Determinants of Behavioral Risk Factors of Hypertensive Disorders in Pregnancy
Article Information
Nkem Ernest Njukang1, 5*, Thomas Obinchemti EGBE2, Martin Sama1, Tah Aldof Yoah1, Joseph Kamgno3, 4
1Department of Public Health and Hygiene, Global Health System Solutions (GHSS), Yaounde, Cameroon
2Department of Obstetrics and Gynaecology, Faculty of Health Sciences, University of Buea, Buea, Cameroon
3Faculty of Medicine and Biomedical Sciences, University of Yaounde I, Yaounde, Cameroon
4Centre for Research on Filariasis and other Tropical Diseases (CRFilMT), Yaounde, Cameroon
5Quality Assurance/Quality Management System Mentor, Global Health System Solutions (GHSS), Yaounde, Cameroon
*Corresponding Author: Nkem Ernest Njukang, Department of Public Health and Hygiene, Quality Assurance/Quality Management System Mentor, Global Health System Solutions (GHSS), Yaounde, Cameroon
Received: 30 July 2020; Accepted: 12 August 2020; Published: 07 September 2020
Citation:
Nkem Ernest Njukang, Thomas Obinchemti EGBE, Martin Sama, Tah Aldof Yoah, Joseph Kamgno. Determinants of Behavioral Risk Factors of Hypertensive Disorders in Pregnancy. Obstetrics and Gynecology Research 3 (2020): 172-189.
View / Download Pdf Share at FacebookAbstract
Background: Hypertensive disorders in pregnancy (HDP) remain a major public health problem worldwide, and its prevalence varies from country to country and from institution to institutions. Few studies have explored the behavioral risk factors of hypertension (HTN) among pregnant women in Cameroon. Thus, this study aimed at determining the prevalence and socio-demographic predictors of behavioral risk factors of HDP in Mezam division.
Methods: A Hospital-based cross-sectional study was conducted in Mezam division. Over 1210 pregnant women were sampled by consecutive sampling. Descriptive statistics, chi-square (χ2) test and multivariate logistic regression were used for analysis.
Results: There was poor uptake of behavioural risk factors: alcohol (21.6%), physical inactivity (49.2%), low fruits/vegetables intake (80.7%), high salt intake (12.4%), stress (48.8%) and overweight/obese (65.5%). Sociodemographic predictors: Age was a risk factor for smoking [Adjusted Odd Ratio (AOR), 95%Confidence interval (CI); (25-29 years): 1.6 (1.1-2.4) & (≥35 years): 2.3 (1.4-3.6)], alcohol [(25-29 years): 1.5 (1.1-2.2) & (≥35 years): 2.5 (1.5-4.1)] and over weight/obesity [(25-29 years): 1.6 (1.2-2.2), (30-34 years): 2.6 (1.8-3.9) & (≥35 years): 2.5 (1.5-4.1)]. Education [secondary school: 1.8 (1.3-2.6) & high school: 2.4 (1.6-3.6)] and Occupation [petit traders: 1.8 (1.4-2.8) & business/employed: 1.9 (1.3-2.9)] were predictors of stress. Religion [Pentecostal: 1.8 (1.1-3.1)] was predictor for high salt consumption.
Conclusion: There was poor uptake of behavioural risk factors of HDP. Age, education, occupation and Religion were predictors of behavioural factors of HTN. There is need for immediate health promotion interventions such as raising community awareness and appropriate antenatal counseling.
Keywords
<p>Behavioral Risk Factors, Hypertension, Hypertensive Disorders in Pregnancy, Pregnant Women, Socio-Demographic Predictors, Mezam</p>
Behavioral Risk Factors articles Behavioral Risk Factors Research articles Behavioral Risk Factors review articles Behavioral Risk Factors PubMed articles Behavioral Risk Factors PubMed Central articles Behavioral Risk Factors 2023 articles Behavioral Risk Factors 2024 articles Behavioral Risk Factors Scopus articles Behavioral Risk Factors impact factor journals Behavioral Risk Factors Scopus journals Behavioral Risk Factors PubMed journals Behavioral Risk Factors medical journals Behavioral Risk Factors free journals Behavioral Risk Factors best journals Behavioral Risk Factors top journals Behavioral Risk Factors free medical journals Behavioral Risk Factors famous journals Behavioral Risk Factors Google Scholar indexed journals Hypertension articles Hypertension Research articles Hypertension review articles Hypertension PubMed articles Hypertension PubMed Central articles Hypertension 2023 articles Hypertension 2024 articles Hypertension Scopus articles Hypertension impact factor journals Hypertension Scopus journals Hypertension PubMed journals Hypertension medical journals Hypertension free journals Hypertension best journals Hypertension top journals Hypertension free medical journals Hypertension famous journals Hypertension Google Scholar indexed journals Hypertensive Disorders in Pregnancy articles Hypertensive Disorders in Pregnancy Research articles Hypertensive Disorders in Pregnancy review articles Hypertensive Disorders in Pregnancy PubMed articles Hypertensive Disorders in Pregnancy PubMed Central articles Hypertensive Disorders in Pregnancy 2023 articles Hypertensive Disorders in Pregnancy 2024 articles Hypertensive Disorders in Pregnancy Scopus articles Hypertensive Disorders in Pregnancy impact factor journals Hypertensive Disorders in Pregnancy Scopus journals Hypertensive Disorders in Pregnancy PubMed journals Hypertensive Disorders in Pregnancy medical journals Hypertensive Disorders in Pregnancy free journals Hypertensive Disorders in Pregnancy best journals Hypertensive Disorders in Pregnancy top journals Hypertensive Disorders in Pregnancy free medical journals Hypertensive Disorders in Pregnancy famous journals Hypertensive Disorders in Pregnancy Google Scholar indexed journals Pregnant Women articles Pregnant Women Research articles Pregnant Women review articles Pregnant Women PubMed articles Pregnant Women PubMed Central articles Pregnant Women 2023 articles Pregnant Women 2024 articles Pregnant Women Scopus articles Pregnant Women impact factor journals Pregnant Women Scopus journals Pregnant Women PubMed journals Pregnant Women medical journals Pregnant Women free journals Pregnant Women best journals Pregnant Women top journals Pregnant Women free medical journals Pregnant Women famous journals Pregnant Women Google Scholar indexed journals Socio-Demographic Predictors articles Socio-Demographic Predictors Research articles Socio-Demographic Predictors review articles Socio-Demographic Predictors PubMed articles Socio-Demographic Predictors PubMed Central articles Socio-Demographic Predictors 2023 articles Socio-Demographic Predictors 2024 articles Socio-Demographic Predictors Scopus articles Socio-Demographic Predictors impact factor journals Socio-Demographic Predictors Scopus journals Socio-Demographic Predictors PubMed journals Socio-Demographic Predictors medical journals Socio-Demographic Predictors free journals Socio-Demographic Predictors best journals Socio-Demographic Predictors top journals Socio-Demographic Predictors free medical journals Socio-Demographic Predictors famous journals Socio-Demographic Predictors Google Scholar indexed journals Mezam articles Mezam Research articles Mezam review articles Mezam PubMed articles Mezam PubMed Central articles Mezam 2023 articles Mezam 2024 articles Mezam Scopus articles Mezam impact factor journals Mezam Scopus journals Mezam PubMed journals Mezam medical journals Mezam free journals Mezam best journals Mezam top journals Mezam free medical journals Mezam famous journals Mezam Google Scholar indexed journals obstetricians articles obstetricians Research articles obstetricians review articles obstetricians PubMed articles obstetricians PubMed Central articles obstetricians 2023 articles obstetricians 2024 articles obstetricians Scopus articles obstetricians impact factor journals obstetricians Scopus journals obstetricians PubMed journals obstetricians medical journals obstetricians free journals obstetricians best journals obstetricians top journals obstetricians free medical journals obstetricians famous journals obstetricians Google Scholar indexed journals gynecologist articles gynecologist Research articles gynecologist review articles gynecologist PubMed articles gynecologist PubMed Central articles gynecologist 2023 articles gynecologist 2024 articles gynecologist Scopus articles gynecologist impact factor journals gynecologist Scopus journals gynecologist PubMed journals gynecologist medical journals gynecologist free journals gynecologist best journals gynecologist top journals gynecologist free medical journals gynecologist famous journals gynecologist Google Scholar indexed journals gestation articles gestation Research articles gestation review articles gestation PubMed articles gestation PubMed Central articles gestation 2023 articles gestation 2024 articles gestation Scopus articles gestation impact factor journals gestation Scopus journals gestation PubMed journals gestation medical journals gestation free journals gestation best journals gestation top journals gestation free medical journals gestation famous journals gestation Google Scholar indexed journals eclampsia articles eclampsia Research articles eclampsia review articles eclampsia PubMed articles eclampsia PubMed Central articles eclampsia 2023 articles eclampsia 2024 articles eclampsia Scopus articles eclampsia impact factor journals eclampsia Scopus journals eclampsia PubMed journals eclampsia medical journals eclampsia free journals eclampsia best journals eclampsia top journals eclampsia free medical journals eclampsia famous journals eclampsia Google Scholar indexed journals
Article Details
1. Background
According to the American college of obstetricians and gynecologist (ACOG), Hypertension (HTN) in pregnancy is defined as: Systolic blood pressure greater than or equal to 140 mmHg and/or diastolic blood pressure greater than or equal to 90 mmHg in two occasions at least 6 h apart after fifth month of gestation for pregnancy induced hypertension or before pregnancy/before 20 weeks of gestation for chronic hypertension. Hypertensive disorders of pregnancy (HDP) refers to categories of conditions characterized by elevated blood pressure and classified as chronic hypertension (of any cause diagnosed before 20 weeks of gestation), gestational hypertension, chronic hypertension with superimposed preeclampsia, preeclampsia and eclampsia [1]. HDP is one of the most common complications in pregnancy forming a triad together with hemorrhage and infection. It affects about 10% of pregnancies [2-4] and contributes for a significant maternal and perinatal mortality [5]. The World Health Organization (WHO) reported that 14.0% of global maternal deaths are attributed to HDP [6]. In Latin-American and Caribbean countries 25.7% of maternal deaths were due to HDP; in Asian and African countries, it contributed to 9.1% of maternal deaths and in fact about 16% in sub-Saharan African countries [5, 6]. HDP is a global public health concern both in developed and developing countries. However, the risk that a woman in a developing country will die of the complications of HDP is approximately 300 times higher than that for a woman in a developed country. A woman who develops pre-eclampsia is three times more likely to progress to eclampsia and if eclampsia is developed, it is up to 14 times more likely to die of eclampsia [7].
HTN raises the risk of heart attacks, cardiac failure, cerebrovascular accidents and renal failure in the mothers. The fetuses of hypertensive mothers are also at increased risks, such as: inappropriate placental oxygen transfer, Intra Uterine Growth Restriction (IUGR), premature delivery, placental abruption, stillbirth, and neonatal death [8]. In Cameroon, HDP occurs in 7.7–8.2% of pregnancies and causes 17.5% (89) of maternal deaths. Egbe et al., [9] showed that 14.5% of maternal deaths in Mezam Division were due to HDP, while in another study by Halle-Ekane et al., [10] in Fako Division, the prevalence of PIH was 5.02% and Maternal Mortality was 1887/100,000 live birth. Cognizant that the disease has no peculiar cause, several studies focusing on risk factors have been conducted in different parts of the globe and identified various risk factors for HDP. These factors can be grouped into modifiable and non-modifiable risk factors [11, 12]. The non-modifiable risk factors are traits or characteristics in the individual that cannot be changed or modified, hence they are out of our control and little or nothing can be done to change them. These factors include age, sex, race, gravidity, preexisting hypertension, family history, genetic composition [13-16] etc. On the other hand, the modifiable risk factors are attributes, characteristics, exposures or life style patterns that can be adjusted or changed to avoid the development of the disease. The modifiable risk factors include; obesity, excessive salt intake, sedentary lifestyle/physical inactivity, high fat diet, tobacco use, alcohol consumption, caffeine [14, 15, 17] etc. Smoking, alcohol intake, poor physical activity and insufficient fruit and vegetable intake are well-known modifiable/behavioral risk factors of HDP [18, 19] Addressing this risk factors is important not just for reducing adverse pregnancy outcomes such as low birth weight, fetal and infant mortalities, and potential congenital defects, but for decrease of HDP among mothers as well [18, 19]. In Nepal, a 2013 non-communicable disease risk factor survey (STEPS) revealed national prevalence of tobacco use to be 30.8%, alcohol intake of 17.4%, insufficient fruit and vegetable intake of 98.9%, and poor physical activity of 3.5% [20]. In addition, a higher percentage (15%) of pregnant women reported to consume alcohol during pregnancy [21]. However, studies of such have been limited in terms of describing the prevalence of behavioral risk factors and their predictors in Mezam division. Identification of the modifiable risk factors of HDP among pregnant women is an important global public health issue, but very limited studies have sought to identify behavioral risk factors of HDP in pregnant women residing in rural settings. In this study, we aimed to determine the prevalence and to identify socio-demographic determinants of behavioral risk factors of HDP among pregnant women living in rural southern Mezam division.
2. Materials and Methods
2.1 Study area/settings
The study was conducted in Mezam Division, NWR of Cameroon. The division is made up of five health districts of which three (Bamenda, Tubah and Santa) were selected for the study. A total of ten facilities were purposefully selected for the study: Bamenda district (Regional hospital, Mulang HC, CMA Nkwen, Azire IHC, St. Blaise and St. Mary Hospital), Santa district (Santa DH and Akum IHC) and Tubah district (Tubah DH and CMA Bambili). The purposeful selection was due to the crisis situation in the NWR. The Division covers an area of 1745 km2, population size of 575, 312 and its elevation above sea level is 1332m (North West Regional Delegation of Public Health, 2014). Bamenda city is the administrative seat of Mezam Division and Regional Headquarters of the North West and the largest town in the North West Region. It is a cosmopolitan city which is the bedrock of Cameroon politics with a very strong traditional set up and extremely powerful Fons and Fondoms. The population though cosmopolitan has a greater majority of local ethnic groups, practice subsistence farming and can boast of a rich variety of food crops. The works of art of the local population is worth emulating as it projects the North West culture nationally and internationally. The colourfully designed traditional attire worn by both men and women depicts a culture which has stood the test of time. The town is endowed with touristic potentials owing to its landscape characterized by waterfalls, craters and traditional palaces.
2.2 Study period
Data was collected for a period of four months i.e. April-July, 2018.
2.3 Study design and population
Health facility based cross-sectional study design with quantitative data collection method was used. All women who attending ANC services in the selected facilities were considered as source population whereas all sampled women were considered as study population.
2.4 Inclusion and exclusion criteria
Living and attending ANC within the study area (Mezam Division), women of reproductive age (12-49 yrs.) and be of gestational age ≥36 weeks were included in the study. Pregnant women with impaired sight/hearing and those with severe mental retardation were excluded from the study.
2.5 Sample size and sampling technique
The sample size for the cross-sectional study was calculated by using a single population proportion sample size calculation formula by considering the prevalence (P) of 8.2% [14]. Because the prevalence is < 10%, the margin of error (precision) will be 8.2%/5= 1.64% [22] at 95% confidence interval (CI). By considering 10% none response rate and an effect size of 1.5, the final sample size will be calculated as follows;
n= Z P (1-P)/d2 [23]
n = Number of participants for the study,
Z= 1.96 at 95% confidence interval.
P= 8.2% = 0.082,
d = 8.2/5 (because sample size is <10%) = 1.64%,
q= 1- p = 1-0.082 = 0.918
n= (1.96)2×0.082× (1-0.082)/ (0.0164)2 = 1,075.2 ≈ 1,076. Considering 10% of 1,076= 107. 6
Therefore 1,076 + 107.6 = 1184. However, the final sample size reached was 1210. The total sample size was proportionally allocated to the ten facilities based on their source population. The source population of each hospital was taken from the Regional Delegation of Health, NWR. Afterwards, the studies participants were consecutively selected from each hospital until the required sample size was reached.
2.6 Data collection
The questions were grouped/arranged according to particular objectives that the study aimed to address. Six data
collectors’ midwives in qualification (fluent in pidgin and English) were recruited to collect data for the study. Preliminary questionnaires were checked, pre-tested in two facilities and necessary modifications made. The questionnaire consisted of two parts: i) baseline characteristics of respondents; and ii) behavioral risk factors of hypertension (tobacco use, alcohol consumption, fruit and vegetable consumption, stress, salt intake, overweight/obesity and physical activity). The participants were asked whether/not they smoke, drank, consumed much salts, frequency of fruits and vegetable consumed (show cards regarding fruits and vegetables intake during pregnancy was used), participants were asked whether/not they were stressed during pregnancy, height and weight were captured from participants ANC cards. The World Health Organization (WHO) global recommendations on PA for health guideline to measure PA level among pregnant women in this study [24]. Activities considered during PA were; farming, walking, jogging, home activities, time spent in walking/sitting at job side. The Compendium of PA was used to calculate total MET/min/week spent (Compendium of PA, 2005). Data was collected by face-to-face interviews of respondents for about 25 minutes. Before data collection, the interviewers were trained for two days on the tools to be used, purpose of the study and how to approach respondents and obtain consent.
2.7 Data management and analysis
Two data supervisors were recruited to monitor data collection daily. The questionnaires were checked for completeness and correctness. The collected data was entered into Excel, cross-checked for accuracy and validity. The computer in which the data was entered was pass worded. Unique Identification Codes (UIC) was used to ensure participants confidentiality. Data was analysed using Statistical Package for Social Sciences (SPSS) version 21 (IBM - SPSS, Inc, Chicago, IL, USA) and Epi INFO version 7 (CDC). Frequencies, proportions and percentages were used for descriptive analysis. Multivariate logistic regression was used to determine the adjusted associations between the demographic variables and the behavioural risk factors of hypertension. It was determined a priori that explanatory variables with p-value ≤ 0.2 were considered for multivariate analysis. Variables with p-value < 0.5 were considered significant for the behavioural risk factors.
2.8 Ethical clearance
Ethical clearance for the study was obtained from the Faculty of Health Sciences institutional review board FHSIRB), university of Buea. In addition, administrative authorizations were obtained from the Regional Delegation of Public Health (RDPH), North West Region (NWR) and the District Health Officers (DHOs) of the Bamenda Health District (BHD), Santa Health District (SHD) and Tubah Health District (THD) and from Directors of all the health facilities included in the study. Potential respondents gave verbal and written/signed consent after they had been given an explicit explanation of the study and an opportunity to ask and respond to any questions. Ascent was also sort from participants less than 18 years old. The questionnaires were anonymous and answers were kept confidential.
3. Results
3.1 Socio-demographic characteristics of participants
Table 1 presents socio-demographic characteristics of 1210 participants who took part in the study. The mean and standard deviation (SD) age of participants was 26.9 (5.41). From the table, over a third of the pregnant women 455 (37.6%) & 390 (32.3%) were between ages ≤24 years & 25-29 years old respectively. The mean/standard deviation (SD) age of participants was 26.9 (5.4) respectively. A fifth 252 (21.6%) of the respondents were single while close to four fifth were 949 (78.4%) were married/cohabiting. Majority of the participants 422 (34.9%) had attended secondary education while an almost equal proportion, 292 (24.1%) and 286 (23.6%) had attended High School and Tertiary education respectively. As concerns occupation, students made up a quarter of the population 321 (26.5%), 210 (17.4%) were peasant/housewives and just 274 (22.5%) were employed or doing large scale business. Residence of Bamenda District were the most represented (69.2%), followed by Santa District (17.0%). Regarding religion, close to three quarter 878 (72.6%) were Catholic/Protestant and almost a quarter 263 (21.7%) Pentecostal (Table 1).
3.2 Prevalence of behavioural risk factors of HDP
As concern lifestyle factors, less than a percent (0.8%) of the pregnant women were smokers. Over a fifth 269 (21.6%) of the pregnant women were consumers of alcohol. As concerns physical activity, close to half 595 (49.2%) the pregnant women were less active with (<600 MET/min/week). For fruits and vegetable intake, four/fifth 976 (80.7%) of the pregnant women consumed <5 servings of fruits/vegetable daily. As concerns daily salt intake, over a fifth 150 (12.4%) of the pregnant women were high salt consumers. Close to half 590 (48.8%) the pregnant women reported been stressed during their pregnancy. Over two-third 798 (65.9%) of the participants were overweight/obsessed.
3.3 Clustering of behavioural risk factors of HDP
The figure 2a depicts the number of behavioural factors suffered by each participant. Eighteen (1.5%) of the pregnant women suffered none of the behavioural factors of HDP. A hundred and ten (9.1%) of the pregnant women suffered just one of the risk factors. Three hundred and thirty-one (27.4%) of the pregnant women suffered two risk factors. Four hundred and fifty-seven percent (37.8%) of the pregnant women had at least three of the risk factors. Two hundred and thirty-nine (19.8%), 49 (4.0%) and 6 (0.5%) of the pregnant women respectively suffered 4, 5 and 6 of the risk factors.
3.4 Socio-demographic predictors of behavioural risk factors of HDP
A number of socio-demographic variables were matched with behavioural risk factors of HDP and the results obtain reveals a number of demographic variables that proves as determinant of behavioural risk factors of HDP (Table 2). For determinants of smoking, participants of ages 25-29 years and those ≥35 years were 1.6 and 2.3 times respectively more likely to smoke compared to those ≤24 years and results were significant. Participants with secondary, high and tertiary level of education were 0.6, 0.5 and 0.3 times respectively less likely to pick up smoking compared to those with none/primary education and the results were all significant. For occupation, students and petit traders were 1.9 and 2.1 times respectively more likely to smoke than of the employed and housewife, though the results were not significant. Lastly, women who were Muslims/non-church goers were 3.4 times more likely to smoke than church goers though the results were not significant. For determinants of alcohol consumption, pregnant women between 25-29 years and those ≥35 years were 1.5 and 2.5 times respectively more likely to take alcohol than those of other age groups and the results were significant.
For education, women who had attended high school and tertiary education were respectively 0.5 and 0.2 times less likely drinkers of alcohol compared to those of secondary level and the non-school goers/primary level and the results were significant. For occupation, women who were employed were 0.5 times less likely to consume alcohol compared to other groups and the result significant. Considering religion, Pentecostal Christians were 0.4 times less likely to consume alcohol compared to the Catholics/Protestants and the Muslims/non-Church goers and result was significant. For determinant of less physical activity (<600MET/min/week), none of the age groups were significantly active as regards exercise. Pregnant women who were single/separated were 0.6 times less likely to exercise compared to married/cohabiting mothers and the result was significant. Women of secondary and high school 1.4 and 1.5 times respectively more active compared to women of primary/tertiary levels and the results were significant. For occupation and religion, none of the groups were significantly active.
For determinants of fruit/vegetable consumption, pregnant women of 30-34 years were 1.4 times better consumers of fruits/vegetables compare to the other age groups though not significant. There wasn’t any difference in fruits/vegetable consumption between single and married groups. As regards educational status, pregnant women of high and tertiary level were 1.7 and 1.9 times respectively better consumers of fruits/vegetables compared to the primary and secondary levels and results were significant. There was no difference in fruits/vegetable consumption among religious group. For determinants of high salt consumption, older participants were less consumers of salt compared to those ≤24 years though just those of ages 25-29 years was significant. For marital and educational status, there was no significant relationship with salt consumption. As for occupation, pregnant women who were student and those doing petit trading were 0.4 and 0.6 times respectively less consumers of salt compared to the housewife and employed and results were significant. For religion, Pentecostal Christians were 1.8 times higher consumers of salt compared to Catholics/Protestants or Muslims/non-church goers and results were significant. For determinants of stress, there is no significant relationship for age group and marital status.
As concerns educational status, pregnant women of secondary, high school and tertiary level were 1.8, 2.4 and 2.8 times respectively more stressed compared to non-school goers/primary school leavers and all were significant. With regards to occupation, petit traders and employed pregnant women were 1.8 and 1.9 times respectively more stressed than housewives and students and the results were significant. For determinants of overweight/obesity, older pregnant women of ages 25-29, 30-34 and ≥35 years were respectively 1.6, 2.6 and 2.5 times of higher BMI (>25 kg/m2) compared to the younger group (≤24 years) and the results were highly significant. For overweight/obese and educational level, pregnant women of secondary, high and tertiary level of education were respectively 0.7, 0.8 and 0.7 times of lower BMI (<25 kg/m2) compared to those of none/primary level of education though only participants of secondary level was significant.
|
Variables |
Respondent |
Percentages (%) |
||
|
Age of participants (years) Mean (SD) ≤24 25-29 30-34 ≥35 |
26.9 (5.4) 455 390 240 125 |
37.6 32.3 19.8 10.3 |
||
|
Marital Status Married/cohabiting Single/separated |
949 261 |
78.4 21.6 |
||
|
Educational Status None/Primary Secondary High school Tertiary |
210 422 292 286 |
17.4 34.9 24.1 23.6 |
||
|
Occupation Peasant/Housewife Student Petit Trader Large scale business/Employed |
210 321 407 272 |
17.4 26.5 33.6 22.5 |
||
|
Area/District of Residence Bamenda district Santa District Tubah District |
873 206 131 |
69.2 17.0 13.8 |
||
|
Religion Catholic/Protestants Pentecostal Muslims |
878 263 69 |
72.6 21.7 5.7 |
||
Table 1: Socio-demographic characteristics of pregnant women (n=1210).
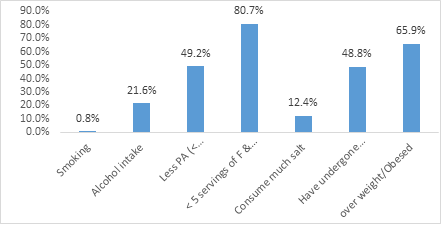
Figure 1: Prevalence of poor behavioural factors of HDP.
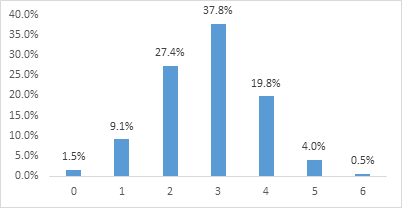
Figure 2a: Clustering of behavioural factors of HDP.
Alco= Alcohol; FV= Fruits/Vegetable < 5 servings/day; PA= Physical Activity < 600MET/Min/Week; Smo= Smoking
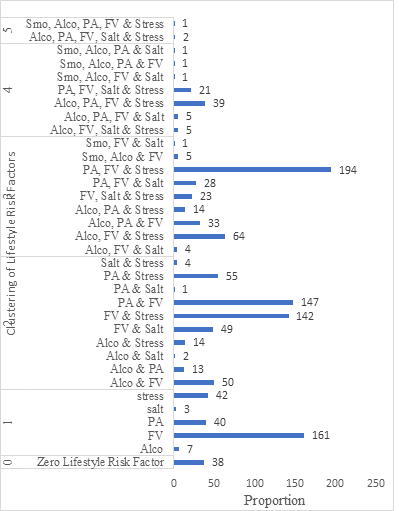
Figure 2b: Proportion of Clustered Risk factors of HDP.
|
Variables |
Smoking |
Alcohol |
Exercise |
Fruit/ Vegetable |
Salt |
Stress |
Obesity |
|
Age range (years) ≤24 25-29 30-34 ≥35 |
1 1.6(1.1-2.4)* 1.2(0.8-1.9) 2.3(1.4-3.6)* |
1 1.5(1.1-2.2)* 1.4(0.9-2.1) 2.5(1.5-4.1)* |
1 1.1(0.8-1.4) 1.1(0.8-1.6) 0.8(0.5-1.3) |
1 1.1(0.8-1.6) 1.4(0.9-2.1) 1.1(0.6-1.8) |
1 0.5(0.3-0.8)* 0.7(0.5-1.2) 0.5(0.3-0.9) |
1 1.2(0.9-1.6) 0.9(0.7-1.4) 1.2(0.8-1.8) |
1 1.6(1.2-2.2)* 2.6(1.8-3.9)* 2.5(1.5-4.1)* |
|
Marital Status Married/Cohabit Single/separated |
1 1.2(0.8-1.7) |
1 1.2(0.8-1.8) |
1 1.6(1.1-2.8)* |
1 1.1(0.8-1.6) |
1 0.8(0.5-1.3) |
1 1.2(0.9-1.6) |
1 0.9(0.7-1.4) |
|
Educational Status None/Primary Secondary High school Tertiary |
1 0.6(0.5-0.9)* 0.5(0.3-0.7)* 0.3(0.2-0.5)* |
1 0.8(0.5-1.2) 0.5(0.3-0.8)* 0.24(0.2-0.7)* |
1 1.3(0.9-1.8) 1.6(1.1-2.3)* 2.3(1.5-3.6)* |
1 0.9(0.6-1.6) 1.7(1.2-2.8)* 1.9(1.1-3.4)* |
1 1.2(0.7-1.9) 0.9(0.6-1.7) 1.3(0.7-2.2) |
1 1.8(1.3-2.6)* 2.4(1.6-3.6)* 0.8(0.4-1.8) |
1 0.7(0.4-0.9)* 0.8(0.5-1.2) 0.7(0.5-1.2) |
|
Occupation Peasant/ Housewife Student Petit Trading Employed |
1 1.9(0.2-17.7) 2.1(0.2-19.4) 1.1(0.1-18.5) |
1 0.8(0.5-1.3) 0.7(0.5-1.1) 0.5(0.3-0.9)* |
1 1.1(0.7-1.7) 1.1(0.8-1.6) 0.8(0.5-1.4) |
1 1.1(0.6-1.7) 1.1(0.7-1.7) 1.2(0.7-2.1) |
1 0.4(0.2-0.7)* 0.6(0.4-0.9)* 1.1(0.7-1.8) |
1 1.4(0.9-2.1) 1.8(1.4-2.8)* 1.9(1.3-2.9)* |
1 0.8(0.5-1.2) 1.3(0.9-1.8) 1.1(0.7-1.7) |
|
Religion Catholic/ Protestant Pentecostal Muslim/others |
1 0.5(0.1-4.3) 3.4(0.7-17.4) |
1 0.4(0.3-0.6)* 0.5(0.3-0.6) |
1 1.2(0.9-1.6) 0.8(0.5-1.4) |
1 1.0(0.7-1.4) 1.9(1.1-3.5)* |
1 1.8(1.1-3.1)* 1.4(0.7-2.8) |
1 1.2(0.8-1.5) 1.3(0.8-2.1) |
1 1.1(0.8-1.5) 0.8(0.5-1.3) |
Table 2: Determinants of lifestyle factors of hypertension.
4. Discussion
This study provides the prevalence and socio-demographic determinants of some major behavioral risk factors of HDP: insufficient fruit and vegetable consumption, insufficient physical activity, use of tobacco, overweight/obesity, stress, excess salt intake and harmful alcohol consumption by pregnant women. We noted this as the first ever study from Mezam division that focused on predicting behavioral risk factors of hypertension in pregnancy among pregnant women. Our study revealed that, less than a percent (0.8%) of pregnant women continued smoking during pregnancy. The result is by far lower than what was reported in England and Wales, 15% [25], in Romania and in Australia 14% [26, 27] and in the United States 13% [28]. These disproportions in prevalence levels might be due to differences between geographic locations or due to hospital setting designs [25]. Furthermore, it should be understood that the true prevalence of smoking during pregnancy may be difficult to determine, because of possible underreporting in studies that depend on self-reporting [25]. Additionally, sociocultural norms discouraging smoking may lead women to fail to reveal their true smoking status during pregnancy. This risky behavior (smoking) contributes significantly towards worsening of maternal and child health, especially among those living with socioeconomic difficulties. Earlier studies have reported that women who continued smoking throughout pregnancy had about twice the risk of adverse pregnancy outcomes, compared to those who don’t smoke or reduced their smoking habit [29]. In the current study, pregnant women of older ages were respectively 1.6 and 2.3 times more likely to take up smoking than the younger age group (≤24 years). This is contrary to studies reported by Sao Paulo, [25] who stated that smoking was more prevalent among the younger age group (≤24 years). This may be explained by the marketing strategies/campaigns designed to target younger age group, forgetting the older ones. Still in our study, it was established that women for whom primary education had twice as much risk of smoking. This is in line with findings reported by Sao Paulo, [25].
The current study established a prevalence of 21.6% alcohol use among pregnant women. This seems to be higher than the proportion reported by Matunga Mpelo et al., [30] in Tanzania and equal to the proportion of 21.5% reported in Northern Tanzania in year 2010 and lower than the observation from other African studies where the prevalence ranged from 30% to 59.2% [31] but rather higher than the prevalence reported in developed countries such as Sweden and Canada with prevalence of 12% and 10.8%, respectively [32, 33]. The difference on the prevalence between this study and the study from Northern Tanzania and Nigeria may partly be explained by the differences in methodology, as this was a cross-sectional design which interviewed women from the antenatal clinic while the Northern Tanzania study was a registry-based study. Furthermore, less than secondary education was identified in this study as a strong determinant of alcohol use during pregnancy. This may be explained by the probable impact of ignorance/poor education on risky lifestyles including alcohol consumption. It is probable that the less educated are less likely to understand the effect of alcohol during pregnancy and hence more likely to consume alcohol during pregnancy. They may equally not be able to read the available information about alcohol in pregnancy and are more likely to have harder social situations with less finances, and family struggles. All the above could make them resort to alcohol as a means of escape [31]. As concerns age and alcohol consumption, older women were respectively 1.5 and 2.5 more likely to drink during pregnancy compared with those aged 24 years or younger. Palma et al. [34] found that it was more common for older study participants to consume alcohol, implying that they are more likely than younger women to have developed drinking habits that may be difficult to break during pregnancy. In addition, participants who were employed were less likely to consume alcohol compared to other groups. The result is contrary to that reported by Alvik et al., [35] who found that higher income was a predictor for drinking. This could be explained by the fact that the employed women have little or no time for themselves and thus are so busy with work and home activities in spite that they have the money. For religious status, Pentecostals were less likely to consume alcohol compared to their counterparts. Alvik et al., [35] reported similar results.
Pentecostals preach against alcohol and this could be the reason of low alcohol consumption. The prevalence of physical inactivity during pregnancy was 49.2%. This is by far higher than the 15% reported in the USA regarding the level of physical activity among pregnant women [36]. This could result from the general belief among grass root women that physical activity leads to miscarriage, poor fetal growth or premature delivery. A systematic review on physical activity during pregnancy suggests that some light-to-moderate physical activity is protective for maternal and child health outcomes such as pre-eclampsia, gestational hypertension and premature birth [19]. The determinants of PA in the current study were marital status and education. Married women were 1.6 times more likely to engage more likely to engage in PA than the married women. To explain, pregnant women who were single were better engaged in PA as compared to those who were married. The result is similar to that of Oliveira-Brochado et al. [37], who also showed that single women are more physically active than married women. This could imply that majority of unmarried persons are younger, had a smaller number of dependents, and had more time to spend on recreation and leisure. To add, married women engaged less in PA possibly owing to their domestic role, relative physical weakness and unfavorable conditions of pregnancy and childbirth. The analysis also reveals that PA increases with education. Pregnant women with high school and tertiary education were 1.6 and 2.3 times more likely to exercise than the less educated. With respect to the argument of Oliveira-Brochado et al., [37] it is thus likely that knowledge of the importance of PA increases with education. This probably indicate that the higher the level of education, the better ones PA level. School education increases the mother’s knowledge on healthy living habits as these are included in standard school curricula. Also, educated mothers are more likely to read comprehensibly and understand health education messages presented in mass media and through other methods more than the less-educated ones.
The most common behavioral risk factor of hypertension in pregnant women is the insufficient intake of fruits and vegetables, which concurs with the findings from other surveys [19]. In this study, average daily intake of <5 servings of FAVs combined was considered to be low and gave a prevalence of 80.7%. This result is similar to the 82.8% of low FAVs intake reported by Singh et al., [38]. Equally similar to the 82% report in Tanzania by Hall et al., [39]. This finding seems to indicate a deterioration in FAV consumption in Mezam division, which may be attributed a nutrition transition dominated by bought and processed foods reducing FAV consumption. The insufficient fruits and vegetables consumption among pregnant women could equally be attributed to poverty that might have caused women not to afford adequate amount of fruits and vegetables. The other studies also supported that low-income level prevents from consuming adequate fruits and vegetables [19]. Another possible reason for less intake of fruits and vegetables during pregnancy could be associated with seasonal and geographical availability of such foods as well as the crisis rocking the North West and South West Regions. Similar reasons were advanced by Paudel et al., [19] Cultural practices to avoid some foods during pregnancy is prevalent due to the belief that pregnancy is considered a delicate state and as such may lead to low FAV consumption [19]. The logistic regression analysis showed that higher educational attainment is associated with higher FAV consumption. Educated pregnant women were respectively 1.7- and 1.9-times better consumers of FAV combined. This study was in agreement with other studies which shows that higher educational attainment is accompanied with better dietary knowledge and skills to implement eating fruits [39]. Several explanations have been used in the literature to justify the disparities in FAV consumption by socio-demographic characteristics [38]. This finding calls for urgent health promotion to increase awareness of the importance of FAV consumption during pregnancy.
The prevalence of stress in the current study was 48.9%. A study in India reported 42% which is slightly lower than the findings of our study [40] while a study in South Africa reported 45% [41]. Rahman, in Pakistan reported a prevalence of antenatal depression of 24% [42]. A study in Malawi showed a prevalence of 21% [43] while in Nigeria, a low prevalence, 9%, was observed in a study by Adewuya et al. [44]. The reason for the high prevalence might be that most pregnant women with stress/depression are not aware their state, and when some complain to their doctors about symptoms of antenatal depression, the doctors could mistake the symptoms for other medical conditions. In this study, higher educational attainment had an impact on stress among pregnant women. Women whom had attained above primary education were more stressed during their pregnancy. The inverse relation of literacy and stressed in this study was similar to findings of other studies [45, 46]. This could be as a result of the fact that highly educated individuals are more sensitive to the symptoms of stress disorders and can easily report them and are not embarrassed about admitting pregnancy related anxiety and depression symptoms. Furthermore, the study revealed that occupation has an impact on stress among pregnant women. Pregnant women who are employed/petit traders were likely to be stressed than the peasants/students. The study by Bloom et al., [47] revealed similar results. This could be explained by the fact that employed women face more social and financial dependence which keeps their minds at all-time working to satisfy family, friends as well as meet up with their financial status. In contrast, other studies from around the world have indicated employment as protective factor [41, 43]. This can be substantiated by the fact that employed women have financial autonomy and are thus able to manage other financial related stress better than their unemployed counterparts.
The results of this study showed that the overall prevalence of overweight and obesity was 65.9% which is higher than the prevalence in most African Universities [48]. In Egyptian tertiary institutions overweight and obesity was found to be 21%, South Africa 19.4%, Namibia 12.3%. The African countries with the least prevalence of overweight and obesity were Ivory Coast and Madagascar with 2.9% and 1.3% respectively [48]. The prevalence was equally higher than 36.8% reported by Tapera et al., [49]. However, the prevalence was lower than the 76% and 77% reported among the Fijian women in 2004 and 2017 respectively [50]. The high prevalence in overweight and obesity among pregnant women in Mezam could be due to their feeding habit as a majority feed on junk food during pregnancy. Low level of physical activity as well as no pre-pregnancy counseling could result to the high over weight/obese pregnant women. Despite our efforts to explore this important topic in our context, the study should be evaluated in the light of some limitations. First, the information we collected was obtained by self-reporting, and thus, is subject to informant bias. Second, our study did not consider information about nutritional status of pregnant women (e.g. under or over nutrition status by body mass index) which is considered to be one of the major factors of understanding these risk factors [50]. Third, because the study was performed in Mezam division, its findings may not be generalizable for the entire region and Cameroon at large. Thus, it would be imperative to conduct similar types of studies in the other divisions of the NWR, taking into consideration the limitations of this study.
5. Conclusions
There was poor uptake of modifiable factors of HTN with smoking (0.8%), alcohol (21.6%), physical inactivity
(49.2%), low fruits and vegetables intake (80.7%), high salt intake (12.4%), stress (48.8%) and overweight/obesity (65.5%). Sixty-two-point one percent of the pregnant women suffered three or more risk factors at a time. For determinants, Age is a significant determinant of smoking, alcohol intake and obesity. Education and occupation were significant determinant of stress and Religion a major determinant of high salt intake. These factors call for immediate health promotion interventions such as community awareness and appropriate antenatal counseling.
Recommendations
Based on the findings of the study, the following can be recommended:
- More sensitization to women of reproductive age should be carried out in Mezam Division so as to increase awareness of behavioural risk factors of hypertension in pregnancy.
- There is need to address the socio-demographic factors that influence behavioural factors of hypertension in pregnancy.
- Community sensitization of pre-pregnancy counselling on the risk and consequences of behavioural risk factors of hypertension should be enhanced.
- Technical support and supervisory visits to all the health facilities in the division/region should be strengthened to ensure adequate health education on the risk and consequences of behavioural risk factors of hypertension in pregnancy.
Acknowledgements
The authors acknowledge the contribution of the pregnant women participated in the study. The authors also acknowledge the assistance of the Regional Delegate of Public health for North West Region, the District Health Officers and Directors of all facilities that were part of the study.
Conflict of Interest
We declare no conflict of interest.
References
- Cunnigham FG, Leveno K, Bloom Sea. Williams Obstetrics. Medical Publishing Division (2010).
- Ghulmiyyah L, Sibai B. Maternal mortality from preeclampsia/Eclampsia. Semin Perinatol 36 (2012): 56-59.
- WHO recommendations for prevention and treatment of pre-eclampsia and eclampsia. World Health Organization, Dept. of Reproductive Health and Research, Dept. of Maternal, Newborn, Child and Adolescent Health, Dept. of Nutrition for Health and Development (2011).
- World Health Organization, UNICEF, UNFPA. Trends in maternal mortality 1990-2008: Estimates developed by WHO, UNICEF, UNFPA and the World Bank. 2010. World Health Organization (2012).
- Khan KS, Wojdyla D, Say L, et al. WHO analysis of causes of maternal death: a systematic review. Lancet 367 (2006): 1066-1074.
- Say L, Chou D, Gemmill A, et al. Global causes of maternal death: a WHO systematic analysis. Lancet Glob Health 2 (2014): 323-333.
- York N. EngenderHealth, Balancing the Scales Expanding Treatment for Pregnant Women with Life-Threatening Hypertensive Conditions in Developing Countries A Report on Barriers and Solutions to Treat Pre-eclampsia & Eclampsia (2007).
- Khosravi S, Dabiran S, Lotfi M, et al. Study of the Prevalence of Hypertension and Complications of Hypertensive Disorders in Pregnancy. Open Journal of Preventive Medicine 4 (2014): 860-867.
- Egbe TO, Therence ND, Gregory EH-E, et al. Determinants of Maternal Mortality in Mezam Division in the North West Region of Cameroon: A Community-based Case Control Study. International journal of tropical disease and health 15 (2016): 1-15.
- Halle-Ekane GE, Che M, Atashili J, et al. Severe Pregnancy Induced Hypertension, a Dreaded Complication in Semi-Urban Area in Fako Division, Cameroon? A Case-Control Study. British Journal of Medicine & Medical Research 12 (2016): 1-9.
- Ahmed NU, Rahman M, Islam MD, et al. Socio-demographic clinical characteristics and status of hypertension control among rural hypertensive patients. Faridpur Medical College Journal 6 (2011): 5-9.
- Mayega RW, Makumbi F, Rutebemberwa E, et al. Modifiable socio-behavioural factors associated with overweight and hypertension among persons aged 35 to 60 years in eastern Uganda. PLoS one 7 (2012): 231-238.
- Leeners B, Neumaier-Wagner PK, Irawan S, et al. Family stability during childhood and risk to develop hypertensive diseases in Journal of Obstetrics and Gynaecology 82 (2006): 441-446.
- Mbouemboue OP, Diallo C, Marcel TT, et al. A Study on Factors Related to Hypertensive Disorders in Pregnancy in Ngaoundere (Adamawa Region, Cameroon). Clinical Medicine Research 5 (2016): 6-12.
- Tebeu PM, Foumane P, Fosso G, et al. Risk Factors for Hypertensive Disorders in Pregnancy: A Report from the Maroua Regional Hospital, Cameroon. J Reprod Infertil 12 (2011): 227-234.
- Wiysonge SCU, Ngu BK, Mbuagbaw JN. Risk factors and complications of hypertension in Yaounde, Cameroon. Cardiovascular Journal of South Africa 15 (2004): 215-219.
- O'Brien TE, Ray JG, Chan WS. Maternal body mass index and the risk of pre-eclamsia: a systematic overview. Epidemiology 14 (2009): 368-374.
- Global health risks: mortality and burden of disease attributable to selected major risks. Geneva: World Health Organization (2009).
- Paudel R, Kwan L, Jitendra KS, et al. Prevalence of behavioral risk factors of cardiovascular diseases and associated socio-economic factors among pregnant women in a rural area in Southern Nepal. BMC Pregnancy and Childbirth 18 (2018): 1-9.
- Aryal K NS, Mehata S, Vaidya A, et al. Non communicable diseases risk factors: STEPS Survey Nepal 2013. Kathmandu: Nepal Health Research Council (2014).
- Niraul S, Jha N, Shyangwa P. Alcohol consumption among women in a district of Eastern Nepal. Health Renaiss 11 (2014): 205-212.
- Pourhoseingholi MA, Vahedi M, Rahimzadeh M. Sample size calculation in medical studies Gastroenterol Hepatol Bed Bench 6 (2013): 14-17
- Naing L, Winn T, Rusli B. Practical issues in calculating the sample size for prevalence studies. Arch Orofac Sci 1 (2006): 9-14.
- Obesity: preventing and managing the global epidemic. Geneva (2000).
- Sao P. Association of risk factors with smoking during pregnancy among women of childbearing age: an epidemiological field study in Turkey. Med J 135 (2017): 100-106.
- Meghea CI, Rus D, Rus IA, et al. Smoking during pregnancy and associated risk factors in a sample of Romanian women. Eur J Public Health 22 (2012): 229-233.
- Seybold DJ, Broce M, Siegel E, et al. Smoking in pregnancy in West Virginia: does cessation/reduction improve perinatal outcomes?. Matern Child Health J 16 (2012): 133-138.
- Tong VT, Jones JR, Dietz PMea. Trends in smoking before, during, and after pregnancy – Pregnancy Risk Assessment Monitoring System (PRAMS), United States, 31 sites, 2000-2005. MMWR Surveill Summ 58 (2009): 1-29.
- Uncu Y, Ozcakir A, Ecran I, et al. Pregnant women quit smoking; what about fathers? Survey study in Bursa Region, Turkey. Croat Med J 46 (2005): 832-837.
- Matunga M, Stephen MK, Fabiola M, et al. Prevalence and Factors Influencing Alcohol Use in Pregnancy among Women Attending Antenatal Care in Dodoma Region, Tanzania: A Cross-Sectional Study. Journal of Pregnancy 8 (2018): 1-7.
- Ordinioha B, Brisibe S. Alcohol consumption among pregnant women attending the ante-natal clinic of a tertiary hospital in South-South Nigeria. Nigerian Journal of Clinical Practice 18 (2015): 13-17.
- Comasco GH E, Helander A, Oreland L, et al. Alcohol consumption among pregnant women in a swedish sample and its efects on the newborn outcomes. Alcoholism: Clinical and Experimental Research 36 (2012): 1779-1786.
- Walker MJ, Al-Sahab B, Islam F, et al. The epidemiology of alcohol utilization during pregnancy: an analysis of the Canadian Maternity Experiences Survey (MES). BMC Pregnancy and Childbirth 11 (2011): 52-58.
- Palma S, Pardo-CrMariscal M, Perez-Iglesias R, et al. Weekday but not weekend alcohol consumption before pregnancy infuences alcohol cessation during pregnancy. European Journal of Public Health 17 (2007): 394-399.
- Alvik A, Haldorsen T, Groholt B, et al. Alcohol Consumption Before and During Pregnancy Comparing Concurrent and Retrospective Reports. Alcohol Clin Exp Res 30 (2006): 510-515.
- Macera CA, Ham SA, Yore MM, et al. Prevalence of physical activity in the United States: Behavioral Risk Factor Surveillance System 2001. Prev Chronic Dis 2 (2005): A17.
- Oliveira-Brochado A, Oliveira-Brochado F, Brito PQ. Effects of personal, social and environmental factors on physical activity behavior among adults. Actividade Física 27 (2010): 7-17.
- Jitendra KS, Dilaram ASG, Mandira A, et al. Socio-Demographic and Diet-Related Factors Associated with Insufficient Fruit and Vegetable Consumption among Adolescent Girls in Rural Communities of Southern Nepal. Int J Environ Res Public Health 16 (2019): 1-11.
- Hall JN, Moore S, Harper SB, et al. Global variability in fruit and vegetable consumption. Am J Prev Med 36 (2009): 403-409.
- Patel V, Rodrigues M, DeSouza N. Gender, poverty, and postnatal depression: a study of mothers in Goa, India. Am J Psychiatry 159 (2002): 43-47.
- Rochat TJ, Tomlinson M, Newel ML, et al. Detection of antenatal depression in rural HIV-affected populations with short and ultrashort versions of the Edinburgh Postnatal Depression Scale (EPDS). Archives of Women’s Mental Health 16 (2013): 401-410.
- Rahman A, Iqbal Z, Harrington R. Life events, social support and depression in childbirth: perspectives from a rural community in the developing world. Psychol Med 33 (2003): 1161-1167.
- Stewart RC, Umar E, Tomenson B, et al. A crosssectional study of antenatal depression and associated factors in Malawi. Archives of Women’s Mental Health 17 (2014): 145-154.
- Adewuya AO, Ola BA, Aloba OO, et al. Prevalence and correlates of depression in late pregnancy among Nigerian women. Depression and Anxiety 24 (2007): 15-21.
- Afusat OB. Prevalence and Associated Factors of Anxiety and Depression Among Pregnant Women. Open Access J Neurol Neurosurg 9 (2018): 1-10.
- Dunkel-Schetter. Stress processes in pregnancy and preterm birth. Curr Directions Psychol Sci 18 (2009): 205-209.
- Tina B, Nancy G, Mary AC, et al. Maternal stress exposures, reactions, and priorities for stress reduction among low-income urban women. J Midwifery Womens Health 58 (2013): 167-174.
- Peltzer K, Pengpid S, Samuels T, et al. Prevalence of overweight/obesity and its associated factors among university students from 22 Countries. International Journal of Environmental Research and Public Health 11 (2014): 7425-7441.
- Roy T, Marogwe TM, Tshephang T, et al. The prevalence and factors associated with overweight and obesity among University of Botswana students. Cogent Medicine 4 (2017): 1-11.
- Pushpa N, Masoud M, Susan P, et al. Prevalence and determinants of overweight and obesity among pregnant women attending thencolonial war memorial hospital (CWMH) antenatal clinic, Suva, Fiji Islands, 2015. MOJ Public Health 6 (2017): 367-371.
