Herb Stevia Rebaudiana’s Functionalities, Safety, and Applications: A Review
Article Information
Ji Li1*, Pradeep Thaker1, Deshou Jiang2, Qingrong Huang3, Chi Tang Ho3
1Country Life LLC, 180 Motor Pkwy, Hauppauge, NY 11788, USA
2Chromocell Corporation, 685 US-1, North Brunswick, NJ 08902, USA
3Food Science Department, Rutgers, the State University of New Jersey, 65 Dudley Rd, New Brunswick, NJ 08901, USA
*Corresponding author: Ji Li, Country Life LLC, 180 Motor Pkwy, Hauppauge, NY 11788, USA.
Received: 19 January 2023; Accepted: 24 January 2023; Published: 06 February 2023
Citation: Ji Li, Pradeep Thaker, Deshou Jiang, Qingrong Huang, Chi Tang Ho. Herb Stevia Rebaudiana’s Functionalities, Safety, and Applications: A Review. Archives of Clinical and Biomedical Research 7 (2023): 64-85.
View / Download Pdf Share at FacebookAbstract
Herb Stevia rebaudiana extract, also known as sweetleaf, was originally discovered for its sweetening function. Researches show that this plant extract is embedded with multiple functionalities such as antioxidant, antidiabetics, anti-inflammation, and antimicrobial. This review article helps re-arrange the scientific affairs of those stevia extract’s functions. Sweetness indexes of steviol glycosides were summarized together for comparison while various in vitro, in vivo approaches were reviewed to quantify those functions’ capacities and to depict the related mechanism. The regulation of steviol glycoside compounds such as rebaudioside A was established by global authorities such as US Food and Drug Administration (FDA) and Joint FAO/ WHO Expert Committee (JECFA) to ensure the safety endorsement prior to commercialization. In applications, we first used sentiment analysis to catch a good command of the stevia-containing products’ market performance. Then, we investigated the product development progress of stevia-containing food products in the categories of beverage, bakery, dairy, and confectionery. Those stevia-containing food consumer goods can be acceptable by certain customers.
Keywords
Antioxidant; Antidiabetics; Anti-Inflammation; Product Development; Safety, Stevia Rebaudiana; Sweetening
Antioxidant articles; Antidiabetics articles; Anti-Inflammation articles; Product Development articles; Safety articles, Stevia Rebaudiana articles; Sweetening articles
Article Details
1. Introduction
Stevia rebaudiana, also known as sweetleaf, is a natural sweet herb originally-found in South America. The natives in Paraguay processed stevia for sweetening beer, tobacco, and other drinks back to early 1900s [1]. Moisés Santiago Bertoni first discovered the sweetening function of stevia herb. In honor of his original contribution, we also called this herb Stevia rebaudiana Bertoni. Stevia rebaudiana was found with multiple economic values including sweetening and additional therapeutic functions. As a short day plant, it is grown in the subtropics with an annual mean temperature 20 oC and an annual precipitation around 1550 mm [2]. Numerous countries in Asia such as China, Japan, Malaysia, Singapore, South Korea, Taiwan, and Thailand massively cultivated this economic plant [3]. During recent years, we observed the profit growth of stevia-related products in the United States of America, Canada, and Europe.
Stevia rebaudiana contains certain amounts of steviol glycosides, fatty acids, and trace amounts of amino acids, polyphenols, vitamins, and minerals [4]. The most important components in stevia are steviol glycosides, which contributes to the high sweetening capability. The simplest approach to extract steviol glycosides from stevia herb is hot water leaching [5]. Other methods such as solvent extraction [6], ultrasound [7], microwave [8], supercritical fluid extraction [9], and enzyme assisted extraction [10] were developed to improve the extraction efficiency of steviol glycosides from Stevia rebaudiana. For instance, Ameer, Chun et al. demonstrated that using response surface methodology (RSM) and artificial neural network (ANN) helped the optimization of extracting steviol glycosides through supercritical fluid extraction [9]. They were able to achieve maximum values for total extract yield, stevioside yield, rebaudioside A yield, and total phenolic content of 15.85%, 95.76 mg/g, 62.95 mg/g and 25.76 mg GAE/g, respectively, which outperformed the results of microwave-assisted steviol glycosides’ extraction conducted by the same group [8]. Other sustainable biotechnological methods such as plant cell or microbial fermentation could help stabilize large-scale manufacture of high potency sweeteners such as steviol glycosides mentioned here [11].
With appropriate processes, we were able to explore active compounds and their functions, evaluate their safety, and prepare regulatory documents, all of which laid a solid foundation for latter product applications. Those in-depth investigations enable us to well-understand specification, scientific labeling, marketing, and education. At the early-discovery stage, conventional chemistry methods including high-performance liquid chromatography (HPLC) [13], nuclear magnetic resonance (NMR) [15], and infrared spectrometer (IR) [16] were used to quantify the composition of Stevia rebaudiana and separate the compounds that interest us the most. Among all of those characterization tools, HPLC-based approaches are the most reliable and straightforward. Furthermore, the optimization of the analytical conditions (e.g., column, mobile phase, and so forth) effectively produce more steviol glycoside compounds with reproducible retention time and specificity. A robust UHPLC-Orbitrap MS method enables the separation and quantification of steviol, isosteviol, and steviol glycosides of more than 10 species referring to majority of stevioside and rebaudioside A, and trace compounds such as dulcoside A, steviolbioside, and rubusoside [17]. Total 30 steviol glycoside species were separated and quantified from dried stevia extracts, sweeteners, and beverages. They meticulously optimized the chromatographic conditions to achieve the best sensitivity and better structural information.
With year’s experiences on the upstream of Stevia rebaudiana, we then witnessed the outburst of its downstream research involving functional exploration, safety evaluation, and product development applications. In this review article, we first summarized and commented on the functions of stevia extracts and their active compounds including sweetening, antioxidant, antidiabetics, anti-inflammation, and antimicrobial. A summary of steviol glycosides’ sweetness indexes was made for future reference, and the antimicrobial activity can be viewed as its secondary function. Later, we summarized the global/regional safety data, certificate, and critical regulatory approvals which ultimately paved the way for the safe stevia applications. The official regulatory milestones of steviol glycosides from US Food and Drug Administration (FDA), Joint FAO/WHO Expert Committee on Food Additives (JECFA), and others were included. Lastly, we took the lead to use sentiment analysis tool to understand the market performance of stevia ingredients and stevia-containing products. In this section, we also reviewed the research reports of stevia-containing products referring to four categories including beverage, bakery, dairy, and confectionery. This review can serve as a comprehensive guidance for researchers in the fields of food, pharmaceutical, and dietary supplement when considering stevia.
2. Functions
2.1 Sweetening
Sweetening is one of the major functions of Stevia rebaudiana. From organoleptic perspective, stevia extracts have a potent sweet taste at the front and a bitter or liquorice like aftertaste when consumed more [17]. The bitter aftertaste in stevia sweetening is probably due to sesquiterpene lactone [18]. Commercial stevia sweeteners are composed of steviol glycosides such as stevioside and rebaudioside A, the two major compounds that have been approved by the US Food and Drug Administration (FDA) as natural sweeteners. They can be blended with other sweeteners such as Luo Han extract, xylitol or dextrose to achieve distinct sensation. So far, more than 40 stevia glycosides have been identified and separated from stevia herbs, including sativoside, steviolbioside, rebaudioside A to E, dulcoside A, and stevioside A [19]. Figure 1 displays the structure of steviol glycoside with aglycone core and steviol derivatives with different side chains on their R1 and R2 sites. Take rebaudioside B as an example. Its functional group R2 is Glc(β1-2)[Glc(β1-3)]Glc(β1- which refers to three glucoses linking each other through β-1,2-glycosidic bond and β-1,3-glycosidic bond.
Those steviol glycosides have different sweetness levels, and their relative sweetness levels could be quantified by different methods. Since sweetness results could be affected by multiple factors such as methods, participants, and resources, it is reasonable to accept a relatively-broad range of sweetness level for steviol glycoside compounds. Table 1 displays a collection of the previously-reported steviol glycosides’ sweetness indexes. The unit of measure for sweetness indexes is times the sweetness of sucrose. Some roughly reported that stevia extract could have the sweetness approximately 250 times of sucrose without any generated energy and a glycemic index of zero [20]. Table 1 shows that the sweetness indexes of stevioside and rebaudioside A could reach 200 up to 400 or 450 folds of sucrose’s sweetness. Fewer reported the sweetness measurements of other steviol glycosides including rebaudioside B – E and dulcoside A. This is probably related to the low yield of those compounds from extraction. We also observed a report which stated that steviol glycosides from stevia extract were only 10 – 15 times sweeter than sucrose [21]. As mentioned-above, the reported sweetness variations of steviol glycosides are attributed to different factors such as concentration, pH, composition, and taste participants. Stevia extract was reported as approximately 300 times sweeter than sucrose in a 0.4% solution, but decreasing to 150 times sweeter in a 10% solution [22]. The relevant organoleptic results showed that stevia extract was 97 times more potent than sucrose at pH 7.0, and 109 times at pH 3.0 when compared with a 10% sucrose solution. Changing pH helps tuning the taste potency of stevia extract. Based on the sweetnesses of different steviol glycosides, Xinfeng Zhao and Wuzhou Yao established a stevia sweetness index equation which could be used to calculate the overall sweetness index of stevia extracts with the known compositions [23]. This empirical approach gives a quick sweetness prediction of unknown stevia extract based on its composition. We notice that many reports of steviol glycosides’ sweetnesses merely cited the taste results from old references. It might be necessary for researchers to update the sweetness values of steviol glycosides based on the new results.
Steviol glycosides are much sweeter than sucrose, however, they still have bitterness and astringency, giving a lingering, unpleasant metallic aftertaste. Researchers developed chemical/enzymatic modifications and processes to improve the gustatory experiences of those steviol glycosides [24–28]. It was found that the glycosyl moieties at C-13 and C-19 positions of the steviol core contributed at least partially to the compounds’ taste sensation [29]. Take the rebaudioside A and stevioside as an example. Empirical data showed that rebaudioside A tasted sweeter than stevioside (See Table 1). Structural modification at those locations can alter the taste quality of steviol glycosides [30]. In early years, Grant et al. reported the synthesis of various stevioside analogues by replacing the R1 group on C- 19 and R2 group on C-13 of the steviolbioside with different ester groups such as sulfopropyl ester and sodiosulfopropyl ester [26,27]. Among all the synthesized analogues, the modified steviolbioside with R1 group (CH2)3SO3Na (Sodiosulfopropyl ester) improved sensory properties of original stevioside [26]. They also demonstrated that no or very limited bitter-taste feature was found when R1 group on C-19 Glyc(b1- ester in stevioside and rebaudioside A were replaced by (CH2)2CH(SO3Na)2 and (CH2)3SO3Na groups, respectively [27]. Other synthesis strategies such as acidic/alkaline hydrolysis [31], change starting materials [32], and alter add-on materials [33] were used to synthesize steviol analogues. The acidic/alkaline hydrolysis of stevioside and rebaudioside A lead to de-esterification at C-19 of the steviol core and saccharide release to form steviolbioside and rebaudioside B, respectively [31]. Multiple step synthesis starting from steviol including acetylation of the C-13 hydroxyl function of steviol with acetic anhydride in pyridine, glycosylation of the C-19 carboxyl group with 2,3,4,6-tetra-O-acetyl-a-Dglucopyranosyl bromide, and subsequent deacetylation were developed to synthesize steviol-19-O-glucoside [32]. Multiple functional groups can be synthesized onto the steviol core to form new steviol analogues which involves organic solvents and intermediate processes.
Compared with chemical synthesis, enzymatic modification of steviol glycosides provides us with a cleaner process requiring less organic solvents and processes. The start materials are subjected to transglucosylation from glycosyl donors with catalysts such as cyclodextrin glucosyltransferase (CGTase) of different resources. More or less discoveries were achieved along the way to optimize the conditions of enzymatic modification of steviol glycosides, which were purposefully to improve the overall taste of end steviol glycoside products [24,30,34,35]. Those findings refer to different aspects of enzymatic synthesis of steviol glycoside derivatives. Darise et al. discovered that the ratio of the number of glucose units at the C-13 of rubusoside to that at the C-19 significantly influenced the sweetness and the taste quality of the synthesized products, and the better sweeteners were made with the ratio of 13-O-glc:19-O-glc more than 1:1. Changes of glucose donors, enzymatic catalysts, reaction conditions, and start materials could alter the end product yield. An optimum condition of 45 oC, pH 6.5-7.5, and stevioside-tocyclodextrin 1:1 resulted in 11.6% ultimate yield of better-tasting product after transglycosylation of stevioside [24]. Besides, conversion of stevioside to rebaudioside A could enhance the sweetness and taste profile of the final stevia extract, but steviol glycosylation beyond a certain number of glucose units reduced sweetness [34]. Recently, Yu et al. reported an optimized culture conditions for CGTase-producing bacteria strain Paenibacillus sp. CGMCC 5316 by using Plackett-Burman design [35]. The monoglycosylated stevioside made from them had a 35.4% sweetness increase, and was endowed with a taste quality better than stevioside. The transglycosylation rate of stevioside could reach 85.6% in the end.
Change pre-/post- processes appropriately can help decrease the bitter aftertaste of steviol glycosides and ameliorate the overall taste quality. New technologies or devices are developed to innovate the processes. Microencapsulation is applied to mask off-flavors [36]. Steviol glycosides encapsulated by maltodextrin (19 DE) and inulin through spray drying presented the appealing sensory and quality characteristics [38]. Research group compared the effects of spray, freeze and oven drying as methods for decreasing bitter aftertaste of steviol glycosides [25]. Considering physicochemical properties and sensorial properties of all the end products, they found that the spray dried steviol glycosides outperformed products from freeze and oven drying. A nanofiltration membrane could effectively concentrate the sweeteners from stevia extracts at high temperature [39]. The preliminary results suggested that certain amounts of bitter-tasting components were washed out from the sweetener concentrate after nanofiltration process operated at 80 oC. Even a brief ethanolic pretreatment could reduce bitter aftertaste of stevia extracts and increase the yield, purity of stevia sweeteners together with ion exchange chromatography and membrane separation approaches [40].
Table 1: Report of steviol glycosides’ sweetness indexes.
2.2 Antioxidant
Oxidation is a process where an atom within a molecule loses electron, an atom within a molecule increases in oxidation state or an organic substance either gains oxygen or loses hydrogen [41]. Oxidative processes can be multiple chain chemical reactions caused by free radicals, the oxygen-containing molecules with an uneven number of electrons. Antioxidants are compounds that are capable of scavenging free radicals and reactive oxygen species (ROS) by transferring electron-free radicals or regulating the antioxidant enzyme system. Typical antioxidants are water-soluble ascorbic acid (vitamin C) and fat-soluble tocopherols (vitamin E). As an electron donor, ascorbic acid exerts powerful antioxidation to protect cellular components from free radical damage [42]. The crude extracts from Stevia rebaudiana have remarkable antioxidant ability due to the abundant content of phenolic compounds, flavonoids, diterpene glycosides (e.g., stevioside and rebaudioside A), condensed tannins, anthocyanins, and phenolic acids [43]. Among those components, the sweet steviol glycosides exhibited low oxygen radical absorbance capacity (ORAC) values compared with the whole herb extract [44]. One report suggested that steviol glycosides and antioxidation were not closely-correlated, and appropriate extraction methods for steviol glycosides and antioxidants from Stevia rebaudiana should be developed separately [45]. Phenolic compounds and flavonoids primarily contribute to the antioxidant function of Stevia rebaudiana. Those phenolic compounds were extracted from different parts of the stevia herb such as leaf, callus, stem [44], and flower [46]. Table 2 lists the representative phenolic profiles from Stevia rebaudiana. The divergence of phenolic profiles could be due to variations of plant species, plant growth conditions (e.g. light exposure), extraction procedures, and units of measure. There is no unification of phenolic report format, either. One reported total phenolic compounds (TPC) were 25.18 mg/g for stevia leaves and 35.86 mg/g for callus on, while the flavonoids were found to be 21.73 and 31.99 mg/g in leaf and callus, respectively [47]. Those discovered flavonoids improved the quantity and quality of antioxidant activities of Stevia rebaudiana [46,48]. Another TPCs of stevia extract were 46.14 mg gallic acid equivalent (GAE)/g from stem and 71.46 mg GAE/g from leaf [49]. Bender et al. reported that the TPCs of stevia extract from different geometries and farms could range from 17.49 to 195.30 mg of chlorogenic acid equivalents/g [44]. Kim et al. reported that total phenolic and flavonoid contents were 130.76 μg catechin and 15.64 μg quercetin for leaves, and 43.99 μg catechin and 1.57 μg quercetin for cellus at mg of water extracts, respectively [48]. Other reported measures of total phenolic contents were 61.50 mg gallic acid equivalent of phenols /g [50] and 56.73 mg/g of phenolic content [51]. Those phenolic components from stevia extract are attributed by various compounds. Karaköse et al. was able to analyze and identify 29 chlorogenic acid derivatives from methanolic stevia extracts by LC-MSn equipped with an ESI ion-trap mass spectrometer [52]. The latter principal component analyses allowed differentiation between the species of different geographical origins and plant varieties. Pyrogallol was identified and reported as the main phenolic compound from leaf extract followed by 4-methoxybenzoic acid [48]. Meantime, quercetin derivatives serve as dominant flavonoids which exert a significant in vitro antioxidant capacity. They are effective in chelating metals, reducing oxidative stress, and regenerating antioxidants.
Various chemical assays such as 1,1-diphenyl-2-picrylhydrazyl (DPPH) assay [47], oxygen radical absorbance capacity (ORAC) assay [53], ferric reducing/antioxidant power (FRAP) assay [54], and 2,2’-azino-bis(3-ethylbenzthiazoline-6-sulfonic acid) (ABTS) assay [55] were utilized to quantify the in vitro antioxidant activities of stevia extracts. Similar to TPC measure, antioxidant effectiveness measure might not be comparable because of differences in extraction procedures, units of measure, and plant parts. Multiple assays are sometimes utilized together to provide a comprehensive quantification. Compared with clinical studies, chemical assays offer us fast and cost-effective preclinical data. Those preclinical data guide us the further exploration of stevia extracts’ functions. Those assays were developed based on chemical reactions to evaluate in vitro anti-radical activities of stevia extracts. DPPH assay is the most common chemical assay to determine the antioxidant capacity of stevia extracts [47,50,51]. The strong purple color from the stable free radical DPPH allows spectrophotometric reading. Color change of DPPH suggests the movement of electron transfer or hydrogen donation. We then use the change in DPPH absorbance after adding a test material as an index of the antioxidant capacity of the material [57]. Tadhadni et al. reported 33.17% to 56.82% DPPH radical inhibition from stevia leaf/callus extracts [47], while Shukla et al. demonstrated the DPPH radical inhibitions of 36.93% to 68.76% and 40% to 72.37% from stevia ethanol extract and aqueous leaf extract, respectively [50,51]. Such antioxidant capability reading render stevia extracts comparable with supplement vitamin C or ascorbic acid (64.26% to 82.58%). As an analytical assay used for many years, DPPH assay has its own drawbacks such as limit for larger molecules and chemical nature not mimicking what occurs in human body. Comparatively, ORAC assay is more suitable for quantifying the antioxidant capacity of a test material in food and physiological systems. This assay gauges the inhibition of the peroxyl radical-induced oxidation initiated by free radical generator such as azo-compounds [58]. The antioxidant activities measured by ORAC assay were different from those of DPPH assay. Study showed that tea catechins with lower antioxidant activities measured by ORAC assay compared with that of DPPH assay was related to a pro-oxidant effect when generating reactive oxygen species in an aqueous physiological condition [59]. Thus, the ORAC/DPPH value indirectly indicates whether a pro-oxidant performs in a biological system. Multiple assays are frequently-used together to compensate test limits of different antioxidant measuring methods. Barba et al. leveraged ORAC and ABTS assays to compare the total antioxidant capacities of Stevia rebaudiana before and immediately after high pressure processing treatment [60]. The two optimized treatments of 300 MPa, 15 min, 2.5 w/v% and 500 MPa, 5 min, 2.5% w/v% enabled the end stevia products to have antioxidant capabilities of 40.90 and 41.35 millimolar trolox equivalents (mM TE from ORAC), respectively. Nevertheless, ORAC values were significantly higher in samples than those measured by ABTS assay, which was attributed to the fact that ORAC method was capable of responding to a larger number of antioxidant compounds than ABTS method in fruit complex mixtures. The antioxidant capacities of stevia extracts depend on various factors such as plant parts and antioxidant components. Bender et al. reported that the stevia leaves extracts had a higher antioxidant activity than stems extract through ORAC and cellular antioxidant activity (CAA) assays [44]. Steviol glycosides, the main sweetening compounds in stevia extracts, exhibited low ORAC values in comparison with plant extracts, and they did not show any CAA value. Their research supported the view that steviol glycosides and stevia antioxidants were negatively correlated as mentioned-before. Other methods including lipid peroxidation [61], reduced glutathione level [62], and oxidative phosphorylation [63] were tailored and applied to measure the antioxidant activities of stevia extracts under various biological systems as well.
Those antioxidant test methods offered low-cost, high-throughput tools for screening dietary antioxidants. Meanwhile, relying solely on colorimetric methods is less encouraged, on the contrary, some biological tests involving cell lines, simulated digestion or in vivo assessments are more preferred [64]. The in vitro and in vivo antioxidant activities can be inconsistent, which is influenced by factors such as metabolism and bioavailability [65]. Sharma et al. studied the synergetic interaction of Stevia rebaudiana together with hyperglycermia and hepatic antioxidant enzymes by using animal models of type 2, noninsulin dependent diabetes [62]. A 28-day treatment of stevia extract resulted in significant increase of superoxide dismutase (SOD), catalase (CAT), and reduced glutathione (GSH) in diabetic treated rats as compared to diabetic control group. However, a 28-day treatment of stevia in normal treated group merely caused slight increase in SOD and no significant change in CAT as compared to control group. Those antioxidant enzymes (SOD and CAT) are the two major scavenging enzymes that remove the toxic free radicals in vivo. Glutathione is an endogenous antioxidant produced by the cells to neutralize free radicals and reactive oxygen compounds. Such results suggested stevia extract have therapeutic potential in indirectly treating diabetes as it improved the activity of oxidative scavenging enzymes dramatically, but mechanism is required for this herb on the enzymes involved in type-2 diabetes. Similar results were found in another report [66]. Prata et al. investigated the insulin-mimetic effect from four different mixtures of steviol glycosides, abundant in stevioside and rebaudioside A in neonatal rat cardiac fibroblasts. Interestingly, their data specifically showed that steviol glycosides could counteract oxidative stress by increasing GSH intracellular levels and upregulating expression and activity of SOD and CAT, although those main compounds from stevia extract had low ORAC values and not any CAA value as mentioned-above [44].
We also compare the antioxidant activities of stevia extracts with those of other plant species, not merely ascorbic acid or tocopherol, which enables us to evaluate their antioxidant performance in an intuitive way. Plant species that have antioxidant activities are peppermint, ginseng, oregano, and etc. Previous report compared the antioxidant capabilities of Siberian ginseng extract, stevia extract, essential oils from oregano, and sage together measured by conventional Bauchamp and Fridovich method [67]. Those plant extracts can targetedly inhibit different free radicals such as superoxide anion radical, nitric oxide, peroxynitrite, and hydroxyl radical. Among all plant extracts, ginseng showed an outstanding ability to scavenge superoxide anion radical and nitric oxide at all the concentrations, while stevia could convert nitric oxide starting from low concentration (20 μg/ml) and quench hydroxyl radical at high concentrations (80 and 100 μg/ml). The polyphenol and antioxidant profiles of peppermint and stevia were compared under organic and conventional fertilization [68]. ABTS and DPPH results clearly displayed that stevia and peppermint had close radical scavenging activities under either organic or conventional fertilization (e.g., 72.78% inhibition for stevia and 74.47% inhibition for peppermint from DPPH measure). Comparatively, stevia had a better antioxidant stability against different fertilizations than peppermint.
|
Main phenolic components |
Total phenolic content* |
Plant part |
Reference |
|
Pyrogallol, 4-Methoxybenzoic acid, p-Coumaric acid, 4- Methylcatechol |
130.67±2.23 µg catechin equivalents/mg |
Leaf |
[48] |
|
5-O-Caffeoylquinic acid, 3,5-O-Dicaffeoylquinic acid, Quercetin-3-O-rhamnoside, Quercetin-3-O-rutinoside |
77.1±0.2 mg/g extract |
Frozen fresh leaf |
[163] |
|
Diosmin, Chlorogenic acid, Ellagic acid, Rutin |
98.4±13.19 mg/g |
Leaf |
[164] |
|
Vanillic acid 4-O-β-D-glucopyranoside, Protocatechuic acid, Caffeic acid, Chlorogenic acid, Cryptochlorogenic acid |
46.14, 71.46 mg GAE/g |
Stem, Leaf |
[49] |
|
Quercitrin, Quercetin-3-O-arabinoside, Kaempferol-3-O- rhamnoside, Luteolin |
0.86 mg gallic acid equivalents/mg |
Leaf |
[61] |
|
29 Chlorogenic acids: mainly 4-Caffeoylquinic acid, 5-Caffeoylquinic acid, 3,5-Dicaffeoylquinic acid, 4,5-Dicaffeoylquinic acid |
30.05 – 57.73 mg chlorogenic acid/g |
Leaf |
[52] |
|
Dicaffeoylquinic acid, Chlorogenic acid, Quercetin 3-Oxyloside, Luteolin 7-O-rutinoside, Caffeic acid |
91 mg/g |
Leaf |
[165] |
|
4-O-Caffeoyl quinic acid, 3,5-di-O-Caffeoyl quinic acid, 4,5-di-O-Caffeoyl quinic acid |
17.49-195.30 mg of chlorogenic acid equivalents/g |
Leaf |
[44] |
Table 2: Phenolic profiles of Stevia rebaudiana extracts.
*The total phenolic contents were calculated on dry basis.
2.3 Antidiabetics
Diabetes mellitus is a group of chronic metabolic diseases characterized by blood glucose elevation irregularly, which can result in serious damage to the heart, blood vessels, eyes, kidneys and nerves in the long run. Type 2 diabetes occur more often in adults. According to the World Health Organization (WHO), approximately 422 million people worldwide have diabetes and 1.6 million lethal events are directly relevant to diabetes each year. It is critical for us to develop affordable, natural therapeutic treatments as alternatives to common medicines (e.g., metformin, thiazolidinediones, and etc.). The antidiabetic potential of stevia extract has been evaluated, and its zero-calorie property can be beneficial to patients suffering from obesity and diabetes. Steviol glycosides have chemical structures similar to those O-glycoside antidiabetic agents studied before [69]. Such chemical structures render us to expect somewhat antidiabetic capability of steviol glycosides. Different in vitro assays and in vivo animal models were utilized to examine and evaluate the antidiabetic function of stevia extracts. Some data collection suggest a promising antidiabetic capability of stevia extracts while others are contradictory with the previous results. The influence of steviol glycosides on the glucose metabolism pathway were investigated in 3T3-L1 cell lines [70]. They demonstrated that stevioside could increase glucose uptake activities by 2.1 times in normal conditions and almost 4.5 times in insulin-resistant states, which was better than what was observed in positive control group treated with rosiglitazone maleate. Bhasker et al. studied the gene regulation of stevioside and steviol in glucose absorption at transcript and protein levels in diabetes induced L6 myotubes and 3T3L1 adipocytes [71]. The qPCR data showed that steviol, stevioside, and the crude stevia extract had direct effect on the activation of GLUT4 transcript for glucose uptake. The increase of GLUT4 protein and glucose update in both cell lines under the same concentration in turn supported the qPCR data. In other words, steviol and stevioside had the similar functionality as insulin in controlling the glucose level in both cell lines. Other relevant in vitro studies refer to the steviol glycosides’ behaviors and metabolisms for regulating insulin. The insulinotropic action of rebaudioside A was investigated with the aid of insulinoma MIN6 cell-based patch-clamp technique [72]. It was found that rebaudioside A (10-9 M) increased the ATP/ADP ratio dramatically, and rebaudioside A stimulated the insulin secretion from MIN6 cells in a dose- and glucose- dependent manner. Steviol glucuronide, a metabolite of steviol glycosides excreted in human urine, was also verified to be able to stimulate insulin secretion in a dose- and glucose-dependent way in mouse islets of Langerhans [73].
The in vivo animal models such as diabetes Wistar rats induced by streptozotocin were used to investigate the antidiabetic properties of steviol glycosides. Stevioside was tested the most, and few reported the antidiabetic studies of other steviol glycosides (e.g., rebaudiana A [74], rebaudiana B-D [75]. Stevia extracts or steviol glycosides were proven to aid high insulin response without negatively affecting plasma glucagon and glucose [76]. Study shows that an 8-week administration of stevia extracts with various concentrations in albino rats resulted in significant decrease of random/fasting blood glucose and glycosylated hemoglobin (HbA1c) while increase of serum insulin and liver glycogen [77]. The random blood glucose levels of 4-group rats administrated with different amounts of stevia extracts was reduced by over 70% after 8-week experiment. Stevia intake decreased the experimental rats’ body weights from 13.84% to 20.55% although regular and diabetic control groups had 23.27% and 19.48% body weight gain, respectively. It was likely due to the lower metabolism of diet glucose or reduce of rat’s food consumption. By contrast, Das et al. reported that stevia crystal at a high daily dose did not have significant impact on body weight of healthy object, and it reduced blood glucose level non-significantly. However it improved body weight loss and reduced blood glucose level significantly in diabetic animal. The final blood glucose levels of stevia-treated groups were between 12 mmol/L and 14 mmol/L, which were close to Amaryl treated group. Meanwhile, the insulin increase after stevia extract intake is likely due to the fact that stevioside acts on pancreatic b cells and stimulates insulin secretion [79]. The research by-product data from histological analysis also suggested that stevioside should protect alloxan-induced oxidative damage of the pancreatic b cells [80], and further lead to the better synthesis of insulin in the pancreas [81].
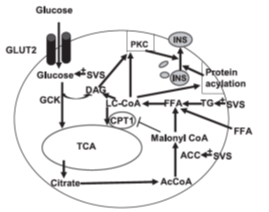
Compared with actual medical treatment, stevia extracts exhibit less effective antidiabetic activities. Therefore, stevia extracts were sometimes used together with medicine for better antidiabetic performance. Abdel-Aal et al. presented an antidiabetic investigation of stevia with saxagliptin, an oral hypoglycemic sold under the brand name Onglyza® [82]. Their research showed that stevia together with saxagliptin produce more significant reduction of fasting blood glucose meanwhile lower total cholesterol (TC), triglyceride (TG), and low-density lipoprotein (LDL). As mentioned before, stevioside was used as model compound for most of the studies including those describing the mechanism of steviol glycosides’ regulation function upon insulin secretion. One mechanism of stevioside’s relieving glucotoxicity in b cells was proposed and shown in Figure 2 [83]. This mechanism showed that stevioside further increased glucose uptake, acetyl-CoA carboxylase (ACC) gene expression and activity, and triglyceride (TG) content when INS- 1E cells were incubated at high glucose condition. Then, stevioside further increased long-chain acyl-CoA which provided substrate for protein acylation, modulated PKC enzyme activity and improved glucose-stimulated insulin secretion. The synthesized byproduct Malonyl CoA inhibited carnitine palmitoyltransferase I (CPT I) and oxidation LC-CoA in mitochondria. This mechanism was then improved by Philippaert et al [79]. Philippaert et al. confirmed that steviol glycosides could enhance pancreatic b-cell function by potentiating TRPM5 channel activity. They emphasized that TRPM5 served as a facilitator of fast glucose-induced Ca2+ oscillation which prompted insulin release. We noticed that steviol glycosides served more as an enhancer while chemical medical treatments such as sulfonylureas could actually block KATP channels directly, push Ca2+ signaling and insulin secretion independent of glucose transport.
2.4 Anti-Inflammation
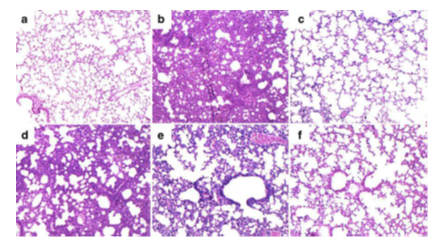
Acute or chronic inflammations attack us frequently during daily life. Acute inflammation reflects our immediate immune response to an injury where manifestations can be warmth, swelling or pain around tissues. When we are injured, our immune system secrets white blood cells to the injured surround and initiates the internal protection mechanism. After the immune system constantly generates white blood cells and chemical messengers, the holistic medical condition becomes chronic inflammation. It has been reported that chronic inflammation is associated with multiple severe health threats such as heart disease, diabetes, cancer, arthritis, and bowel diseases [84]. Sourcing natural therapeutics confronting inflammation becomes useful to provide patients with affordable medical treatments to alleviate sufferings. Studies show promising anti-inflammatory profiles of crude stevia extracts, steviol glycosides, and other extracted compounds. Table 3 presents the selected reports of anti-inflammatory activities of steviol glycosides based on different experimental models. Those experimental models refer to in vitro cell models and in vivo animal models. Those cell models for quantifying steviol glycosides’ anti-inflammatory activities are similar to each other despite of different cell lines. One example is quantifying steviol glycosides’ anti-inflammatory activities by incubating lipopolysaccharide (LPS)- stimulated THP-1 cells with the test compounds [85]. The results showed that stevioside (1 mM) significantly suppressed the release of multiple pro-inflammatory cytokines such as tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and interleukin-1β (IL-1β) in LPS-stimulated THP-1 cells without placing any direct toxic effect. Only stevioside induced TNF-α, IL-1β, and nitric oxide release in unstimulated THP-1 cells. Li et al. then confirmed that stevioside could inhibit the expression of TNF-α, IL-6, and IL-1β in LPS stimulated RAW264.7 cells in a dose-dependent manner [86]. The experimental records of in vivo animal models are more sporadic than in vitro experiments due to the complexity of inflammatory stimulation, however the relevant observations should be more comprehensive and direct. Early, Mizushina et al. synthesized isosteviol, a hydrolysis product of stevioside, and 11 derivative compounds by microbial transformation and chemical conversion [87]. They found that 500 μg isosteviol could cause a 53% reduction in TPA (12-O-tetradecanoylphorbol-13-acetate)-induced inflammation in mouse ear. More recently, Nie et al. evaluated the anti-inflammatory activities of stevioside in LPS-induced acute lung injury in male BALB/c mice along with positive control dexamethasone [88]. They observed that stevioside could reduce the inflammation cells (neutrophils and macrophages) and largely alleviated the LPS-induced histological lesions including collapse of air alveoli, emphysematous and pulmonary congestion. Figure 3 displays the histological changes in lung tissue from LPS-induced ALI mice treated with stevioside. Figure 3b showed feature histological changes in LPS-induced ALI mice including areas of inflammatory infiltration, focal area of fibrosis with collapse of air alveoli, thickening of alveolar wall and pulmonary congestion. Such histological morphologies were largely relieved by 12.5 to 50 mg/kg stevioside (Figure 3d-f) and 5 mg/kg dexamethasone treatment (Figure 3c).
Stevioside is the mostly-investigated stevia compound with anti-inflammatory properties (Nie et al., 2012; Wang, Song, et al., 2014) while others reported the anti-inflammatory capabilities of crude stevia leaf extract [91], rebaudiana A [92], and other compounds extracted from stevia such as austroinulin [93]. Rebaudiana A could effectively prevent the liver injuries brought by thioacetamide (TAA) in rats where negative changes such as upregulation of α-smooth muscle actin, transforming growth factor-β1, nuclear factor kappaB and downregulation of nuclear erythroid factor 2 could occur [92]. Austroinulin (AI) and 6-O-acetyl-austroinulin (6-OAAI), the non-steviol compounds from stevia extracts were also demonstrated to inhibit NO production and iNOS expression by blocking the activation of STAT1, interferon regulatory factor-3 (IRF3), and NF-kB in LPSstimulated RAW264.7 macrophages [93]. The mechanisms of steviol glycosides’ anti-inflammatory activities are complicated, which depend on different reasons. However, it is reasonably speculated that steviol glycosides should participate in inhibiting the inflammation-promoting critical pathways such as regulation of NF-kB, activation of ERK1/2, JNK, and p38 MAPK [86], and inhibition of DNA polymerases or topoisomerase [87]. Both in vitro and in vivo anti-inflammatory investigations of steviol glycosides were often performed together to give comprehensive mechanism analysis. The antiinflammation of steviol glycosides could have positive impacts on treating liver disease, obesity, and type 2 diabetes together. The effects of stevioside, rebaudiana A, and steviol on liver steatosis and hepatic effects of lipotoxicity using a mouse model, and insulin resistance were systematically investigated [94]. Those tested compounds could improve glucose metabolism, fat catabolism, bile acid metabolism, and lipid storage and transport despite of different impacts on glucose and lipid metabolism, inflammation and oxidative stress, therefore, those steviol glycosides helped attenuate hepatic steatosis giving us confidence in their potentials of mild treatments to those diseases.
Figure 3: Effects of stevioside on histopathological changes in lung tissues in LPS-induced ALI mice. Mice were given an intraperitoneal injection of stevioside (12.5, 25, and 50 mg/kg) 1 h prior to an i.n. administration of LPS. Lungs (n=4–6) from each experimental group were processed for histological evaluation at 7 h after LPS challenge. Representative histological changes of lung obtained from mice of different groups. a Control group, b LPS group, c LPS+DEX group, d LPS+Stevioside (12.5 mg/kg) group, e LPS+Stevioside (25 mg/kg) group f LPS+Stevioside (50 mg/kg) group (Hematoxylin and eosin staining, magnification 200Å~) (Reprint with permission).
|
Year |
Compounds |
Methods |
Results |
Reference |
|
2005 |
Isosteviol and other 11 derivatives |
Application to mouse with TPA-induced inflammation |
Isosteviol inhibited mammalian DNA polymerases and human DNA topoisomerase II; caused a marked reduction in TPA-induced inflammation. |
[87] |
|
2006 |
Stevioside, steviol |
Incubating LPS-stimulated THP-1 cells with stevioside, steviol |
Stevioside (1 mM) significantly inhibited LPS-induced release of TNF-α and IL-1β and slightly reduced nitric oxide release but steviol at 100 μM did not. |
[85] |
|
2012 |
Stevioside |
Incubating LPS-stimulated RAW264.7 cells with stevioside |
Stevioside inhibited the expression of tumor necrosis factor- α, interleukin-6, and interleukin-1β; suppressed LPS-induced NF-κB activation, IκBα degradation, phosphorylation of ERK, JNK, and P38. |
[86] |
|
2013 |
Stevioside and steviol |
Incubating LPS-stimulated Caco-2 cells with stevioside or steviol |
Stevioside and steviol attenuated LPS-induced proinflammatory cytokines such as TNF-α, IL-1β, and IL-6. |
[166] |
|
2014 |
Stevioside |
Intraperitoneal injection of mouse with S. aureus-induced mastitis |
Stevioside significantly decreased the inflammatory cell penetration and the levels of TNF-α, IL-1β, and IL-6 and the expression of their mRNA. |
[167] |
|
2017 |
Stevia extract and stevioside |
Oral administration of Male BALB/cN mice with cisplatin (CP)-induced kidney injury |
Stevia extract and stevioside relieved CP nephrotoxicity by reducing oxidative stress, inflammation, and apoptosis which involves ERK1/2, STAT3, and NF-κB depression. |
[123] |
Table 3: Anti-inflammatory activities of steviol glycosides.
2.5 Antimicrobial
Infections of pathogen microorganism post severe threats to the public health. Infectious diseases could result in more lethal events than other causes. Great efforts have been put forward to battle against those infections. Those infectious threats are brought up by bacteria, viruses, and fungi which can be found almost everywhere. As part of multi-functions exploration, the antimicrobial activities of stevia extracts have been meticulously examined and compared. Stevia leaf extracts were reported with certain antimicrobial capabilities, nevertheless, their antimicrobial activity is limited compared with their other functions such as antioxidant activity. Previously, the antimicrobial potentials of açaí (Euterpe oleracea), stevia (Stevia rebaudiana Bertoni), and ginseng (Panax quinquefolius L.) were assessed under various process treatments [95]. Ginseng was the most effective antimicrobial agent with thermal, microwave, and pulsed electric field treatments, and microwave-treated ginseng extracts achieved the largest inhibition zone against E. coli O157:H7 in the study (15.11 ± 0.11 mm). As comparison, stevia displayed lower in vitro antimicrobial effectiveness at all concentrations against non-sporeforming and sporeforming bacterias, which could be improved with the aid of pulsed electric field treatment. In another report, the efficacy of Equisetum arvense and stevia extracts against 6 mycotoxigenic moulds at different concentrations were investigated and compared [96]. 3% E. arvense extract effectively suppressed the growth of the mycotoxigenic fungi under all conditions, however, 2-3% stevia could not produce significant fungi depress. Change extracting solvents can alter the antimicrobial activities of the end stevia products. The solvent effects on the antifungal and antibacterial activities of stevia extracts were assessed against 10 microorganisms such as E. coli, B. subtilis, and P. aeruginosa [97]. Different solvents resulted in different antimicrobial activities of the end stevia extracts. Only a few strains could be decreased more effective by petroleum ether-aided stevia extract, the optimum solvent, than standard streptomycin and cotrimazole. The antimicrobial function of stevia extracts has somewhat strain-selectivity.
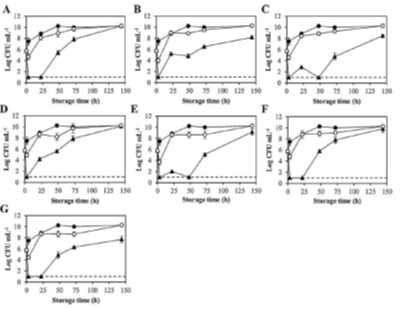
Stevia extracts have limited antimicrobial capacity, hence we view it as their secondary function. If needed, we can enhance its comprehensive antimicrobial activity by using approaches such as solvent, physical, and chemical treatments. Those approaches bring more or less inevitable changes to the stevia products. Residue solvents might occur in end stevia extracts while additional physical processes interact with stevia’s other functions. Among those methods, chemical modification is the most direct approach to alter the antimicrobial capacity of stevia compounds. Sharipova et al. firstly synthesized the glucuronosyl derivative compounds of the glycoside steviolbioside from stevia extract [98]. The synthesized steviolbioside displayed selective bacteriostatic and bactericidal activity against S. aureus ATCC 209p with minimum inhibitory concentration of 62.5 μg/mL. Compared with chemical synthesis, physical processes are milder in condition setup. High pressure treatments of stevia aqueous extracts were studied by using response surface methodology to enable maximum retention nutritional properties [60]. The research group demonstrated that the optimum treatment of 453 MPa for 5 min with a 2.5% (w/v) of stevia could inactivate more than 5 log cycles of L. monocytogenes and reach highest inactivation of polyphenoloxidase (PPO) and peroxidase (POD). The stevia extracts are complex materials which are composed of different compounds, which makes it challenge to identify their antimicrobial attributes. 18 functional compounds such as catechin, caffeic acid, and quercetin could be identified in stevia water and methanol-water (50/50 v:v) extracts [99]. A small amount of essential oils was also retained during extraction. Among all the extract samples, stevia methanol-water extract was the most effective in inhibiting various bacteria strains, especially Escherichia coli and Staphylococcus aureus. Even drying methods of stevia leaves had impacts on the post extracts’ antioxidant, antimicrobial and anti-inflammatory capabilities [100]. Figure 4 displays the antimicrobial activities of stevia extracts against Listeria innocua growth. Those stevia extracts were produced under different drying methods including freeze drying, convective drying, vacuum drying, and etc. All stevia leaf extracts inhibited Listeria innocua growth in a variable but significant manner while none of the tested extracts was able to inhibit Saccharomyces cerevisiae growth. The inhibition behaviors of stevia leaf extracts were similar to each other.
Figure 4: Antimicrobial activity of Stevia extracts against Listeria innocua growth. (A) Freeze drying, (B) Convective drying, (C) Vacuum drying, (D) Microwave drying, (E) Infrared drying, (F) Sun drying and (G) Shade drying. (●) No Stevia addition, (▲ ) 0.1 mg/mL, (▲) 1 mg/mL, (——) Detection limit (Reprint with permission).
3. Safety
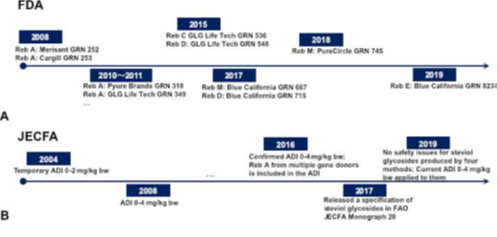
Stevia leaf extracts or steviol glycosides have been approved for food use for more than a decade in different regions and countries including US, China, South Korea, and so forth. Historically, authorities or institutes reviewed and revised regulations pertinent to steviol glycosides on different timelines. US Food and Drug Administration (FDA) has issued and revised Import Alert #45-06 since 1995 which provides the criteria to determine whether steviol glycosides or their food products may be imported into the United States. US FDA evaluated over 50 GRAS notices through its GRAS “Generally Regarded as Safe” Notification program for various steviol glycosides with no question letter between 2008 and 2018 [101]. Figure 5A displays the representative milestones of steviol glycosides’ FDA GRAS approvals from different suppliers. We can see that early steviol glycosides’ GRAS applications were focused on rebaudioside A. Those rebaudioside A GRAS notices reflect the historical research and policy-making of steviol glycosides from different organizations. Cargill commented on the historical research gaps and their resolutions of steviol glycosides in GRAS notice 253 involving lack of consistent food-grade specification, lack of toxicity demonstration, safety for consumers with diabetes and low blood pressure, and shortage of adverse or physiological effects of stevioside or steviol on kidney [102]. Numerous clinical studies were reviewed to assess the tolerability of purified steviol glycosides and stevia extracts with single- or repeat- dose administration. For instance, no fluctuations of blood glucose or insulin were observed in hypertensive subjects treated with stevioside at daily dose of 1500 mg during a 2-year randomized clinical study (RCT) [103]. By contrast, oral stevioside significantly reduced systolic and diastolic blood pressure compared with placebo in this 2-year study for patients with mild hypertension. But no clinical significance on blood pressure were observed in a 3-day study in which healthy subjects received 750 mg stevioside daily [104]. In addition to Cargill, GLG Life Tech Ltd. also contributed to the stevia’s regulatory enrichment. In GRAS notice 349, GLG Life Tech Ltd. reported an independent safety evaluation of their BlendSureTM preparation which was composed of highly-purified rebaudioside A and stevioside [105]. GLG Life Tech thoroughly reviewed the regulatory history of stevia & stevia-derived sweeteners supported by multiple sets of safety data including absorption, distribution, metabolism & excretion (ADME) studies, acute toxicity studies, subchronic toxicity studies, clinical studies and other reports in human. Among those ADME studies, Wheeler et al. found that rebaudioside A and stevioside shared similar metabolic pathways with steviol glucuronide excreted mostly in the urine and steviol in the feces [106]. Their pharmacokinetics analysis indicated a rapid first-pass conjugation of steviol prior to urinary excretion. Steviol glucuronide was a metabolite found in blood and urine while steviol was detected only in feces. Rebaudioside A or stevioside was converted in the colon to steviol, which was then quickly converted to steviol glucuronide. Later on, GLG Life Tech obtained the GRAS approval for rebaudioside C and rebaudioside D in 2015 [107,108], while the GRAS for rebaudioside M and rebaudioside E were secured by Blue California in 2017 and 2019, respectively [109,110] (Figure 5A).
Similar but differently, Figure 5B shows the selected milestones of steviol glycosides’ evaluations and announcements from the Joint FAO/WHO Expert Committee on Food Additives (JECFA). From Figure 5B, we see that JECFA established a temporary ADI (acceptable daily intake) of 0-2 mg/kg bw for steviol glycosides at its 63rd meeting in 2004 [111]. After assessing clinical studies with steviol glycosides, JECFA removed the temporary status of the ADI and updated the ADI to 0-4 mg/kg bw/day for steviol glycosides at the 69th meeting in 2008 [112]. The new ADI was established based on 4 new toxicological studies with rebaudioside A in experimental animals and clinical trials. The results suggested no negative effects when taken at 4 mg/kg per day for up to 16 weeks by individuals with type 2 diabetes and those with low-normal blood pressure for 4 weeks. JECFA reaffirmed the updated ADI 0-4 mg/kg bw/day at the 82nd meeting in 2016 [113]. The committee considered new information to support the safety of rebaudioside A produced by fermentation in a strain of the yeast Yarrowia lipolytica and hydrolysed products of steviol glycosides by colonic microflora. The report of the 82nd meeting of JECFA comprehensively measured dietary exposure conducted by the European Food Safety Authority (EFSA), Food Standards Australia New Zealand (FSANZ), and other studies [114]. With years of evaluation, JECFA then released an official specification of steviol glycosides in FAO JECFA Monograph 20 in 2017 covering the authorized analytics and chemical structures [115]. In 2012, the European Union (EU) approved stevia extracts containing at least 95% steviol glycosides as food additives (E960) for food applications [116]. 75% of the final products should consist of stevioside and/or rebaudioside A. The amendment of specifications concerning steviol glycosides was submitted in the late 2013 [117]. In the amendment, rebaudioside M was first requested to be added to the list of permitted steviol glycosides with the updated chemical name, molecular weight, CAS number, and other relevant information. The previously-mentioned minimum quantity of 75% stevioside and/or rebaudioside A is then removed. In Oceania, FSANZ approved Cargill’s application A1037 to increase the allowed maximum permitted level (MPL) of steviol glycosides in ice cream, water based flavored drinks, brewed soft drink, formulated beverages in 2011 [118]. Similarly, FSANZ established an ADI for steviol glycosides of 4 mg/kg bw/day (expressed as steviol equivalents) based on the toxicological and other relevant data. After considering the public health, safety, and other information, FSANZ approved the MPL of steviol glycosides for 6 beverage sub-categories of with precise toxicology records. According to their approval, fruit and vegetable juice products are allowed with MPL = 125 mg/kg while fruit and vegetable preparations including pulp have MPL = 208 mg/kg. The approval of application A1037 provides solid guidance on the product development of stevia-containing beverage products in Australia and New Zealand in detail.
In addition to global authorities’ regulations, recent researches concerning about steviol glycosides’ safety and toxicity were accomplished by different research groups. Those researches covered the assessments of cytotoxicity, genotoxicity, nephrotoxicity, and etc. Sharif et al. confirmed none cytotoxicity and genotoxicity of stevioside on human colon carcinoma cell HCT 116 and human colon derived CCD18Co myofibroblast cell lines [119]. They found that only the highest concentration (200 μM) caused 20% to 30% cell death on CCD18Co and HCT 116 cells, and such high concentrations on CCD18Co and HCT 116 cell line would not cause DNA damage. It is worth-mentioning that genotoxicity refers to the properties of chemicals that can damage the genetic information within cells, which further develop into severe diseases such as cancer. Genotoxicity study should be considered prior to using new compounds, and steviol glycosides are no exception. Steviol glycosides might elicit in vitro genotoxic effects. Steviol might be mutagenic in cultured mammalian cells. Steviol could be negative in standard Ames assays but positive in others [120]. Therefore, genotoxicity studies continued to ease those worries. Zhang et al. updated the in vivo genotoxicity assay results for steviol, stevioside, and rebaudioside A against the early negative assay data [121]. Their maximum experimented rats’ oral administration could reach up to 300 times of the recommended daily intake. Oral administration with dietary concentrations of 1.04%, 2.08% and 3.12% for 90 days did not cause significant behavioral, hematological, clinical, or histopathological changes in rats. The cholesterol reduction was observed in female animals of high dose group, which was due to the lipid-lowering effect of chlorogenic acid. The major summaries from a comprehensive review find no evidence of genotoxicity activity of stevioside and steviol [122]. Those steviol compounds have not been shown to interact directly with DNA or cause genotoxic jeopardy. It is convincing that those steviol substances do not exert a genetic damage risk following human consumption. Additionally, the impact of stevia leaves extract and stevioside on cisplatin nephrotoxicity was investigated by using Male BALB/cN mice model [123]. The research group found that stevia extract treatment effectively suppressed the expression of oxidative stress markers such as p53, caspase-9, p21, Bax together with antioxidant cytoprotective enzyme HO-1. Besides, stevia extract and stevioside attenuated cisplatin-induced apoptosis and expression of ERK1/2 and STAT3 in a dose-dependent manner. Stevioside was reported to have similar renoprotective activity as stevia extract, which could provide us with some supplement ideas in the prevalence of metabolic disorders such as type II diabetes and obesity. Even new approaches such as inverse virtual screening was developed for a rapid safety evaluation of stevia glycosides on farnesoid X receptor. The presumably safe profile of known compounds such as steviol were critically discussed [124].
4. Applications
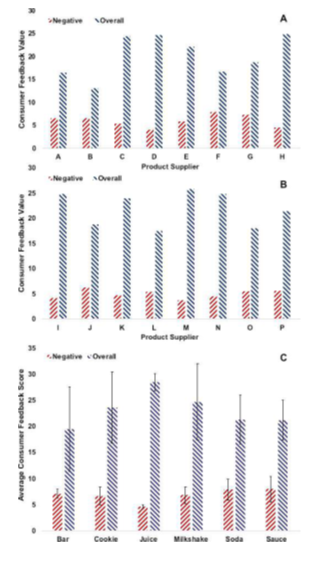
Stevia extracts or stevia glycosides have commercialization values in food and nutritional industries. From product perspective, Mintel once tracked over 200 launches of products containing stevia extracts under multiple brands in 2012, a significant soar compared with previous records [125]. Among those launches, chocolate confectionery occupied 50 launches with soft drinks 45. Recently, Coca-Cola debuted its ready-to-drink (RTD) product, Fuze tea in Europe in early 2018 [126]. This beverage product helped the company escape sugar tax due to the use of stevia as sweetener. From safety perspective, sugar reduction has been frequently advocated for processed beverages and foods because excessive consumption of sugar is associated with the health risks of obesity, cardiovascular disease (CVD), metabolic syndrome, type 2 diabetes, and dental decay [127,130,131]. Appropriate applications of stevia extracts in the developments of foods and beverages ensure us an effective solution as part of sugar reduction strategy. It is worth-mentioning that stevia usage level in products should be optimized in terms of taste, nutrition, and safety. Low dosage is commonly recommended as a good start in products such as beverages while high dosage should be reviewed when used in other categories such as bakery. Meanwhile, Kamarulzaman et al. studied the consumer acceptance of stevia and factors influencing the acceptance of stevia-based products [132]. Their research showed that consumers showed positive attitudes toward stevia-based products. They emphasized that it was critical for business to continuously enhance the awareness of stevia among consumers by appropriate education and propaganda. A well-designed promotion helps enhance consumer recognition of stevia and its products. In order to assess product quality from customers’ perspective, we carried out sentiment analysis of customers’ online reviews for selected stevia ingredients and stevia-containing products. The sentiment analysis technique enables us to empower product applications based on the meticulous scrutiny of users’ opinions [133]. Its major advantage lies in collection of the product reviews in real time. The current customers’ reviews of stevia ingredients and products were extracted with the aid of Web Scraper Chrome extension for further analysis [134]. Figure 6 displays the consumer feedback scores of representative stevia/xylitol ingredients and average consumer feedback scores of selected products formulated with stevia. The data provide us with qualitative customers’ feedbacks of stevia-relevant products on the current market. 50% of the sampled stevia ingredients had customer feedback scores higher than 20, whose situation is similar to the customer feedback profile of the sampled xylitol ingredients. Meanwhile we observed that the negative scores of stevia ingredients were slightly higher than those of xylitol ingredients. It is probably due to the unique taste profile of stevia described as a potent sweet taste at the front and a bitter or liquorice-like aftertaste when consumed more (Part 2.1. Sweetening). Stevia became a topic or event for those consumers while less close to table sugar. We analyzed the consumers’ reviews for stevia-containing products to understand stevia’s applications from consumers’ perspective as well. As we see, stevia has been used as sweetener in multiple food categories including bar, cookie, juice, milkshake, soda, and sauce. (Figure 6C). We found that almost all the product categories with stevia obtained average consumer feedback scores higher than 20, which suggested stevia a compatible ingredient in various food matrixes. Besides, we observed high feedback consistence in soda and large feedback variations in bar, cookie, and milkshake which is probably attributed to the dynamics of consumer behaviors. We then confirmed that stevia helped sweetening food products and those food products were acceptable by consumers.
Prior to be commercialized as sellable products, stevia product developments have been researched in laboratories as well. Not every stevia product research is identical or similar with each other. Stevia has been utilized to produce beverage, dairy, bakery, and confectionery goods. As mentioned-previously, it is not surprising that stevia, as a potent sweetener, is compatible in highly-sweet products. Stevia is not only a sweetener but also a table sugar substitute. We understand that consumers could ignore the sensorial satisfaction and accept some negative sensory changes in a healthier diet [135]. Researchers noticed that stevia could effectively protect the bakery products’ sweet profile during thermal process. Developing oatmeal cookies with aqueous extract of Stevia rebaudiana Bertoni (var. Morita II) is a good example [136]. The oatmeal cookies were developed by replacing 25% to 100% sucrose with stevia extract. The sensory acceptance of oatmeal cookies formulated with stevia was close but lower than those cookies with sucrose. Regarding purchasing intention, the cookies which incorporated 25% to 50% of the stevia extract were predominantly qualified as “May buy”. And we cannot completely diminish sweet taste since it plays a key role in regulating the metabolism of carbohydrates by triggering a series of anticipatory physiological responses [127]. Hence, it is worth formulating products with the combination of stevia and table sugar from taste and cost perspective. A latter-developed gluten-free cookie with almond flour and stevia could retain 43–73% more of total phenolics than those with rice flour and sucrose [137]. Almond flour and stevia produced finished goods with more acceptable physicochemical, nutrition and sensorial properties’ perspective. Another example in bakery is muffin products composed of stevia and cocoa powder [138]. Developers demonstrated that 50% stevia replacement of sucrose enabled muffins to have textures similar to a control muffin, while further increase of stevia could negatively affect the firmness, volume, and texture. Besides, sugar replacement significantly reduced in vitro predictive glycemic response of muffins by up to 55% of the control.
Beverage is another large category for developing stevia-containing products. In low viscous product system, stevia not only help sweeten the drinks but also somewhat retain or enhance the nutrients abundance. The green stevia extract enabled chokeberry juice to retain vitamin C content increased up to two times, higher total phenolics, and better antioxidant capacity compared with original juice [139]. However, the bioactive compounds and antioxidant capacity of chokeberry juice were significantly reduced during shelf life study with the exception of steviol glycosides content. With the functions mentioned previously, stevia-containing beverage helps reduce the risk of cardiovascular diseases and pathophysiological situations such as obesity and diabetes mellitus when consuming appropriate quantity. The influence of stevia and sucrose on phytochemical content of a citrus maqui beverage was monitored and compared [140]. The physicochemical attributes such as pH, titratable acidity, total soluble solids, and color remained stable during shelf life test nevertheless the flavanones of the stevia-containing citrus maqui beverages exhibited a higher loss under light conditions than sucrose beverage. Stevia extract was even incorporated into Russian currant, berry blended wine products [141]. The blended wine based on black currant and blackberry was more replete with vitamins, minerals, and organic acids. There were two times more thiamine as vitamin B1 and lactoflavin as vitamin B2 in the blended wines than in the control specimen of the unblended black currant or blackberry wines.
Stevia was also added into dairy products such as milk, yoghurt, and others, however it should be treated differently since dairy products are dynamic edibles composed of fat, proteins, minerals, and carbohydrates. Adding stevia into those systems likely change the original balance of ingredient-ingredient interactions, which sometimes deteriorates the quality of dairy products. For instance, replacing sugar with stevia significantly could accelerate sedimentation and texture shinning of chocolate milk [142]. The optimized formula containing sucrose/stevia at 1:1 ratio and 6% inulin produced the lowest precipitation while having a non-significant texture deviation from the control. Other sucrose/stevia ratio such as 75:25 was also reported to achieve the best mean liking score of stevia-containing fruit milk shakes [143]. Monk fruit can be used together with stevia in sugar reduce milk products. Mahato et al. confirmed a 50% sugar reduction in chocolate milk using stevia and monk fruit, which reflected their active scientific response to ease the health concerns from high-sugar foods and beverages [144]. Those health concerns about high sugar intake were raised in considerable epidemiological evidences [145,146]. As a matter of fact, such sweetness synergy was helpful to mask the bitter, metallic aftertaste and enhance the overall sensory attributes of the product compared with the control. A popularity investigation of skim chocolate milk with stevia and monk fruit was conducted by applying power function and 2-alternative forced choice studies [147]. The chocolate milk solely-sweetened by those non-nutritive sweeteners was less popular among young adults compared with milk sweetened by sucrose. The researchers accidentally found that label-conscious parents preferred chocolate milk with non-nutritive sweeteners for their children while traditional parents would pick sucrose-sweetened chocolate milk. In addition to milk, stevia was formulated and investigated in other dairy products such as yoghurt, dessert, and ice cream [148–150]. Stevia-containing chocolate ice-cream was an interesting low-calorie dairy food product among them [149]. The sensory panelists liked the agreeable texture and mouthfeel of the ice cream samples with stevia and cocoa to others. Such better taste experience was associated with the highest overrun ratio (20.17%) and longest first melting time (3460 s) of sample with cocoa and stevia.
Figure 6: Consumer feedback scores of representative (A) Stevia, (B) Xylitol ingredients, and average consumer feedback scores of (C) selected food products formulated with Stevia; The legends in Figures A, B, and C represent overall and negative scores of product online reviews ranging from -100 to +100.
5. Conclusion
This article reviewed major functions of herb Stevia rebaudiana extract including but not limited to sweetening, antioxidant, antidiabetics, anti-inflammation, and antimicrobial. The sweetness indexes of major steviol glycosides such as stevioside, rebaudioside A-E, and dulcoside A, the sweetening compounds in stevia, were summarized for records and comparison. The total phenolic contents and representative compounds of distinct stevia extracts from multiple plant locations were discussed, and in vitro, in vivo antioxidant capacities of those stevia extracts were quantified by assays such as DPPH, ORAC, and ABTS. Stevia extracts’ antidiabetics and anti-inflammation were demonstrated based on the empirical experiments with different cell lines such as Caco-2 and 3T3-L1 cells and animal models such as mouse with cisplatin-induced kidney injury and diabetes Wistar rats induced by streptozotocin. Mechanisms supporting stevia’s functions such as antidiabetics were put forward after analysis. The safety of steviol glycosides starting from rebaudioside A was evaluated and approved by multiple global authorities such as US Food and Drug Administration (FDA), Joint FAO/WHO Expert Committee (JECFA), and Food Standards Australia New Zealand (FSANZ). Sentiment analysis of stevia ingredients and stevia containing products was performed to help understand the stevia applications from customer perspective. With a unique potent sweet taste and liquorice-like aftertaste, stevia can help sweetening food products in the categories of beverage, bakery, dairy, and confectionery. Those stevia-containing food products were acceptable by consumers or researchers. However, stevia is still not identical to sucrose or table sugar that we use during daily life.
Acknowledgements
We thank Professor Dr. Arthur A. Boni’s proof-reading of this review manuscript and providing helpful comments.
References
- Lewis WH, Rawat AS, Pharswan AS, et al. Notes on economic plants. Econ. Bot 46 (1992): 336-340.
- Udo K. Einfluss von Bewässerung und Schnittfolge auf den Ertrag von Stevia rebaudiana in Südspanien. Göttinger Beiträge Zur Land- Und Forstwirtschaft Den Tropen Und Subtrop 84 (1993): 4-108.
- Anbazhagan M, Kalpana M, Rajendran R, et al. In vitro production of stevia rebaudiana Bertoni. Emirates J. Food Agric 22 (2010): 216-222.
- Gasmalla MAA, Yang R, Amadou I, et al. Nutritional composition of Stevia rebaudiana bertoni leaf: Effect of drying method. Trop. J. Pharm. Res 13 (2014): 61-65.
- Vanek T, Nepovím A, Valícek P. Determination of stevioside in plant material and fruit teas. J. Food Compos. Anal 14 (2001): 383-388.
- Jentzer JB, Alignan M, Vaca-Garcia C, et al. Response surface methodology to optimise Accelerated Solvent Extraction of steviol glycosides from Stevia rebaudiana Bertoni leaves. Food Chem 166 (2015): 561-567.
- Rouhani M. Modeling and optimization of ultrasound-assisted green extraction and rapid HPTLC analysis of stevioside from Stevia Rebaudiana. Ind. Crops Prod 132 (2019): 226-235.
- Ameer K, Bae SW, Jo Y, et al. Optimization of microwave-assisted extraction of total extract, stevioside and rebaudioside-A from Stevia rebaudiana (Bertoni) leaves, using response surface methodology (RSM) and artificial neural network (ANN) modelling. Food Chem 229 (2017): 198-207.
- Ameer K, Chun BS, Kwon JH. Optimization of supercritical fluid extraction of steviol glycosides and total phenolic content from Stevia rebaudiana (Bertoni) leaves using response surface methodology and artificial neural network modeling. Ind. Crops Prod 109 (2017): 672-685.
- Puri M, Sharma D, Barrow CJ. Enzyme-assisted extraction of bioactives from plants. Trends Biotechnol 30 (2012): 37-44.
- Philippe RN, De Mey M, Anderson J, et al. Biotechnological production of natural zero-calorie sweeteners. Curr. Opin. Biotechnol 26 (2014): 155-161.
- Kovylyaeva GI, Bakaleinik GA, Strobykina IY, et al. Glycosides from Stevia rebaudiana. Chem. Nat. Compd 43 (2007): 81-85.
- Woelwer-Rieck U, Lankes C, Wawrzun A, et al. Improved HPLC method for the evaluation of the major steviol glycosides in leaves of Stevia rebaudiana. Eur. Food Res. Technol 231 (2010): 581-588.
- Yamasaki K, Tanaka O. Application of 13C NMR Spectroscopy to Chemistry of Natural Glycosides: Rebaudioside-C, a New Sweet Diterpene Glycoside of Stevia rebaudiana. Chem. Pharm. Bull (1977).
- Sakamoto I, Yamasaki K, Tanaka O. Application of 13C NMR Spectroscopy to Chemistry of Plant Glycosides: Rebaudiosides-D and -E, New Sweet Diterpene- Glucosides of Stevia rebaudiana bertoni. Chem. Pharm. Bull (1977).
- Kujundzic D, Jambrak AR, Vukušic T, et al. Near-infrared spectroscopic characterization of steviol glycosides extracted from Stevia rebaudiana Bertoni using high-power ultrasound and gas-phase plasma. J. Food Nutr. Res 56 (2017): 109-120.
- Gardana C, Simonetti P. Determination of steviol glycosides in commercial extracts of Stevia rebaudiana and sweeteners by ultra-high performance liquid chromatography Orbitrap mass spectrometry. J. Chromatogr. A 1578 (2018): 8-14.
- Soejarto DD, Kinghorn AD, Farnsworth NR. Potential sweetening agents of plant origin. III. organoleptic evaluation of Stevia leaf herbarium samples for sweetness. J. Nat. Prod 45 (1982): 590-599.
- Gerwig GJ, te Poele EM, Dijkhuizen L, et al. Stevia Glycosides: Chemical and Enzymatic Modifications of Their Carbohydrate Moieties to Improve the Sweet- Tasting Quality. in: D. Baker (Ed.), Adv. Carbohydr. Chem. Biochem (2016): 1-72.
- Gardana C, Simonetti P, Canzi E, et al. Metabolism of stevioside and rebaudioside A from Stevia rebaudiana extracts by human microflora. J. Agric. Food Chem 51 (2003): 6618-6622.
- Chattopadhyay S, Raychaudhuri U, Chakraborty R. Artificial sweeteners - A review. J. Food Sci. Technol 51 (2014): 611-621.
- Cardello HM, Da Silva MA, Damasio MH. Measurement of the relative sweetness of stevia extract, aspartame and cyclamate/saccharin blend as compared to sucrose at different concentrations. Plant Foods Hum. Nutr 54 (1999): 119-129.
- Zhao X, Yao W. A method for evaluating the quality of stevia extract, CN 104049047 B (2015).
- Abelyan VA, Balayan AM, Ghochikyan VT, AA, et al. Transglycosylation of stevioside by cyclodextrin glucanotransferases of various groups of microorganisms. Appl. Biochem. Microbiol 40 (2004): 129-134.
- Chranioti C, Chanioti S, Tzia C. Comparison of spray, freeze and oven drying as a means of reducing bitter aftertaste of steviol glycosides (derived from Stevia rebaudiana Bertoni plant) - Evaluation of the final products. Food Chem 190 (2016): 1151-1158.
- DuBois GE, Dietrich PS, Lee JF, et al. Diterpenoid Sweeteners. Synthesis and sensory evaluation of stevioside analogues nondegradable to steviol. J. Med. Chem 24 (1981): 1271-1277.
- DuBois GE, Stephenson RA. Diterpenoid Sweeteners. Synthesis and sensory evaluation of stevioside analogues with improved organoleptic properties. J. Med. Chem 28 (1985): 93-98.
- Nguyen TT, Seo C, Kwak SH, et al. Enzymatic production of steviol glucosides using β-glucosidase and their applications. in: Enzym. Food Biotechnol. Prod. Appl. Futur. Prospect (2019): 405-418.
- Ibrahim MA, Rodenburg DL, Alves K, et al. Minor diterpene glycosides from the leaves of stevia rebaudiana. J. Nat. Prod 77 (2014): 1231-1235.
- Darise M, Mizutani K, Kasai R, et al. Enzymic transglucosylation of rubusoside and the structure-sweetness relationship of steviol-bisglycosides. Agric. Biol. Chem 48 (1984): 2483-2488.
- Chaturvedula VS, Prakash I. Acid and alkaline hydrolysis studies of stevioside and Rebaudioside A. J. Appl. Pharm. Sci 1 (2011): 104-108.
- Chaturvedula VS, Klucik J, Upreti M, et al. Synthesis of ent-kaurane diterpene monoglycosides. Molecules 16 (2011): 8402-8409.
- Kamiya S, Konishi F, Esaki S. Synthesis and Taste of Some Analogs of Stevioside. Agric. Biol. Chem 43 (1979): 1863-1867.
- Purkayastha S, Markosyan A. Glucosylated steviol glycoside as a flavor modifier. US 9107436 B2 (2015).
- Yu X, Yang J, Li B, et al. High efficiency transformation of stevioside into a single mono-glycosylated product using a cyclodextrin glucanotransferase from Paenibacillus sp. CGMCC 5316. World J. Microbiol. Biotechnol 31 (2015): 1983-1991.
- Laokuldilok N, Thakeow P, Kopermsub P, et al. Optimisation of microencapsulation of turmeric extract for masking flavour. Food Chem 194 (2016): 695-704.
- Tatar F, Kahyaoglu T. Microencapsulation of Anchovy (Engraulis encrasicolusL.) Oil: Emulsion Characterization and Optimization by Response Surface Methodology. J. Food Process. Preserv 39 (2015): 624-633.
- Chranioti C, Chanioti S, Tzia C. Microencapsulation of steviol glycosides (Stevia rebaudiana Bertoni) by a spray drying method - Evaluation of encapsulated products and prepared syrups. Int. J. Food Stud 4 (2015): 212-220.
- Zhang SQ, Kumar A, Kutowy O. Membrane-based separation scheme for processing sweeteners from stevia leaves. Food Res. Int 33 (2000): 6176-20.
- Formigoni M, Milani PG, da Silva Avíncola A, et al. Pretreatment with ethanol as an alternative to improve steviol glycosides extraction and purification from a new variety of stevia. Food Chem 241 (2018): 452-459.
- Silverstein TP. Oxidation and reduction: Too many definitions? J. Chem. Educ 88 (2011): 279-281.
- Beyer RE. The role of ascorbate in antioxidant protection of biomembranes: Interaction with vitamin E and coenzyme Q. J. Bioenerg. Biomembr 26 (1994): 349-358.
- Ameer K, Jiang GH, Amir RM, et al. Chapter 33 - Antioxidant potential of Stevia rebaudiana (Bertoni). in: V.R.B.T.-P. Preedy (Ed.), Pathol. Oxidative Stress Diet. Antioxidants, Academic Press (2020): 345-356.
- Bender C, Graziano S, Zimmermann BF. Study of Stevia rebaudiana Bertoni antioxidant activities and cellular properties. Int. J. Food Sci. Nutr 66 (2015): 553-558.
- Barba FJ, Grimi N, Vorobiev E. Evaluating the potential of cell disruption technologies for green selective extraction of antioxidant compounds from Stevia rebaudiana Bertoni leaves. J. Food Eng 66 (2015): 553-558.
- Can Z, Baltas N. Bioactivity and Enzyme Inhibition Properties of Stevia rebaudiana. Curr. Enzym. Inhib 12 (2016): 188-194.
- Tadhani MB, Patel VH, Subhash R. In vitro antioxidant activities of Stevia rebaudiana leaves and callus. J. Food Compos. Anal 20 (2007): 323-329.
- Kim IS, Yang M, Lee OH, et al. The antioxidant activity and the bioactive compound content of Stevia rebaudiana water extracts. LWT - Food Sci. Technol 44 (2011): 1328-1332.
- Yu H, Yang G, Sato M, et al. Antioxidant activities of aqueous extract from Stevia rebaudiana stem waste to inhibit fish oil oxidation and identification of its phenolic compounds. Food Chem 232 (2017): 379-386.
- Shukla S, Mehta A, Bajpai VK, et al. In vitro antioxidant activity and total phenolic content of ethanolic leaf extract of Stevia rebaudiana Bert. Food Chem. Toxicol 47 (2009): 2338-2343.
- Shukla S, Mehta A, Mehta P, et al. Antioxidant ability and total phenolic content of aqueous leaf extract of Stevia rebaudiana Bert. Exp. Toxicol. Pathol 64 (2012): 807-811.
- Karaköse H, Müller A, Kuhnert N. Profiling and quantification of phenolics in stevia rebaudiana leaves. J. Agric. Food Chem 63 (2015): 9188-9198.
- Criado MN, Barba FJ, Frígola A, et al. Effect of Stevia rebaudiana on Oxidative Enzyme Activity and Its Correlation with Antioxidant Capacity and Bioactive Compounds. Food Bioprocess Technol 7 (2014): 1518-1525.
- Hajihashemi S. Physiological, biochemical, antioxidant and growth characterizations of gibberellin and paclobutrazol-treated sweet leaf (Stevia rebaudiana B.) herb. J. Plant Biochem. Biotechnol 27 (2018): 237-240.
- Rao GN. Antioxidant Activity of Stevia (Stevia rebaudianaL.) Leaf Powder and A Commercial Stevioside Powder. J. Food Pharm. Sci 2 (2014): 32-38.
- Ahmad N, Ahmad N, Rab A. Light-induced biochemical variations in secondary metabolite production and antioxidant activity in callus cultures of Stevia rebaudiana (Bert). J. Photochem. Photobiol. B Biol 64 (2016): 807-811.
- Sharma OP, Bhat TK. DPPH antioxidant assay revisited. Food Chem 113 (2009): 1202-1205.
- Cao G, Alessio HM, Cutler RG. Oxygen-radical absorbance capacity assay for antioxidants. Free Radic. Biol. Med 14 (1993): 303-311.
- Roy MK, Koide M, Rao TP, et al. ORAC and DPPH assay comparison to assess antioxidant capacity of tea infusions: Relationship between total polyphenol and individual catechin content. Int. J. Food Sci. Nutr 61 (2010): 109-114.
- Barba FJ, Criado MN, Belda-Galbis CM, et al. Stevia rebaudiana Bertoni as a natural antioxidant/antimicrobial for high pressure processed fruit extract: Processing parameter optimization. Food Chem 148 (2014): 261-267.
- Ghanta S, Banerjee A, Poddar A, et al. Oxidative DNA damage preventive activity and antioxidant potential of Stevia rebaudiana (bertoni) bertoni, a natural sweetener. J. Agric. Food Chem 55 (2007): 10962-10967.
- Sharma R, Yadav R, Manivannan E. Study of effect of Stevia rebaudiana bertoni on oxidative stress in type-2 diabetic rat models. Biomed. Aging Pathol 2 (2012): 126-131.
- Bracht AK, Alvarez M, Bracht A. Effects of Stevia rebaudiana natural products on rat liver mitochondria. Biochem. Pharmacol 34 (1985): 873-882.
- Granato D, Shahidi F, Wrolstad R, et al. Antioxidant activity, total phenolics and flavonoids contents: Should we ban in vitro screening methods? Food Chem 264 (2018): 471-475.
- Xu XY, Zheng J, Meng JM, et al. Effects of food processing on in vivo antioxidant and hepatoprotective properties of green tea extracts. Antioxidants 8 (2019): 572.
- Prata C, Zambonin L, Rizzo B, et al. Glycosides from stevia rebaudiana Bertoni possess insulin-mimetic and antioxidant activities in rat cardiac fibroblasts. Oxid. Med. Cell 3724545 (2017): 1-13.
- Vaško L, Vašková J, Fejercáková A, et al. Comparison of some antioxidant properties of plant extracts from Origanum vulgare, Salvia officinalis, Eleutherococcus senticosus and Stevia rebaudiana. Vitr. Cell. Dev. Biol. - Anim 50 (2014): 614-622.
- Garcia-Mier L, Meneses-Reyes AE, Jimenez-Garcia SN, et al. Polyphenol content and Antioxidant Activity of Stevia and Peppermint as a Result of Organic and Conventional Fertilization. J. Food Qual 6620446 (2021): 1-6.
- Palasz A, Ciez D, Trzewik B, et al. In the search of glycoside-based molecules as antidiabetic agents. Top. Curr. Chem 377 (2019): 19.
- Mohd-Radzman NH, Ismail WI, Jaapar SS, et al. Stevioside from Stevia rebaudiana Bertoni increases insulin sensitivity in 3T3-L1 adipocytes. Evidence- Based Complement. Altern. Med 938081 (2013): 1-8.
- Bhasker S, Madhav H, Chinnamma M. Molecular evidence of insulinomimetic property exhibited by steviol and stevioside in diabetes induced L6 and 3T3L1 cells. Phytomedicine (2015).
- Abudula R, Matchkov VV, Jeppesen PB, et al. Rebaudioside A directly stimulates insulin secretion from pancreatic beta cells: A glucose-dependent action via inhibition of ATP-sensitive K+-channels. Diabetes, Obes. Metab 10 (2008): 1074-1085.
- Gu W, Rebsdorf A, Anker C, et al. Steviol glucuronide, a metabolite of steviol glycosides, potently stimulates insulin secretion from isolated mouse islets: Studies in vitro. Endocrinol. Diabetes Metab 2 (2019): e00093.
- Saravanan R, Babu KV, Ramachandran V. Effect of Rebaudioside A, a diterpenoid on glucose homeostasis in STZ-induced diabetic rats. J. Physiol. Biochem 68 (2012): 421-431.
- Aranda-González I, Moguel-Ordóñez Y, Chel-Guerrero L, et al. Evaluation of the antihyperglycemic effect of minor steviol glycosides in normoglycemic and induced-diabetic Wistar rats. J. Med. Food 19 (2016): 844-852.
- Jeppesen PB, Gregersen S, Alstrup KK, et al. Stevioside induces antihyperglycaemic, insulinotropic and glucagonostatic effects in vivo: Studies in the diabetic Goto-Kakizaki (GK) rats. Phytomedicine 9 (2002): 9-14.
- Ahmad U, Ahmad RS. Anti-diabetic property of aqueous extract of Stevia rebaudiana Bertoni leaves in Streptozotocin-induced diabetes in albino rats. BMC Complement. Altern. Med 18 (2018): 179-189.
- Das S, Istiak A, Hazra P, et al. Effects of Crystal Derived from Stevia rebaudiana leaves on Alloxan Induced Type-1 Diabetic Mice. Br. J. Pharm. Res 17 (2017): 1-11.
- Philippaert K, Pironet A, Mesuere M, et al. Steviol glycosides enhance pancreatic beta-cell function and taste sensation by potentiation of TRPM5 channel activity. Nat. Commun 8 (2017): 14733.
- Ilic V, Vukmirovic S, Stilinovic N, et al. Insight into anti-diabetic effect of low dose of stevioside. Biomed. Pharmacother 90 (2017): 216–221.
- Assaei R, Mokarram P, Dastghaib S, et al. Hypoglycemic effect of aquatic extract of Stevia in pancreas of diabetic rats: PPARγ -dependent regulation or antioxidant potential. Avicenna J. Med. Biotechnol 8 (2016): 65-74.
- Abdel-Aal RA, Abdel-Rahman MS, Al Bayoumi S, et al. Effect of stevia aqueous extract on the antidiabetic activity of saxagliptin in diabetic rats. J. Ethnopharmacol 265 (2021): 113188.
- Chen J, Jeppesen PB, Nordentoft I, et al. Stevioside improves pancreatic β-cell function during glucotoxicity via regulation of acetyl-CoA carboxylase. Am. J. Physiol. - Endocrinol. Metab 292 (2007): E1906–E1916.
- Laveti D, Kumar M, Hemalatha R, et al. Anti-Inflammatory Treatments for Chronic Diseases: A Review. Inflamm. Allergy-Drug Targets 12 (2013): 349-361.
- Boonkaewwan C, Toskulkao C, Vongsakul M. Anti-Inflammatory and immunomodulatory activities of stevioside and its metabolite steviol on THP-1 cells. J. Agric. Food Chem 54 (2006): 785-789.
- Fengyang L, Yunhe F, Bo L, et al. Stevioside suppressed inflammatory cytokine secretion by downregulation of NF-κB and MAPK signaling pathways in LPS- stimulated RAW264.7 cells. Inflammation 35 (2012): 1669-1675.
- Mizushina Y, Akihisa T, Ukiya M, et al. Structural analysis of isosteviol and related compounds as DNA polymerase and DNA topoisomerase inhibitors. Life Sci 77 (2005): 2127-2140.
- Nie Y, Wang Z, Lin J et al. Stevioside protects LPS-induced acute lung injury in mice. Inflammation 36 (2012): 242-250.
- Wang T, Song X, Zhang Z, et al. Stevioside inhibits inflammation and apoptosis by regulating TLR2 and TLR2-related proteins in S. aureus-infected mouse mammary epithelial cells. Int. Immunopharmacol 22 (2014): 192-199.
- Meng J, Zhou C, Hu B, et al. Stevioside Prevents Wear Particle-Induced Osteolysis by Inhibiting Osteoclastogenesis and Inflammatory Response via the Suppression of TAK1 Activation. Front. Pharmacol 9 (2018): 1053.
- Ramos-Tovar E, Flores-Beltrán RE, Galindo-Gómez S, et al. Stevia rebaudiana tea prevents experimental cirrhosis via regulation of NF-κB, Nrf2, transforming growth factor beta, Smad7, and hepatic stellate cell activation. Phyther. Res 32 (2018): 2568-2576.
- Casas-Grajales S, Reyes-Gordillo K, Cerda-García-Rojas CM, et al. Rebaudioside A administration prevents experimental liver fibrosis: an in vivo and in vitro study of the mechanisms of action involved∘ J. Appl. Toxicol 39 (2019): 1118-1131.
- Cho BO, Ryu HW, So Y, et al. Anti-inflammatory effect of austroinulin and 6-O-acetyl-austroinulin from Stevia rebaudiana in lipopolysaccharide-stimulated RAW264.7 macrophages. Food Chem. Toxicol 62 (2013): 638-644.
- Holvoet P, Rull A, García-Heredia A, et al. Stevia-derived compounds attenuate the toxic effects of ectopic lipid accumulation in the liver of obese mice: A transcriptomic and metabolomic study. Food Chem. Toxicol 77 (2015): 22-33.
- Pina-Pérez MC, Rivas A, Martínez A, et al. Effect of thermal treatment, microwave, and pulsed electric field processing on the antimicrobial potential of açaí (Euterpe oleracea), stevia (Stevia rebaudiana Bertoni), and ginseng (Panax quinquefolius L.) extracts. Food Control 90 (2018): 98-104.
- Garcia D, Garcia-Cela E, Rasmos A, et al. Mould growth and mycotoxin production as affected by Equisetum arvense and Stevia rebaudiana extracts. Food Control 22 (2011): 1378-1384.
- Ghosh S, Subudhi E, Nayak S. Antimicrobial assay of Stevia rebaudiana Bertoni leaf extracts against 10 pathogens. Int. J. Intergrative Biol 2 (2008): 27-31.
- Sharipova RR, Andreeva OV, Strobykina IY, et al. Synthesis and antimicrobial activity of glucuronosyl derivatives of steviolbioside from stevia rebaudiana. Chem. Nat. Compd 53 (2017): 1107-1111.
- Muanda FN, Soulimani R, Diop B, et al. Study on chemical composition and biological activities of essential oil and extracts from Stevia rebaudiana Bertoni leaves. LWT - Food Sci. Technol 44 (2011): 1865-1872.
- Lemus-Mondaca R, Vega-Gálvez A, Rojas P, et al. Antioxidant, antimicrobial and anti-inflammatory potential of Stevia rebaudiana leaves: effect of different drying methods. J. Appl. Res. Med. Aromat. Plants 11 (2018): 37-46.
- Perrier J, Mihalov J, Carlson S. FDA regulatory approach to steviol glycosides. Food Chem. Toxicol 122 (2018): 132-142.
- GRN No. 253 Rebaudioside A purified from Stevia rebaudiana (Bertoni) Bertoni. GRAS Not. Invent 253 (2008).
- Hsieh MH, Chan P, Sue YM, et al. Efficacy and tolerability of oral stevioside in patients with mild essential hypertension: A two-year, randomized, placebo- controlled study. Clin. Ther 25 (2003): 2797-2808.
- Geuns JM, Buyse J, Vankeirsbilck A, et al. Metabolism of stevioside by healthy subjects. Exp. Biol. Med 232 (2007): 164-173.
- GRN No. 349 Purified steviol glycosides with rebaudioside A and stevioside as the principal components. GRAS Not. Invent 349 (2011).
- Wheeler A, Boileau AC, Winkler PC, et al. Pharmacokinetics of rebaudioside A and stevioside after single oral doses in healthy men. Food Chem. Toxicol 46 (2008): S54-S60.
- GRN No. 536 High purity rebaudioside C. GRAS Not. Invent 536 (2015).
- GRN No. 548 High purity rebaudioside D. GRAS Not. Invent 548 (2015).
- GRN No. 667 Rebaudioside M. GRAS Not. Invent 667 (2017).
- GRN No. 823 Rebaudioside E. GRAS Not. Invent 823 (2019).
- Evaluation of certain food additives: sixty-third report of the Joint FAO/WHO Expert Committee on Food Additives. WHO Tech. Rep. Ser 928 (2005).
- Evaluation of certain food additives and contaminants: sixty-ninth report of the Joint FAO/WHO Expert Committee on Food Additives. WHO Tech. Rep. Ser 952 (2009).
- Evaluation of certain food additives: eighty-second report of the Joint FAO/WHO Expert Committee on Food Additives. WHO Tech. Rep. Ser 1000 (2016).
- Renwick AG. The use of a sweetener substitution method to predict dietary exposures for the intense sweetener rebaudioside A. Food Chem. Toxicol 46 (2008): S61-S69.
- Steviol glycosides from Stevia rebaudiana Bertoni. FAO JECFA Monogr 20 (2017).
- Commission Regulation (EU) No 231/2012. Specifications for food additives in Annexes II and III to Regulation (EC) No 1333/2008 of the European Parliament and of the Council. OJ Eur. Union (2012): L 83/1.
- Commission Regulation (EU) No 2016/1814. Specifications for food additives listed in Annexes II and III to Regulation (EC) No 1333/2008 of the European Parliament and of the Council as regards specifications for steviol glycosides (E 960). OJ Eur. Union (2016): L 278/37.
- Application A1037 Steviol glycosides -Increase in permitted use levels approval report (2011).
- Sharif R, Chan KM, Ooi TC, et al. Cytotoxicity and genotoxicity evaluation of stevioside on CCD18Co and HCT 116 cell lines. Int. Food Res. J 24 (2017): 341-345.
- Urban JD, Carakostas MC, Brusick DJ. Steviol glycoside safety: Is the genotoxicity database sufficient? Food Chem. Toxicol 51 (2013): 386-390.
- Zhang Q, Yang H, Li Y, et al. Toxicological evaluation of ethanolic extract from Stevia rebaudiana Bertoni leaves: Genotoxicity and subchronic oral toxicity. Regul. Toxicol. Pharmacol 86 (2017): 253-259.
- Brusick DJ. A critical review of the genetic toxicity of steviol and steviol glycosides. Food Chem. Toxicol 46 (2008): S83-S91.
- Potocnjak I, Broznic D, Kindl M, et al. Stevia and stevioside protect against cisplatin nephrotoxicity through inhibition of ERK1/2, STAT3, and NF-κB activation. Food Chem. Toxicol 107 (2017): 215-225.
- Potenza M, Cavalluzzi MM, Milani G, et al. Inverse Virtual Screening for the rapid re-evaluation of the presumed biological safe profile of natural products. The case of steviol from Stevia rebaudiana glycosides on farnesoid X receptor (FXR). Bioorg. Chem 111 (2021): 104897.
- Addy R. Mintel: stevia-containing products show “significant uplift” in number. Foodnavigator.Com (2012).
- Arthur R. Coca-Cola launches Fuze Tea in Europe. Beveragedaily.com (2018).
- Hutchings SC, Low JY, Keast RS. Sugar reduction without compromising sensory perception. An impossible dream? Crit. Rev. Food Sci. Nutr 59 (2019): 2287-2307.
- Bray GA. Energy and Fructose From Beverages Sweetened With Sugar or High- Fructose Corn Syrup Pose a Health Risk for Some People. Adv. Nutr 4 (2013): 220-225.
- Stanner SA, Spiro A. Public health rationale for reducing sugar: Strategies and challenges. Nutr. Bull 45 (2020): 253-270.
- Stanhope KL. Sugar consumption, metabolic disease and obesity: The state of the controversy. Crit. Rev. Clin. Lab. Sci 53 (2016): 52-67.
- Neelakantan N, Park SH, Chen GC, et al. Sugar-sweetened beverage consumption, weight gain, and risk of type 2 diabetes and cardiovascular diseases in Asia: a systematic review. Nutr. Rev 80 (2022): 50-67.
- Kamarulzaman NH, Jamal K, Vijayan G, et al. Will consumers purchase stevia as a sugar substitute?: An exploratory study on consumer acceptance. J. Food Prod. Mark 20 (2014): 122-139.
- Ridzwan Yaakub M, Iqbal Abu Latiffi M, Safra Zaabar L. A review on sentiment analysis techniques and applications. IOP Conf. Ser. Mater. Sci. Eng 551 (2019): 12070.
- Li J, Lowe D, Wayment L, et al. Text mining datasets of β-hydroxybutyrate (BHB) supplement products’ consumer online reviews. Data Br 30 (2020): 105385.
- Sabbe S, Verbeke W, Deliza R, et al. Effect of a health claim and personal characteristics on consumer acceptance of fruit juices with different concentrations of açaí (Euterpe oleracea Mart.). Appetite 53 (2009): 84-92.
- Góngora Salazar VA, Vázquez Encalada S, Corona Cruz A, et al. Stevia rebaudiana: A sweetener and potential bioactive ingredient in the development of functional cookies. J. Funct. Foods 44 (2018): 183-190.
- Yildiz E, Gocmen D. Use of almond flour and stevia in rice-based gluten-free cookie production. J. Food Sci. Technol 58 (2021): 940-951.
- Gao J, Guo X, Brennan MA, et al. The potential of modulating the reducing sugar released (and the potential glycemic response) of muffins using a combination of a stevia sweetener and cocoa powder. Foods 8 (2019).
- Šic Zlabur J, Dobricevic N, Galic A, et al. The influence of natural sweetener (Stevia rebaudiana Bertoni) on bioactive compounds content in chokeberry juice. J. Food Process. Preserv 42 (2018): e13406.
- Salar FJ, Agulló V, García-Viguera C, et al. Stevia vs. sucrose: Influence on the phytochemical content of a citrus–maqui beverage—A shelf life study. Foods 9 (2020): 219.
- Nazarova NE, Zubova EV, Zaletova TV, et al. Blended fruit wines based on stevia essence. IOP Conf. Ser. Earth Environ. Sci 640 (2021): 52027.
- Homayouni Rad A, Delshadian Z, Arefhosseini SR, et al. Effect of inulin and stevia on some physical properties of chocolate milk. Heal. Promot. Perspect 2 (2012): 42-47.
- Alizadeh M, Azizi-lalabadi M, Hojat-ansari H, et al. Effect of stevia as a substitute for sugar on physicochemical and sensory properties of fruit based milk shake. J. Sci. Res. Reports 3 (2014): 1421-1429.
- Mahato DK, Keast R, Liem DG, et al. Optimisation of natural sweeteners for sugar reduction in chocolate flavoured milk and their impact on sensory attributes. Int. Dairy J 115 (2021): 104922.
- Assy N, Nasser G, Kamayse I, et al. Soft drink consumption linked with fatty liver in the absence of traditional risk factors. Can. J. Gastroenterol 22 (2008): 810961.
- Bremer AA, Auinger P, Byrd RS. Sugar-sweetened beverage intake trends in US adolescents and their association with insulin resistance-related parameters. J. Nutr. Metab 196476 (2010).
- Li XE, Lopertcharat K, Drake MA. Parents’ and children’s acceptance of skim chocolate milks sweetened by monk fruit and stevia leaf extracts. J. Food Sci 80 (2015): S1083-S1092.
- Morais EC, Morais AR, Cruz AG, et al. Development of chocolate dairy dessert with addition of prebiotics and replacement of sucrose with different high-intensity sweeteners. J. Dairy Sci 97 (2014): 2600-2609.
- Ozdemir C, Arslaner A, Ozdemir S, et al. The production of ice cream using stevia as a sweetener. J. Food Sci. Technol 52 (2015): 7545-7548.
- Ribeiro MN, Rodrigues DM, Rocha RA, et al. Optimising a stevia mix by mixture design and napping: A case study with high protein plain yoghurt. Int. Dairy J 110 (2020): 104802.
- Prakash I, Chaturvedula VS. Steviol Glycosides: Natural Noncaloric Sweeteners. In: Sweeteners. Springer (2018): 1-28.
- Ferrazzano GF, Cantile T, Alcidi B, et al. Is stevia rebaudiana bertoni a non-cariogenic sweetener? A review. Molecules 21 (2016): 38.
- Marcinek K, Krejpcio Z. Stevia Rebaudiana bertoni - chemical composition and functional properties. Acta Sci. Pol. Technol. Aliment 14 (2015): 145-152.
- Gantait S, Das A, Mandal N. Stevia: A comprehensive review on ethnopharmacological properties and in vitro Sugar Tech 17 (2015): 95-106.
- Urban JD, Carakostas MC, Taylor SL. Steviol glycoside safety: Are highly purified steviol glycoside sweeteners food allergens? Food Chem. Toxicol 75 (2015): 71-78.
- Gasmalla MA, Yang R, Hua X. Stevia rebaudiana Bertoni: An alternative sugar replacer and its application in food industry. Food Eng. Rev 6 (2014): 150-162.
- Christaki E, Bonos E, Giannenas I, et al. Stevia rebaudiana as a novel source of food additives. J. Food Nutr. Res 52 (2013): 195-202.
- Brown RJ, Rother KI. Non-nutritive sweeteners and their role in the gastrointestinal tract. J. Clin. Endocrinol. Metab 97 (2012): 2597-2605.
- Coban I, Sargin S, Celiktas MS, et al. Bioethanol production from raffinate phase of supercritical CO2 extracted Stevia rebaudiana leaves, Bioresour. Technol 120 (2012): 52-59.
- Yadav SK, Guleria P. Steviol glycosides from stevia: Biosynthesis pathway review and their application in foods and medicine. Crit. Rev. Food Sci. Nutr 52 (2012): 988-998.
- Kochikyan VT, Markosyan AA, Abelyan LA, et al. Combined enzymatic modification of stevioside and rebaudioside A, Appl. Biochem. Microbiol 42 (2006): 31-37.
- Brandle JE, Starratt AN, Gijzen M. Stevia rebaudiana: Its agricultural, biological, and chemical properties. Can. J. Plant Sci 78 (1998): 527-536.
- Barroso M, Barros L, Rodrigues MÂ, et al. Stevia rebaudiana Bertoni cultivated in Portugal: A prospective study of its antioxidant potential in different conservation conditions. Ind. Crops Prod 90 (2016): 49-55.
- Covarrubias-Cárdenas AG, Martínez-Castillo JI, Medina-Torres N, et al. Antioxidant capacity and uplc-pda esi-ms phenolic profile of stevia rebaudiana dry powder extracts obtained by ultrasound assisted extraction. Agronomy 8 (2018): 170-182.
- Shivanna N, Naika M, Khanum F, et al. Antioxidant, anti-diabetic and renal protective properties of Stevia rebaudiana. J. Diabetes Complications 27 (2013): 103-113.
- Boonkaewwan C, Burodom A. Anti-inflammatory and immunomodulatory activities of stevioside and steviol on colonic epithelial cells. J. Sci. Food Agric 93 (2013): 3820-3825.
- Wang T, M. Guo M, X. Song X, et al. Stevioside plays an anti-inflammatory role by regulating the NF-κB and MAPK pathways in S. aureus-infected mouse mammary glands. Inflammation 37 (2014): 1837-1846.