Glucose-6-Phosphate Dehydrogenase (G6PD) Deficiency in Neonates Presenting with Indirect Hyperbilirubinemia in Neonatal Intensive Care Unit of Tertiary Health Care Center of Pakistan: Is the Trend Changing with Change in Consanguineous Marriages?
Article Information
Shahzeen1, Abid Ali Jamali2*, Aasma Kayani3, Anam Zubair1, Rabia Noreen1, Sanober Fatima3
1Civil Hospital Karachi, Pakistan
2Aga Khan University hospital, Karachi, Pakistan
3National Institute of Child Health Karachi, Pakistan
*Corresponding author: Abid Ali Jamali, Aga Khan University Hospital, Pakistan.
Received: 06 August 2022; Accepted: 17 August 2022; Published: 30 August 2022
Citation: Shahzeen, Abid Ali Jamali, Aasma Kayani, Anam Zubair, Rabia Noreen, Sanober Fatima. Glucose-6-Phosphate Dehydrogenase (G6PD) Deficiency in Neonates Presenting with Indirect Hyperbilirubinemia in Neonatal Intensive Care Unit of Tertiary Health Care Center of Pakistan: Is the Trend Changing with Change in Consanguineous Marriages?. Fortune Journal of Health Sciences 5 (2022): 510-516
View / Download Pdf Share at FacebookAbstract
Introduction: G6PD is an X-linked enzyme that catalyzes the first step in the HMP pathway to produces NADPH which is required for regeneration of the reduced form of glutathione (GSH). GSH is essential for the detoxification of hydrogen peroxide, especially in RBCs, which rely only on this pathway. There has been some declining trend in consanguineous marriages since last 3 decades in Pakistan, so we are conducting this study to find out whether this change lead to decrease in the frequency of G6PD deficiency in the country.
Materials and Methods: This was a prospective cross-sectional study, using non-probability consecutive sampling technique, conducted at the Neonatal Unit of Civil Hospital, Karachi during 2016-17, on 144 neonates with indirect hyperbilirubinemia. The study was approved by the Ethical Review Committee of the Civil Hospital, Dow University of Health Sciences Karachi.
Results: The mean age ± SD of neonates was 10.63±3.48 days and the majority were males (68%). The majority of neonates presented after the 5th day of their life i.e. 87.5%. G6PD deficiency was present in 8.3% of the neonates with indirect hyperbilirubinemia. Consanguinity was present in around 50% of the neonates who presented with neonatal jaundice. The overall mean total serum bilirubin, hemoglobin, hematocrit and reticulocyte count were 15.57±2.87 (mg/dl), 14.22±0.48 g/dl, 45.57±3.31%, and 0.84±0.32 % respectively. Most of the mothers (22.2%) have blood groups AB +ve and B +ve and the most babies (28.5%) have A +ve blood group.
Conclusion: Male gender and age 5 days or less had significantly high G6PD deficiency. Although consanguineous marriages have decreased only slightly, no significant effect on G6PD deficiency is found.
Keywords
Consanguineous Marriages; G6PD Deficiency; Hyperbilirubinemia; Neonatal Jaundice; Neonatal Indirect
Consanguineous Marriages articles; G6PD Deficiency articles; Hyperbilirubinemia articles; Neonatal Jaundice articles; Neonatal Indirect articles
Article Details
1. Introduction
Neonatal jaundice is a common physiological occurrence and around 60-80% of otherwise healthy newborns develop jaundice in their early neonatal period [1,2]. It is a medical condition that occurs in newborns as a result of an imbalance between the production and elimination of bilirubin. Jaundice is a result of the increased breakdown of red blood cells and/or decreased hepatic excretion of bilirubin [3]. Neonatal jaundice commonly presents in the first week of life [4]. More than 60% of healthy newborns develop clinical jaundice associated with increased concentration of total serum bilirubin (TSB) during the first week of life. However, bilirubin (TSB) above 95th percentile for age in hours (high-risk zone) occurs in 8-9% of infants during the first week with approximately 4% affected after 72 hours [5,6]. The etiology in the majority of neonatal jaundice includes idiopathic causes, hemolytic disorders, inadequate Breastfeeding and Glucose-6-phosphate dehydrogenase (G6PD) deficiency [7]. The prevalence rate of G6PD varies from as high as 62% among Kurdish Jews to as low as 0.1% in Japan, while it ranges from 3 to 6.9% in Pakistan, Southern China and Southern Russia [4]. G6PD is a cytoplasmic enzyme that plays a pivotal role in the hexose monophosphate shunt (HMP shunt) of glucose metabolism. It catalyzes the first step in the HMP pathway i.e. conversion of Glucose-6-phosphate to 6-phosphogluconic acid. HMP shunt produces NADPH which is required for reactions of various biosynthetic pathways as well as for the stability of catalase and the preservation and regeneration of the reduced form of glutathione (GSH) [8,9]. Catalase and GSH are essential for the detoxification of hydrogen peroxide [10]. Red blood cells (RBCs) are exquisitely sensitive to oxidative damage, as other NADPH-producing enzymes are lacking within RBCs and they rely only on the HMP shunt for GSH and catalase production. G6PD deficiency is the most commonly known inherited disorder in man and is estimated to affect 400 million people worldwide. Males are more affected than females due to X-linked inheritance and the condition is common in malaria-endemic regions [11]. G6PD deficiency is genetically heterogeneous with the majority of mutations disrupting the enzyme structure, thereby reducing overall enzyme activity [12,13]. The residual enzyme activity of G6PD variants ranges from <1% to 100%. The majority of G6PD deficient individuals are asymptomatic most of the time. Symptoms are induced when red blood cells are exposed to exogenous oxidative stresses against which they cannot defend themselves, like certain drugs or infectious agents (hydrogen peroxide is generated by activated polymorphonuclear neutrophils) [14,15]. Clinically it may manifest as neonatal jaundice, acute haemolytic anaemia and drug-induced haemolysis [16]. G6PD deficiency is diagnosed on the basis of clinical history and haematological findings, including anaemia, reticulocytosis and characteristic red cell changes (for example, 'bite' cells and Heinz bodies, Assays of G6PD activity depend on measuring the rate of production of NADPH from NADP in red cells, and the assay may be performed on a sequestrene (EDTA) or heparinized blood sample. Enzyme activity declines with red cell age and is highest in reticulocytes. Assay results obtained after an acute hemolytic episode should always be confirmed during the steady-state, as reticulocytosis may rarely lead to a false-negative result. Most haematology laboratories in the UK utilize screening tests, which are rapid and can reliably distinguish between affected men and heterozygous females; formal biochemical characterization involves enzyme purification from red cells, an assay of activity by spectrophotometry and enzyme electrophoresis, and is only necessary for selected [17]. Pakistan has one of the highest reported rates of consanguineous marriages in the world. Although over the last 3 decades there has been some declining trend. Consanguineous marriage ratio decreased from over 60% before 2000 AD to slightly less than 50 % in 2017-2018 [18,19]. We are conducting this study to find out whether this change in consanguineous marriages has affected the frequency of G6PD deficiency in Pakistani children.
2. Materials and Methods
2.1 Study Design and Setting
This was a prospective cross-sectional study, conducted at the Neonatal Unit of the pediatric ward, Civil Hospital, Dow University of Health Sciences, Karachi, during 2016-17. The study was conducted on admitted neonates with indirect hyperbilirubinemia.
2.2 Inclusion and Exclusion Criteria (participants)
All the admitted neonates, of either gender and between 1-28days, with indirect hyperbilirubinemia of any duration (that is peak serum bilirubin more than 12mg/dl in full-term more than 15mg/dl in preterm neonates) were included in the study, and their status for G6PD deficiency was checked (The condition was characterized by abnormally low levels of glucose-6-phosphate dehydrogenase). In this study G6PD activity below 4.0 IU/gHb was considered as G6PD Deficiency. Whereas babies with conjugated hyperbilirubinemia (confirmed by direct bilirubin level that is more than 20% of total bilirubin level), neonates with sepsis (confirmed by examination i.e. poor moro and sucking reflex and by blood culture, if needed), neonates who have already received a blood transfusion and /or undergone exchanged transfusion and those neonates whose parents refused for admission or did not give consent to be enrolled in the study were excluded.
2.3 Sampling and Sample Size
Non-probability consecutive sampling technique was used for sample collection and the sample size was calculated by WHO software, by using P=16%, 5 and d=6%, was 144 neonates at 95% confidence interval.
2.4 Ethical Consideration
The study was approved by the Ethical Review Committee of the Civil Hospital, Dow University of Health Sciences Karachi, Pakistan. Written informed consent was taken from the guardian (mother or father), after explaining to them the purpose and procedure of the study in detail and ensuring their confidentiality. Patients and parents/guardians were given full authority to withdraw from the study at any time and they were assured that withdrawal from the study will not affect their treatment or follow-ups.
2.5 Data Collection
The neonates admitted to the neonatal intensive care unit (NICU) with indirect hyperbilirubinemia and fulfilling the inclusion criteria were enrolled in this study. The informed consent was taken with the parents. Neonate’s clinical history and physical examination were done by the principal investigator. The data included the neonate’s registration number, age, gender, gestational age, birth weight, and laboratory data. All the laboratory investigations were done in the laboratory of Civil Hospital Karachi. Laboratory tests for Serum Bilirubin (Direct, Indirect), Hemoglobin/Hematocrit, Reticulocyte count, Blood group of baby and mother, Coombs test and G6PD assay were done in all neonates because these tests were routinely performed in all the neonates with hyperbilirubinemia to see the rate of hemolysis and to exclude other causes of hemolysis i.e. ABO & RH incompatibility. The red cell G6PD activity, expressed as units per gram of haemoglobin (U/gHb), was determined for the quantitative determination of G6PD deficiency. Based on the frequency distribution of activity levels, the critical level for diagnosing G6PD deficiency is considered 4.0 U/gHb. Any neonate with activity below this value was diagnosed as G6PD deficient. Biasness and confounding variables were controlled by strictly following inclusion and exclusion criteria.
2.6 Data Analysis
Patients’ data was compiled and analyzed through the Statistical Package for Social Sciences (SPSS) Version 21. Frequency and percentage were computed for qualitative variables like gender, G6PD deficiency and blood group. Mean±SD was calculated for quantitative variables i.e. age, hemoglobin level, serum bilirubin, and G6PD activity. The stratification was done on gender, age, consanguinity and length of hospital stay to see the effect of these modifiers on the outcome using the Chi-square test. P≤0.05 was considered significant.
3. Results
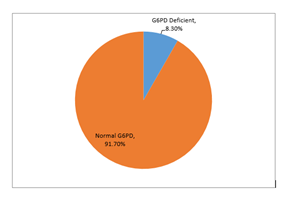
The mean age ± SD of neonates presenting with indirect hyperbilirubinemia was 10.63±3.48 days. Age is further stratified into two groups i.e. ≤ 5 days and > 5 days old. The detailed descriptive statistics of age are presented in Table I. In our study, the majority of the neonates were males (68%), with a male to female ratio of approximately 2:1. The majority of neonates presented after the 5th day of their life i.e. 87.5% as compared to only 12.5% presenting before the 5th day of life. Consanguinity was present in around 50% of the neonates who presented with neonatal jaundice. Eighty-four per cent of the neonates were discharged within 5 days and the coombs test was positive in approximately 7% of the neonates, Table II. The overall mean total serum bilirubin level of our study subjects was 15.57±2.87 (mg/dl). The overall mean hemoglobin, hematocrit and reticulocyte count was 14.22±0.48 g/dl, 45.57±3.31%, and 0.84±0.32 % respectively. The detailed descriptive statistics of hemoglobin, hematocrit, reticulocyte count and total serum bilirubin level are presented in Table III. Most of the mothers (22.2%) have blood groups AB +ve and B +ve and most babies (28.5%) have A +ve blood group. The detailed frequency distribution of the blood group of mother and baby is presented in Table IV. In our study, G6PD deficiency was present in 8.3% of the neonates with indirect hyperbilirubinemia. Graph I. The descriptive statistics of mean total serum bilirubin level, hemoglobin, hematocrit and reticulocyte count in relation to G6PD activity are presented in Table V. Stratification with respect to gender, age, consanguinity and length of hospital stay was done to observe the effect of these modifiers on G6PD deficiency. P-value ≤0.05 was considered significant. The results showed that there was a significant association between G6PD deficiency with gender (p=0.010) and age (p=0.000) while no significant association was found with the length of hospital stay (p=0.691). Surprisingly our study did not show a significant association with consanguinity. The detailed results of associations are presented in Table VI.
|
Mean ±SD |
95%CI |
Median (IQR) |
Range |
Minimum |
Maximum |
|
|
Age |
10.63±3.48 |
10.05 to 11.20 |
11.00 (5) |
14 |
4 |
18 |
|
≤5 days (n=18) |
4.55±0.51 |
4.30-4.80 |
5.00 (1) |
1.00 |
4.00 |
5.00 |
|
>5days (n=126 |
11.50±2.79 |
11.00-11.99 |
12.00 (4) |
12 |
6 |
18 |
Table I: Detailed descriptive statistics of age (days). (n=144)
|
Variable |
Frequency |
Percentage |
|
|
Gender |
Male |
98 |
68% |
|
Female |
46 |
32% |
|
|
Total |
144 |
100% |
|
|
Age |
≤ 5 days old |
18 |
12.5% |
|
>5 days old |
126 |
87.5% |
|
|
Total |
144 |
100% |
|
|
Consanguinity |
Yes |
71 |
49.3% |
|
No |
73 |
50.7% |
|
|
Total |
144 |
100% |
|
|
Length of hospital stay |
≤ 5 days |
121 |
84% |
|
>5days |
23 |
16% |
|
|
Total |
144 |
100% |
|
|
Coombs test |
Positive |
10 |
6.9% |
|
Negative |
134 |
93.1% |
|
|
Total |
144 |
100% |
|
Table II: Frequency and percentage of neonates presenting with indirect hyperbilirubinemia with respect to Gender, Age, Consanguinity, Length of hospital stay and coomb’s test.
|
Hemoglobin (g/dl) |
Hematocrit (%) |
Reticulocyte count (%) |
Total serum bilirubin level (mg/dl) |
|
|
Mean ±SD |
14.22±0.48 |
45.57±3.31 |
0.84±0.32 |
15.57±2.87 |
|
95%CI |
14.14-14.30 |
45.03-6.12 |
0.79-0.89 |
15.10-16.05 |
|
Median (IQR) |
14.20 (0.90) |
44.90 (2.93) |
0.80 (0.30) |
14.80 (3.50) |
|
Range |
1.60 |
12.80 |
1.40 |
10.20 |
|
Minimum |
13.40 |
41.50 |
0.50 |
12.20 |
|
Maximum |
15.00 |
54.30 |
1.90 |
22.40 |
Table III: Descriptive statistics of, hemoglobin (g/dl), hematocrit (%), reticulocyte count (%) and total serum bilirubin level (mg/dl). (n=144)
|
Blood Group |
Mother Blood Group |
Baby Blood Group |
||
|
Frequency (n) |
Percentage (%) |
Frequency (n) |
Percentage (%) |
|
|
A –ve |
5 |
3.47 |
6 |
4.17 |
|
A +ve |
24 |
16.67 |
41 |
28.47 |
|
AB –ve |
3 |
2.08 |
4 |
2.78 |
|
AB +ve |
17 |
11.81 |
18 |
12.50 |
|
B –ve |
7 |
4.86 |
9 |
6.25 |
|
B +ve |
30 |
20.83 |
38 |
26.39 |
|
O –ve |
15 |
10.42 |
8 |
5.56 |
|
O +ve |
43 |
29.86 |
20 |
13.88 |
|
TOTAL |
144 |
100% |
144 |
100% |
Table IV: Frequency distribution of mother blood group and baby blood group (n=144).
Table V: Descriptive statistics of hemoglobin, hematocrit, reticulocyte count and total serum bilirubin level according to G6PD deficiency. (n=144)
|
Variables |
G6PD deficiency |
Total |
p-Value |
||
|
Yes (n=12) |
No (n=132) |
||||
|
Gender |
Male |
12 |
86 |
98 |
0.010* |
|
Female |
0 |
46 |
46 |
||
|
Total |
12 |
132 |
144 |
||
|
Age |
≤5 days (n=18) |
10 |
8 |
18 |
0.000* |
|
>5 days (n=126) |
2 |
124 |
126 |
||
|
TOTAL |
12 |
132 |
144 |
||
|
Consanguinity |
Yes |
5 |
66 |
71 |
0.580 |
|
No |
7 |
66 |
73 |
||
|
Total |
12 |
132 |
144 |
||
|
Length of hospital stay |
≤5 Days (n=121) |
11 |
110 |
121 |
0.691 |
|
>5 Days (n=23) |
1 |
22 |
23 |
||
|
Total |
12 |
132 |
144 |
||
Table VI: Frequency and association of G6PD deficiency according to gender, *Age*, cousin marriages and length of hospital stay. (n=144)
Chi-Square Test was applied.
*P-value≤0.05 considered significant.
4. Discussion
G6PD deficiency is an X-linked recessive condition. This deficiency makes red blood cells hypersensitive to exposure to oxidants, leading to various clinical manifestations such as neonatal jaundice [20]. Studies performed in different parts of the world have reported various prevalence rates of G6PD deficiency. The reported prevalence in Singapore, Spain, France and most of the Latin American and Caribbean countries is below 2 %, which is considerably low [21,22]. In contrast, prevalence rates across sub-Saharan Africa and the Arabian Peninsula may cross over 30% of the population [23]. In our study, the prevalence of g6pd deficiency was 8.3%. A study conducted by Kumar et al. From Pakistan reported a prevalence of 9.3% which is quite near to our results [24]. Other studies from Asia have reported different results like Khodashenas et al study from Iran, the prevalence of g6pd deficiency was estimated at 3.5%, which was lower than our study [25]. Study by Ellela et al. from Egypt reported a prevalence of 4.3% that is also lower as compared to Pakistan data [26]. This is most probably due to the lower ratio of consanguineous marriages in these countries. Consanguineous marriages in Iran are 37.4% [27] and in Egypt, it is around 40 % [28]. In our study, consanguinity was present in 49.3% of the parents of neonates who presented with indirect hyperbilirubinemia. These findings are comparable to the national demographic data conducted by NIPS [19]. G6PD deficiency is quite higher in the Arabian peninsula as well (18%) [23]. Due to the higher prevalence of cousin marriage. A recent survey conducted in Saudi Arabia showed that even educated adults were having consanguineous marriages up to 40% [29]. In our study, the most common mother blood group (MBG) was O+ve followed by B+ve and A+ve respectively. Among Rh-negative blood groups, O-ve was the most common. As compared to MBG, the most common baby blood group (BBG) was A+ve, followed by B+ve and AB+ve respectively, indicating that ABO blood group incompatibility was one of the common causes of hyperbilrubimeina in neonatal age. In our study, all G6PD deficient neonates were males and no female was found to be deficient in enzyme levels. Over 80% of the G6PD deficient neonates presented within 5 days of life. A study conducted by Akhtar et al. also reported that the majority of the G6PD deficient neonates presented on 3rd day of life [30]. Over 50% of the neonates with G6PD deficiency were the products of non-consanguineous marriages. Length of hospital stay in G6PD deficient neonates was less than 5 days in almost all of the patients. No significant difference in length of stay of neonates with or without G6PD deficiency was found. Atay et al. [31] reported similar results for length of hospital stay in their study around 15 years back.
5. Strengths and Limitations of the Study
This study was conducted in one of the busiest pediatric neonatology units in the city. The study center is a public health sector hospital, so almost all categories of socio-economic patients present here, ranging from very poor to upper and middle-upper class families. The main limitations of the present study include a single-center experience, low female representation and a nonrandomized study design. It was conducted with small sample size and in an urban environment therefore, the results might not be generalizable to larger populations.
6. Conclusion
G6PD enzyme deficiency is common among neonates, admitted to the hospital due to jaundice. Male gender and age 5 days or less had significantly high G6PD deficiency. Although consanguineous marriages have decreased only slightly, no significant effect on G6PD deficiency is found. We need large population-based studies to find out the exact relation of G6PD with change in consanguinity.
Recommendations
G6PD deficiency can cause malignant hyperbilirubinemia, which might lead to kernicterus if not diagnosed and treated promptly. Therefore, we recommend that the establishment of an early G6PD screening program for neonates is essential. Such programs are required to prevent subsequent complications by timely diagnosis and treatment.
Declarations
Acknowledgements
Not applicable.
Funding
Not funded.
Author’s contributions
SS was the lead investigator who designed this study and collected data. AJ helped in writing study protocol and analyzing the results along with writing the final manuscript. AK helped in data analysis and converting it into tabulations and graphic forms. AZ and RN both collected data and interpreted lab investigations. SF helped in manuscript writing, specially adding the references in Vancouver style.
Competing Interests
The authors declare that they have no competing interests.
Informed Consent
Informed consent is obtained from parents/guardians of the patient.
Ethical Approval
This study was approved by the Ethical Review Committee of the Civil Hospital, Dow University of Health Sciences Karachi, Pakistan
References
- Ullah S, Rahman K, Hedayati M. Hyperbilirubinemia in neonates: types, causes, clinical examinations, preventive measures and treatments: a narrative review article. Iran. J. Public Health 45 (2016): 558.
- Brits H, Adendorff J, Huisamen D, et al. The prevalence of neonatal jaundice and risk factors in healthy term neonates at National District Hospital in Bloemfontein. Afr J Prim Health Care Fam Med 10 (2018): 1-6.
- Scrafford CG, Mullany LC, Katz J, et al. Incidence of and risk factors for neonatal jaundice among newborns in southern Nepal. Trop Med Int Heal 18 (2013): 1317-1328.
- Ali Q, Aqeel M, Iqbal H. Frequency of glucose 6 phosphate dehydrogenase deficiency in neonatal jaundice. J Med Sci 19 (2011): 167-170.
- Hussain M, Irshad M, Kalim M, et al. Glucose-6-phosphate dehydrogenase deficiency in jaundiced neonates. J Postgrad Med Inst 24 (2010): 122-126.
- Jan AZ, Zahid SB, Ahmad S. Frequency of G6PD deficiency and its severity in neonatal jaundice in Rehman Medical Institute, Peshawar. Khyber Med Uni J 5 (2013): 36-39.
- WR Hansen T. Neonatal Jaundice (2017).
- Stanton RC. Glucose-6-phosphate dehydrogenase, NADPH, and cell survival. IUBMB life 64 (2012): 362-369.
- Burgoine KL, Bancone G, Nosten F. The reality of using primaquine. Malaria J. 9, 376. Burka, E.R., Weaver, Z., Marks, P.A., 1966. Clinical spectrum of hemolytic anemia associated with glucose-6-ghosphate dehydrogenase deficiency. Ann Intern Med 64 (2010): 817-825.
- Stincone A, Prigione A, Cramer T, et al. The return of metabolism: biochemistry and physiology of the pentose phosphate pathway. Biol 90 (2015): 927-963.
- Bubp J, Jen M, Matuszewski K. Caring for glucose-6-phosphate dehydrogenase (G6PD)–deficient patients: implications for pharmacy. Pharmacol. Ther 40 (2015): 572.
- Sarker SK, Islam MT, Eckhoff G, et al. Molecular analysis of glucose-6-phosphate dehydrogenase gene mutations in Bangladeshi individuals. PloS one 11 (2016): e0166977.
- Gómez-Manzo S, Terrón-Hernández J, la Mora-De la Mora D, et al. The stability of G6PD is affected by mutations with different clinical phenotypes. Int. J. Mol. Sci 15 (2014): 21179-21201.
- Boonyuen U, Chamchoy K, Swangsri T, et al. A tradeoff between catalytic activity and protein stability determines the clinical manifestations of glucose-6-phosphate dehydrogenase (G6PD) deficiency. Int. J. Biol. Macromol 104 (2017): 145-156.
- Gómez-Manzo S, Marcial-Quino J, Ortega-Cuellar D, et al. Functional and biochemical analysis of glucose-6-phosphate dehydrogenase (G6PD) variants: Elucidating the molecular basis of G6PD deficiency. Catalysts 7 (2017): 135.
- Mortazavi Y, Mirzamohammadi F, Ardestani TM, et al. Glucose 6-phosphate dehydrogenase deficiency in Tehran, Zanjan and Sistan-Balouchestan provinces: prevalence and frequency of Mediterranean variant of G6PD. Iran J Biotechnol 8 (2010): 229-233.
- Luzzatto L, Nannelli C, Notaro R. Glucose-6-phosphate dehydrogenase deficiency. Hematol. Oncol 30 (2016): 373-393.
- Hussain R, Bittles A. The prevalence and demographic characteristics of consanguineous marriages in Pakistan. J. Biosoc. Sci 30 (1998): 261-275.
- Pakistan demographic and health survey 2017-18 (2019).
- Piazza AJ, Stoll BJ. Jaundice and Hyperbilirubinemia in the Newborn. In: Kliegman RM, Behrman RE, Jenson HB, Stanton BF, editors. Nelson text book of Pediatrics: 19th Ed. New Delhi; Saunders Elsevier 1 (2012): 603-612.
- Monteiro WM, Val FF, Siqueira AM, et al. G6PD deficiency in Latin America: systematic review on prevalence and variants. Mem. Inst. Oswaldo Cruz 109 (2014): 553-568.
- Khodashenas E, Kalani MF, Araghi Z, et al. Glucose-6-phosphate dehydrogenase deficiency and neonatal hyperbilirubinemia. Iran. J. Neonatol 6 (2015): 28-31.
- Howes RE, Piel FB, Patil AP, et al. G6PD deficiency prevalence and estimates of affected populations in malaria endemic countries: a geostatistical model-based map. PLoS medicine 9 (2012): e1001339.
- Kumar K, Sohaila A, Tikmani SS, et al. Screening for G6PD deficiency among neonates with neonatal jaundice admitted to tertiary care center: a need in disguise. JCPSP 25 (2015): 625.
- Khodashenas E, Kalani-Moghaddam F, Araghi Z, et al. Glucose-6-Phosphate Dehydrogenase Deficiency and Neonatal Hyperbilirubinemia. Iranian J Neonatol IJN 6 (2015): 28-31.
- Elella SA, Tawfik M, Barseem N, et al. Prevalence of glucose-6-phosphate dehydrogenase deficiency in neonates in Egypt. Annals of Saudi medicine 37 (2017): 362-365.
- Hosseini-Chavoshi M, Abbasi-Shavazi MJ, Bittles AH. Consanguineous marriage, reproductive behaviour and postnatal mortality in contemporary Iran. Human heredity 77 (2014): 16-25.
- Keeping it in the family. The Economist (2016).
- Mahboub SM, Alsaqabi AA, Allwimi NA, et al. Prevalence and pattern of consanguineous marriage among educated married individuals in Riyadh. Journal of Biosocial Science 52 (2020): 768-775.
- Akhter N, Habiba U, Mazari N, et al. Glucose-6-phosphate dehydrogenase deficiency in neonatal hyperbilirubinemia and its relationship with severity of hyperbilirubinemia. Isra Med J 11 (2019): 237-241.
- Atay E, Bozaykut A, Ipek IO. Glucose-6-phosphate dehydrogenase deficiency in neonatal indirect hyperbilirubinemia.J Trop Pediatr 52 (2006): 56-58.