Endocarditis at Prior Venous Cannulation Site after Sternal Wound Infection: Case Report
Article Information
Jared Cappelli, MD¹*, Amber Edwards, MD²
1University of Tennessee Medical College - Nashville, General Surgery Residency, Nashville, Tennessee, USA
2Ascension Saint Thomas Hospital West, Department of Surgery, Nashville, Tennessee, USA
*Corresponding Author: Jared Cappelli, University of Tennessee Medical College - Nashville, General Surgery Residency, Nashville, Tennessee, USA
Received: 25 May 2023; Accepted: 12 June 2023; Published: 28 June 2023
Citation: Jared Cappelli, Amber Edwards. Endocarditis at Prior Venous Cannulation Site after Sternal Wound Infection: Case Report. Archives of Clinical and Medical Case Reports. 7 (2023): 299-301.
View / Download Pdf Share at FacebookAbstract
Endocarditis originating from a prior venous cannulation site is undescribed in the current literature. Infections of the heart pose significant morbidity and mortality to patients, therefore prompt recognition, diagnosis, and treatment are critical. Our patient underwent coronary artery bypass grafting (CABG) and developed a postoperative sternal wound infection with methicillin resistant staphylococcus aureus (MRSA). After failing nonoperative management, redo-sternotomy was performed with atrial wall debridement and patch repair. During this procedure, two unexpected small discrete abscess pockets of the right atrial epicardium were discovered. One of the abscess pockets fistulized into the right atrium and was noted to be at his prior venous cannulation site for cardiopulmonary bypass as evidenced by neighboring prolene suture. The patient had an uneventful recovery and was discharged home on postoperative day 7. Transthoracic echo was obtained 6 weeks after his second operation and did not show any recurrence of endocarditis. We present a unique case of persistent cardiac infection with a complicated course and management strategy.
Keywords
Epicarditis; Endocarditis; Epicardial Abscess; MRSA; Atrial Vegetation; Redo Sternotomy; Mediastinitis; Pericarditis; Coronary Artery Bypass Grafting
Epicarditis articles; Endocarditis articles; Epicardial Abscess articles; MRSA articles; Atrial Vegetation articles; Redo Sternotomy articles; Mediastinitis articles; Pericarditis articles; Coronary Artery Bypass Grafting articles
COVID-19 articles COVID-19 Research articles COVID-19 review articles COVID-19 PubMed articles COVID-19 PubMed Central articles COVID-19 2023 articles COVID-19 2024 articles COVID-19 Scopus articles COVID-19 impact factor journals COVID-19 Scopus journals COVID-19 PubMed journals COVID-19 medical journals COVID-19 free journals COVID-19 best journals COVID-19 top journals COVID-19 free medical journals COVID-19 famous journals COVID-19 Google Scholar indexed journals Epicarditis articles Epicarditis Research articles Epicarditis review articles Epicarditis PubMed articles Epicarditis PubMed Central articles Epicarditis 2023 articles Epicarditis 2024 articles Epicarditis Scopus articles Epicarditis impact factor journals Epicarditis Scopus journals Epicarditis PubMed journals Epicarditis medical journals Epicarditis free journals Epicarditis best journals Epicarditis top journals Epicarditis free medical journals Epicarditis famous journals Epicarditis Google Scholar indexed journals Endocarditis articles Endocarditis Research articles Endocarditis review articles Endocarditis PubMed articles Endocarditis PubMed Central articles Endocarditis 2023 articles Endocarditis 2024 articles Endocarditis Scopus articles Endocarditis impact factor journals Endocarditis Scopus journals Endocarditis PubMed journals Endocarditis medical journals Endocarditis free journals Endocarditis best journals Endocarditis top journals Endocarditis free medical journals Endocarditis famous journals Endocarditis Google Scholar indexed journals MRSA articles MRSA Research articles MRSA review articles MRSA PubMed articles MRSA PubMed Central articles MRSA 2023 articles MRSA 2024 articles MRSA Scopus articles MRSA impact factor journals MRSA Scopus journals MRSA PubMed journals MRSA medical journals MRSA free journals MRSA best journals MRSA top journals MRSA free medical journals MRSA famous journals MRSA Google Scholar indexed journals Ultrasound articles Ultrasound Research articles Ultrasound review articles Ultrasound PubMed articles Ultrasound PubMed Central articles Ultrasound 2023 articles Ultrasound 2024 articles Ultrasound Scopus articles Ultrasound impact factor journals Ultrasound Scopus journals Ultrasound PubMed journals Ultrasound medical journals Ultrasound free journals Ultrasound best journals Ultrasound top journals Ultrasound free medical journals Ultrasound famous journals Ultrasound Google Scholar indexed journals treatment articles treatment Research articles treatment review articles treatment PubMed articles treatment PubMed Central articles treatment 2023 articles treatment 2024 articles treatment Scopus articles treatment impact factor journals treatment Scopus journals treatment PubMed journals treatment medical journals treatment free journals treatment best journals treatment top journals treatment free medical journals treatment famous journals treatment Google Scholar indexed journals CT articles CT Research articles CT review articles CT PubMed articles CT PubMed Central articles CT 2023 articles CT 2024 articles CT Scopus articles CT impact factor journals CT Scopus journals CT PubMed journals CT medical journals CT free journals CT best journals CT top journals CT free medical journals CT famous journals CT Google Scholar indexed journals Mediastinitis articles Mediastinitis Research articles Mediastinitis review articles Mediastinitis PubMed articles Mediastinitis PubMed Central articles Mediastinitis 2023 articles Mediastinitis 2024 articles Mediastinitis Scopus articles Mediastinitis impact factor journals Mediastinitis Scopus journals Mediastinitis PubMed journals Mediastinitis medical journals Mediastinitis free journals Mediastinitis best journals Mediastinitis top journals Mediastinitis free medical journals Mediastinitis famous journals Mediastinitis Google Scholar indexed journals Redo Sternotomy articles Redo Sternotomy Research articles Redo Sternotomy review articles Redo Sternotomy PubMed articles Redo Sternotomy PubMed Central articles Redo Sternotomy 2023 articles Redo Sternotomy 2024 articles Redo Sternotomy Scopus articles Redo Sternotomy impact factor journals Redo Sternotomy Scopus journals Redo Sternotomy PubMed journals Redo Sternotomy medical journals Redo Sternotomy free journals Redo Sternotomy best journals Redo Sternotomy top journals Redo Sternotomy free medical journals Redo Sternotomy famous journals Redo Sternotomy Google Scholar indexed journals Pericarditis articles Pericarditis Research articles Pericarditis review articles Pericarditis PubMed articles Pericarditis PubMed Central articles Pericarditis 2023 articles Pericarditis 2024 articles Pericarditis Scopus articles Pericarditis impact factor journals Pericarditis Scopus journals Pericarditis PubMed journals Pericarditis medical journals Pericarditis free journals Pericarditis best journals Pericarditis top journals Pericarditis free medical journals Pericarditis famous journals Pericarditis Google Scholar indexed journals
Article Details
1. Introduction
Post-CABG mediastinal infections significantly increase hospital length of stay, cost, and patient mortality [1]. The overall incidence of deep sternal wound infections (DSWI) following median sternotomy is reported to be between 0.2% and 3% [2]. Some researchers have found DSWIs to be associated with an almost tripled health care cost and doubled hospital length of stay, reinforcing our current understanding of the significant economic impact [3].
There are currently no described cases of endocarditis of a previous cannulation site for cardiopulmonary bypass. We describe a case in which a patient developed a MRSA sternal wound infection after coronary bypass that was complicated by persistent bacteremia and right atrial vegetation ultimately requiring redo sternotomy with intraoperative discovery of two epicardial abscess pockets eroding through the atrial wall. One of the abscess pockets communicated directly with the right atrium and was noted to be at the prior venous cannulation site for cardiopulmonary bypass as evidenced by neighboring prolene suture. Our goal is to bring awareness to a previously undescribed cardiac infection to assist other clinicians in promptly diagnosing and treating this complicated disease process.
2. Case Report
2.1 History
The patient is a 56-year-old male with a history of hypertension, hyperlipidemia, atrial fibrillation, and coronary artery disease status post four vessel CABG. His initial postoperative course was complicated by a delayed MRSA positive sternal wound infection requiring debridement and bilateral pectoralis major flaps three months after his index operation. At that time he was also found to have MRSA bacteremia. He was discharged on a prolonged course of intravenous antibiotics with clearing of his blood cultures. Unfortunately, he developed recurrent fevers, and repeat blood cultures and a transthoracic echocardiogram were obtained. Blood cultures again were positive for MRSA positivity and echo showed right atrial/tricuspid valve vegetation concerning for MRSA endocarditis. The patient was readmitted and restarted on broad spectrum IV antibiotics. A transesophageal echocardiogram was obtained that confirmed large multi lobular mobile vegetation on the anterior portion of the right atrial free wall and extending into the right atrial appendage, measuring 3.0 cm x 1.7 cm. His ejection fraction at that time was 52%. He underwent catheter aspiration of the right atrial vegetation with the Angiovac™ system with an estimated 80% extraction during that procedure. He was able to clear his blood cultures and was discharged home with IV antibiotics and plan for 1 month follow up blood cultures and echocardiogram. Unfortunately, his follow up blood cultures were once again positive and his echo redemonstrated mobile right vegetation. He was subsequently readmitted and started on vancomycin. Confirmatory transesophageal echo was performed that showed a large 3.1 cm x 1.4 cm mobile mass in the right atrium. At this point, it was decided the patient would require operative intervention to clear his infection and atrial vegetation. He underwent a preoperative left heart catheterization which demonstrated widely patent bypass grafts.
The patient was taken to the operating room and underwent reoperative sternotomy. There were significant scarring/adhesions of the pericardium and epicardial tissues. Prior bypass grafts were noted and avoided. Arterial access for cardiopulmonary bypass was the ascending aorta and venous accesses were the superior vena cava and inferior vena cava. The inferior vena cava was accessed percutaneously through the right common femoral vein. During the dissection two small pockets of epicardial abscess were identified and cultured. The right atrium was opened and the vegetation identified and removed. It seemed to be at the area of the prior venous cannulation site and near the two small epicardial abscesses with one of the abscess pockets communicating directly with the right atrium. All involved areas of the right atrium were removed. The tricuspid valve was evaluated and did not appear infected or to have structural defects. The right atrial defect was too large to close primarily, and a bovine pericardium patch reconstruction was used. Final cultures from the operation were positive for MRSA. The patient’s postoperative course was uneventful and he was discharged home on postoperative day 7 with 6 weeks of IV antibiotics. He had repeat blood cultures and a transthoracic echocardiogram four weeks after discharge which were all negative for recurrent infection.
3. Discussion
While relatively rare, the U.S. sees between 40,000 and 50,000 new endocarditis cases per year [4]. The evolving risk factors of this well described infection have been characterized in generations of cardiac literature. Prosthetic valve replacement, immunosuppression, and IV drug abuse are the principal risk factors [5]. Despite advances in medicine, endocarditis is still associated with a 15-20% in-hospital mortality [6]. Endocarditis after CABG is a known risk factor for the development of a DSWI, but the development of endocarditis following DSWI for non-valvular surgery is rare [7]. We have presented a unique case of persistent cardiac infection with a complicated course and management strategy. There are currently no documented cases of epicardial abscess originating from a prior cannulation site. It is important for clinicians to be aware of this disease process and the risk it poses to patients with recurrent mediastinal infections after cardiopulmonary bypass. We suspect the non-absorbable suture used to close this cannulation site in the standard fashion may have acted as a nidus for recurrent infection in a patient whose postoperative course was complicated by MRSA mediastinitis. This may have allowed bacteria to erode through the atrial wall posing significant risk of hemorrhage. An awareness of this disease process may lean the clinician towards earlier reoperation and potentially prevent catastrophic hemorrhage.
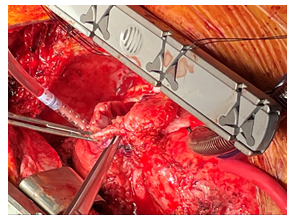
Figure 1: Forceps placed through atrial wall erosion into the atrial cavity.
References
- Hollenbeak, et al. The Clinical and Economic Impact of Deep Chest Surgical Site Infections Following Coronary Artery Bypass Graft Surgery. CHEST 118 (2000): 397-402.
- Phoon, et al. Deep Sternal Wound Infection: Diagnosis, Treatment and Prevention. J of Card and Vasc Anest 34 (2020): 1602-1613.
- Graf K, et al. Economic aspects of deep sternal wound infections. Euro J of Card-Thorac Surg 37 (2010): 893-896.
- Bor D, et al. Infective Endocarditis in the U.S, 1998-2009: A Nationwide Study. Plos ONE 8 (2013): 1-8.
- Slipczuk L, et al. Infective Endocarditis Epidemiology Over Five Decades: A Systematic Review. Plos ONE 8 (2013): 1-17.
- Murdoch D, et al. Clinical Presentation, Etiology, and Outcome of Infective Endocarditis in the 21st Century. Arch of Intern Med 169 (2009): 463-473.
- Toumpoulis I, et al. The Impact of Deep Sternal Wound Infection on Long-Term Survival after Coronary Artery Bypass Grafting. CHEST 127 (2005): 464-471.
