Effects of Palm Oil Mill Effluent on the Histology of Gill, Liver, Kidney, Heart and Muscle and its Heamatological Effects on African Cat Fish, Clarias gariepinus
Article Information
Oluniyi AA1; Solomon OA1; Folakemi DA2; Oluwaseun AI1
1Department of Pure and Applied Biology, Ladoke Akintola University of Technology, PMB. 4000, Ogbomoso, Nigeria
2Department of Animal Production and Health, Ladoke Akintola University of Technology,PMB. 4000, Ogbomoso, Nigeria
*Corresponding Author: Oluniyi A. Afolabi, Department of Pure and Applied Biology, Ladoke Akintola University of Technology, PMB. 4000, Ogbomoso, Nigeria
Received: 14 March 2023; Accepted: 23 March 2023; Published: 00 July 2023.
Citation: Oluniyi A. Afolabi; Solomon O. Adewoye; Folakemi D. Afolabi; Oluwaseun A. Ishola. Effects of palm oil mill effluent on the histology of gill, liver,kidney, heart and muscle and its heamatological effects on African cat fish, Clarias gariepinus. Archives of Nephrology and Urology. 6 (2023): 90-98.
View / Download Pdf Share at FacebookAbstract
Palm oil mill effluent (POME) is one of the major sources of pollutant produced during oil palm processing. POME in water significantly affects the ability of fishes to detect and respond to chemical stimulus. The aim of this study isto examine the toxicological effects of POME with emphasis on the histopathology of the gills, liver, kidney, heart and muscle as well as the heamatological effects. African catfish, Clarias gariepinus of mean weight 300g and standard length 18-21cm were exposed to varying concentrations 0.01, 0.03, 0.05, 0.07 and 0.09 ml/l of POME in duplicates for 14 days to determine the toxicity of POME. 110 healthy fishes were randomly placed into eleven rearing tanks of 100 litres capacity (dimension 50 cm x 25 cm x 25 cm) and filled with 40 litres non-chlorinated water at 10 fishes per tank (one tank for control and two tanks each for each concentration) Histopathological examination of the liver after 14 days of exposure revealed lesions which ranged from necrosis, vacuolation and diffused congestion of cells that deteriorate more with increase in POME effluent concentration. Toxic reactions were observed during the experiment. Haematological values of RBC, PCV and Hb decreases with increase in POME concentration while WBC registered an initial increase before declining at concentrations 0.07 and 0.09 ml/l. POME is harmful to aquatic organisms, according to the findings of this study, hence its discharge into bodies of water without sufficient treatment should be avoided.
Keywords
Clarias gariepinus; Heamatology; Histopathology; Toxicity; Lethal test; POME; Sublethal test.
Clarias gariepinus articles; Haematology articles; Histopathology articles; Toxicity articles; Lethal test articles; POME articles; Sublethal test articles.
Clarias gariepinus articles Clarias gariepinus Research articles Clarias gariepinus review articles Clarias gariepinus PubMed articles Clarias gariepinus PubMed Central articles Clarias gariepinus 2023 articles Clarias gariepinus 2024 articles Clarias gariepinus Scopus articles Clarias gariepinus impact factor journals Clarias gariepinus Scopus journals Clarias gariepinus PubMed journals Clarias gariepinus medical journals Clarias gariepinus free journals Clarias gariepinus best journals Clarias gariepinus top journals Clarias gariepinus free medical journals Clarias gariepinus famous journals Clarias gariepinus Google Scholar indexed journals Heamatology articles Heamatology Research articles Heamatology review articles Heamatology PubMed articles Heamatology PubMed Central articles Heamatology 2023 articles Heamatology 2024 articles Heamatology Scopus articles Heamatology impact factor journals Heamatology Scopus journals Heamatology PubMed journals Heamatology medical journals Heamatology free journals Heamatology best journals Heamatology top journals Heamatology free medical journals Heamatology famous journals Heamatology Google Scholar indexed journals Histopathology articles Histopathology Research articles Histopathology review articles Histopathology PubMed articles Histopathology PubMed Central articles Histopathology 2023 articles Histopathology 2024 articles Histopathology Scopus articles Histopathology impact factor journals Histopathology Scopus journals Histopathology PubMed journals Histopathology medical journals Histopathology free journals Histopathology best journals Histopathology top journals Histopathology free medical journals Histopathology famous journals Histopathology Google Scholar indexed journals Toxicity articles Toxicity Research articles Toxicity review articles Toxicity PubMed articles Toxicity PubMed Central articles Toxicity 2023 articles Toxicity 2024 articles Toxicity Scopus articles Toxicity impact factor journals Toxicity Scopus journals Toxicity PubMed journals Toxicity medical journals Toxicity free journals Toxicity best journals Toxicity top journals Toxicity free medical journals Toxicity famous journals Toxicity Google Scholar indexed journals Lethal test articles Lethal test Research articles Lethal test review articles Lethal test PubMed articles Lethal test PubMed Central articles Lethal test 2023 articles Lethal test 2024 articles Lethal test Scopus articles Lethal test impact factor journals Lethal test Scopus journals Lethal test PubMed journals Lethal test medical journals Lethal test free journals Lethal test best journals Lethal test top journals Lethal test free medical journals Lethal test famous journals Lethal test Google Scholar indexed journals Palm oil mill effluent articles Palm oil mill effluent Research articles Palm oil mill effluent review articles Palm oil mill effluent PubMed articles Palm oil mill effluent PubMed Central articles Palm oil mill effluent 2023 articles Palm oil mill effluent 2024 articles Palm oil mill effluent Scopus articles Palm oil mill effluent impact factor journals Palm oil mill effluent Scopus journals Palm oil mill effluent PubMed journals Palm oil mill effluent medical journals Palm oil mill effluent free journals Palm oil mill effluent best journals Palm oil mill effluent top journals Palm oil mill effluent free medical journals Palm oil mill effluent famous journals Palm oil mill effluent Google Scholar indexed journals Sublethal test articles Sublethal test Research articles Sublethal test review articles Sublethal test PubMed articles Sublethal test PubMed Central articles Sublethal test 2023 articles Sublethal test 2024 articles Sublethal test Scopus articles Sublethal test impact factor journals Sublethal test Scopus journals Sublethal test PubMed journals Sublethal test medical journals Sublethal test free journals Sublethal test best journals Sublethal test top journals Sublethal test free medical journals Sublethal test famous journals Sublethal test Google Scholar indexed journals mean corpuscular volume articles mean corpuscular volume Research articles mean corpuscular volume review articles mean corpuscular volume PubMed articles mean corpuscular volume PubMed Central articles mean corpuscular volume 2023 articles mean corpuscular volume 2024 articles mean corpuscular volume Scopus articles mean corpuscular volume impact factor journals mean corpuscular volume Scopus journals mean corpuscular volume PubMed journals mean corpuscular volume medical journals mean corpuscular volume free journals mean corpuscular volume best journals mean corpuscular volume top journals mean corpuscular volume free medical journals mean corpuscular volume famous journals mean corpuscular volume Google Scholar indexed journals periodic acid Schiff' articles periodic acid Schiff' Research articles periodic acid Schiff' review articles periodic acid Schiff' PubMed articles periodic acid Schiff' PubMed Central articles periodic acid Schiff' 2023 articles periodic acid Schiff' 2024 articles periodic acid Schiff' Scopus articles periodic acid Schiff' impact factor journals periodic acid Schiff' Scopus journals periodic acid Schiff' PubMed journals periodic acid Schiff' medical journals periodic acid Schiff' free journals periodic acid Schiff' best journals periodic acid Schiff' top journals periodic acid Schiff' free medical journals periodic acid Schiff' famous journals periodic acid Schiff' Google Scholar indexed journals Pack Cell Volume articles Pack Cell Volume Research articles Pack Cell Volume review articles Pack Cell Volume PubMed articles Pack Cell Volume PubMed Central articles Pack Cell Volume 2023 articles Pack Cell Volume 2024 articles Pack Cell Volume Scopus articles Pack Cell Volume impact factor journals Pack Cell Volume Scopus journals Pack Cell Volume PubMed journals Pack Cell Volume medical journals Pack Cell Volume free journals Pack Cell Volume best journals Pack Cell Volume top journals Pack Cell Volume free medical journals Pack Cell Volume famous journals Pack Cell Volume Google Scholar indexed journals
Article Details
1. Introduction
The incorrect handling of huge amounts of trash generated by diverse anthropogenic activities is one of the most serious problems faced by emerging countries. The hazardous disposal of these pollutants into the environment is even more difficult. Freshwater reservoirs are particularly vulnerable making these natural resources more frequently inappropriate for both main and secondary use [1]. River systems are the principal means of removing pollution from nearby industry, particularly effluents. These industrial effluents have a significant impact on water pollution; they can change the physical, chemical, and biological qualities of the water body [2]. Pollution stress on surface waters has increased as a result of increased industrial activity, as well as pollution from agricultural and home sources [3]. Palm oil is made from the oil palm Elaeis guineens, which originated in West Africa but has since spread to other tropical lowland areas like Malaysia. The African oil palm, Elaeis guineensis belongs to the Palmae family. It’s specific epithet guineensis indicates that the first specimen identified was taken in Guinea, West Africa. Palm oil is now one of the most widely traded vegetable oils, accounting for about half of the world's 36 million metric tons of trade [4]. Oil palm is processed both locally and industrially in Nigeria's oil palm belt, which stretches from South-South region of the country to the South- Western states. Palm oil mill effluent (POME), a by-product of palm oil production, is capable of harming the aquatic environment, particularly aquatic life [5]. After fishes have been exposed to water containing a range of noxious substances, such as pesticides, phenol, and heavy metals, reported histopathological changes in the gills are hyperplasia and hypertrophy, epithelial lifting, aneurysm, and increased mucus secretion [6]. Also, since liver is an essential organ for breaking down toxins, the cells of the liver are frequently part of those that are harmed by hazardous chemicals. [7] studied the impacts of glyphosate herbicide on Tilapia fish, finding filament cell proliferation, lamellar fusion, lamellar cell hyperplasia, and epithelial lifting. The most evident effects on the gills were oedema, epithelial lifting, thickening of the primary lamellar epithelium, and fusion of secondary lamellae. In the gills of Oreochromis niloticus fingerlings exposed to POME, [8] reported lamellar degeneration and cell hypertrophy. [9] reported haematological changes due to exposure of Tilapia guineensis to different levels of effluent under a static condition showed a significant different in all the parameters of exposed fish. He reported that there was a significant increase in the values of white blood cells, mean corpuscular volume (MCV), neutrophils, monocytes and glucose compared to the control value. Therefore, the aim of this study is to examine the toxic effect of POME on Clarias gariepinus, with an emphasis on histopathology and haematology, due to the need for understanding of POME's aquatic adverse effects
2. RESEARCH METHODOLOGY
2.1 TEST ORGANISM SOURCE AND COLLECTION
Kazeem farm, a private farm in Igbole, Surulere Local Government of Ogbomoso, Oyo state, provided Clarias gariepinus with an average weight of 300 g and a standard length of 18-21 cm. Clarias gariepinus was chosen because of its capacity to withstand stress and its great commercial worth in Nigeria influenced the decision.
2.2 Fish acclimatization:
Fishes were kept in non-chlorinated water in glass tanks measuring 100 cm × 25 cm x 25 cm. They were given 14 days to acclimatize in the lab and fed twice a day with commercial feed so that they can adjust to the experimental circumstances. Waters were changed at two days interval to remove feaces and uneaten feed and the fishes were fed, during acclimatization
2.3 Test Effluents:
(POME) was collected in a 25-litres keg from Oke-awon palm oil mill in Surulere Local Government of Ogbomoso, Oyo State.
2.4 Experimental Set Up
POME sublethal concentrations were discovered using 5 different concentrations within the range obtained during presumptive test. 0.01, 0.03, 0.05, 0.07 0.09 mg/L of POME preparations were used. Each of the prepared concentrations was given to ten healthy fishes. Every 48 hours, the solutions were replaced. The test lasted for 14 days.
2.5 HISTOPATHOLOGICAL STUDIES
The fishes were killed and sacrificed by a blow to the head with a hammer on conclusion of the experiments, and the liver, gill, heart and kidney components were removed. Then the organs were put in 10% formalin for 3 days, then they were subjected to dehydration in periodic acid Schiff's reagent (PAS) within 3 days in alcohol, according to [10] so that the paraffin wax will permeate the tissue while embedded. After that, melted wax was used to encapsulate the organs. Using a rotator microtome, tissue was sectioned into thin sections (5-7 m), was allowed to dehydrate, and then stained using Harris haematoxyllin-Eosin (H&E) stain. Bancroff and a microtone were then used to clear each portion, which was then placed in water that is warm (380 °C), clean slide was used to pick it and then oven dried at 580 °C for half an hour to get the wax melted for five minutes, the segment was stained with haematoxyline Eosin. A light microscope was then used to examine the stained slides with the aid of an Olympus BH2 microscope with a photographic attachment, portions were inspected and photographed at 400 magnifications (Olympus PM C35 AD4).
2.6 HAEMATOLOGY
Survivors of the sublethal assay were decapitated and bled by cardiac puncture using 2 ml sterile plastic disposable syringe fitted with 0.8 mm x 38 mm hypodermic needles. The blood samples were rocked gently in the vial to allow thorough mixing of its contents. Three capillary tubes were filled with well-mixed anticoagulant-treated blood from each specimen and centrifuged for 5 minutes at 10,500 revolutions per minute (rev/min). Pack Cell Volume (PCV) was determined by filling three capillary tubes with well-mixed anticoagulant-treated blood from each specimen and centrifuged for 5 minutes at 10,500 revolutions per minute (rev/min). PCV was measured by the average obtained from three tubes and was recorded for each specimen at various concentrations. Haemoglobin concentration was measured using a Cyanmethaematocrit method. Three microhaematocrit tubes were filled with blood after sealing one end of the capillary tube and allowed to stand for 10 minutes; this gave sufficient time for complete conversion of haemoglobin (Hb) into cyanmet haemoglobin. Transmittance was read from a corning colorimeter 253 at wavelength of 540 mm. This was then converted to haemoglobin concentration in g/dl-1 by using the commercially available cyanmet haemoglobin standards. Erythrocyte (RBC) and Leucocyte (WBC) counts were determined using an electronic cell counter (Coulter Electronic Ltd, England) standardized by an improved Neubauer haemocytometer after the blood had been diluted with dacie’s fluid and stained with brilliant cresyl blue.& For R.B.C count a dilution of 1:2000 was used while for WBC a dilution of 1:1500 was used.
3. RESULTS
3.1 LIVER
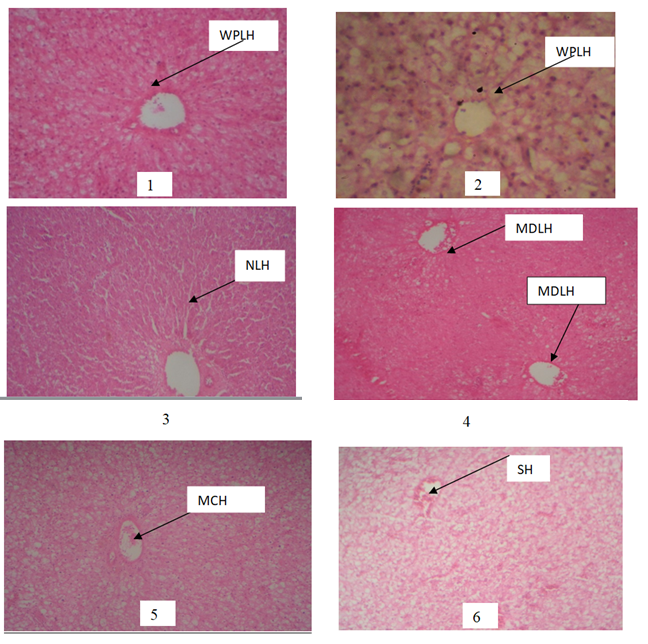
Plate 1 (control) and 2 showing WPLH: Well Preserved Liver Hepatocytes Photomicrograph of the liver showing well preserved architecture of Liver cells with no areas of inflammatory changes. H/E. Plate 3 NLH: Normal Liver Hepatocytes Photomicrograph of the liver showing normal architecture of liver hepatocytes with no areas of inflammatory changes when compared with the control (Plate 1). SDLH: Slightly Distorted Liver Hepatocytes Photomicrograph of the liver exposed to 0.05mg/L showing slightly distorted architecture of liver hepatocytes. Plate 4 MDLH: Mildly Distorted Liver Hepatocytes(0.07mg/L) Photomicrograph of the liver showing mild distortion of the liver Hepatocytes architecture when compare with the control (Plate 1). Plate 5 MCH: Mildly Congested Hepatocytes Photomicrograph of the liver showing mild congestion of central vein when compared with the control (Plate 1). Plate 6 SH: Shrink Hepatocytes Photomicrograph of the liver showing pronounced distortion of liver hepatocytes architecture when compared with the control (Plate 1)
3.2 GILLS
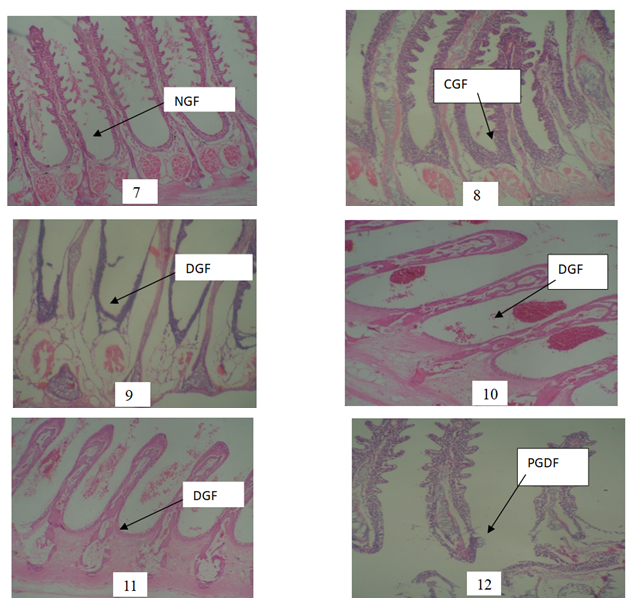
NGF: Normal Gill Filament PLATE 7(control) NGF: A photomicrograph of gill showing normal and well vascularised gill Filaments. Gill filaments architecture is well preserved. Plate 8 CGF: Curl Gill Filament:photomicrograph of gill showing well vascularised and slightly distorted gill filaments after exposure to 0.01mg/L of POME. Plates 9, 10 and 11 DGF: Distorted Gill Filament A photomicrograph of gill showing vascularised and distorted gill Filaments after exposure to 0.03, 0.05 and 0.07mg/L POME concentrations respectively. Plate 12 PDGF: Poorly Degenerated Gill Filament A photomicrograph of gill (E) showing poorly vascularized and grossly degenerated gill filaments after exposure to 0.09mg/L POME.
3.3 KIDNEY
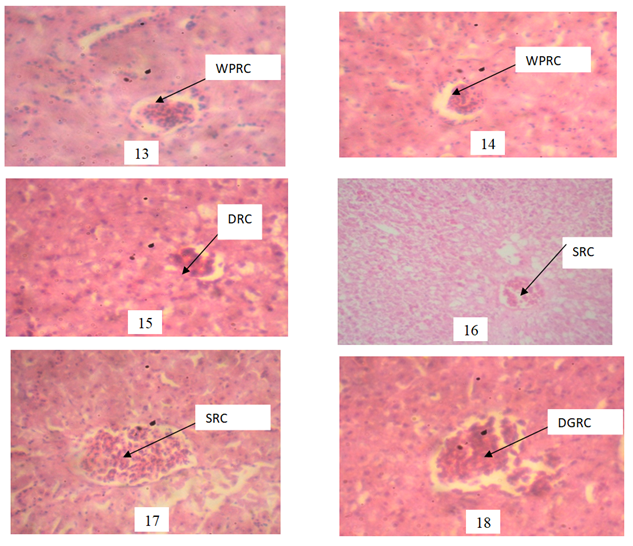
Plate 13 and 14 WPRC: Well Preserved Renal Corpuscle: Photomicrograph of the kidney showing well preserved architecture of the renal corpuscle with a wide Bowman’s space. Plate 15 DRC: Distorted Renal Corpuscle:(0.03mgl-1) Photomicrograph of the kidney showing distortion of the renal corpuscle with a narrow Bowman’s space when compared with the Control. Plate 16: Shrink Renal Corpuscle: (0.05mgl-1) Photomicrograph of the kidney showing distortion of the renal corpuscle with a narrow Bowman’s space when compare with the Control. SRC: Shrink Renal Corpuscles (0.07mgl-1) Photomicrograph of the kidney showing inflamed renal corpuscles with narrow Bowman’s space when compare with the control. Plate 18 DGRC: Degenerated Renal Corpuscles: (0.09mgl-1) Photomicrograph of the kidney showing degenerated renal corpuscles when compared with the control. H/E Stain.mag. X400.
3.4 MUSCLE
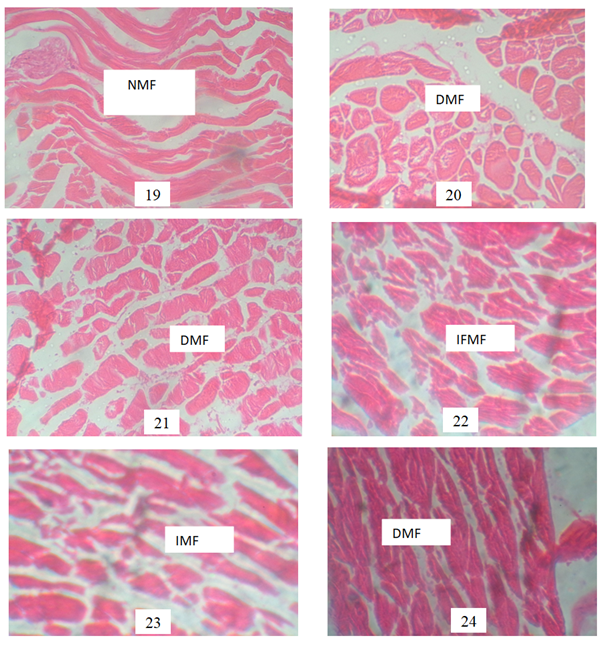
Plate 19 NMF:(control) Photomicrograph showing normal appearance of the muscle fibres with wider interfibre spaces. Plate 20 DMF: Distorted Muscle Fibre: A photomicrograph showing mild morphological distortion of muscle fibres after exposure to 0.01mg/L POME. Plate 21 and 24 DMF: Distorted Muscle Fibre . A photomicrograph showing further distortion of muscle fibre after exposure to 0.03 and 0.05mg/l POME. Plate 22 IFMF: Irregular & Fragmented Muscle Fibre A photomicrograph showing distorted, irregular and fragmented Musclefibres of fish exposed to 0.09mg/L POME concentration. Plate 23 IMF ; Irreguar muscle Fibre of fish exposed to 0.07mg/L POM
Table 1: haematology changes in c. Gariepinus exposed to different concentration of pome for 14 days
Values with same alphabet are not significantly different otherwise they do at p≤0.05
There was a significant increase in the values of white blood cells at concentration 0.01 mgl-1,0.03 mgl-1 and 0.05 mgl-1 compared to control while a decline in the values were recorded at higher concentrations 0f 0.07 mgl-1 and 0.09 mgl-1. Exposure of Clarias gariepinus to effluents caused a concentration dependent reduction in the values of Hb, RBC and PCV
4. DISCUSSION
In fish, the gill, kidney, and liver are in charge of vital activities like respiration, excretion, and the accumulation and biotransformation of xenobiotics. One of the most significant advantages of utilizing histopathological biomarkers in environmental monitoring is the ability to examine these specific target organs [11].
Normal cellular pattern, normal central vein, hepatic plate, and hepatocytes were seen in a transverse section across the liver. In the control experiment (Plate 1) and fish subjected to a concentration of POME 0.01 ppt, no lesion, necrosis, pigmentation, malignancy, or inflammation were seen (Plate 2). In the livers exposed to higher POME concentrations, there were areas of mild lesion, necrosis, malignancy, and inflammation in concentrations 0.03 ppt, 0.05 ppt, 0.07 ppt and 0.09 ppt (Plate 3, 4, 5 and 6). In POME-treated fish at a concentration of 0.07 ppt, vacuolation and tissue disarray were observed. Cellular shrinkage and hyperplasia were also found. At the greatest dose of 0.09 ppt, complete degeneration of tissue was seen. Histopathological alterations in C. gariepinus tissues subjected to sublethal concentrations of POME effluents revealed lesions ranging from necrosis and vacuolation to widespread cell congestion. This supports [12] results that pollutant cause varying degrees of damage to liver cells depending on exposure period and test media concentration. In the investigation of the histological effect of POME on juveniles of Oreochromis niloticus, [8] reported areas of lesions, necrosis, malignancy, and inflammation in the livers. He also said that fish exposed to higher amounts experienced vacuolation, cell shrinkage, and total degeneration.
4.1 GILL
Histopathological changes seen in the gill tissues of C. gariepinus experimented on showed a considerable dependence on POME concentration. The gradual and complete vascularization of the gill filaments of C. gariepinus coupled with traces to severe necrosis could be attributed to their ability to act as primary site of absorption of pollutants, which degraded the gill tissues. Similarly, [8] found enlargement of the gill arch and total filament degeneration in juvenile of Oreochromis niloticus subjected to 50.0 mg/l pome for 96 hours.
4.2 HEART
The histological changes observed in the heart tissues of fishes exposed to the effluent showed distorted muscle fibre and congestion of the heart wall. The degree of the alterations could be attributed to the continuous and rapid movements of mouth and the gulping for air which might weaken and impair the heart tissues, as a result of the accumulation of pollutants. This conformed with the submission of [12] that the level of the degeneration of the heart tissue depends on the concentrations of the pollutant and period of exposure.
4.3 KIDNEY
There was a distortion of the renal corpuscle with a narrow Bowman’s space from treatments with concentration 0.03 mg/l by POME effluents when compared with treatment experiment. Shrinkage of Bowman's capsule and vacuolation formed around the capsule were also noticed. This also conformed with the work of [13] who reported that the kidney of Channa punctatus revealed varying degree of distortion on exposing the fish to tannery effluent.
4.4 MUSCLE
The observed histological changes in the muscles examined could be attributed to the destructive effects of the varying concentrations of POME effluents and the exposure time. The observed histological changes agreed with the submission of [14] that the higher the concentration of pollutant the more the degree of damages to the tissue. [15] however opined that the alteration in tissues morphology may probably lead to several physiological stresses in fish.
4.5 HAEMATOLOGY
The total white blood counts registered an initial significant increase as compared to the control but at higher concentrations of 0.09 mgl-1 and 0.07 mgl-1, there was a decline in the counts. The revealed increase in total white blood counts could be traced to an attempt of the fish to build up defence against stress which resulted to behavioural responses of C. gariepinus to POME effluents. This observation agreed with the submission of [16] and Sowumi (2003) that the increase could be attributed to stress reaction when fish tried to restore disturbed homeostasis. Similarly, [17] opined that the increase was due to initiation of pathogenic condition, most likely in the form of irritation, injury to the cells and formation of tissue debris and occurrence of secondary infection in the fish body. The decrease was attributed to limited role in exercising protective and immune responses under effluent stress [17]. It was observed that the red blood and haemoglobin (HB) counts were found to decrease with increase in concentration of POME effluents except those exposed to 0.01 mg/l. This may be due to in ability of the C. gariepinus to cope with the metabolic stress as a result of increase in demand for oxygen. Hence, there might be damage counts cells to the tissues which in turn impair the production of erythrocytes and haemoglobin. This conformed with the submission of [18]. [19] noticed the same trend and opined that the decline in red blood cells and haemoglobin counts was indicative of worsening state of health and possibly of aneamia development in the organisms. The values of the derived variables of the packed cell volume (PCV) in the experimental C. gariepinus were lower than the control apart from fish exposed to concentration 0.01 ppt. Generally, there were significant decline in the values of these parameters as concentration of the POME effluent increases with exception to concentration 0.01 ppt. The observed reduction in the derived indices could as well be attributed to the cause of decline in red blood cell counts, that metabolic stresses might lead to aneamic situation as a result of damaged tissues. Similarly, [20] and [19] submitted that high concentrations of pollutant and long-term exposure of fish to pollutant usually result in decrease of the derived indices and that worsens the fish health status and indicates anemia.
5. CONCLUSION
POME is hazardous to fish organs and causes histopathological abnormalities in many organs such as the gills and liver, according to the findings of this study. As a result, indiscriminate discharge into water bodies by palm oil producers should be prohibited, particularly inland rivers.
Ethical approval
I hereby declare that this work is original and conducted to ethical standards.
Consent to participate
The authors participated in the research process and give their consent to further questions on this work
Consent to publish
The authors give their consent to publishing this work.
Author’s contribution
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Oluniyi Adediran AFOLABI, Solomon Olayinka ADEWOYE and Folakemi Dupe AFOLABI, Oluwasheun Adebayo. The first draft of the manuscript was written by Oluniyi Adediran AFOLABI and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
The authors did not receive support from any organization for the submitted work.
Competing Interest
The authors declare no competing interest.
Availability of data and materials
Not applicable.
References
- Osibanjo O, Daso AP and Gbadebo AM. “The impact of industries on surface water quality of River Ona and River Alaro in Oluyole Industrial Estate, Ibadan, Nigeria”, Afr. J. Biotech 10 (2011): 696-702.
- Akoteyan and Soladoye O. Groundwater quality assessment in Eti-Osa, Lagos-Nigeria using multivariate Analysis. J. of Applied Science Environ. Manage 15 (2011): 121-125.
- Kanu I, Achi O. Industrial effluents and their impact on water quality of receiving rivers in Nigeria. Journal of Applied technology in Environmental Sanitation 1 (2011): 75-86.
- Agricultural Research Data Book Malaysian Council on Agricultura Research: Oil Palm Production (2004).
- Singh RP, Embrandiri A, Ibrahim MH, et al. Management of biomass residues (2011).
- Nwanna LO Fagbenro & E Ogunlowo. Acute mortality of Clarias gariepinus & Heterobranchus bidorsalis exposed to textile effluent. Journal of Sustainable Tropical Agricultural Research 11 (2018): 36-42.
- Ayoola SO. Histopathological effects of glyphosate on juvenile African catfish (Clarias gariepinus). American-Eurasian Journal of Agricultural & Environmental Science 4 (2008): 362-367.
- Akinsorotan AM. Histopathological Effects of Acutely Toxic Levels of Palm Oil Mill Effluents on Gill and Liver of Nile Tilapia. International Journal of Scientific and Engineering Research 4 (2013): 6-7.
- Akinrotimi O, Orlu E and Gabriel U. Haematological Responses of Tilapia guineensisTreated with Industrial Effluents. Applied Ecology and Environmental Sciences 1 (2013): 10-13.
- Hughes GM & Perry SF Morphemetric study of trout gills. A light microscopic method for the evaluation of pollutant action. J. Exp. Biol 63 (1976): 447-460.
- Thophon S, Kruatrachue S, Upathan E, et al. Histopathological alterations of white seabass, Lates calcarifer in acute and subchronic cadmium exposure. Environ Pollut 121 (2003): 307-320.
- Fafioye OO, Adebisi AA and Fagade SO. Toxicity of Parkia biglobosa and Raphiavinifera extracts on ClariasgariepinusJuveniles. African Journal of Biotechnology 3 (2004): 627-630.
- MK Mohanta, Salam MA, AK Saha, et al. Effects of Tannery Effluents on Survival and Histopathological Changes in Different Organs of Channa punctatus, ASIAN J. EXP. BIOL. SCI 1 (2010): 294-302.
- Bassey BO. Histopathological and Biochemical Response of Chrysichthys nigrodigitatus to Environmental Stressors from Two Polluted Lagoons, Southwest Nigeria. J Toxicol Risk Assess 5 (2019): 025.
- Fafioye OO, Adebisi AA and Fagade SO. Toxicity of Parkia biglobosa and Raphia vinifera extracts on Clarias gariepinus Juveniles. African Journal of Biotechnology 3 (2001): 627-630.
- Fange R. Fish blood cells. In: Fish Physiology Vol. XII, Part B. The Cardiovascular system.Eds: Hoar, W.S., Randall, D.J and Farrell, A.P. Pub. A cademic Press, Inc. London (1992): 1-54.
- Devi G, Baruah BK and Das M. Study on the effect of paper mill effluent on haematologial profile of Heteropneustesfossilis(Bloch) Poll. Res 23 (2004): 611-614.
- Bhatia, S., Othman, Z. and Ahmad, A. L., (2006): Palm Oil Mill Effluent Pretreatment Using Moringa oleifera Seeds as an Environmentally Friendly Coagulant: Laboratory and Pilot Plant Studies. J. Chem.Technol. Biotechnol 81 (2006): 1852-1858.
- Vosyliene MZ. The effect of heavy metals on haematological indices of fish. Science Research Reporter 1 (1999): 165-170.
- Oladimeji AA and Ologunmeta RT. (1987): Toxicity of water borne lead to Tilapia niloticus Nig. Journal Appl. Fish. Hydrobiol 2 (1987): 19-24.
- Davies JB and Reilly PJA. Reviewof Current Palm Oil Mill Effluent POME, Summary of treatment methods 35 (2004): 85-96.
- generated from palm oil mill: vermicomposting a sustainable option. Resour Conserv Recycl 55 : 423-434.
