Development and Evaluation of Nutraceutical products from Soybean, Sorghum and Basil leaf using Response Surface Methodology
Article Information
Abiodun Olapade, Uchenna Umeohia*
Department of Food Technology, Faculty of Technology, University of Ibadan, Oyo State, Nigeria
*Corresponding Author: Uchenna Umeohia, Department of Food Technology, Faculty of Technology, University of Ibadan, Oyo State, Nigeria
Received: 23 May 2021; Accepted: 31 May 2021; Published: 21 June 2021
Citation: Abiodun Olapade, Uchenna Umeohia. Development and Evaluation of Nutraceutical Products from Soybean, Sorghum and Basil Leaf Using Response Surface Methodology. Journal of Food Science and Nutrition Research 4 (2021): 144-160.
View / Download Pdf Share at FacebookAbstract
A central composite orthogonal experimental design (CCOD) was used to develop a nutraceutical product from soy protein isolate, sorghum pyrodextrin and basil instant soluble powder using their levels incorporation as the independent factors while functional, and antinutritional parameters served as the responses. The haematological and biochemical parameters of albino wistar rat fed with the preferred nutraceutical product were determined. Sensory profiling by 30 panelists using a 9-point hedonic scale was conducted. The response surface analysis of the dependent variables of the products revealed both quadratic and linear fitted effects (P<0.05).The developed nutraceutical product had water absorption capacity, 1.69g/g, oil absorption capacity, 0.78g/cm3 bulk density, 6.82 pH, 1.40mg/g trypsin inhibitor, 0.73mg/100g phytate, 0.54mg/100g tannin, and 2.15HU/mg haemagglutinins. The nutraceutical product boosted the homopeitic activities, reduced the fasting blood glucose and cholesterol of the experimental rats, while biochemical analysis indicated no toxicological implication. A regression graph of the observed and predicted physicochemical properties of the product showed a highly fitted model of R2 = 0.999. Consumer acceptability tests showed that the developed nutraceutical product was accepted by the panelists.
Keywords
Nutraceuticals, Pyrodextrin, Basil, Bioassay, Soy isolate, Response Surface Methodology (RSM), Regression analysis, Model, Fitting, Regression, Optimization
Nutraceuticals articles; Pyrodextrin articles; Basil articles; Bioassay articles; Soy isolate articles; Response Surface Methodology (RSM) articles; Regression analysis articles; Model articles; Fitting articles; Regression articles; Optimization articles
Nutraceuticals articles Nutraceuticals Research articles Nutraceuticals review articles Nutraceuticals PubMed articles Nutraceuticals PubMed Central articles Nutraceuticals 2023 articles Nutraceuticals 2024 articles Nutraceuticals Scopus articles Nutraceuticals impact factor journals Nutraceuticals Scopus journals Nutraceuticals PubMed journals Nutraceuticals medical journals Nutraceuticals free journals Nutraceuticals best journals Nutraceuticals top journals Nutraceuticals free medical journals Nutraceuticals famous journals Nutraceuticals Google Scholar indexed journals Pyrodextrin articles Pyrodextrin Research articles Pyrodextrin review articles Pyrodextrin PubMed articles Pyrodextrin PubMed Central articles Pyrodextrin 2023 articles Pyrodextrin 2024 articles Pyrodextrin Scopus articles Pyrodextrin impact factor journals Pyrodextrin Scopus journals Pyrodextrin PubMed journals Pyrodextrin medical journals Pyrodextrin free journals Pyrodextrin best journals Pyrodextrin top journals Pyrodextrin free medical journals Pyrodextrin famous journals Pyrodextrin Google Scholar indexed journals Basil articles Basil Research articles Basil review articles Basil PubMed articles Basil PubMed Central articles Basil 2023 articles Basil 2024 articles Basil Scopus articles Basil impact factor journals Basil Scopus journals Basil PubMed journals Basil medical journals Basil free journals Basil best journals Basil top journals Basil free medical journals Basil famous journals Basil Google Scholar indexed journals Bioassay articles Bioassay Research articles Bioassay review articles Bioassay PubMed articles Bioassay PubMed Central articles Bioassay 2023 articles Bioassay 2024 articles Bioassay Scopus articles Bioassay impact factor journals Bioassay Scopus journals Bioassay PubMed journals Bioassay medical journals Bioassay free journals Bioassay best journals Bioassay top journals Bioassay free medical journals Bioassay famous journals Bioassay Google Scholar indexed journals Soy isolate articles Soy isolate Research articles Soy isolate review articles Soy isolate PubMed articles Soy isolate PubMed Central articles Soy isolate 2023 articles Soy isolate 2024 articles Soy isolate Scopus articles Soy isolate impact factor journals Soy isolate Scopus journals Soy isolate PubMed journals Soy isolate medical journals Soy isolate free journals Soy isolate best journals Soy isolate top journals Soy isolate free medical journals Soy isolate famous journals Soy isolate Google Scholar indexed journals Response Surface Methodology articles Response Surface Methodology Research articles Response Surface Methodology review articles Response Surface Methodology PubMed articles Response Surface Methodology PubMed Central articles Response Surface Methodology 2023 articles Response Surface Methodology 2024 articles Response Surface Methodology Scopus articles Response Surface Methodology impact factor journals Response Surface Methodology Scopus journals Response Surface Methodology PubMed journals Response Surface Methodology medical journals Response Surface Methodology free journals Response Surface Methodology best journals Response Surface Methodology top journals Response Surface Methodology free medical journals Response Surface Methodology famous journals Response Surface Methodology Google Scholar indexed journals Regression analysis articles Regression analysis Research articles Regression analysis review articles Regression analysis PubMed articles Regression analysis PubMed Central articles Regression analysis 2023 articles Regression analysis 2024 articles Regression analysis Scopus articles Regression analysis impact factor journals Regression analysis Scopus journals Regression analysis PubMed journals Regression analysis medical journals Regression analysis free journals Regression analysis best journals Regression analysis top journals Regression analysis free medical journals Regression analysis famous journals Regression analysis Google Scholar indexed journals Model articles Model Research articles Model review articles Model PubMed articles Model PubMed Central articles Model 2023 articles Model 2024 articles Model Scopus articles Model impact factor journals Model Scopus journals Model PubMed journals Model medical journals Model free journals Model best journals Model top journals Model free medical journals Model famous journals Model Google Scholar indexed journals Fitting articles Fitting Research articles Fitting review articles Fitting PubMed articles Fitting PubMed Central articles Fitting 2023 articles Fitting 2024 articles Fitting Scopus articles Fitting impact factor journals Fitting Scopus journals Fitting PubMed journals Fitting medical journals Fitting free journals Fitting best journals Fitting top journals Fitting free medical journals Fitting famous journals Fitting Google Scholar indexed journals Regression articles Regression Research articles Regression review articles Regression PubMed articles Regression PubMed Central articles Regression 2023 articles Regression 2024 articles Regression Scopus articles Regression impact factor journals Regression Scopus journals Regression PubMed journals Regression medical journals Regression free journals Regression best journals Regression top journals Regression free medical journals Regression famous journals Regression Google Scholar indexed journals
Article Details
1. Introduction
The increase in the spate of health-related diseases coupled with emerging trend in the demand for functional foods had fueled in the past few years the evolving aggregated interest by scientists geared towards producing foods targeted at meeting this current challenge. The world food market is currently interested in food that provide not only nutritive values but that also promote health benefits beyond those provided by their inherent components [1]. Due to risk of toxicity or adverse effect of drugs, consumers are turning massively to food supplements to improve health where pharmaceutical fails. This resulted in a worldwide nutraceutical revolution [2].
The term ‘‘nutraceutical’’ was coined from ‘‘nutrition’’ and ‘‘pharmaceutical’’ in 1989 by Stephen DeFelice. Nutraceutical can be defined as, a food or part of a food that provides medical or health benefits, including the prevention and/or treatment of a disease [3]. Nutraceuticals is a broad term used to describe any product derived from food sources that provides extra health benefits in addition to the basic nutritional value found in foods [4]. These nutraceuticals normally contain the required amount of vitamins, lipids, proteins, carbohydrates, minerals, or other necessary nutrients, depending on their emphases [5]. The presence of these bioactive ingredients in the processed foods has become quintessential in order to satisfy the demands of health conscious consumers. At present, nutraceuticals represent the fastest growing segment of today’s food industry [6]. Soybeans, Glycine max (L.) Merr. (Leguminosae) are one of the most important legumes [7]. Soybeans also contain many nutritious and functional phytochemicals such as isoflavones, phytic acids, saponins and oligosaccharides [8, 9]. Due to its high biological value and content good numbers of essential amino acids it can be used to prevent protein-calorie malnutrition among vulnerable groups in the community [10]. Several recent scientific studies [11] have shown that regular intake of traditional soya foods may help to prevent breast cancer, prostate cancer, colon cancer and menopausal problems of women. Due to presence of isoflavones and phytoestrogen in soyabean, it helps to prevents cancer by inhibiting the growth of existing tumor cells, and the risk of endometrial cancer. Regular intake of soya product helps to prevent disease by lowering total cholesterol, low density lipoprotein, blood pressure and prevent plague built up in arteries (atherocleorosis) [12]. Soy protein isolate (SPI), a soybean derivative protein powder, is one of the most important products in food processing as well as many other industrial uses [13]. Its preference is attributed to its ability to enhance nutritional (especially protein) and functional qualities of food products to which it is used as an ingredient [14-16]. Sorghum (Sorghum bicolor (L.) Moench) is a cultivated tropical cereal grass. It is the only viable food grain for many of the world’s most food insecure people [17]. Diversification of the use of this important grain through conversion to nutraceutical commodity like dextrin will open new vistas of application of sorghum and thereby enhance utilization. Eliminating low molecular-weight fractions can make the dextrin sugar-free and significantly improve its digestive tolerance and reduce hygroscopicity. An ideal soluble dietary fiber should exhibit instant and complete water solubility, low viscosity, neutral taste, high-fiber content, high digestive tolerance, excellent stability (heat and acid), and easy processability (good flow and direct compressibility). A novel soluble dextrin fiber, possessing all the above-mentioned characteristics, can overcome the stability and application limitations of most soluble fibers [18]. Ocimum gratissimum L. (Lamiaceae) is an herbaceous perennial plant commonly known as scent leaf because of its aromatic smell. The genus, Ocimum with the general name Basil, belongs to the family of plants known as Labiatae [19]. The plant is known to contain alkaloids, tannins, flavonoids and oligosaccharides [20]. It is used in the treatment of various diseases like cancer [21], antinociceptive, anti-inflammatory [22], antidiahorreal [23], antibacterial [24], antifungal [25], wound-healing [26] and as nephroprotective [20]. Its ethanolic extract has shown various activities like analgesic [27], antifungal [28], aphrodisiac [29], hepatoprotective [30], antioxidant [31,32] and anti-diabetic activity [33]. The specific objective of this study was to develop effective nutraceutical products from combinations of locally sourced plant material using response surface methodology as a means of optimization of the desirable responses.
2. Materials and Methods
2.1 Procurement of materials
The seeds of soybean (Glycine max) and sorghum (Sorghum bicolor L. (Merr.)) were purchased from Bodija market, Ibadan, Oyo State, Nigeria. Freshly cuts leaves of African Basil (Ocimum gratissimum) were procured from Ojo market, Ibadan, Oyo State, Nigeria.
2.2 Production of soy protein isolates (SPI)
The flow chart of preparation of soy protein isolate (SPI) is shown in Figure 1. The defatted soybean meal was produced using method of [34]. Briefly, soybean seeds (12kg) were sorted, cleaned and washed thoroughly with clean water. The washed seeds were soaked in 1:5 ratio of water for 8 hours and subsequently drained and dehulled. The dehulled seeds where dried, milled into fine flour using hammer mill and was deffated using soxhlet extraction method. The defatted meal was converted to isolate by the method reported by Zhongjiang et al. [35]. Defatted soy bean flakes where suspended in 10-fold water and adjusted to a pH of 7 with 2mol/L NaOH. After stirring for 1hour, the suspension was centrifuged at 8000rpm for 30minutes. The supernatant was further subjected to isoelectric precipitation by adjusting the pH to 4.5 with 2mol/L HCL. The protein precipitate was obtained by centrifugation (5,000 rpm, 30 minutes), resuspended in water and adjusted to a pH of 7 with 2mol/L NaOH. After removing a small amount of insoluble substances by centrifuging at 10,000 rpm for 30minutes, the protein solution was freeze dried and ground to yield SPI powder.
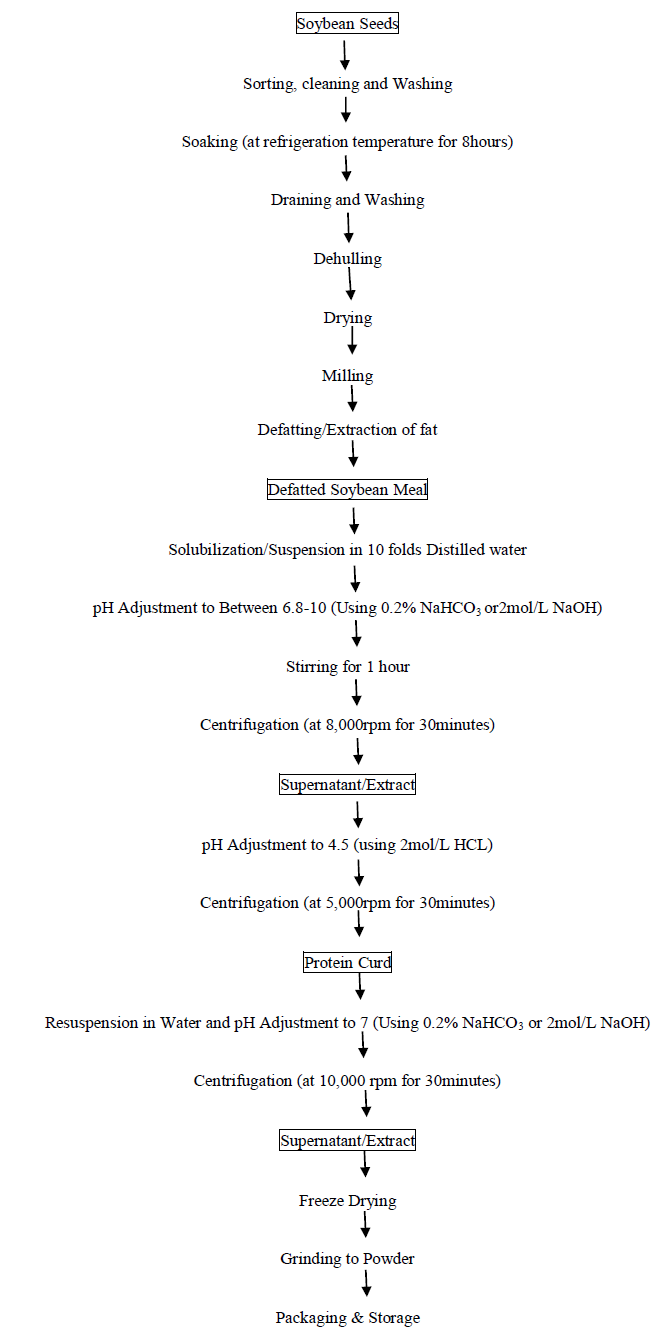
Figure 1: Flow chart for the production of soybean protein isolate (SPI) [35]
2.3 Production of sorghum pyrodextrin
2.3.1 Isolation of sorghum starch: Sorghum starch was extracted by using the methods of Beta and Singh [36, 37] as shown in Figure 2. Sorghum grain (10kg) was steeped in 10 litres of NaOH (0.25%, w/v) at 5oC for 24hours. The steeped grains were washed and ground with an equal volume of water using a blender for 3minutes. The slurry was filtered through a 200-mesh screen. The material remaining on the sieve was rinsed with water. Grinding and filtering processes were repeated on this material. After rinsing, the material still remaining on the sieve was discarded. The filtrate was allowed to stand for 1hour. The filtrate was centrifuged at 10,000rpm for 19 minutes. The grey coloured, top protein-rich layer was removed using a spatula. Excess water was added to re-suspend the sample, and centrifugation was done for 5minutes. Washing and centrifugation was repeated several times until the top starch layer was white. The starch was dried for 24 hours at 40oC.
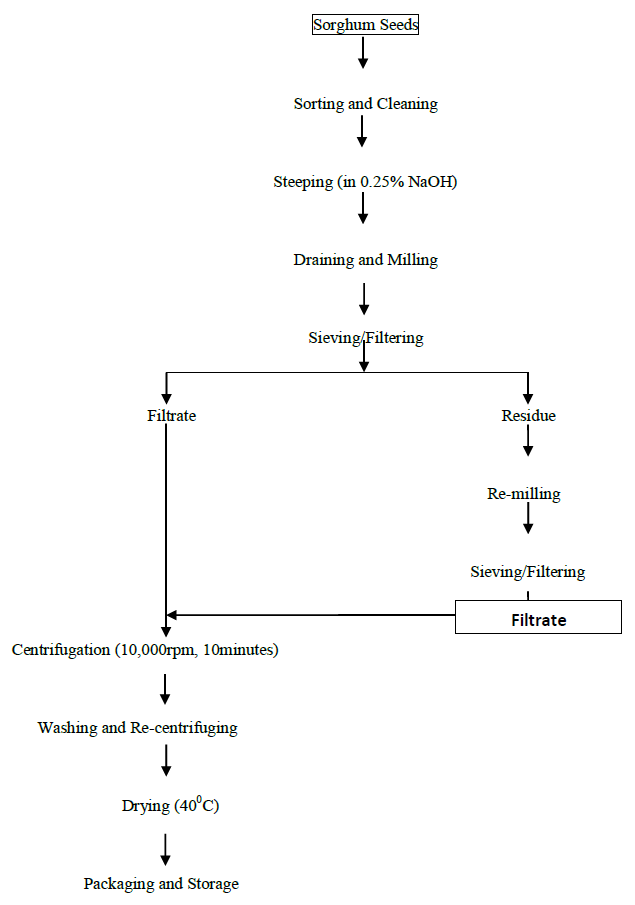
Figure 2: Flow chart of production of pyrodextrin from sorghum
2.3.2 Pyrodextrinization of sorghum starch: The method of Sankhon [38] and Ghali [39], with slight modification was employed in the production of sorghum pyrodextrin. Briefly, 5kg of starch was placed in stainless trays and 2.2 M hydrochloric acid was added. The starch/HCl ratio was 80:1 (w/v). The acid was dispersed on the starch and the mixture was allowed to react for 16 hours at room temperature. After that the mixture was dried in an oven at 110 °C for 3 hours and grinded to pass through a 100 μm sieve.
2.3.3 Production of basil instant (Soluble) powder: The instant soluble basil powder was produced from freshly cut leaves of the Ocimum gratissimum as shown in Figure 3. Basil instant (soluble) powder was produced using the method of Sinija [40, 41], with slight modification. Fresh basil leaves were steamed immediately after plucking to arrest the fermentation reaction. The leaves were crushed and the juice was expressed out manually with the aid of cheese cloth for production of instant soluble powder. The juice with total solids of 7-9% was freeze dried to obtain basil instant soluble powder.
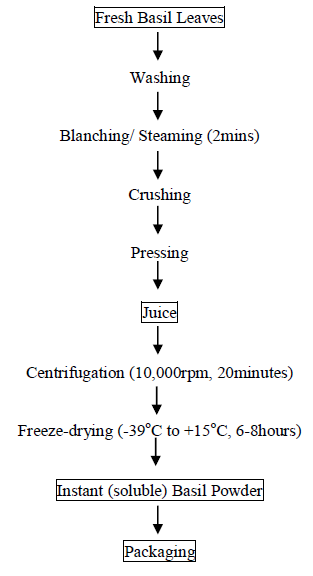
Figure 3: Production of Ocimum gratissimum instant (soluble) powder
2.4 Nutraceutical product formulation
Nutraceutical products were prepared in the laboratory. Each sample of the nutraceutical products were formulated from soy proteins isolates, sorghum pyrodextrin and basil instant soluble powder. The ingredients were measured accurately with weighing balance and mixed thoroughly using Kenwood Mixer according to experimental design in Table 1.
2.5 Experimental design for the nutraceutical product formulation
Response surface methodology (RSM) was used to determine optimum formulation for the responses of interest. A central composite orthogonal experimental design (CCOD) was created with 20 runs, 8 factorial points, 6 center points, and 6 axial points, with α value =1.52456. Response surface methodology (RSM) with a five-level three-factor mixture design was used to optimize the formula of the nutraceutical product. The effects of three independent variables: soy proteins isolate (A; 54.75-85.25g), sorghum pyrodextrin (B; 14.75-45.25g) and Basil instant (soluble) powder (C; 0.95-7.05g) on the physicochemical, functional, and sensory parameters of the nutraceutical products as well as effects on the haematological and biochemicall parameters on albino wistar rats after standardisation test were determined. The range and center point values of three independent variables presented in Table 1 was based on the results of preliminary experiments. Each variable to be optimized was coded at five levels (−α, −1, 0, +1, +α). Star points were carried out using α of 1.52465. Twenty randomized experiments including six replicates as the centre points were assigned based on CCOD.
In brief, the specified amount of the ingredients (soy protein isolate, sorghum pyrodextrin and basil instant tea powder) were measured accurately for each run and mixed thoroughly for even distribution of the component in a laboratory mixer.
The relationship of the independent variables and the response was calculated by the second-order polynomial Equation (1).
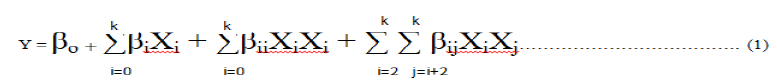
Y is the predicted response; βo is a constant; βi is the linear coefficient; βij is the cross product coefficient; and K the number of factors [42]. The second order polynomial coefficients was calculated using Statease design expert software package version 7.0.
2.5.1 The optimization process: A stepwise multiple regression analyses were conducted on the data from the Central Composite Ortogonal Design to relate amounts of soy protein isolate, sorghum pyrodextrin and basil instant (soluble) powder to the functional, antinutritional, and colour parameters of the samples. The response surface models were generated and presented as 3-dimensional plots in the function of 3 factors (soy protein isolate, sorghum pyrodextrin basil instant soluble powder). Adequacy of the model equation for predicting optimum response values was tested in the experiment using the soy protein isolate of 54.75–82.25g, sorghum pyrodextrin of 14.75-45.25g and amount of basil instant (soluble) powder of 0.95–7.05g. The optimal formulation of the nutraceutical products was determined from the mathematical models. In order to get these optimal values, the first partial derivatives of the regression equations was done according to X1 (A), X2 (B) and X3 (C) and sorted.
|
Soy Protein Isolate (g) A |
Sorghum Pyrodextrin (g) B |
Basil Instant Tea Powder (g) C |
||||
|
Runs |
Coded |
Uncoded |
Coded |
Uncoded |
Coded |
Uncoded |
|
1 |
-1.52465 |
54.75 |
0 |
30.00 |
0 |
4.00 |
|
2 |
0 |
70.00 |
0 |
30.00 |
0 |
4.00 |
|
3 |
0 |
70.00 |
0 |
30.00 |
0 |
4.00 |
|
4 |
0 |
70.00 |
+1.52465 |
45.25 |
0 |
4.00 |
|
5 |
-1 |
60.00 |
+1 |
40.00 |
-1 |
2.00 |
|
6 |
-1 |
60.00 |
-1 |
20.00 |
-1 |
2.00 |
|
7 |
+1 |
80.00 |
-1 |
20.00 |
-1 |
2.00 |
|
8 |
+1.52465 |
85.25 |
0 |
30.00 |
0 |
4.00 |
|
9 |
-1 |
60.00 |
+1 |
40.00 |
+1 |
6.00 |
|
10 |
0 |
70.00 |
0 |
30.00 |
-1.52465 |
0.95 |
|
11 |
0 |
70.00 |
0 |
30.00 |
0 |
4.00 |
|
12 |
+1 |
80.00 |
+1 |
40.00 |
+1 |
6.00 |
|
13 |
0 |
70.00 |
0 |
30.00 |
0 |
4.00 |
|
14 |
0 |
70.00 |
-1.52465 |
14.75 |
0 |
4.00 |
|
15 |
0 |
70.00 |
0 |
30.00 |
+1 |
7.05 |
|
16 |
0 |
70.00 |
0 |
30.00 |
0 |
4.00 |
|
17 |
0 |
70.00 |
0 |
30.00 |
0 |
4.00 |
|
18 |
+1 |
80.00 |
+1 |
40.00 |
1 |
2.00 |
|
19 |
-1 |
60.00 |
-1 |
20.00 |
1 |
6.00 |
|
20 |
+1 |
80.00 |
-1 |
20.00 |
1 |
6.00 |
Table 1: Levels of dependent and independent variables
2.6 Analyses of the nutraceutical products
Functional, antinutritional and colour parameters analyses were done on the samples of the nutraceutical products. The optimized sample of nutraceutical products was feed to albino wistar rats and the effects recorded through bioassay and subsequently subjected to sensory analysis to ascertain the level of acceptability of the developed product.
2.6.1 Functional properties: Water absorption capacity and oil absorption capacity was determined by the method of Sosulski [43], while bulk density was determined by the method of Pan [44]
2.6.2 Determination of anti-nutritional factors and phytochemicals: Trypsin inhibitor activity was determined by the method of Kakade [45], phytate by the method of Maga [46], tannin was determined by the method of Kirk and Sawyer [47], while haemagglutinin was determined by the method of Pull [48]
2.6.3 Bioassay: The optimized nutraceutical product was selected and used for bioassay study using 20 albino rats, their cholesterol level and blood sugar was determined by the method described by Sood [49], biochemical and haematological parameters were determined by Coles [50], liver and kidney function test was determined by Burtis and Ashwood [51].
2.6.4 Sensory analysis: Sensory evaluation of the samples was carried out using a 30-member panel; for various sensory attributes. A 9- point hedonic scale, where 9- like extremely, 8- like very much, 7- like moderately, 6 – like slightly, 5- neither like nor dislike, 4- dislike slightly, 3- dislike moderately, 2- dislike very much, 1- dislike extremely, as described by Ihekoronye and Ngoddy [52] was used to rate the taste, colour, flavour, mouthfeel and overall acceptability of the samples.
2.6.5 Statistical analysis: All the statistical analysis and graphical presentations were done using Statease Design Expert Software Package version 7.0. The significant probability was set at P < 0.05.
3. Results and Discussion
3.1 Effects of independent variables on the functional and antinutritional properties of the nutraceutical products
The functional and antinutrional properties of the nutraceutical products as affected by the varying proportions of soy protein isolate, sorghum pyrodextrin and basil instant soluble powder is presented in Table 2. From the results, water absorption capacity ranged between 2.698g/g and 3.366g/g, oil absorption capacity, 1.538g/g and 1.775g/g, bulk density, 0.715g/ml and 0.809g/ml, pH, 6.594 and 6.920, trypsin inhibitor activity, 1.132mg/g and 1.577mg/g, phytate, 0.609mg/100g and 0.810mg/100g, tannin, 0.532mg/100g and 0.610mg/100g and haemagglutinins, 1.774HU/mg and 2.399HU/mg.
Table 2: Functional properties and antinutritional components of the nutraceutical products
The dependency of functional and antinutritional properties of the nutraceutical samples on the independent variables of soy protein isolate, sorghum pyrodextrin and basil instant soluble powder is shown in the regression equations in Table 3. From the regression equations, water and oil absorption capacities, bulk density and pH possessed linear relationships, while trysin inhibitor activity, phytate, tannin and haemagglutinins had a mix of linear and quadratic effects. The effects of the independent factors on the functional and antinutrional properties of the nutraceutical products is depicted in 3D response plots in Figure 4.
|
Water Absorsption Capacity |
+3.04332 +9.37746E-003*A - 0.019708*B -0.019789*C |
0.9789 |
|
Oil Absorption capacity |
+1.65768 + 3.31444E-003*A - 6.38082E-003*B - 0.011273*C |
0.9341 |
|
Bulk Density |
+0.76621 +1.32577E-003*A - 2.55233E-003*B - 4.50927E-003*C |
0.9529 |
|
pH |
+6.74500 + 2.56898E-003*A - 6.75805E-003*B - 2.03283E-003*C |
0.5453 |
|
Trypsin Inhibitor |
+1.29970 + 0.014052*A - 0.025860*B - 0.024941*C + 6.68408E-005*A*B + 7.27080E-005*A*C + 2.57688E-004*B*C -7.21194E-005*A2 + 1.16853E-004*B2 -1.33931E-004*C2 |
0.9989 |
|
Phytate |
+0.73755 + 5.07859E-003*A - 0.012170*B - 0.013002*C +3.00784E-005*A*B +3.27186E-005*A*C +1.15959E-004*B*C -2.35613E-005*A2 +6.14764E-005*B2 + 1.62042E-004*C2 |
0.9993 |
|
Tannin |
+0.55876 -1.12125E-003*A + 4.59850E-003*B - 8.83440E-003*C - 1.50547E-005 *A* B +5.10070E-005*A*C +2.32601E-005*B*C + 4.96553E-006* A2 -2.33804E-005*B2 -3.40112E-007 *C2 |
0.9991 |
|
Haemagglutinins |
+2.14497 + 0.016680*A - 0.037402*B - 0.043190*C +9.21405E-005*A*B +1.19481E-004*A*C +3.78452E-004*B*C -7.92431E-005*A2 +1.85319E-004*B2 +3.91564E-004*C2 |
0.9997 |
Table 3: Regression equations of functional and antinutritional properties of the nutraceutical product formulations
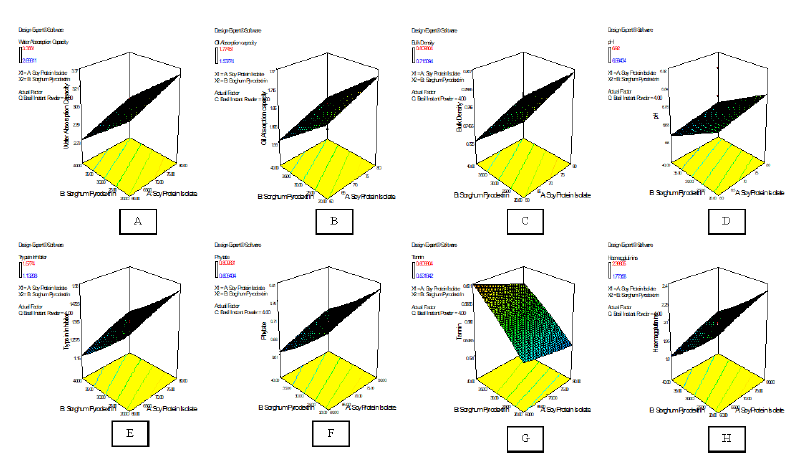
Figure 4: 3D Response surface plots of the effects of independent factors on the functional and antinutrional composition of the nutraceutical products
*A= water absorption capacity, B= oil absorption capacity, C= bulk density, D= pH, E= trypsin inhibitor, F= phytate, G= tannin, H= haemagglutinin
3.2 Optimization and validation of nutraceutical product formulation
In order to determine the optimum formulation, the regions of acceptability in the contour plot for each attribute were superimposed. Optimization was performed on the basis of a multiple response method called desirability. This procedure involved transforming scores on each of the dependent variables into desirability scores that could range from 0.0 for undesirable to 1.0 for very desirable. Thus considering the above mentioned approach, the objective was to hold in range functional, antinutritional, and colour parameters. Superimposition of contour plot regions of interest (within which, each attribute received ratings greater than or equal to 3.0) resulted in optimum regions for nutraceutical product formulation. The optimum nutraceutical product formulation were determined as all combinations from 54.75g to 85.25g soy protein isolate, from 14.75g to 45.25g sorghum pyrodextrin and from 0.96g to 7.05g basil instant soluble powder. Based on the superimposed plots, the selected optimal ingredient (independent variable) levels were 60.0g soy protein isolate, 20.0g sorghum pyrodextrin, and 4.83g basil instant soluble powders. The calculated desirability for this formulation was 0.679 and resulted to nutraceutical product of good quality. In order to verify the optimum formulation, the nutraceutical product using the optimal ingredient level was analyzed and the results were statistically compared to the predicted values of the mathematical model. The predicted response values and the actual obtained response values for the optimized products were within the range and found to be not statistically different at the 95% confidence level. The predicted and actual responses of the developed nutraceutical product are shown in Table 4.
Table 4: Predicted and Observed values for the physicochemical properties of the nutraceutical product properties.
The matching quality of the data obtained by the optimization model proposed was evaluated considering the correlation coefficient, R2, between the experimental and modeled data. The mathematical adjustment of those values generated a R2 = 0.999, revealing that the model could not explain only 1% of the overall effects, showing that it is a robust statistical model. The parity plot shows a satisfactory correlation between the experimental and predictive values (Figure 5).

Figure 5: Parity plot showing the distribution of experimental versus predicted values by the mathematical model of the y values
3.3 Haematological and Biochemichemical parameters of albino wistar rats fed with the developed nutraceutical product
The various biochemical and haematological parameters investigated in this study are useful indices of evaluating the toxicity of plant extract in animals [53]. Assessment of haematological parameters cannot only be used to determine the extent of deleterious effect of extracts on the blood of an animal, but it can also be used to explain blood relating functions of a plant extract or its products [54]. Analysis of blood parameters is relevant in risk evaluation as changes in the haematological system have higher predictive value for human toxicity when the data are translated from animal studies [55]. The comparative effects of the developed nutraceutical product and controlled diet on the haematological and biochemical parameters of the albino wistar rat is presented in the bar charts in Figures 6 and 7 respectively.
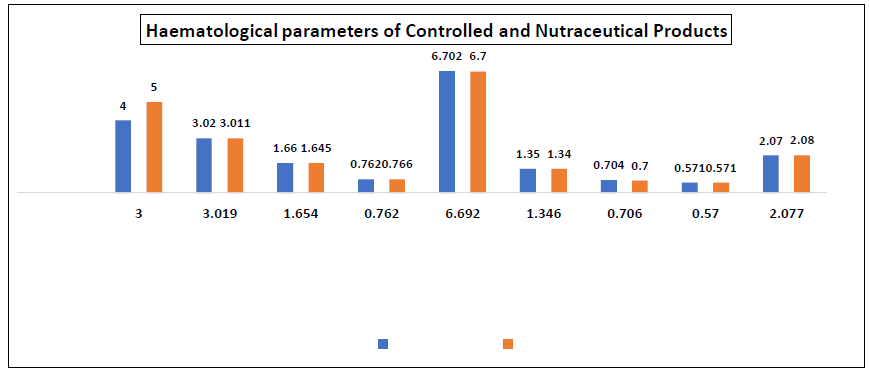
Figure 6: Haematological parameters of albino wistar rat fed with the nutraceutical product and controlled diet
*WBC = White blood cell count, RBC = red blood cell count, PVC = packed cell volume, MCH = mean corpuscular haemoglobin, MCHC = mean corpuscular haemoglobin concentration and MCV= mean corpuscular volume
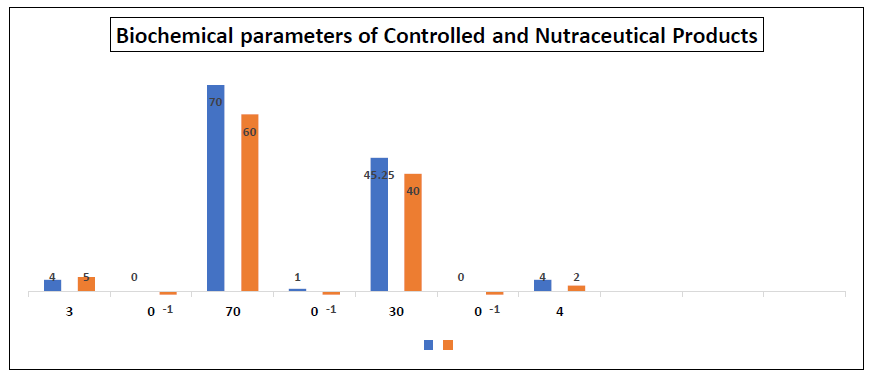
Figure 7: Biochemical parameters of albino wistar rat fed with the nutraceutical product and controlled diet
*ALP = alkaline phosphatase, ALT = alanine aminotransferase, and AST = aspartate aminotransferase
The result showed that there was non-significant effect of the nutraceutical product on the haematological parameters of the rats as compared to the controlled sample. The non-significant effect of the extract on the haematological parameters may be an indication that the balance between the rate of production (erythropoiesis) and destruction of the blood corpuscles was not altered. From the result, there was slight increase in the haematological parameters of rats fed with the nutraceutical product compared with the controlled sample, indicating that the developed nutraceutical product could be homeopetic and helps in building of blood. This could be as a result of the relative high iron content of the nutraceutical product and iron had been known to be an integral part of haemoglobin moiety. Alkaline phosphatase (ALP), alanine aminotransferase (ALT), aspartate aminotransferase (AST), bilirubin, albumin, total protein, urea and creatinine which measured the extent of alteration of normal biochemical reactions in the animals were significantly same for both groups. This may imply that the developed nutraceutical product had no toxicity implication as the biochemical parameters of the rats fed with it were similar to the ones fed with normal rat chows. On the contrary, the blood glucose level and cholesterol of the rats fed with the nutraceutical product significantly reduced. This implied that the developed nutraceutical product could had blood sugar and cholesterol lowering potentials.
3.4 Sensory profile of the nutraceutical product
The sensory analysis of the developed nutraceutical product using hedonic rating system revealed that the product was accepted by the panelists. It was observed that the mean rating for overall acceptability of the product was 7.17 (moderately liked). The mean rating for colour was 7.63 (like moderately), mouthfeel/texture had 7.43 (moderately liked), while taste had 6.97 (slightly liked). Flavour was rated the lowest having mean value of 6.90 (slightly liked).
4. Conclusion
Response surface methodology (RSM) was successfully applied to optimize the development of soy-sorghum-basil nutraceutical product. The three variables employed in the study had a significant effect on the functional, and antinutritional parameters of the nutraceutical products. Modelling of experimental data allowed the generation of useful regression equations for general use, to predict the behaviours of the products under different factor combination. The nutraceutical product developed from the models had no toxicological effects and significantly lowered the blood glucose and cholesterol levels of the albino wistar rats fed with it. The product was equally accepted by the sensory evaluation panelists.
References
- Torres-Giner S, Martinez-Abad A, Ocio MJ, et al. Stabilization of a nutraceutical omega-3 fatty acid by encapsulation in ultrathin electro-sprayed zein prolamine. Journal of Food Science 75 (2010): 69-79.
- Rohan SG, Bhartesh RK, Kiran AW, et al. Nutraceuticals: The Medicinal Key of Living Life Health. Journal of Pharmaceutical Research and Clinical Practice 1 (2011): 121-129.
- Brower V. Nutraceuticals: poised for a healthy slice of the healthcare market. Nat Biotechnology 16 (1998): 728-731.
- Tank DS, Sanket G, et al. Nutraceuticals. Portmanteau of science and nature. International Journal of Pharmaceutical Sciences Review and Research 5 (2010): 33-38.
- Whitman M. Understanding the perceived need for complementary and alternative nutraceuticals: lifestyle issues. Clinical Journal of Oncology Nursing 5 (2001)190-194.
- Kou J, Saxena DC. Studies on the Development of Nutraceutical Foods Using Extrusion Technology – A Review. Austin Journal of Nutrition and Food Sciences 2 (2014): 1-7.
- Yang JH, Mau JL, Ko PT, et al. Antioxidant properties of fermented soybean broth. Food Chemistry 71 (2000): 249-254.
- Anderson RL, Wolf WJ. Compositional changes in trypsin inhibitors, phytic acid, saponin, and isoflavones related to soybean processing. Journal of Nutrition 125 (1995) 581S-588S.
- Kwak CS, Lee MS, Park SC. Higher antioxidant properties of chungkookjang, a fermented soybean paste, may be due to increased aglycone and malonylaglycone isoflavone during fermentation. Nutritional Research 27 (2007): 719-727.
- Ghatge N. Supplementation of Nutraceutical Food to Malnourished Preschool Children and its Impact on Biochemical Examination. Research Journal of Family, Community and Consumer Sciences 1 (2013): 2-6.
- Messina MJ, Loprinzi CL. Soy intake and cancer risk. A critical Review of the literature and invivodata, Journal of Nutritional. 131 (1994): 3095S-108S.
- Messina MJ. Soyfood their role in disease prevention and treatment in Liu Keshun. Editor Soybean chemistry.Technolog and utilization. Chapmanand Hall, New York. (1997): 443-447.
- Moses VM, Yufei H, Quirino D, et al. Effects of Synthetic and Natural Extraction Chemicals on Functional Properties, Polyphenol Content and Antioxidant Activity of Soy Protein Isolates Extracted from Full-Fat and Defatted Flours. Advance Journal of Food Science and Technology 5 (2013):1443-144.
- Kinsella JE. Functional properties of soy protein isolate. Journal of the American Oil Chemists' Society. 56 (1979): 242-258.
- Mariotti F, Mahé S, Benamouzig R, et al. Nutritional value of [15N]-soy protein isolate assessed from ileal digestibility and postprandial protein utilization in humans. Journal of Nutrition 129 (1999): 1992-1997.
- L’hocine L, Boye JI, Arcand Y. Composition and functional properties of soy protein isolate prepared using alternative defatting and extraction procedures. Journal of Food Science 71 (2006): 137-C145.
- Taylor JR. Overview: Importance of Sorghum in africa: In Proceedings of AFRIPRO Workshop on the Proteins of Sorghum And Millets: Enhancing Nutritional and Functional Properties for Africa. Pretoria, South Africa (2003): 1-29.
- Roquette F. Nutriose: A New Soluble Fiber for Nutraceutical and Clinical Formulations, Roquette Technical literature (2014): 1-30.
- Ighodaro OM, Agunbiade SO, Akintobi OA. Phytotoxic and Antimicrobial Activities of Flavonoids in Ocimum grattisumum’, European Journal of Applied Sciences 2 (2010): 37-40.
- Ezeonwu VU, Dahiru D. Protective Effect of Bi-Herbal Formulation of Ocimum gratissimum and Gongronema latifolium Aqueous Leaf Extracts On Acetaminophen-Induced Hepato-Nephrotoxicity in Rats. American Journal of Biochemistry 3 (2013): 18-23.
- Filip A, Clichici S, Daicoviciu D, et al. Photochemoprevention of Cutaneous Neoplasia Through Natural Products. Experimental Oncology 31 (2009): 9-15.
- Tanko Y, Yaro AH, Mohammed KA, et al. Antineociceptive and Anti-Inflammatory Activities of Methanol Leaves Extract of Ocimum gratissimum in Mice and Rats. Journal of Pharmacy and Biological Sciences 4 (2012): 2278-3008.
- Ezekwesili CN, Obiora KA, Ugwu OP. Evaluation of Anti- Diarrhoeal Property of Crude Aqueous Extract of Ocimum gratissimum L. (Labiatae) in Rats. Biokemistri 16 (2004): 122-131.
- Sastry KP, Kumar RR, Kumar AN, et al. Morpho-Chemical Description and Antimicrobial Activity of Different Ocimum Species. Journal of Plant development 19 (2012): 53-64.
- Nguefack J, Lekagne JB, Dakole CD, et al. Food Preservative Potential of Essential Oils and Fractions from Cymbopogon citratus, Ocimum gratissimum and Thymus vulgaris Against Mycotoxigenic Fungi. International Journal of Food Microbiology 131 (2009): 151-156.
- Orafidiya LO, Fakoya FA, Agbani EO, et al. Vascular Permeability- Increasing effect of the leaf essential oil of ocimum gratissimum linn as a mechanism for its wound healing property. African Journal of Traditional, Complementary and alternative medicine 2 (2005): 253-258.
- Okiemy NA, Miguel ML, Etou AW, et al. Analgesic Effect of Aqueous and Hydroalcoholic Extracts of Three Congolese Medicinal Plants: Hyptis suaveolens. Nauclea latifolia and Ocimum gratissimum. Pakistan Journal of biological sciences 7 (2004) 1613-1615.
- Ijato JY. Inhibitory Effects of Two Indigenous Plant Extracts (Zingiber officinale and Ocimum gratissimum) on Post Harvest Yam (Dioscorea rotundata Poir) Rot, In Vitro. Journal of American Science 7 (2011): 43-47.
- Pande M, Pathak A. Effect of Ethanolic Extract of Ocimum gratissimum (ram tulsi) on Sexual Behaviour in Male Mice. International Journal of Pharmtech Research 1 (2009): 468-473.
- George S, Chaturvedi P, Kamau JM. Hepatoprotective Potential of Ocimum gratissimum on Ethanol Induced Hepatotoxicity in Albino Rats. Asian Journal of Pharmaceutical and Biological Research 2 (2012): 27-32.
- Oboh G. Antioxidant and Antimicrobial Properties of Ethanolic Extract of Ocimum gratissimum Leaves. Journal of Pharmacology and Toxicology 5 (2006): 47-53.
- Njoku OU, Joshua PE, Chidozie V, et al. Antioxidant Properties of Ocimum gratissimum (Scent Leaf). New York Science Journal4 (2011): 98-103.
- Oguanobi NI, Chijioke CP, Ghasi S. Anti-Diabetic Effectof Crude Leaf Extracts of Ocimum gratissimum in Neonatal Streptozotocin-induced Type-2 Model Diabetic Rats. International Journal of Pharmacy and Pharmaceutical Sciences 4 (2012): 77-83.
- Enwere NJ. Foods of Plant Origin. Afro-Orbis Publications LTD, Nsukka, Enugu Nigeria (1998) 56-60.
- Zhongjiang W, Yang L, Lianzhou J, et al. Relationship between Secondary Structure and Surface Hydrophobicity of Soybean Protein Isolate Subjected to Heat Treatment. Journal of Chemistry (2014): 1-10.
- Beta T, Corke H, Rooney LW, Taylor JRN. Starch properties as affected by sorghum grain chemistry. Journal of the Science of Food and Agriculture 81 (2001): 245-251.
- Singh H., Sodhi NS, Singh N. Structure and Functional Properties of Acetylated Sorghum Starch. International Journal of Food Properties 15 (2012): 312-325.
- Sankhon A, Yao W, Amadou I, et al. Effect of Pyrodextrinization, Crosslinking and Heat-Moisture Treatment on In vitro Formation and Digestibility of Resistant Starch from African Locust Bean (Parkia biglobosa). Tropical Journal of Pharmaceutical Research 12 (2013): 173-179.
- Ghali Y, Ibrahim N, Gabr S, et al. Modification of corn starch and fine flour by acid and gamma irradiation. Part 1. Chemical investigation of the modified product. Starch/Starke 31 (1979): 325-332.
- Sinija VR, Mishra HN, Bal S. Process technology for production of soluble tea powder. Journal of Food Engineering 82 (2008): 276-283.
- Sinija VR, Mishra HN. Moisture sorption isotherms and heat of sorption of instant (soluble) green tea powder and green tea granules. Journal of Food Engineering 86 (2007): 494-500.
- Bezerra MA, Santelli RE, Oliveira EP, et al. Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 76 (2008): 965-977.
- Sosulski F. Young CG. Yield and functional properties of air-classified protein and starch fractions from eight legume flours. Journal of American Oil and Chemical Society, 56 (1979): 292-295.
- Pan Z, Zhnag S, Jane J. Effects of extrusion variables and chemicals on the properties of starch-based binder and processing conditions. Cereal Chemistry. 75 (1998): 541-546.
- Kakade MC, Rackis JJ, Mcghee JC, et al. Determination of trypsin inhibitor actually of soya products: A collaborative analysis of an improved procedure. Cereal Chemistry 51 (1974): 376-384.
- Maga JA. Phytates: Its chemistry, occurrence, food interactions, nutritional significance and method of analysis. Journal of Agricultural Food Chemistry 30 (1982): 1-7.
- Kirk R, Sawyer R. Pearson’s composition and analysis of foods. Churchill Livingstone, Edinburgh (1998): 211-212.
- Pull SP, Puekke SG, Hymowitz T. et al. Soybean Lines Lacking the 120,000-Dalton Seed Lectin. Science 200 (1978): 1277-1279.
- Sood R. Textbook of medical laboratory technology. Jaypee Brothers Medical Publishers Ltd, New Delhi, India (2006): 1281.
- Coles EH. Veterinary Clinical Pathology. W.B. Saunders Company, Philadelphia 4 (1986):1-486.
- Burtis AC, Ashwood ER. Tietz Textbook of Clinical Chemistry.3rd Ed. AACCW.B. Sounders co. London 564, (1999): 1-1917.
- Ihekoronye AI, Ngoddy PO (1985) ‘Integrated Food Science and Technology for the Tropics’, Macmillan Education Ltd., London (1985):180-189.
- Yakubu MT, Akanji MA, Oladiji AT. Alterations in serum lipid profile of male rats by oral administration of aqueous extract of Fadogia argrestis stem. Research Journal of Medicinal Plant 2 (2008). 66-73.
- Yakubu MT, Akanji MA, Oladiji AT. Haematological evaluation in male albino rats following chronic administration of aqueous extract of Fadogia agrestis stem. Pharmacognosy Magazine 3 (2007): 34-38.
- Olson H. Betton G. Robinson D. et al. Concordance of toxicity of pharmaceuticals in humans and in animals. Regulatory Toxicology Pharmacology 32 (2000).
