Efficacy of the Super-Retinal Imaging Display Based on Scanning Laser Technology in Visual Impairment Induced by Corneal Diseases
Article Information
Hisataka Fujimoto*, Junichi Kiryu
Department of Ophthalmology, Kawasaki Medical School, Kurashiki, Okayama, Japan
*Corresponding author: Hisataka Fujimoto, Department of Ophthalmology, Kawasaki Medical School, Kurashiki, Okayama, Japan
Received: 30 August 2021; Accepted: 06 September 2021; Published: 08 September 2021
Citation:
Hisataka Fujimoto, Junichi Kiryu. Efficacy of the Super-Retinal Imaging Display Based on Scanning Laser Technology in Visual Impairment Induced by Corneal Diseases. Journal of Ophthalmology and Research 4 (2021): 263-278.
View / Download Pdf Share at FacebookAbstract
We investigated the effectiveness of wearable laser-eyewear (LEW) in visual impairment owing to corneal disease, including low vision (LV) eye. LEW was performed in 35 patients (38 eyes) with irregular corneal astigmatism. Best corrected visual acuity (BCVA) and visual acuity (VA) with LEW were measured using Landolt rings. Corneal topography was measured using anterior segment optical coherence tomography; corneal opacity grade was observed using slit lamp examination. A significant negative correlation between Logarithm of the Minimum Angle of Resolution (LogMAR) VA improvement using LEW and basal BCVA was observed. In 20 eyes of the LV, no significant correlation was observed between VA improvement by LEW and basal BCVA. The LogMAR VA improvement using LEW was 0.27±0.32 (-0.48−0.70) in ×1 0.53±0.37 (-0.17−1.18) in ×2. The most significant factor that determined VA improvement in eyes with LV using LEW was the corneal opacity grade, while the corneal topographic parameters, including keratometric refractive parameters (Kmax, Ks, Kf, and Cylinder), higher-order aberrations (HOAs), and central corneal thickness (CCT), showed no or limited impact on the VA improvement effect by LEW in LV. Therefore, LEW improves vision regardless of irregular corneal astigmatism; however, corneal opacity limits the LEW effect in the LV eye.
Keywords
<p>Laser-eyewear; Low vision; Corneal opacity</p>
Laser-eyewear articles; Low vision articles; Corneal opacity articles
Article Details
1. Introduction
Low vision (LV) is a chronic visual impairment resulting from several visual system disorders that cannot be corrected with glasses, medical treatment, or surgery. Such defects influence individuals’ quality of life (QOL) by interfering with free actions [1]. Age-related macular degeneration (AMD), diabetic retinopathy, and glaucoma are some of the reasons for LV and blindness [2]. Other diseases affecting the anterior segment of the eye can also lead to blindness. These include LV owing to corneal diseases, such as keratokonus, corneal dystrophies, opacities caused by infections, or corneal epithelial stem cell deficiency. Not all patients receive successful treatment with surgery, such as keratoplasty or limbal-stem-cell transplantation. Few patients are do not undergo surgery owing to ocular risk factors or rejection of surgery [3,4]. In addition, limited donor supply results in waiting periods of months or years for patients for keratoplasty [5]. A higher prevalence of depression and loss of quality of life are associated with patients of LV [6,7]. Therefore, supporting the patient´s visual impairment and increasing their autonomy are imperative. Vision can be enhanced by bypassing the diseased anterior part of the eye in patients with impairment of the optic media and preserved sensory function [4].
A new type of retinal imaging wearable laser-eyewear (LEW) might be capable of delivering such a bypass, and allows projection of images directly onto the retina, thereby, improving the visual acuity (VA) of patients [8]. More specifically, the RGB (red, green, and blue)-laser image was projected onto the retina using the microelectromechanical (MEMS) system developed by QD Laser, Inc. (Kawasaki, Japan). In contrast to the typical video displays in which optical systems are generated for eyes, the greatest advantage of QD laser eyewear is that it employs a Maxwellian view optical system [9,10]. In this system, a real image of a light source is focused on the pupil of the eye, and the image reaches the retina without defocusing, irrespective of the axial length or refractive indices of the ocular parts. Thus, correction of the refractive error is not required. The Maxwellian view optical system is often used to present stimuli for eye examinations, for example, to analyze the optical density of the lens [11], macular pigment [12], color appearance [13], cone function [14], photo stress recovery related to colored intraocular lenses [15], light adaptation [16], and localization of photophobia [17]. Moreover, the resolution of the projector of the QD laser eyewear was as high as 1280 × 720, and that of the camera was 2 million pixels. The laser used in our study device was classified as a Class-1-laser according to IEC60825-1. The low energy output could be shown to be lower than that of a fluorescent lamp and is therefore harmless to the eye [18,19]. In the laser, the low-power red, green, and blue laser light oscillate; the blue light wavelength is at 1/40, and that of red and green is at 1/400 the power of the maximum levels permitted by the Japanese Industrial Standard and International Electrotechnical Commission.
In the present study, we sought to assess whether LEW may have an effect on visual impairment induced by corneal diseases, including severe LV eye. We assessed this using LEW in patients with irregular astigmatism induced by corneal disease. We also investigated the relationship between the LEW effect and the corneal topographic parameters or corneal opacity.
2. Materials and Methods
This retrospective observational study was approved by the Institutional Review Board of Kawasaki Medical School Hospital (approval number 5182-00). Informed consent was obtained from all the study participants, and the study was conducted in accordance with the Declaration of Helsinki. This study did not have patient or public involvement in its design, participant recruitment, or conduct.
For a consecutive series of 35 patients (38 eyes) ranging from 21 to 89 years (mean: 53.5 years), who had irregular corneal astigmatism that could not be corrected by glasses, and the best corrected VA (BCVA) was less than 20/20.
The causes of corneal astigmatism were keratoconus (KC, 9 eyes in 7 patients), corneal opacity induced by corneal dystrophy or limbal stem cell deficiency (8 eyes in 7 patients), scarring induced by corneal infection (3 eyes in 3 patients), post-keratoplasty (16 eyes in 16 patients), and bullous keratopathy (BK, two eyes in two patients) (Table 1). Keratoplasty was performed in 14 eyes as penetrating keratoplasty (PKP) and in 2 eyes as deep anterior lamellar keratoplasty in (DALK) two eyes.
The inclusion criteria were as follows: patients with irregular corneal astigmatism, but without other anterior segment ocular disorders, such as epithelial erosions, active infections, conjunctivitis, and nasolacrimal duct obstruction. In contrast, the exclusion criteria included any fundus disease including retinal or optic nerve disease, active systemic diseases such as cardiovascular disease and/or renal disease. Patients with diabetes mellitus and systemic hypertension, which were controlled through the administration of oral medications, were included in the final cohort.
The outcome measures of interest were BCVA and VA using the LEW (RETISSA Medical, QD Laser, Inc. Kawasaki, Japan) clinically approved in Japan. VA was measured using the Landolt rings. Corneal topography was measured using anterior segment optical coherence tomography (AS-OCT). In addition, corneal opacity grade was observed by slit-lamp examination.
Corneal tomographic measurements were obtained by AS-OCT using CASIA 2 (Tomey Corporation, Inc., Nagoya, Japan) with an infrared light wavelength of 1310 nm. Measurements were obtained along the vertex normal, and the images were centered on the corneal vertex. The analysis software (Tomey Corporation, Inc., Nagoya, Japan) was used to identify and digitize both the anterior and posterior corneal surfaces and to align the reference axis of the measurement with the vertex normal. Tomographic and pachymetric maps were calculated from 16 radial cross-sectional images through the central 10-mm diameter of the cornea obtained in 0.34 seconds.
Swept-source OCT measurements have been shown to have adequate repeatability in healthy eyes, as well as in eyes with corneal disease such as keratoconus [20]. Total (i.e., including both the anterior and posterior) keratometric refractive parameters (Kmax, Ks, Kf, Kave, and Cylinder, in Diopter), and corneal total higher-order aberrations in 4mm (HOAs, in μm) were measured. The other corneal topographic parameters, such as the central corneal thickness (CCT, in μm), were measured. Each measurement satisfied the following conditions: over 96.5% of the data within the central 6-mm diameter had adequate contrast to trace the corneal border, all 16 radial scans within the central 4-mm diameter topographic analysis could be performed for both the anterior and posterior corneal surfaces, the distance from the center of the image to the corneal vertex was less than 0.86 mm, and over 93% of the scans inside the central 6-mm diameter topographic analysis could be obtained for both the anterior and posterior surfaces.
Corneal opacity grade was observed by slit-lamp examination: Grade 0, no opacity was observed in the cornea; grade 1, 100% of the pupil is observable in diffuser illumination; grade 2, 1–99% of the pupil is observable in diffuser illumination; grade 3, pupil cannot be observed in diffuser illumination but can be observed in slit lamp illumination; and grade 4, pupil cannot be observed under slit lamp illumination. Nuclear hardness graded by the Emery-Little classification was also examined in phakic eyes.
2.1 Statistical analysis
Data distributions of Logarithm of the Minimum Angle of Resolution (LogMAR) VA were assessed for normality using the Shapiro-Wilk test. Following the assumption that all data on outcomes of interest followed a parametric distribution, the paired t-test was used for LogMAR VA using LEW at magnifications of ×1 and ×2 (LogMAR LEW VA ×1 and LogMAR LEW VA ×2, respectively) and BCVA. The VA improvement by LEW wear was calculated as LogMAR change = –(LogMAR LEW VA – LogMAR BCVA). Correlations between the LogMAR change and corneal topographic parameters or corneal opacity grade were analyzed using the Pearson correlation coefficients in linear regression, and the levels of significance of the correlations were set at p < 0.05. The ANOVA and Tukey HSD posthoc tests were performed to compare the LogMAR VA in BCVA, LEW use, rigid gas-permeable contact lens. All analyses were conducted using SPSS version 25.0 (IBM Corp. Released 2017. IBM SPSS Statistics for Windows, version 25.0. Armonk, NY: IBM Corp. USA).
3. Results
The study included 38 eyes of 35 patients. Of these, 20 eyes from 18 patients had LV eyes with 20/60 BCVA. The mean participant age was 53.5 ± 21.1 years (Table 1).
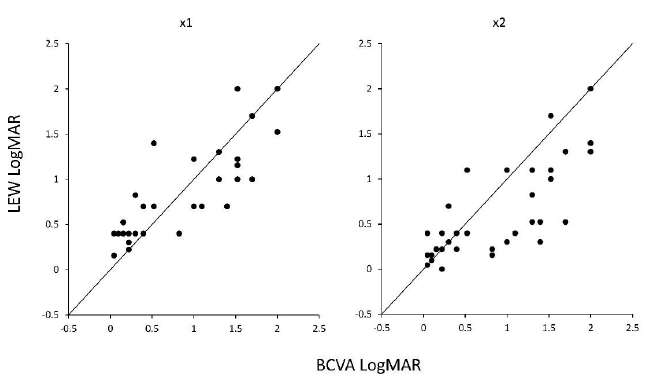
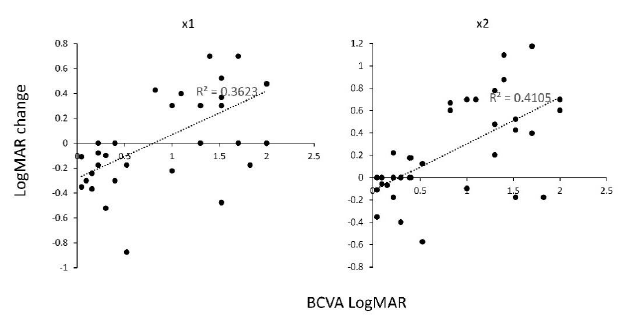
The LogMAR VA was 0.87 ± 0.67 in BCVA; by LEW use, this changed to 0.84 ± 0.52 in ×1 magnification (p = 0.66, paired t-test) and significantly improved to 0.61 ± 0.51 in ×2 magnification (p = 0.0009, paired t-test) (Figure 1, Supplementary Figure 1). A significant negative correlation was observed between LogMAR VA improvement by LEW and basal BCVA (Figure 2). As previously reported [8], in eyes with originally good BCVA, relatively few visual impairments were desired.
BCVA, best corrected visual acuity; CCT, central corneal thickness; D, diopter; HOAs, higher-order aberrations; IOL, intraocular lens; LEW, laser eye wear; LogMAR, Logarithm of the Minimum Angle of Resolution; LV, low vision; VA, visual acuity
Table 1: BCVA and VA in LEW use and the background parameters in all and the LV group
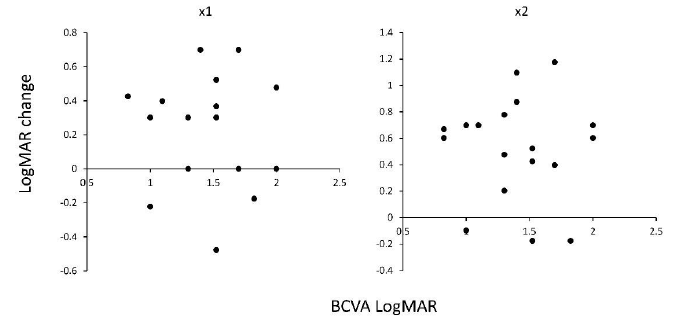
Thus, we focused on eyes of LV under 20/60. In the LV eye, the LogMAR BCVA was 1.41 ± 0.34; by LEW use, this significantly improved to 1.15 ± 0.50 in ×1 magnification (p = 0.0014) and to 0.89 ± 0.51 in ×2 magnification (p = 0.00004). We observed no significant correlation between LogMAR VA improvement by LEW and basal BCVA in the LV eye (Figure 3, Table 2), suggesting that LEW improves the VA regardless of the basal BCVA and severity of corneal abnormality.
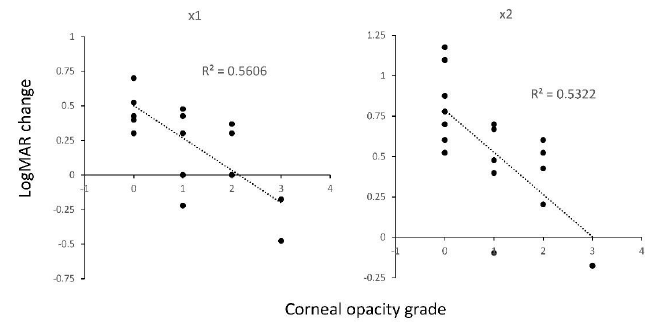
Next, we investigated the effect of corneal opacity on VA improvement by LEW in the LV eyes. We observed a strong correlation (R2 = 0.561 in x1, and R2 = 0.532 in ×2) between LogMAR VA improvement by LEW and corneal opacity (Figure 4, Table 2). This suggests that corneal opacity limits the LEW effect in the LV eye. In grade 0 of the no corneal opacity eye (N = 8 eye), LogMAR improvement by LEW use was up to 0.51 ± 0.17 in ×1 magnification and 0.78 ± 0.25 in ×2 magnification. Calculated as fractional VA, the average BCVA was 20/450, which improved to 20/140 in LEW ×1 and to 20/70 in LEW ×2 in eyes with grade 0 corneal opacity.
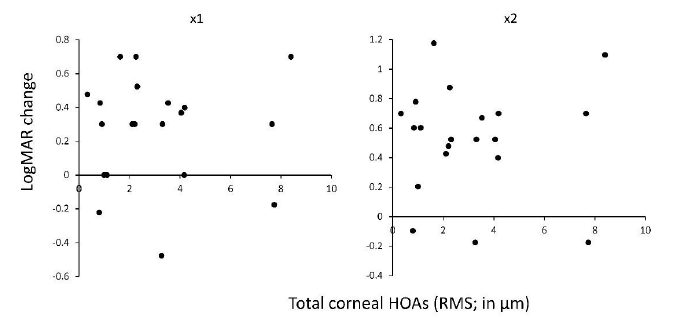
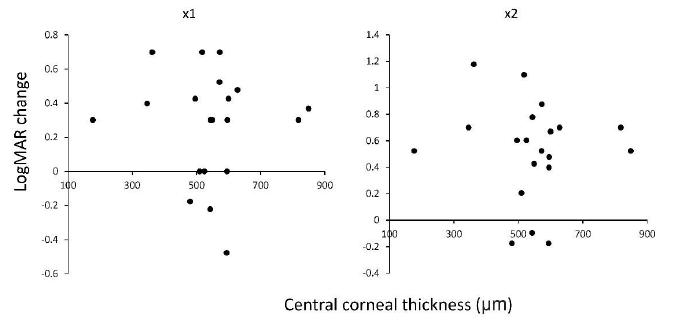
Next, we investigated the correlation between corneal topographic effect and irregular corneal astigmatism on VA improvement by LEW. We observed no correlation (p = 0.78 in ×1, and p = 0.84 in ×2) between LogMAR VA improvement by LEW and HOAs (Figure 5, Table 2). This suggests that LEW improves the VA, regardless of irregular corneal astigmatism. We observed no correlation (p = 0.28, ×1, and p = 0.17 in ×2) between LogMAR VA improvement by LEW and another corneal topographic parameter, CCT (Figure 6, Table 2).
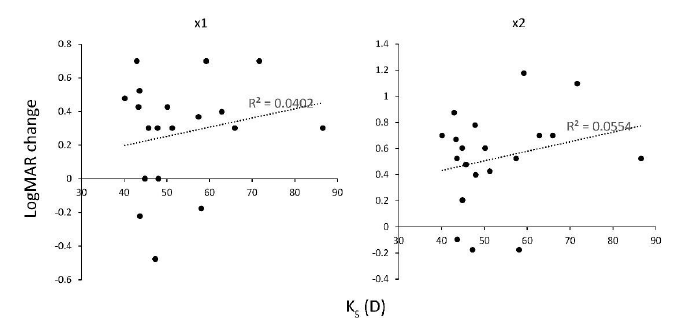
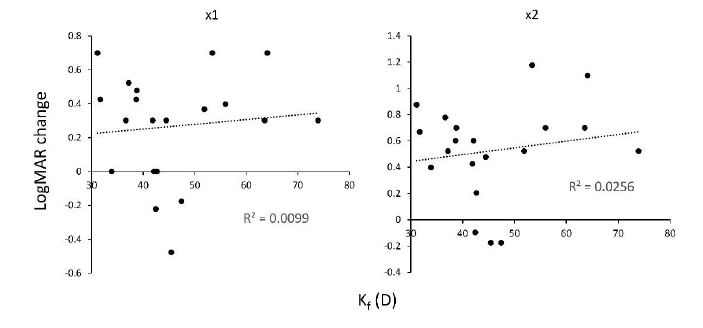
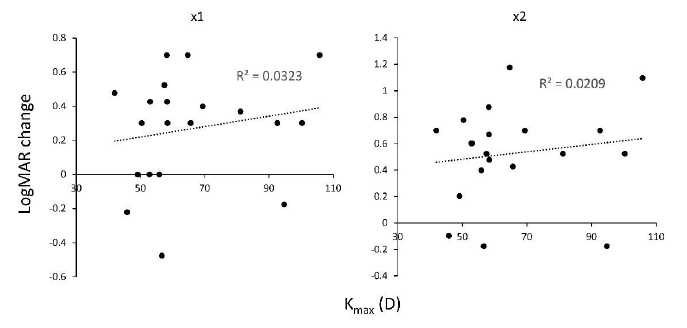
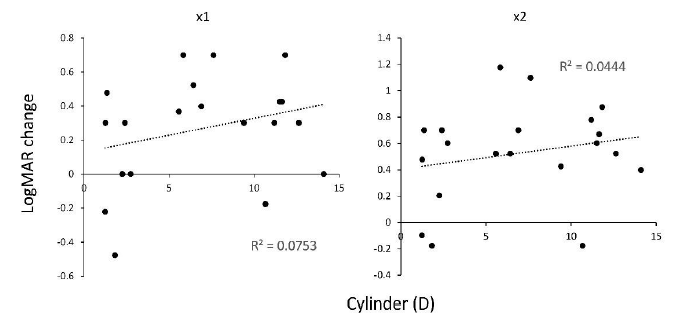
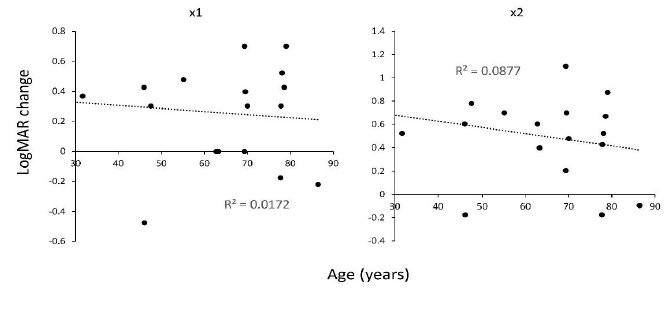
Next, we measured the corneal refractive parameters on VA improvement by LEW (Table 2). We observed a weak correlation between LogMAR VA improvement by LEW and Ks (R2 = 0.040 for ×1 and R2 = 0.055 for ×2, Figure 7) , Kf (R2 = 0.010 in x1 and R2 = 0.026 in ×2, Figure 8), Kmax (R2 = 0.032 in x1 and R2 = 0.021 in ×2, Figure 9), and cylinder (R2 = 0.075 in ×1 and R2 = 0.044 in ×2, Figure 10). This suggests that the refractive power has a low impact on LEW and improves VA. In addition, we measured the background parameters of VA improvement by LEW (Table 2). We observed weak or no correlation between LogMAR VA improvement by LEW and age (R2 = 0.017 in ×1 and R2 = 0.088 in ×2, Figure 11) as previously reported8, gender (p = 0.57 · in ×1, and p = 0.58 · in ×2), or cataract grade estimated by nuclear hardness graded by the Emery-Little classification (p = 0.21, ×1, and p = 0.31, ×2) and LogMAR VA improvement by LEW (Table 2).
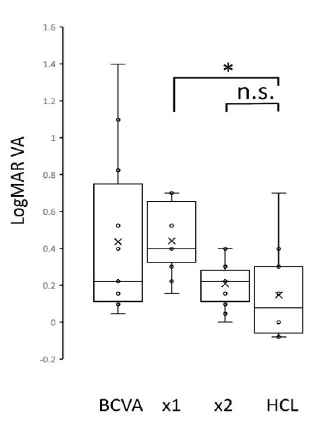
Finally, we measured the effectiveness of LEW comparing to rigid gas-permeable contact lens in 12 contact lens users among these participants. LogMAR VA was 0.44 ± 0.44, 0.48 ± 0.23, 0.20 ± 0.12 and 0.20 ± 0.30, in BCVA, LEW x1, LEW x2 and rigid gas-permeable contact lens use, respectively (Figure 12). LogMAR VA in LEW x2 use was not significantly different in rigid gas-permeable contact lens use (p = 0.86, ANOVA and Tukey HSD posthoc tests).
BCVA, best corrected visual acuity; CCT, central corneal thickness; D, diopter; HOAs, higher-order aberrations; IOL, intraocular lens; LEW, laser eye wear; LogMAR, Logarithm of the Minimum Angle of Resolution; LV, low vision; VA, visual acuity
Table 2: Correlation of VA improvement by LEW use and the corneal topographic, corneal opacity, and background parameters in the 20 eyes of low vision (LV, BCVA < 20/60) group.
Figure 2: Correlation between BCVA and VA improvement in LogMAR scale using the LEW in all 38 eyes. The left panel shows the visual acuity using LEW in ×1 magnification, and the right panel shows the visual acuity using LEW in ×2 magnification. BCVA, best corrected visual acuity; LEW, laser eye wear; LogMAR, Logarithm of the Minimum Angle of Resolution; VA, visual acuity
Figure 3: Correlation between BCVA and VA improvement in LogMAR scale using the LEW in 20 eyes under 20/60 of LV. The left panel shows the VA using LEW in ×1 magnification, and the right panel shows the VA using LEW in ×2 magnification. No significant correlation was observed (p ≥ 0.05) in both of the ×1 and ×2 magnification. BCVA, best corrected visual acuity; LEW, laser eye wear; LV, low vision; LogMAR, Logarithm of the Minimum Angle of Resolution; VA, visual acuity
Figure 4: Correlation between the corneal opacity grade and VA improvement in LogMAR scale using the LEW in 20 eyes under 20/60 of LV. The left panel shows the VA using LEW in ×1 magnification, and the right panel shows the VA using LEW in ×2 magnification. Strong negative correlation was observed (p < 1.0 ×10-12) in both of the ×1 and ×2 magnification. LEW, laser eye wear; LogMAR, Logarithm of the Minimum Angle of Resolution; LV, low vision; VA, visual acuity
Figure 5: Correlation between total corneal HOAs measured by AS-OCT and VA improvement in LogMAR scale using the LEW in 20 eyes of LV. No significant correlation was observed (p ≥ 0.05) in both of the ×1 and ×2 magnification. AS-OCT, anterior segment optical coherent tomography; HOAs, higher-order aberrations; LEW, laser eye wear; LogMAR, Logarithm of the Minimum Angle of Resolution; LV, low vision, VA, visual acuity
Figure 6: Correlation between CCT measured by AS-OCT and VA improvement in LogMAR scale using the LEW in 20 eyes of LV. No significant correlation was observed (p ≥ 0.05) in both of the ×1 and ×2 magnification. AS-OCT, anterior segment optical coherent tomography; CCT, central corneal thckness; LEW, laser eye wear; LogMAR, Logarithm of the Minimum Angle of Resolution; LV, low vision; VA, visual acuity
Figure 7: Correlation between keratometric refractive parameter Ks measured by AS-OCT and VA improvement in LogMAR scale using the LEW in 20 eyes of LV. Weak significant positive correlation was observed in both of the ×1 and ×2 magnification. AS-OCT, anterior segment optical coherent tomography; LEW, laser eye wear; LogMAR, Logarithm of the Minimum Angle of Resolution; LV, low vision; VA, visual acuity
Figure 8: Correlation between keratometric refractive parameter Kf measured by AS-OCT and VA improvement in LogMAR scale using the LEW in 20 eyes of LV. Weak significant positive correlation was observed in both of the ×1 and ×2 magnification. AS-OCT, anterior segment optical coherent tomography; LEW, laser eye wear; LogMAR, Logarithm of the Minimum Angle of Resolution; LV, low vision; VA, visual acuity
Figure 9: Correlation between keratometric refractive parameter Kmax measured by AS-OCT and VA improvement in LogMAR scale using the LEW in 20 eyes of LV. Weak significant positive correlation was observed in both of the ×1 and ×2 magnification. AS-OCT, anterior segment optical coherent tomography; LEW, laser eye wear; LogMAR, Logarithm of the Minimum Angle of Resolution; LV, low vision; VA, visual acuity
Figure 10: Correlation between keratometric cylindrical power measured by AS-OCT and VA improvement in LogMAR scale using the LEW in 20 eyes of LV. Weak significant positive correlation was observed in both of the x1 and x2 magnification. AS-OCT, anterior segment optical coherent tomography; LEW, laser eye wear; LogMAR, Logarithm of the Minimum Angle of Resolution; LV, low vision; VA, visual acuity
Figure 11: Correlation between age and VA improvement in LogMAR scale using the LEW in 20 eyes of LV. Weak significant negative correlation was observed in both of the ×1 and ×2 magnification. LEW, laser eye wear; LogMAR, Logarithm of the Minimum Angle of Resolution; LV, low vision; VA, visual acuity
Figure 12: Comparison among VAs in LogMAR scale using the LEW in 12 eyes of rigid gas-permeable contact lens use. LogMAR VA in LEW x2 use was not significantly different in rigid gas-permeable contact lens use. The horizontal lines in the box and whisker plots represent the median values, and the bottom and top of the boxes represent the lower and upper quartiles, respectively. The x represents the mean and the bars represent the minimum and maximum values within 1.5 times the lower and upper quartiles. * p < 0.001, ANOVA and Tukey HSD posthoc tests. HCL, rigid gas-permeable contact lens; LEW, laser eye wear; LogMAR, Logarithm of the Minimum Angle of Resolution; VA, visual acuity
4. Discussion
Our study revealed that LEW improves the VA regardless of irregular corneal astigmatism; however, the corneal opacity limits the LEW effect in the LV eye. In addition, LEW showed effectiveness relatively independent of refractive errors, consistent with previously reported studies in relatively good VA (not LV) [8].
Currently, there is no comparable device that uses the principle of the Maxwellian view. LEW effectiveness was prominent, especially in LV eyes without corneal opacity; 20/450 BCVA improved to 20/140 (×1 magnification) and 20/70 (×2 magnification) by LEW use on average. This is three times (×1) and seven times (×2) greater improvement in the resolution. On the other hand, the effectiveness of LEW was predominantly limited by corneal opacity and quenched in eyes with severe corneal opacity of grade 3 (figure 4).
The LEW effect was confirmed to be independent of irregular corneal astigmatism measured by HOAs (Figure 5). The theoretical independence described by Sugawara et al. could be confirmed [21]. Moreover, this is indicative that LEW, using the principle of Maxwellian view, had unique efficacy in eyes with severe irregular astigmatism due to corneal disease, especially in eyes without surgical indication for several reasons, such as rejection or mental retardation (Figure S1). A weak relationship was observed between LogMAR VA improvement by LEW and age (Figure 11), as previously reported. In accordance with previous trials, one of the reasons could be that VA generally decreases with age, even in healthy individuals [8].
We observed only a weak positive correlation between LogMAR VA improvement by LEW and Ks, Kf, Kmax, and cylinder (Figures 7–10) in LV eyes with BCVA < 20/60. A previous study reported that LEW VA was not significantly affected by the amount of refractive error in eyes with BCVA ≥ 20/25 [8]. In our study of the LV eye, basal BCVA was relatively low; thus, the possibility of VA improvement could remain in severe cornea. Overall, refractive power has a low impact on the improvement of VA by LEW. In addition, in rigid gas-permeable contact lens uses, LEW in x2 is comparable to the rigid gas-permeable contact lens effect on visual acuity improvement (Figure 12).
The main limitations of the present study are its retrospective design and small sample size. Our results need to be verified by prospective, randomized, masked studies in the future. The small sample size reflects the fact that our patient groups needed to include patients with corneal diseases. The data here suggest that examining a large cohort of patients, with and without LV, would be important to confirm that the use of LEW has a broad effect in patients with impairment of the optic media, including the cornea.
5. Conclusions
Our results suggest that LEW using the Maxwellian view could help improve the VA in patients with irregular corneal astigmatism. The efficacy of LEW was prominent in severe cases of the LV eye. However, accompanying corneal opacity limits the effect of LEW on VA improvement. In addition, the LEW effect is relatively independent of the refractive error, especially in irregular corneal astigmatism. These findings suggest that LEW could be beneficial for LV aid in eyes with severe corneal diseases.
Funding
This research was funded by Research Project Grant R02B-010 from Kawasaki Medical School (to H.F.), and JSPS KAKENHI grants 25861630 and 16K18378 (to H.F.)
Acknowledgements
We thank K. Haruishi, S. Araki, S. Goto, A. Masuda, and Y. Mito for discussions, advice, and criticisms that greatly benefited this project.
Conflicts of Interests
Hisataka Fujimoto, Alcon (F), Otsuka (F), Kowa (F, P), Santen (F), Novartis (F), Junichi Kiryu, (F), Kowa: (F), Alcon: (F), Novartis: (F), Otsuka: (F)
References
- West SK, Rubin GS, Broman AT, et al. How does visual impairment affect performance on tasks of everyday life? The SEE Project. Salisbury Eye Evaluation. Archives of Ophthalmology 120 (2002): 774-780.
- Chan TC, Friedman DS, Bradley C, Massof RW. Estimates of incidence and prevalence of visual impairment, low vision, and blindness in the United States. JAMA Ophthalmology 136 (2018): 12-19.
- Oeverhaus M, Stöhr M, Hirche H, et al. Visual rehabilitation of patients with corneal diseases. BMC Ophthalmology 82 (2020): 20.
- Nguyen NX, Seitz B, Martus P, et al. Long-term topical steroid treatment improves graft survival following normal-risk penetrating keratoplasty. American Journal of Ophthalmology 144 (2007): 318-319.
- Pauklin M, Steuhl KP, Meller D. Limbal stem cell deficiency after chemical burns: investigations on the epithelial phenotype and inflammation status. Der Ophthalmologe 106 (2009): 995-998.
- Brody BL, Gamst AC, Williams RA, et al. Depression, visual acuity, comorbidity, and disability associated with age-related macular degeneration. Ophthalmology 108 (2001): 1893-1900.
- Trousdale ER, Hodge DO, Baratz KH, et al. Vision-related quality of life before and after keratoplasty for Fuchs’ endothelial dystrophy. Ophthalmology 121 (2014): 2147-2152.
- Stöhr M, Dekowski D, Bechrakis N, et al. First Evaluation of a Retinal Imaging Laser Eyewear System Based Low Vision Aid. Clinical Ophthalmology 14 (2020): 4115-4123.
- Maxwell JC. On the Theory of Compound Colours, and the Relations of the Colours of the Spectrum, Philosophical Transactions of the Royal Society 150 (1860): 57-84.
- Westheimer G. “The Maxwellian View,” Vision Research 6 (1966): 669-682.
- Stringham JM, Hammond BR Jr. The glare hypothesis of macular pigment function. Optometry and Vision Science 84 (2007): 859-864.
- Stringham JM, Garcia PV, Smith PA, et al. Macular Pigment and Visual Performance in Low-Light Conditions. Investigative Ophthalmology and Vision Science 56 (2015): 2459-2468.
- Gordon J, Abramov I. Color appearance: Maxwellian vs. Newtonian views. Vision Research 48 (2008): 1879-1883
- Carmichael Martins A, Vohnsen B. Analysing the impact of myopia on the Stiles-Crawford effect of the first kind using a digital micromirror device. Ophthalmic and Physiological Optics 38 (2018): 273-280.
- Hammond BR, Bernstein B, Dong J. The Effect of the AcrySof natural lens on glare disability and photostress. American Journal of Ophthalmology 148 (2009): 272-276.
- Gloriani AH, Matesanz BM, Barrionuevo PA, et al. Influence of background size, luminance and eccentricity on different adaptation mechanisms. Vision Research 125 (2016): 12-22.
- Stringham JM, Fuld K, Wenzel AJ. Spatial properties of photophobia. Investigative Ophthalmology and Vision Science 45 (2004): 3838-3848.
- Sugawara M, Suzuki M, Miyauchi N. 14-5L: late-News Paper: retinal Imaging Laser Eyewear with Focus-Free and Augmented Reality. Digest of Technical Papers - SID International Symposium 47 (2016): 164-167.
- Sugawara M, Suzuki M, Miyauchi N. Image quality of retinal projection laser eyewear: how to achieve high resolution and free focus in proper balance. Yokohama, Japan: The 6th Laser Display and Lighting Conference 2017 (LDC 17) 2017.
- Szalai E, Berta A, Hassan Z, Módis L Jr. Reliability and repeatability of swept-source Fourier-domain optical coherence tomography and Scheimpflug imaging in keratoconus. Journal of Cataract and Refractive Surgery 38 (2012): 485-494.
- M Sugawara, Miyauchi N. Retinal imaging laser eye wear with focus-free and augmented reality. Digest of Technical Papers - SID Internation Symposium 47 (2016): 164-167.