Early Treatment with a Peptide Derived from the Human Heat-Shock 60 Protein Avoids Progression to Severe Stages of COVID-19
Article Information
Julio E Baldomero Hernández1, Amaurys Del Río Martín1, Leticia del Rosario Cruz2,Rafael Venegas Rodríguez2, Mabel Hernández Cedeño3, Anabel Serrano Diaz3, Rubén Peña Ruiz2, Idelsis Esquivel Moynelo2, Tania Fernández Sordo4, Alejandro Martínez Adan4, Joanne Graña Mir5, Dariel Suñol Mulet5, Fidel Sánchez Villanueva6, Rodolfo Domínguez Rosabal 7, Zoe Roca Núñez8, Yordanys Salina-Caballero8, Adriel Viera Paz9, Odalis Marrero Martinez10, Gillian Martínez Donato3, Verena L Muzio González1, Gerardo Guillen Nieto3, María del Carmen Dominguez Horta3*
1Department of Clinical Research, Center for Genetic Engineering and Biotechnology, Havana, Cuba
2Luis Díaz Soto Hospital. Avenida Monumental km 2, Havana, Cuba
3Department of Biomedical Research, Center for Genetic Engineering and Biotechnology, Havana, Cuba
4Salvador Allende Hospital, Havana, Cuba
5Fermín Valdés Domínguez Hospital, Holguín, Cuba
6Mario Muñoz Monroy Hospital, Matanzas, Cuba
7Amalia Simoni Hospital, Camagüey, Cuba
8Octavio de la Concepción y de la Pedraja Hospital, Camagüey, Cuba
9Héroes de Baire Hospital, Isla de la Juventud, Cuba
10Pedro Kouri Hospital , Havana, Cuba
*Corresponding Author: María del Carmen Domínguez, Department of Biomedical Research, Center for Genetic Engineering and Biotechnology, P.O. Box 6162, Havana 11300, Cuba
Received: 10 December 2021; Accepted: 18 December 2021; Published: 30 December 2021
Citation:
Hernández JEB, Martín ADR, Cruz LDR, Rodríguez RV, Cedeño MH, Diaz AS, Ruiz RP, Moynelo IE, Sordo TF, Adan AM, Mir JG, Mulet DS, Villanueva FS, Rosabal RD, Núñez ZR, Salina-Caballero Y, Paz AV, Martinez OM, Donato GM, González VLM, Nieto GG, Horta MCD. Early Treatment with a Peptide Derived from the Human Heat-Shock 60 Protein Avoids Progression to Severe Stages of COVID-19. Journal of Biotechnology and Biomedicine 4 (2021): 196-210.
View / Download Pdf Share at FacebookAbstract
Hyperinflammatory response induced by SARS-CoV-2 characterizes COVID-19 patients progressing to severe conditions. CIGB-258 is an immunoregulatory peptide with anti-inflammatory properties derived from the human stress protein 60 (HSP60). This peptide has been used in the treatment of serious and critically ill COVID-19 patients with positive results. This study is aimed to describe the outcomes of a cohort of moderately ill COVID-19 patients treated with CIGB-258. Clinical assessment and inflammation biomarkers indicated that these patients were progressing to the hyperinflammation phase. In addition, this study displays the outcomes of two other cohorts of COVID-19 patients in serious and critical conditions, treated with this molecule. One hundred and four patients with COVID-19 in moderate (18.3%), serious (60.6%) and critical (21.1%) conditions were enrolled in this study. None of the moderate patients progressed to the severe stage of the disease. Out of sixty-three seriously ill patients, only eleven progressed to a critical condition. These patients had several comorbidities that aggravated their clinical conditions.
Despite this, six of the patients who progressed to the critical condition recovered and were discharged from the hospitals. Out of twenty-two critically ill patients, only two died. Inflammatory biomarkers decreased after seven days of treatment. Also, IL-6 significantly decreased at 96 hours of the treatment. These results indicate that the early administration of CIGB-258 can improve the condition of moderately ill patients and avoid their progression to severe stages of COVID-19. This study confirms the capacity of this peptide to reduce the hyperinflammation characterizing this disease.
Keywords
COVID-19, CIGB-258, HSP60, Hyperin-flammation, Jusvinza
COVID-19 articles COVID-19 Research articles COVID-19 review articles COVID-19 PubMed articles COVID-19 PubMed Central articles COVID-19 2023 articles COVID-19 2024 articles COVID-19 Scopus articles COVID-19 impact factor journals COVID-19 Scopus journals COVID-19 PubMed journals COVID-19 medical journals COVID-19 free journals COVID-19 best journals COVID-19 top journals COVID-19 free medical journals COVID-19 famous journals COVID-19 Google Scholar indexed journals CIGB-258 articles CIGB-258 Research articles CIGB-258 review articles CIGB-258 PubMed articles CIGB-258 PubMed Central articles CIGB-258 2023 articles CIGB-258 2024 articles CIGB-258 Scopus articles CIGB-258 impact factor journals CIGB-258 Scopus journals CIGB-258 PubMed journals CIGB-258 medical journals CIGB-258 free journals CIGB-258 best journals CIGB-258 top journals CIGB-258 free medical journals CIGB-258 famous journals CIGB-258 Google Scholar indexed journals HSP60 articles HSP60 Research articles HSP60 review articles HSP60 PubMed articles HSP60 PubMed Central articles HSP60 2023 articles HSP60 2024 articles HSP60 Scopus articles HSP60 impact factor journals HSP60 Scopus journals HSP60 PubMed journals HSP60 medical journals HSP60 free journals HSP60 best journals HSP60 top journals HSP60 free medical journals HSP60 famous journals HSP60 Google Scholar indexed journals Hyperin-flammation articles Hyperin-flammation Research articles Hyperin-flammation review articles Hyperin-flammation PubMed articles Hyperin-flammation PubMed Central articles Hyperin-flammation 2023 articles Hyperin-flammation 2024 articles Hyperin-flammation Scopus articles Hyperin-flammation impact factor journals Hyperin-flammation Scopus journals Hyperin-flammation PubMed journals Hyperin-flammation medical journals Hyperin-flammation free journals Hyperin-flammation best journals Hyperin-flammation top journals Hyperin-flammation free medical journals Hyperin-flammation famous journals Hyperin-flammation Google Scholar indexed journals Jusvinza articles Jusvinza Research articles Jusvinza review articles Jusvinza PubMed articles Jusvinza PubMed Central articles Jusvinza 2023 articles Jusvinza 2024 articles Jusvinza Scopus articles Jusvinza impact factor journals Jusvinza Scopus journals Jusvinza PubMed journals Jusvinza medical journals Jusvinza free journals Jusvinza best journals Jusvinza top journals Jusvinza free medical journals Jusvinza famous journals Jusvinza Google Scholar indexed journals pneumonia articles pneumonia Research articles pneumonia review articles pneumonia PubMed articles pneumonia PubMed Central articles pneumonia 2023 articles pneumonia 2024 articles pneumonia Scopus articles pneumonia impact factor journals pneumonia Scopus journals pneumonia PubMed journals pneumonia medical journals pneumonia free journals pneumonia best journals pneumonia top journals pneumonia free medical journals pneumonia famous journals pneumonia Google Scholar indexed journals Tumor Necrosis Factor articles Tumor Necrosis Factor Research articles Tumor Necrosis Factor review articles Tumor Necrosis Factor PubMed articles Tumor Necrosis Factor PubMed Central articles Tumor Necrosis Factor 2023 articles Tumor Necrosis Factor 2024 articles Tumor Necrosis Factor Scopus articles Tumor Necrosis Factor impact factor journals Tumor Necrosis Factor Scopus journals Tumor Necrosis Factor PubMed journals Tumor Necrosis Factor medical journals Tumor Necrosis Factor free journals Tumor Necrosis Factor best journals Tumor Necrosis Factor top journals Tumor Necrosis Factor free medical journals Tumor Necrosis Factor famous journals Tumor Necrosis Factor Google Scholar indexed journals Lactate Dehydrogenase articles Lactate Dehydrogenase Research articles Lactate Dehydrogenase review articles Lactate Dehydrogenase PubMed articles Lactate Dehydrogenase PubMed Central articles Lactate Dehydrogenase 2023 articles Lactate Dehydrogenase 2024 articles Lactate Dehydrogenase Scopus articles Lactate Dehydrogenase impact factor journals Lactate Dehydrogenase Scopus journals Lactate Dehydrogenase PubMed journals Lactate Dehydrogenase medical journals Lactate Dehydrogenase free journals Lactate Dehydrogenase best journals Lactate Dehydrogenase top journals Lactate Dehydrogenase free medical journals Lactate Dehydrogenase famous journals Lactate Dehydrogenase Google Scholar indexed journals infection articles infection Research articles infection review articles infection PubMed articles infection PubMed Central articles infection 2023 articles infection 2024 articles infection Scopus articles infection impact factor journals infection Scopus journals infection PubMed journals infection medical journals infection free journals infection best journals infection top journals infection free medical journals infection famous journals infection Google Scholar indexed journals Patients articles Patients Research articles Patients review articles Patients PubMed articles Patients PubMed Central articles Patients 2023 articles Patients 2024 articles Patients Scopus articles Patients impact factor journals Patients Scopus journals Patients PubMed journals Patients medical journals Patients free journals Patients best journals Patients top journals Patients free medical journals Patients famous journals Patients Google Scholar indexed journals
Article Details
1. Introduction
In December 2019, a novel pneumonia was reported in Wuhan, Hubei province in China. On February 11th, 2020, the World Health Organization (WHO) officially named it Coronavirus Disease 2019 (COVID-19), which is caused by Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2) [1]. The first case of COVID-19 in Cuba was reported on March 11th, 2020, just three months after first cases were reported in China [2]. Hyperinflammatory response induced by SARS- CoV-2 is the main cause of disease severity and death in infected patients [3, 4]. Several studies suggest that the pathophysiological basis of this severe inflammatory response is similar to the cytokine release syndrome [5-8].
During the progression to severe stages, patients significantly increased inflammatory biomarkers, Including Lactate Dehydrogenase (LDH), C-reactive Protein (CRP) and serum interleukin-6 [9-11]. Another severity biomarker is the Neutrophil/Lymphocyte Ratio (NLR). These predictors can identify patients who may progress towards severe phases of the disease [3, 12]. Drugs that inhibit inflammation at early stages are crucial to avoid progression to the severe phase of COVID-19 [13, 14]. CIGB-258 is an altered peptide ligand derived from human cellular stress protein 60 (HSP60). This peptide inhibited the inflammation in several experimental models of Rheumatoid Arthritis (RA). CIGB-258 reduced the levels of the Tumor Necrosis Factor (TNFα), Interleukin (IL)-17 (IL-17) and interferon-γ (IFNγ) in preclinical studies [15-17] and in a phase I clinical trial with RA patients. Likewise, this molecule induced a significant decrease in autoanti-bodies against citrullinated self-proteins in RA patients [18]. Furthermore, this peptide increased the frequency of regulatory T cells (Treg) and their suppressive capacity against antigen-responding effector CD4+T cells from RA patients [19].
This peptide has been used in the treatment of serious and critically ill COVID-19 patients with positive outcomes [20, 21]. The drug received the Authorization for Emergency Use by the Cuban Regulatory Authority for the treatment of COVID-19 patients [22]. After this authorization, CIGB-258 received the name of Jusvinza. The inclusion of CIGB-258 in the Cuban national protocol approved by the Ministry of Public Health for COVID-19 took place on April 27th, 2020 [23]. The use of this drug has helped decrease fatality rate in Cuba. In April 2020, the fatality rate in Cuba was 4.16 [2] and in March 2021 it was 0.46 [24]. The therapy with CIGB-258 induces the inhibition of the activity of monocytes, macrophages and neutrophils in severe COVID-19 patients. This inhibition decreased IL-6, TNFa and IL-10 levels, and improved the NLR.
The therapeutic effects of CIGB-258 may also be enhanced by the progressive expansion of Treg. These cells migrate to inflammation sites inhibiting auto-immune damage on the endothelium, which is induced during SARS-CoV-2 infection [21]. These above results indicate that the early administration of CIGB-258 may avoid the progression to severe stages of COVID-19. Consistent with this idea, a cohort of moderate COVID-19 patients was included in this study. Clinical assessment and inflammation markers indicated that this cohort of patients was progressing to the hyperin-flammation phase. The treatment with CIGB-258 stops progression to severe stages of COVID-19 in these patients. Furthermore, this study confirms the capacity of this peptide to reduce hyperinflammation in seriously and critically ill COVID-19 patients.
2. Materials and Methods
2.1 Patients
One hundred and four patients confirmed as SARS-CoV-2 positive by real-time reverse transcription polymerase chain reaction were enrolled in this study. Patients were recruited for the study between April 27th and October 7th, 2020, from eight Hospitals in Cuba. The patients’ data were anonymously recorded to ensure confidentiality. The patients or their legal represent-tatives signed the Informed Consent. All patients included in the study were in a moderate, severe or critical condition, according to the Cuban national prot-ocol approved by the Ministry of Public Health for COVID-19. The patients were classified according to the following criteria.
Moderately ill patients:
- Fever or cough. Evidence of lower respiratory disease during clinical assessment or imaging. SpO2 ≥94%. Neutrophil-to-lymphocyte ratio (NLR) > 3
Seriously ill patients:
- Fever, cough and polypnea. SpO2 <94 % in room air at sea level, a respiratory rate >30 breaths/min, PaO2/FiO2 <300 mm Hg, or lung infiltrates >50%. NLR > 5
Critically ill patients:
- Acute respiratory distress syndrome (ARDS) evidenced by PaO2/FiO2 ≤ 300 mm Hg.
- Sequential Organ Failure Assessment (SOFA) >2 Bilateral multilobar interstitial pattern < 50% in chest x-rays and CT Sepsis. Septic Shock
The patients also received the standard therapy established in the above-mentioned protocol [23]. They received oxygen therapy or mechanical ventilation, according to their clinical and respiratory condition. This study was conducted following the Helsinki Declaration for research in humans [25] and the guidelines of the International Conference on Harmoni-zation [26]. The Ethics and Scientific Committees of each study site and the Cuban Regulatory Authority (CECMED, http://www.cecmed.cu/) approved the protocol. The study was registered as RPCEC00000313 at the Cuban Clinical Trial Registry (www. registroclinico.sld.cu).
2.2 Design of the study
The information on each patient including sex, clinical classification and comorbidities, laboratory tests and clinical outcomes were obtained from their medical records. Data on safety were collected according to Regulation 45/2007 from the Cuban Regulatory Autho-rity: “Requirements for reporting adverse events in on-going clinical trials, based on WHO regulations.” This regulation conforms to the “National Cancer Institute Common Toxicity Criteria Adverse Event version 3.0” (National Cancer Institute, Frederick, MD, USA).
The peptide was administered intravenously every 12 hours. The dose of CIGB-258 was 1 mg for moderately and seriously ill patients and 2 mg for critically ill patients. Irrespective of their clinical condition, obese patients received 2 mg every 12 hours. Patients were treated until their clinical and radiological conditions were resolved, as evidenced by the decrease in NLR. Figure 1 shows the design of this study.
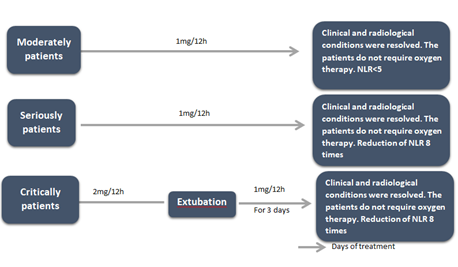
Figure 1: Diagram of CIGB-258 treatment. Moderately ill and seriously ill patients were treated with 1mg of CIGB-258 every 12 h. Critically ill patients were treated with 2 mg of CIGB-258 every 12 h. After extubation, the critically ill patients received 1mg of CIGB-258 daily for another three days. Obese patients received 2 mg every 12 hours, irrespective of their clinical condition. The peptide was administered intravenously. Moderately ill patients were treated until their clinical and radiological conditions were resolved and they did not need oxygen therapy and theirs NLR decreased below 5. The seriously and critically ill patients were treated until their clinical and radiological conditions were resolved and they did not need oxygen therapy and the NLR decreased below 5, compared to the value of this biomarker before starting treatment. NLR: neutrophil-lymphocyte ratio.
2.3 Biomarkers assessments
NLR in peripheral blood, serum values of LDH and CRP were quantified in all patients enrolled in the study before and during the CIGB-258 treatment. A Sysmex automated hematology analyzer was used to carry out blood counts according to the manufacturer's protocol (SysmexPartec, Milan, Italy). Serum samples were analyzed on a fully automated clinical chemistry analyzer (Beckman Olympus, Beckman, Germany), according to the manufacturer’s instructions.
2.4 IL-6 assessments
Serum samples were obtained before the CIGB-258 treatment and 96 hours after the first inoculation. IL-6 concentrations in sera were assessed by ELISA (Quantikine®, R&D Systems, USA) according to the recommendations of the manufacturer, with lower limits of quantification of 3.13pg/ mL.
2.5 Statistical analysis
Continuous variables (Table 1) were expressed as median (range) and compared using the Mann-Whitney U test; categorical variables (moderate, severe and critical patients) were expressed as number (%) and compared by the χ2 test. NLR, LDH, CRP and IL-6 were analyzed using GraphPad Prism version 7.04. (GraphPadSofware, San Diego California, USA). Samples were examined for normality and equal variance with Kolmogorov-Smirnov and Bartlett’s tests, respectively. The Mann-Whitney U test was used for NLR, CPR, LDH, PaO2/FiO2 and SpO2. IL-6 levels were analyzed using the two-way ANOVA and Bonferroni’s multiple comparisons test P<0.05.
|
Characteristics |
All patients |
Moderate |
Seriuos |
Critical |
|
(n=104) |
(n=19, 18.3%) |
(n=63, 60.6%) |
(n=22, 21.1%) |
|
|
Age, mean (range) |
61 (23-88) |
60 (27-88) |
61 (23-87) |
59 (42-80) |
|
Sex |
||||
|
Female |
45 (43.3%) |
9 (47.4) |
28 (44.4%) |
8 (36.4%) |
|
Male |
59 (56.7%) |
10 (52.6%) |
35 (55.6%) |
14 (63.6%) |
|
Comorbidities |
||||
|
Arterial Hypertension |
66 (73.3%) |
13 (86.7%) |
40 (71.4%) |
13 (68.4%) |
|
Cardiovascular diseases (ischemic heart, thromboembolism, heart failure, peripheral arterial insufficiency, cardiac arrhythmia) |
23 (25.6%) |
6 (40%) |
11 (19.6%) |
6 (31.6%) |
|
Diabetes mellitus |
26 (28.9%) |
5 (33.3%) |
15 (26.8%) |
6 (31.6%) |
|
Chronic obstructive pulmonary disease |
13 (14.4%) |
3 (13.3%) |
9 (16.1%) |
2 (10.5%) |
|
Bronchial asthma |
11 (12.2%) |
- |
9 (16.1%) |
2 (10.5%) |
|
Obesity |
14 (15.6%) |
1 (6.7%) |
8 (14.3%) |
5 (26.3%) |
|
Malignant tumors |
8 (8.9%) |
- |
5 (8.9%) |
3 (15.8%) |
|
Chronic kidney disease |
6 (6.7%) |
1 (6.7%) |
4 (7.1%) |
1 (5.3%) |
|
Others (bronchiectasis, gout, cerebrovascular disease, hydrocephalus, AIDS, anemia, liver cirrhosis, Hepatitis virus C) |
11 (12.2%) |
1 (6.7%) |
9 (16.1%) |
1 (5.3%) |
|
Patients with two comorbidities |
27 (30.0%) |
6 (40.0%) |
18 (32.1%) |
3 (15.8%) |
|
Patients with three or more comorbidities |
33 (36.7%) |
5 (33.3%) |
20 (35.7%) |
8 (42.1%) |
|
None |
14 (13.5%) |
4 (21.1%) |
7 (11.1%) |
3 (13.6%) |
|
*AIDS: Acquired Immunodeficiency Syndrome |
||||
Table 1: Demographic characteristics of COVID-19 patients treated with CIGB-258.
3. Results
3.1 Demographic characteristics and clinical description of patients
One hundred and four patients with COVID-19 in moderate, serious or critical conditions were included in this study. Demographic characteristics, clinical classi-fication and comorbidities of patients are summarized in Table 1. The median age (min-max) of patients was 61 (23-88) years old. Ninety patients had one or more comorbidities, including hypertension and other cardiovascular diseases, diabetes, chronic obstructive pulmonary disease, bronchial asthma, obesity, cancer and chronic kidney disease. Other diseases found in 12.2% of the patients were: bronchiectasis, gout, cerebrovascular disease, hydrocephalus, AIDS, anemia, liver cirrhosis and hepatitis C. In 30 % of the patients, two comorbidities were present and in 36.7 %, there were more than two comorbidities. Out of 104 patients, only fourteen did not have comorbidities. Nineteen patients were classified as moderately ill, sixty-three as seriously ill and twenty-two as critically ill. Moderately ill patients had a mild respiratory disease shown by clinical and radiological assessment. Ten of these patients did not require any kind of oxygen support and nine patients needed oxygen therapy, including nasal cannula or an oxygen mask (Table 2). Seriously ill patients had fever, cough, fatigue and polypnea as the most common symptoms. Fifty-three were treated with oxygen therapy and only nine patients in this condition did not require oxygen therapy. Although these nine patients had SpO2> 94%, they were classified as severe based on their clinical assessment and comorbidities. One seriously ill patient needed non-invasive mechanical ventilation in the continuous positive airway pressure (CPAP) mode. Before the CIGB-258 treatment, twenty critically ill patients had acute respiratory distress syndrome (ARDS), according to the Berlin criteria [27]. These patients were under mechanical ventilation when starting with CIGB-258. Only two patients in critical conditions did not require mechanical ventilation, one of these patients had hepatic encephalopathy [28] and the other patient had extreme bradycardia due to ischemic heart disease. One critically ill patient received non-invasive ventilation in the CPAP mode (Table 2).
3.2 Therapy outcomes
Out of all patients included in this study, 93.3% recovered from the disease and were discharged from the hospital. The average treatment duration with CIGB-258 was seven and eleven days for moderately and seriously or critically ill patients, respectively (Table 2). All moderately ill patients improved their clinical status, after 96 hours of treatment with CIGB-258. None of these patients progressed to severe disease stages. CRP and LDH decreased significantly (P=0.0014 and P=0.0426 respectively) after seven days of treatment. The NLR decreased below 5, after seven days (Table 3). All moderately ill patients were discharged from the hospital (Table 2). Out of sixty-three seriously ill patients, fifty-two did not progress to a critical condi-tion. These patients expressed a marked improvement in their clinical condition after 96 hours of treatment with CIGB-258. The improvement in functional or radio-logical respiratory parameters agreed with clinical improvement. On the seventh day, the NLR was less than 5 in most of these patients. CRP decreased significantly (P=0.0151) as compared to the baseline and LDH tended to normalize (Table 3).
|
All patients (n=104) |
Moderate (n=19) |
Seriuos (n=63) |
Critical (n=22) |
|
|
Days treated with CIGB-258 |
10 (2-35) |
7 (4-9) |
11(2-35) |
11 (6-20) |
|
Oxygen support |
||||
|
Invasive mechanical ventilation |
19 (22.9%) |
- |
- |
19 (95.0%) |
|
Non-invasive ventilation |
2 (2.4%) |
- |
1 (1.9%) |
1 (5.0%) |
|
Oxygen therapy |
62 (74.7%) |
9 (100%) |
53 (98.1%) |
- |
|
No type of oxygen support required |
21 (20.2%) |
10 (52.6%) |
9 (14.3%) |
2 (9.1%) |
|
Clinical outcomes |
||||
|
Alive, discharged from the hospital |
97 (93.3%) |
19 (100%) |
58 (92.1%) |
20 (90.9%) |
|
Death |
7 (6.7%) |
- |
5 (7.9%) |
2 (9.1%) |
Table 2: Days of treatment with CIGB-258, characteristics of oxygen support and outcomes by clinical stages of patients.
Table 3: Inflammation biomarkers and ventilation parameters of COVID-19 patients under CIGB-258 Treatment.
NLR: neutrophil-lymphocyte ratio; CRP: C-reactive protein; LDH: lactate dehydrogenase; ns: non-significant difference. Differences were analyzed using the Mann-Whitney test
Eleven seriously ill patients progressed to the critical phase of the disease. Ten of them had more than one comorbidity and seven were over 70 years of age. Six patients that had progressed to a critical condition recovered and were discharged from the hospital and five patients died. The patients who died had more than two comorbidities. One of the patients had metastatic prostate cancer. Three patients had chronic obstructive pulmonary disease and two patients had chronic kidney disease (Table 4). Fifty-eight seriously ill patients recovered and were discharged from the hospital (Table 2). Twenty of the critically ill patients recovered from their ARDS and were extubated.
During the treatment with CIGB-258, the patients improved their oxygen uptake efficiency. CRP decreased significantly (P=0.0033) compared to the baseline and LDH tended to normalize by the seventh day of the treatment (Table 3). These patients were discharged from the hospital (Table 2). Two patients from this group died (Table 4). No adverse events associated with CIGB-258 were reported during treatment or in the follow-up stage.
3.3 Effect of CIGB-258 on inflammation biomarkers
Laboratory parameters linked with inflammation including NLR in peripheral blood, LDH and CRP, gradually normalized during the treatment with CIGB-258 (Table 3). Furthermore, quantified IL-6 levels in these patients (moderate, severe and critical conditions) are included in this study. IL-6 was measured in the sera of patients before the treatment and at day four after starting the treatment with CIGB-258. As shown in figure 2, the use of CIGB-258 led to a significant reduction of this cytokine in seriously (P=0.0137) and critically ill patients (P=0.0134). Likewise, IL-6 levels were analyzed independently in moderate, serious, and critically ill patients. The levels of this cytokine were significantly higher (P=0.0154) in critically ill patients than in moderate and seriously ill patients, before the treatment with CIGB-258. However, no significant difference was detected between the three groups at 96 hours after starting the CIGB-258 treatment (Figure 2).
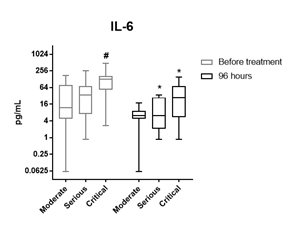
Figure 2: IL-6 levels among clinical states and at 96 hours after CIGB-258 treatment. Data are presented as means ± standard deviation. # represents significant differences between clinical stages. * represents significant differences between times. Serum samples were obtained before treatment (light bars) and at 96 hours (dark bars). Differences were analysed using the two-way ANOVA and Bonferroni’s multiple comparisons test. P<0.05.
|
Case |
Clinical classification |
Sex |
Age |
Comorbidities |
Clinical outcomes |
|
1 |
Seriously ill |
M |
56 |
Arterial hypertension. Diabetes mellitus. Chronic kidney failure. Prostate cancer |
Death |
|
2 |
Seriously ill |
M |
84 |
Arterial hypertension. Diabetes mellitus. Ischemic heart disease. Chronic obstructive pulmonary disease |
Death |
|
3 |
Seriously ill |
M |
58 |
Diabetes mellitus. Obesity |
Alive, discharged from Hospital |
|
4 |
Seriously ill |
F |
54 |
Chronic lymphoid Leukemia |
Alive, discharged from Hospital |
|
5 |
Seriously ill |
M |
72 |
Arterial hypertension. Diabetes mellitus |
Alive, discharged from Hospital |
|
6 |
Seriously ill |
M |
78 |
Arterial hypertension. Ischemic heart disease. Chronic obstructive pulmonary disease. Chronic kidney failure |
Death |
|
7 |
Seriously ill |
M |
85 |
Arterial hypertension. Ischemic heart disease |
Death |
|
8 |
Seriously ill |
F |
80 |
Arterial hypertension. Chronic obstructive pulmonary disease |
Death |
|
9 |
Seriously ill |
F |
74 |
Diabetes mellitus. Peripheral arterial insuf-ficiency |
Alive, discharged from Hospital |
|
10 |
Seriously ill |
M |
58 |
Arterial hypertension. Diabetes mellitus. Deep vein thrombosis. Obesity |
Alive, discharged from Hospital |
|
11 |
Seriously ill |
M |
75 |
Arterial hypertension. Ischemic cerebrovas-cular disease. Operated hip fracture |
Alive, discharged from Hospital |
|
12 |
Critically ill |
M |
53 |
Arterial hypertension. Schizophrenia. Malnut-rition |
Death |
|
13 |
Critically ill |
F |
65 |
Arterial hypertension. Chronic obstructive pulmonary disease. Obesity |
Death |
Table 4: Demographic characteristics of COVID-19 patients treated with CIGB-258, in which inflammation progresses
4. Discussion
The pathophysiology of COVID-19 is highly multi-faceted but three stages associated with severity can be described. Stage I is associated with early infection, which is characterized by symptoms of upper respiratory tract infection and stage II is the pulmonary phase when the patients progress to full-blown pneumonia. The last phase corresponds to hyperinflammation, when patients develop ARDS, sepsis and multiple organ failures [29]. Anti-inflammatory therapies for autoimmune diseases are under consideration for the control of this hyperin-flammation. These therapies include monoclonal antibo-dies against proinflammatory cytokines such as IL-1 (Anakinra) and IL-6 (Tocilizumab) [30, 31]. Other alternatives are Bruton kinase inhibitors and Janus kinase inhibitors [32, 33]. These drugs may reduce hyperinflammation, but will cause immunosuppression. However, immunosuppression is not beneficial in the course of viral infections; on the contrary, in this con-dition a robust immune response is needed to eliminate the virus.
CIGB-258 has shown, in previous studies, that it can inhibit hyperinflammation, in severe phases of COVID-19, while the patients treated with CIGB-258 did not show symptoms of possible immunosuppression asso-ciated with the drug. This treatment restores the NLR and produces a favorable outcome for the patients [20, 21]. Here, the treatment with CIGB-258 was applied to cohorts of patients in different stages of inflammation. The patients were classified as moderate, severe and critical, according to the Cuban classification criteria for COVID-19 patients, which are in line with the WHO criteria. Similar to previous reports, signs of immune-suppression and adverse effects associated with CIGB-258 were not detected in this study. Most patients included in this study had comorbidities and many of them had two or more of these pathological conditions that complicate the course of COVID-19. Many authors have described that arterial hypertension, cardiovascular diseases, diabetes mellitus, obesity, and others, affect the progression towards the severe phases of COVID-19 [9, 34,35]. In this study, the percentages of patients with comorbidities were similar in the three groups.
The patients who progress toward a severe phase of COVID-19 gradually increase biomarkers of inflame-mation, such as CRP, LDH and NLR. The increases in CRP concentrations have been linked to an unfavorable development of COVID-19 disease [6, 9]. LDH is considered a good biomarker associated with the prog-ression of COVID-19 pneumonia [10]. The NLR has been widely used as a severity predictor for COVID-19 [36]. Additionally, patients who progress towards the severe phases of COVID-19 gradually increase the levels of pro-inflammatory cytokines such as IL-6 [37]. All patients classified as moderate in this study had increased levels of NLR, CRP and LDH. Also, their clinical and radiological assessment indicated that these patients were progressing to the hyperinflammation phase. These criteria were the basis for starting the CIGB-258 treatment. CRP and LDH were significantly reduced, and the NLR decreased below 5, after seven days of treatment. The reduction in these biomarkers was in agreement with the clinical improvement of the patients. Besides, IL-6 levels tended to decrease after 96 hours of treatment. None of the patients with a moderate classification in this study progressed to the serious stages of the disease. This outcome is very interesting since it reinforces the therapeutic potential of CIGB-258. The result indicates that CIGB-258 prevents the progression to the severe phase in COVID-19 patients. These results contrast with other drugs used in patients with moderate COVID-19 such as dexamethasone. The recovery trials showed no benefit with dexamethasone in moderately ill patients not requiring oxygen. Moreover, the use of dexame-thasone can be harmful for these patients [38].
In this study, fifty-two seriously ill patients did not progress to the critical condition. These patients improved and the inflammation biomarkers diminished. Consequently, IL-6 levels significantly decreased after 96 hours of the treatment. High systemic concentrations of IL-6 have been correlated with disease severity [7]. These results are similar to our previous reports, in which TNF and IL-10 also decreased [20, 21]. Out of sixty-three seriously ill patients, only eleven patients progressed to a critical condition. These patients had several comorbidities that worsened their clinical condition. Despite this, six of these patients recovered from their critical condition and were discharged from the hospital. Twenty critically ill patients under treat-ment showed clinical improvement, which was associated with a normalization of CRP and LDH. Notably, a decrease in circulating IL-6 was found in these patients. In this study, IL-6 levels were higher in critically ill patients than in moderate and seriously ill patients. IL-6 has been involved in the progression of several viral infections [39]. During CIGB-258 treat-ment, IL-6 levels were significantly reduced, indicating the decline in inflammation. Out of twenty-two critically ill patients, two died. Both patients had several comorbidities that hampered their recovery.
The results presented in this study indicate that the early administration of CIGB-258 can improve the condition of moderately ill patients and avoid their progression to severe stages. This effect is in line with the molecular mechanism of this peptide. CIGB-258 inhibits the activity of monocytes, macrophages and neutrophils. This inhibition contributes to the restoration of normal levels of neutrophils and lymphocytes. Also, CIGB-258 has the ability to increase the frequency of Treg [21]. These facts contribute to the resolution of hyperin-flammation and the positive outcome of the patients. The main limitation of this study is the lack of a concurrent placebo-control group. The assessment of the efficacy of the CIGB-258 treatment will need rando-mized, placebo-controlled trials. Nevertheless, the current study ratifies that this peptide can reduce hyperinflammation. These results indicate the thera-peutic potential of CIGB-258 and sustain the rationale for additional research on this drug for the treatment of hyperinflammation in other diseases.
Acknowledgments
This study was sponsored by the CIGB and the Ministry of Public Health of Cuba. We thank for the CIGB-258 Study Group: Eduardo Pentón b, Miriam Ribasb, Klaudia Urrutia Pérez,a Karen Urrutia Pérez,a Raúl Santana Sánchez,b Beatriz Santiesteban Licea,b Deily Chacón Montano,b Pedro R. Aroche de Dios,b Sara María Martínez Martín,b Adriana del Carmen Sin Mayor,b Maura García Sánchez,b Marisol Díaz Gálvez,b Armando Armenteros Amador,b Ana Gloria Galarraga Lazcano,b Jesús Pérez Escribano,b María Elena Fuerte Calvo,b Raiko Vaillant Padrón,b Silvio Rodríguez Martín,d Nelson Yero Díaz,d Nancy Pastor Garofalo,d Ariel Blandy Malmierca,d Sergio Orizondo,d Maria E. Tacoronte Morales,d José D J Rego Hernández,d Teresa Albelo Larrinaga,d Alberto R Piriz Assa,e José A Romero García,e Pedro Noris Noris,e Boris L Carballo Hechavarría,e Reinier R. Paz Carballosa,e Juan Carlos Ortiz e, Jorge F. Núñez Martinez,f Ernesto de la Paz Carmona,h Yosvany Ortiz Jimenez,j Mariano Valle Vargas,i Lilia M Ortega González,j Alina Barcala Nuñez,j Karem Catasus Alvarez,a Gustavo Perera Viamontes,c Angela Damary Tuero Iglesias,a Marel Alonso Valdés,a Ana Campal Espinosa,a José A. Buxadó,1 Eulogio Pimentel Vazquez,a Martha Ayala Avila, a and Eduardo Martínez Diaz.a
Conflict of Interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
References
- Zhu N, Zhang D, Wang W et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. New Engl J Med 382 (2020): 727-733.
- Galbán-García E, Más-Bermejo P. COVID-19 in Cuba: Assessing the National Response. MEDICC Review 22 (2020): 29-34.
- Chen G, Wu D, Guo W. et al. Clinical and immunological features of severe and moderate coronavirus disease 2019. J. Clin. Invest 130 (2020): 2620-2629.
- Qin C, Zhou L, Hu Z, et al .Dysregulation of immune response in patients with COVID-19 in Wuhan, China. ClinInfectDis 71 (2020): 762-768.
- Xu Z, Shi L, Wang Y, et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med 8 (2020): 420-422.
- Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retro-spective cohort study. Lancet 395 (2020): 1054-1062.
- Mehta P, McAuley DF, Brown M, et al. COVID-19: consider cytokine storm syndro-mes and immunosuppression. Lancet 395 (2020): 1033-1034.
- De Maio A, Hightower LE. COVID-19, acute respiratory distress syndrome (ARDS), and hyperbaric oxygen therapy (HBOT): Cell Stress Chaperones 25 (2020): 717-720.
- Wu C, Chen X, Cai Y, et al. Risk Factors Associated With Acute Respiratory Distress Syndrome and Death in Patients With Coro-navirus Disease 2019 Pneumonia in Wuhan, China. JAMA Intern Med 180 (2020): 934-943.
- Wu M, Yao L, Wang Y, et al .Clinical evaluation of potential usefulness of serum lactate dehydrogenase (LDH) in 2019 novel coronavirus (COVID-19) pneumonia. Respir Res 21 (2020):171.
- Herold T, Jurinovic V, Arnreich C, et al. Elevated levels of IL-6 and CRP predict the need for mechanical ventilation in COVID-19. J Allergy Clin Immunol 146 (2020): 128-136.
- Wang F, Hou H, Luo Y, et al. The laboratory tests and host immunity of COVID-19 patients with different severity of illness. JCI insight 5 (2020): 137799.
- Soy M, Keser G, Atagündüz P, et al. Cytokine storm in COVID-19: pathogenesis and overview of anti-inflammatory agents used in treatment. Clin. Rheumatol 39 (2020); 2085-2094.
- Xu X, Han M, Li T, et al. Effective treatment of severe COVID-19 patients with tocilizumab. Proc. Natl. Acad. Sci. USA 117 (2020); 10970-10975.
- Domínguez MC, Lorenzo N, Barbera A, et al. An altered peptide ligand corresponding to a novel epitope from heat-shock protein 60 induces regulatory T cells and suppresses pathogenic response in an animal model of adjuvant induced arthritis. Autoimmunity 44 (2011): 471-482.
- Barberá A, Lorenzo N, Garrido G, et al. APL-1, an altered peptide ligand derived from human heat-shock protein 60, selectively induces apoptosis in activated CD4+ CD25+ T cells from peripheral blood of rheumatoid arthritis patients. Int Immunopharmacol 17 (2013): 1075-1083.
- Lorenzo N, Altruda F, Silengo L, et al. APL-1, an altered peptide ligand derived from heat-shock protein, alone or combined with metho-trexate attenuates murine collagen induced arthritis. Clin Exp Med 17 (2017): 209-216.
- Corrales O, Hernandez L, Prada D et al. CIGB-814, an altered peptide ligand derived from human heat-shock protein 60, decreases anti-cyclic citrullinated peptides antibodies in patients with rheumatoid arthritis .Clinical Rheumatol 38 (2019): 955-960.
- Barberá A, Lorenzo N, van Kooten P, et al. APL1, an altered peptide ligand derived from human heat-shock protein 60, increases the frequency of Tregs and its suppressive capacity against antigen responding effector CD4+T cells from rheumatoid arthritis patients. Cell Stress Chaperones 21 (2016): 735-744.
- Venegas-Rodriguez R, Santana-Sanchez R, Peña-Ruiz R, et al. CIGB-258 Immuno-modulatory Peptide: Compassionate Use for Critical and Severe COVID-19 Patients. Austin J Pharmacol Ther 8 (2020): 1119.
- Hernandez-Cedeño M, Venegas-Rodriguez R, Peña-Ruiz R et al. CIGB-258, a peptide derived from human heat-shock protein 60, decreases hyperinflammation in COVID-19 patients. Cell Stress Chaperones 26(2021): 515-525.
- CECMED Jusvinza, Emergency Use Author-ization for the treatment of patients with COVID-19 (Autorizo de Uso de Emergencia a Jusvinza, para el tratamiento de pacientes con COVID-19) (2020).
- Ministry of Public Health of Cuba .Protocolo de Actuación Nacional para la COVID-19 Versión 1.5 ;2020. [cited 2020 August] .
- Ministry of Public Health of Cuba .Parte de cierre del día 31 de marzo a las 12 de la noche;2020 [cited 2020 April 1].
- World Medical Association. World medical declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA 310 (2013): 2191-2194.
- Dixon JR. The international conference on harmonization good clinical practice guideline. Qual Assur 6 (1999): 65-74.
- Ranieri VM, Rubenfeld GD, Thompson BT, et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA 307 (2012): 2526-2533.
- González-Zorrilla M, González-Mesana R, Hernández-Cedeño M et al. CIGB-258, An Immunomodulatory Peptide for the Treatment of a COVID-19-associated Hepatic Encepha-lopathy: A Case Report. Preprints. (2020); 2020090240.
- Wiersinga WJ, Rhodes A, Cheng AC,Peacock SJ, Prescott HC. Pathophysiology, Transmi-ssion, Diagnosis, and Treatment of Coronavirus Disease 2019 (COVID-19). JAMA 324 (2020): 782-793.
- Huet T, Beaussier H, Voisin O, et al. Anakinra for severe forms of COVID-19: a cohort study. Lancet Rheumatol 2 (2020): 393-400.
- Capraa R, DeRossia N, Mattiolib F, et al. Impact of low dose tocilizumab on mortality rate in patients with COVID-19 related pneu-monia . Eur J Intern Med 76 (2020): 31-35.
- Roschewski M, Lionakis MS, Sharman JP et al. Inhibition of Bruton tyrosine kinase in patients with severe COVID-19. Sci Immunol 5 (2020): 0110.
- Peterson D, Damsky W, King B. The use of Janus kinase inhibitors in the time of SARS-CoV-2. J Am Acad Dermatol 82 (2020): 223-226.
- Doglietto F, Vezzoli M, Gheza F, et al. Factors Associated With Surgical Mortality and Complications Among Patients With and Without Coronavirus Disease 2019 (COVID-19) in Italy. JAMA Surg 155 (2020): 1-14.
- Gasmi A, Peana M, Pivina L et al. Interrelations between COVID-19 and other disorders. Clin Immunol 224 (2021): 108651.
- Liu J, Liu Y, Xiang P et al. Neutrophil to lymphocyte ratio predicts critical illness patients with 2019 coronavirus disease in the early stage. J Transl Med 18 (2020): 206.
- Smetana K, Brábek J. Role of Interleukin-6 in Lung Complications in Patients With COVID-19: Therapeutic Implications. In Vivo 34 (2020): 1589-1592.
- Horby P, Lim WS, Emberson JR et al. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med 384 (2021): 693-704.
- Dienz O, Rincon M. The effects of IL-6 on CD4 T cell responses. Clin. Immunol 130 (2009): 27-33.
