipomatosis (Madelung’s disease). European Journal of Plastic Surgery 35 (2012): 557-560.
- Ko MJ, Chiu HC. Madelung's disease and alcoholic liver disorder. Hepatology 51 (2010): 1466-1467.
- Ahuja AT, King AD, Chan ES. Ultrasound, CT and MRI in patients with multiple symmetric lipomatosis. Clin Radiol 55 (2000): 79.
- Che K, Lyu QW, Ma GE. Madelung Disease Type Ia: Lipectomy or Liposuction? Ann Plast Surg 85 (2020): 332.
- Friedl C, Sampl E, Prandl EC, et al. Osteoporosis, weight gain and atypical fat accumulations - a typical feature not only for Cushing's, but also Madelung's disease: A case report. Wien Klin Wochenschr 124 (2012): 188-192.
- Bowman JD, Kim H, Bustamante JJ. Drug-induced gynecomastia. Pharmacotherapy 32 (2012): 1123-1140.
- Quintanilla-Dieck L, Penn EB, Jr. Congenital Neck Masses. Clin Perinatol 45 (2018): 769-785.
- Tan E, Jaya J. An approach to neck masses in adults. Aust J Gen Pract 49 (2020): 267-271.
- Lagathu C, Béréziat V, Gorwood J, et al. Metabolic complications affecting adipose tissue, lipid and glucose metabolism associated with HIV antiretroviral treatment. Expert Opin Drug Saf 18 (2019): 829-840.
- Barton N, Moore R, Prasad K, et al. Excisional lipectomy versus liposuction in HIV-associated lipodystrophy. Arch Plast Surg 48 (2021): 685-690.
- Brea-García B, Cameselle-Teijeiro J, Couto-González I, et al. Madelung's disease: comorbidities, fatty mass distribution, and response to treatment of 22 patients. Aesthetic Plast Surg 37 (2013): 409-416.
- Lin FY, Yang TL. Madelung disease. Cmaj 185 (2013): E79.
- Saiz-Hervas EMLM, Lopez alvarez J. Peripheral neuropathy as the first manifestation of Madelung’s disease. Br J Dermatol 143 (2000): 684.
- Chopra K, Tiwari V. Alcoholic neuropathy: possible mechanisms and future treatment possibilities. British journal of clinical pharmacology 73 (2012): 348-362.
- Chan HF, Sun Y, Lin CH, et al. Madelung's disease associated with polyneuropathy and symptomatic hypokalemia. J Formos Med Assoc 112 (2013): 283-286.
- Khosla S, Amin S, Orwoll E. Osteoporosis in men. Endocr Rev 29 (2008): 441-464.
- Segsarnviriya C, Chirakalwasan N. A case of Madelung's disease presenting with obstructive sleep apnea. J Clin Sleep Med 16 (2020): 1603-1605.
- Tizian C, Berger A, Vykoupil KF. Malignant degeneration in Madelung's disease (benign lipomatosis of the neck): case report. Br J Plast Surg 36 (1983): 187-189.
- Durand J, Thomine J, Tayrot J, et al. Liposarcome au cours d’une maladie de Launois-Bensaude. Rev Rhum Mal Ostoartic 40 (1973): 287-291.
- Fischer M, Wohlrab J, Taube KM, et al. Intralesional injection of enoxaparin in benign symmetrical lipomatosis: an alternative to surgery? Br J Dermatol 144 (2001): 629-630.
- Chen CY, Fang QQ, Wang XF, et al. Madelung's Disease: Lipectomy or Liposuction? Biomed Res Int 2018 (2018): 3975974.
- González-García R, Rodríguez-Campo FJ, Sastre-Pérez J, et al. Benign symmetric lipomatosis (Madelung's disease): case reports and current management. Aesthetic Plast Surg 28 (2004): 108-113.
- Lee DH, Lim SC, Lee JK. Laryngeal involvement in Madelung disease. Otolaryngol Head Neck Surg 144 (2011): 481-482.
- Pinto V, Morselli PG, Tassone D, et al. A case of severe obstructive sleep apnoea in Madelung's disease treated by lateral pharyngoplasty. J Laryngol Otol 131 (2017): 834-837.
- Chia Hsuan L, Kang K-T, Ko J-Y, et al. Laryngeal involvement in Madelung's disease with acute airway compromise. The Kaohsiung journal of medical sciences 28 (2012): 462-463.
