Cranberry Extract for Preventing Recurrent Urinary Tract Infections: An Outcome-Specific Meta-Analysis of Prospective Trials
Article Information
Jana Havranova1, Steven Cardio1, Matthew Krinock1, Max Widawski1, Rachel Sluder1, Ashish Kumar3, John Hippen1, Harsh Goel1,2*
1Department of Medicine, St. Luke’s University Hospital, PA, USA
2Associate Professor of Medicine, Lewis Katz School of Medicine, Temple University School of Medicine, PA, USA
3Department of Medicine, Wellspan York Hospital, PA, USA
*Corresponding author: Harsh Goel, Department of Medicine, St Luke’s University Hospital, 801 Ostrum Street, EW-4, Bethlehem, PA, USA
Received: 11 July 2020; Accepted: 23 July 2020; Published: 05 August 2020
Citation:
Jana Havranova, Steven Cardio, Matthew Krinock, Max Widawski, Rachel Sluder, Ashish Kumar, John Hippen, Harsh Goel. Cranberry Extract for Preventing Recurrent Urinary Tract Infections: An Outcome-Specific Meta-Analysis of Prospective Trials. Journal of Women’s Health and Development 3 (2020): 222-242.
View / Download Pdf Share at FacebookAbstract
Background: Urinary tract infection (UTI) is one of the most common bacterial infections, representing enormous healthcare-cost and morbidity burdens. Though cranberries have long been used to prevent recurrent UTI (r-UTI), extant evidence is inconsistent. Potentials reasons for inconsistency include using different cranberry preparations (juice versus tablet/capsule), heterogenous populations, and outcome definition. Importantly, juice hardly a feasible long-term therapeutic option and there are no meta-analyses specifically examining tablet/capsule form of cranberry. We conducted an updated meta-analysis to address these inconsistencies.
Methods: MEDLINE was systematically searched for, i) placebo-controlled clinical trials, ii) restricted to adults, iii) exclusively investigating cranberry extract in tablet/capsule form to prevent r-UTI, and iv) clearly reporting incidence of any of the three outcomes (detailed subsequently) in treatment and placebo groups. Three outcome measures, i.e culture-confirmed UTI, asymptomatic pyuria/bacteriuria, and symptomatic UTI were meta-analyzed separately.
Results: 15 RCTs met inclusion criteria. Ten trials reported culture-confirmed UTI, seven reported asymptomatic pyuria/bacteriuria, and 5 trials reported symptomatic but not culture-confirmed UTI as primary outcome, yielding twelve (n=2391 subjects), ten (n=2565 urine cultures), and seven (n=1325 subjects) independent cohorts, respectively. Meta-analysis revealed a 30% reduced risk of culture-confirmed UTI (pooled RR 0.70, 95% CI 0.54, 0.91; I2=59%), 23% reduction in asymptomatic bacteriuria/pyuria (pooled RR 0.77, 95% CI 0.69, 0.86, I2=75%), and 14% reduction in symptomatic UTI (RR 0.86, 95% CI 0.75, 0.98; I2=48%). Excluding low-risk patients and those with neurogenic bladder having indwelling/intermittent catheterization reduced heterogeneity among culture-confirmed UTI trials revealing baseli
Keywords
Urinary tract infection
Urinary tract infection articles Urinary tract infection Research articles Urinary tract infection review articles Urinary tract infection PubMed articles Urinary tract infection PubMed Central articles Urinary tract infection 2023 articles Urinary tract infection 2024 articles Urinary tract infection Scopus articles Urinary tract infection impact factor journals Urinary tract infection Scopus journals Urinary tract infection PubMed journals Urinary tract infection medical journals Urinary tract infection free journals Urinary tract infection best journals Urinary tract infection top journals Urinary tract infection free medical journals Urinary tract infection famous journals Urinary tract infection Google Scholar indexed journals infections articles infections Research articles infections review articles infections PubMed articles infections PubMed Central articles infections 2023 articles infections 2024 articles infections Scopus articles infections impact factor journals infections Scopus journals infections PubMed journals infections medical journals infections free journals infections best journals infections top journals infections free medical journals infections famous journals infections Google Scholar indexed journals Meta-Analysis articles Meta-Analysis Research articles Meta-Analysis review articles Meta-Analysis PubMed articles Meta-Analysis PubMed Central articles Meta-Analysis 2023 articles Meta-Analysis 2024 articles Meta-Analysis Scopus articles Meta-Analysis impact factor journals Meta-Analysis Scopus journals Meta-Analysis PubMed journals Meta-Analysis medical journals Meta-Analysis free journals Meta-Analysis best journals Meta-Analysis top journals Meta-Analysis free medical journals Meta-Analysis famous journals Meta-Analysis Google Scholar indexed journals Prospective articles Prospective Research articles Prospective review articles Prospective PubMed articles Prospective PubMed Central articles Prospective 2023 articles Prospective 2024 articles Prospective Scopus articles Prospective impact factor journals Prospective Scopus journals Prospective PubMed journals Prospective medical journals Prospective free journals Prospective best journals Prospective top journals Prospective free medical journals Prospective famous journals Prospective Google Scholar indexed journals heterogeneity articles heterogeneity Research articles heterogeneity review articles heterogeneity PubMed articles heterogeneity PubMed Central articles heterogeneity 2023 articles heterogeneity 2024 articles heterogeneity Scopus articles heterogeneity impact factor journals heterogeneity Scopus journals heterogeneity PubMed journals heterogeneity medical journals heterogeneity free journals heterogeneity best journals heterogeneity top journals heterogeneity free medical journals heterogeneity famous journals heterogeneity Google Scholar indexed journals asymptomatic articles asymptomatic Research articles asymptomatic review articles asymptomatic PubMed articles asymptomatic PubMed Central articles asymptomatic 2023 articles asymptomatic 2024 articles asymptomatic Scopus articles asymptomatic impact factor journals asymptomatic Scopus journals asymptomatic PubMed journals asymptomatic medical journals asymptomatic free journals asymptomatic best journals asymptomatic top journals asymptomatic free medical journals asymptomatic famous journals asymptomatic Google Scholar indexed journals pyuria articles pyuria Research articles pyuria review articles pyuria PubMed articles pyuria PubMed Central articles pyuria 2023 articles pyuria 2024 articles pyuria Scopus articles pyuria impact factor journals pyuria Scopus journals pyuria PubMed journals pyuria medical journals pyuria free journals pyuria best journals pyuria top journals pyuria free medical journals pyuria famous journals pyuria Google Scholar indexed journals cystitis articles cystitis Research articles cystitis review articles cystitis PubMed articles cystitis PubMed Central articles cystitis 2023 articles cystitis 2024 articles cystitis Scopus articles cystitis impact factor journals cystitis Scopus journals cystitis PubMed journals cystitis medical journals cystitis free journals cystitis best journals cystitis top journals cystitis free medical journals cystitis famous journals cystitis Google Scholar indexed journals analyses articles analyses Research articles analyses review articles analyses PubMed articles analyses PubMed Central articles analyses 2023 articles analyses 2024 articles analyses Scopus articles analyses impact factor journals analyses Scopus journals analyses PubMed journals analyses medical journals analyses free journals analyses best journals analyses top journals analyses free medical journals analyses famous journals analyses Google Scholar indexed journals non-culture articles non-culture Research articles non-culture review articles non-culture PubMed articles non-culture PubMed Central articles non-culture 2023 articles non-culture 2024 articles non-culture Scopus articles non-culture impact factor journals non-culture Scopus journals non-culture PubMed journals non-culture medical journals non-culture free journals non-culture best journals non-culture top journals non-culture free medical journals non-culture famous journals non-culture Google Scholar indexed journals
Article Details
1. Introduction
Urinary tract infections (UTIs) are one of the most common bacterial infections, amounting to >5 million office visits, over 1.5 million emergency department visits, and almost 400,000 hospital admissions, costing ≈$2.8 billion in the US alone [1, 2]. Much more common in women, 50-60% of all women experience a UTI in their lifetime, with 20-30% of those suffering recurrence within 6 months [3]. On a population basis, incidence of recurrent UTI (r-UTI) increases from 100/100,000 women 18-34 years old to 189/100,000 women aged 55-64 years [4]. Undoubtedly, r-UTI represents a significant individual and societal burden, both in cost and morbidity. Though antibiotic prophylaxis against r-UTI is effective, it is costly, comes with risk of adverse events, and raises legitimate concerns regarding anti-microbial resistance among uropathogens [5]. Hence, non-antimicrobial prophylaxis against r-UTI remains a topic of avid research. Cranberries have been used for over a century to prevent and treat UTIs. Acidification of urine, based on increased urinary hippuric acid secretion was long thought to mediate this benefit. However, combined with increasing awareness of bacterial adhesion in pathogenesis of UTI, in vitro studies in the 1980s revealed that cranberries have significant anti-adhesion properties [6, 7] Recently, anti-adhesion properties of cranberries have been attributed to plant polyphenols called proanthocyanidins (PACs), present in high concentrations in cranberries [8–10]. Numerous clinical trials over the last two decades have investigated the impact of cranberries in preventing r-UTIs. Unfortunately, inconsistent findings have resulted in conflicting conclusions and recommendations [11]. Multiple meta-analyses have frustratingly also come to conflicting conclusions [5, 12-15]. The most recent comprehensive meta-analysis was performed almost a decade ago, and concluded cranberry juice to be ineffective in preventing r-UTIs [13]. Several prospective studies since have reported outcomes with cranberry extract in tablet/capsule form, hence the need for an updated analysis. Apart from inconsistent findings, admittedly due to heterogeneity among individual studies themselves, previous meta-analyses have suffered methodologic problems.
Firstly, all of them have included studies using cranberry in any form i.e juice or tablet, obtaining pooled outcomes with “cranberry product”. This is problematic because bioavailability of plant flavonoids including anthocyanins is significantly influenced by the specific food matrix and chemical/heat/mechanical processing [16]. Hence, bioactivity of cranberry extract may well differ significantly from cranberry juice, and pooling these two interventions would skew results of a meta-analysis if one is ineffective or significantly different in efficacy from the other. Distinguishing between efficacy of the two cranberry formulations is critical because juice, even if beneficial, is hardly a viable therapeutic option due to high cost, well-known unpalatability amply demonstrated by a high attrition rates in several trials [15, 17, 18] and adverse effects of excess fluid and/or caloric intake that inevitably accompanies juice consumption, for example in diabetics and those with hypervolemic states. Moreover, there is inherent bias in cranberry juice studies, since most of these have used “placebo” juice as control, either flavored water or other fruit juices. This is problematic since hydration per se reduces UTI risk, making these studies essentially comparisons of two interventions, rather than true-placebo controlled trials. Given expected benefit in “placebo” groups from hydration, benefit of cranberry juice is likely to be muted. On the other hand, cranberry tablet/capsule trials have used true inert placebo as comparator. Hence, pooling trials using the two formulations risks distorting pooled effects in a meta-analysis. A second major issue with previous meta-analyses is pooling studies measuring diverse outcomes, i.e culture-confirmed UTI, asymptomatic bacteriuria/pyuria, or symptoms alone. Obviously, UTI definition majorly impacts incidence, usually higher defined based on symptoms alone rather than confirmed by urine cultures. Obviously, variability in incidence due to differing definitions per se will result in variable effect sizes and risk introducing avoidable heterogeneity in a meta-analysis, not to mention impact on the direction of pooled results. The “gold-standard” of diagnosing a UTI is positive urine cultures in a patient with symptoms consistent with a UTI, making this specific outcome of primary importance. Though it could be argued that treatment decisions in most uncomplicated UTIs are based on symptoms alone, from a scientific stand-point, the effects of a novel preventative must first be confirmed using a “gold-standard” definition. Hence, the efficacy of the only viable cranberry preparation i.e extract in tablet/capsule form, in modifying both culture-confirmed and symptomatic UTI is of interest. To address these issues, we conducted a meta-analysis limited to studies investigating the role of cranberry extract in preventing r-UTI, stratified by outcome definition a priori.
2. Methods
2.1 Study identification
We searched PUBMED/MEDLINE for clinical trials using terms “cranberry”, “cranberries”, or “cranberry extract”
AND “urinary tract infection”, “UTI”, “cystitis”, “pyuria”, “bacteriuria”, or “dysuria”. We did not search conference abstracts or clinical trial registries. Bibliographies of published systematic reviews and individual studies were also searched. We included, i) prospective randomized controlled trials (parallel group or cross-over design), ii) restricted to adults, iii) comparing cranberry extract in tablet/capsule form to placebo or non-placebo control, and iv) clearly defining and reporting each or any of the three end-points in treatment and placebo/control groups. Studies recruiting subjects with neurologic injury/disease, and hence indwelling catheters or undergoing intermittent catheterization, were included. We excluded trials i) using cranberry juice, ii) with treatment duration <30 days, iii) those where incidence of outcome in treatment and placebo groups were not reported as participants with ≥1 UTI (with culture-confirmed UTI or symptomatic UTI as outcome) or number of positive urine cultures (with pyuria/bacteriuria as outcome), iv) studies with active control (antibiotics/other experimental therapies), and finally, v) studies including cranberry combined with another fruit/natural product.
2.2 Study selection, data extraction, and quality assessment
After eliminating duplicates, titles and abstracts of remaining studies were scanned to identify potential candidates. Full-text manuscripts of studies, thus identified, were assessed per the specified inclusion/exclusion criteria for eligibility. Conflicts or differences of opinion were settled by consensus among all authors. Five authors independently extracted data from eligible studies into a pre-designed spreadsheet. Extracted data included study design (parallel/cross-over), duration (months), dosing frequency, population characteristics, outcome definition, and outcome incidence, among others. Studies were sub-divided by one of three outcomes, 1) culture-confirmed UTI, expressed as incidence of ≥1 UTI, 2) asymptomatic pyuria and/or bacteriuria, expressed as number of positive urine cultures, and 3) symptomatic UTI, expressed as for culture-confirmed UTI. The Cochrane risk of bias tool was used to assess the risk of bias for each included study, and graded as “high-risk”, “low-risk”, or “unclear risk” for each of, 1) random sequence generation, 2) allocation concealment, 3) blinding, 4) incomplete outcomes data, 5) selective reporting, and 6) other bias [19]. Risk of bias was assessed by two authors (HG, JH) and any conflicts were settled by group consensus
2.3 Data analysis
Included studies were divided by outcome definition into three categories, i.e. culture-confirmed UTI, pyuria and/or bacteriuria, and symptomatic UTI. These three outcomes were analyzed separately. Some studies had multiple parallel arms, either based on population characteristics (low-risk versus high-risk), or based on cranberry extract dosing (low-dose versus high-dose). In these cases, all arms were analyzed as discreet cohorts. Individual study effect sizes were computed as risk-ratios (RRs) with 95% confidence intervals. Heterogeneity was tested using the I2 statistic, where I2≥50% indicated significant heterogeneity, and the chi-square test, with P ≤ 0.05 indicating significant heterogeneity. Weighted pooled effect-size estimates were obtained using random-effects model (DerSimonian-Laird method) when either test indicated heterogeneity. Fixed-effects (Mantel-Haenszel method) summary estimates were obtained in case of low heterogeneity (I2 <50%, and P>0.05). Meta-analysis was performed using Review Manager (RevMan), version 5.3 (Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014). Publication bias was assessed using funnel plots. Funnel plot asymmetry was assessed using Egger’s test and Kendall’s rank correlation test. In case of significant funnel plot asymmetry, we planned on considering fill and trim adjustment to funnel plots. Case-wise diagnostics were performed by visual inspection of the funnel plots, radial plots, and by influence measures statistics, to identify outliers contributing to heterogeneity. Among influence measures, we were largely interested in Cook’s distance, which is the distance between effect-size with the study included compared to when it is excluded, hence being a measure of how strongly each study influences the overall pooled effect size. Cook’s distance <1.0 is usually accepted as cut-off below which any single study is not deemed to have a predominant influence. The second measurement of interest was the covariance ratio, which indicates (when <1.0) whether removing the study would lead to a more precise effect-size estimate. Heterogeneity diagnostics and funnel plot analysis were performed using the statistical software JASP (JASP Team (2020). JASP (Version 0.12.2) [Computer software].
3. Results
3.1 Study characteristics
Fifteen studies met inclusion criteria for quantitative synthesis [20-34]. PRISMA flow-diagram of the literature search is depicted in Figure 1. Study characteristics are summarized in Table 1, stratified by outcome. Briefly, 5 studies reported symptomatic UTI (not culture-confirmed) as the primary outcome [26-28, 32, 33], yielding 7 comparison cohorts, since one study (Caljouw et al) had 3 intervention arms based on baseline UTI risk, i.e. low risk (no history of recurrent UTI and non-diabetic), high-risk (diabetics or history of r-UTI, but not catheterized) and catheterized patients. Ten studies reported culture-confirmed symptomatic UTI as the primary outcome, yielding 12 comparison groups (Caljouw et al having three parallel groups, as above) [21, 23-25, 27-31, 34]. Seven studies reported incidence of asymptomatic pyuria and/or bacteriuria, yielding 10 comparison groups (Sengupta et al having two independent PAC-dose groups, and Bianco et al having three independent PAC-dose groups) [20, 22, 23, 26, 30-32]. Five trials exclusively enrolled patients with neuropathic bladder, three including patients with spinal cord injury [26, 30, 33], and two including patients with multiple sclerosis [29, 34]. The SINBA trial (Lee 2007) had three intervention arms besides placebo, i.e. cranberry+methenamine-hippurate, cranberry alone, and methenamine-hippurate alone [33]. For our analysis, we only included the cranberry alone versus placebo groups. Only one trial was cross-over [30], the rest being parallel design. Figure 2 summarizes individual study quality and risk of bias assessment. Just over half the studies (8/15) were well-randomized and double-blinded, with the rest not clearly reporting their randomization process. Most studies had low risk of attrition bias (<20% attrition).
3.2 Quantitative synthesis
3.2.1 Impact of cranberry extract on culture-confirmed UTI: There were 2391 subjects across 10 trials (totaling 12 cohorts) reporting culture-confirmed symptomatic UTI, 1174 in cranberry group and 1217 in placebo (PBO) group. There was significant heterogeneity (I2=59%, P=0.005). Pooled random-effects summary estimates revealed a significant reduction in risk of r-UTI with cranberry extract versus PBO (RR: 0.70, 95% CI: 0.54, 0.91, p=0.008, figure 3, upper panel). Funnel plot analysis (Egger’s test: z=0.080, p=0.936; Kendall’s rank correlation test: τ=-0.061, p=0.841) revealed no evidence of significant publication bias (figure 5, upper panel, table S1), hence lack of need for trim-fill analysis. Influence measures analysis did not reveal any one study having undue influence on overall effect size (Cook’s distance <1.0 for all studies, table S2). This was also evident with sensitivity analysis, performed using the leave-one-out method, which revealed stable effect sizes (figure 4, upper panel). Caljouw et al [27] (catheterized cohort), and Singh et al [23] had covariance ratios <1.0, and were also outliers on visual inspection of the funnel plots, indicating that omitting these studies would likely lead to more precise effect-size estimates.
Indeed, repeating the analysis after excluding these two cohorts revealed persistent benefit from cranberry extract (RR: 0.72, 95% CI: 0.57, 0.90, p=0.004), this time with low heterogeneity (I2=36%, P=0.12). Subgroup analysis provided further notable insights as to causes of heterogeneity among the literature (Table 2). Firstly, benefit of cranberry extract seemed limited to patients at high-risk of UTI, with no benefit in low-risk patients, i.e those without history of r-UTI. Similary, neither did there appear to be benefit in patients at an extremely high-risk, i.e. those with neurogenic bladder and/or having indwelling or chronic intermittent catheterization. Hence, in the middle of the risk spectrum, i.e those with history of r-UTI without neurogenic bladder and/or bladder catheterization, the benefit of cranberry extract not only became stronger (RR: 0.54, 95% CI: 0.39, 0.74), there also was a significant drop in heterogeneity (I2 = 42%, P=0.14).
Hence, further sub-group analyses were performed excluding trials limited to these two populations i.e low-risk of r-UTI and catheterized. Initially, cranberry extract seemed to exert no benefit among longer (≥6-month) studies, in contrast to studies <6 months. However, excluding catheterized/neurogenic bladder and low-risk cohorts revealed benefit regardless of study duration. Similarly, once daily versus more frequent dosing had no impact on benefit when catheterized and low-risk patients were excluded. Finally, studies exclusively recruiting females showed a significant benefit with cranberry extract, with no heterogeneity among studies. Pooling studies recruiting both genders (n=6) revealed no benefit with cranberry extract. However, 3 of these included catheterized or low-risk patients. Excluding these cohorts revealed a significant benefit in bi-gender studies, though there seemed to be some attenuation of effect, and significant heterogeneity. Notably, there were no studies exclusively enrolling males, hence the last sub-group comparison was essentially between studies exclusive to females, and studies enrolling both genders. Finally, only two trials recruited healthy community-dwelling females, totaling 276 patients (133 cranberry versus 143 PBO) [24, 25], again demonstrating significant benefit with cranberry extract (RR: 0.49, 95% CI: 0.29, 0.80; p=0.005, I2=0%, p=0.57).
3.2.2 Impact of cranberry extract on asymptomatic bacteriuria and/or pyuria: All studies in this group collected multiple urine samples per subject throughout the study period, reporting proportions of positive urine cultures in treatment and placebo arms. In total, 2565 urine samples were collected across 7 trials (totaling 10 cohorts), 1319 in cranberry group and 1246 in placebo group. There was significant heterogeneity among studies (I2=75%, p<0.0001). Pooled summary estimates (figure 3, middle panel) revealed a significant benefit with cranberry versus placebo in reducing asymptomatic pyuria/bacteriuria (RR: 0.77, 95% CI: 0.69, 0.86, p<0.0001). Funnel lot analysis did not indicate presence of significant publication bias (Egger’s test: z=-1.356, p=0.175; Kendall’s τ=-0.333, P=0.216) as noted in figure 5, middle panel, and table S1. Influence measures analysis revealed Singh et al with a Cook’s distance of >1.0, also evident as an outlier on visual inspection of the funnel plot, and radial plot indicating significant influence of this one study of pooled effect size. Additionally, Singh et al [23] also had a covariance ratio <1.0. Not surprisingly, excluding this study resulted in attenuated (but still statistically significant) effect size among remaining 5 trials (RR: 0.87, 95%CI: 0.76, 0.99, p=0.04). Moreover, excluding Singh et al resulted in elimination of heterogeneity (I2=4%, P=0.39), indicating this one study driving most of the observed pooled effect-size as well as most of the heterogeneity. Since the study by Ledda et al was also an outlier on radial and funnel plot inspection, along with a covariance ratio just under 1.0, we repeated the analysis excluding these two studies [23, 32]. This time, effect size was further attenuated, now remaining borderline significant, with elimination of heterogeneity (RR 0.89, 95% CI 0.79-1.01, p=0.06, I2 = 0%, P = 0.99).
Sensitivity analysis revealed stable effect estimates (figure 4, middle panel). Notably, the trial by Singh et al was generally low-quality-with poorly-defined outcomes, and selective reporting, while the study by Leda et al was a small (n=22 in each group) pilot registry study where both intervention and control groups received “lifestyle advice”. Two trials exclusively enrolled patients with neurogenic bladder due to spinal cord injury [26, 30]. There was no benefit with cranberry extract in this subgroup (RR: 0.87, 95% CI: 0.65, 1.15, p=0.32) and no heterogeneity (I2=0%, P=0.32). On the other hand, there was significant RR-reduction in non-catheterized and non-neurologic injury patients (RR: 0.75, 95% CI: 0.67, 0.84, p<0.00001), but with significant heterogeneity (I2=80%, P<0.0001). Two trials were conducted in nursing home dwelling elderly females [20, 31] totaling 1389 patients (683 cranberry versus 706 PBO), revealing reduced risk with cranberry extract, but with high heterogeneity (RR: 0.75, 95% CI: 0.66-0.85, p<0.0001, I2=86%, P<0.0001). Finally, three trials conducted in young healthy females [22, 23, 32], totaling 540 subjects (306 cranberry versus 234 PBO), also revealed significant benefit with cranberry extract (RR 0.56, 95% CI: 0.46-0.67, p<0.0001, I2=82%, P=0.0007).
3.2.3 Impact of cranberry extract on symptomatic, non-culture confirmed UTI: Five trials yielded 7 cohorts, totaling 1325 patients, 658 in cranberry and 667 in PBO groups. There was borderline heterogeneity (I2=48%, P=0.07). Pooled estimates (figure 3, lower panel) revealed significant benefit with cranberry extract (RR: 0.86, 95% CI: 0.75, 0.98, p=0.03). Funnel plot analysis again revealed no evidence of publication bias (Eggers test: z=-0.007, p = 0.995; Kendall’s τ = 0.048, p=1.000), though it is worth noting that these tests are generally deemed underpowered to detect publication bias with fewer than 10 studies (figure 5, lower panel, table S1). There were no clear outliers either on visual inspection of funnel plot or radial plot (figure 5, lower panel).
Results were unstable on sensitivity analysis (figure 4, lower panel). However, influence measures showed Caljouw [27] (low-risk cohort) and Foxman et al, [28] having a covariance ratio <1.0, indicating excluding these two studies could yield more precise results (table S1). Indeed, excluding these two studies yielded significant pooled effect size with elimination of heterogeneity (RR 0.83. 95% CI, 0.71-0.97, p=0.02, I2 = 0%, P = 0.41). On sub-group analysis, cranberry extract seemed to benefit non-catheterized patients with history of r-UTI (RR: 0.75, 95% CI: 0.61, 0.91, p=0.004) with low heterogeneity (I2=0%, P=0.58), though there were only two trials in this sub-group, totaling 464 subjects (226 cranberry vs 238 PBO) [27, 32]. There was no benefit in catheterized/neurogenic bladder patients (RR: 0.84, 95% CI: 0.67, 1.04, p=0.11).
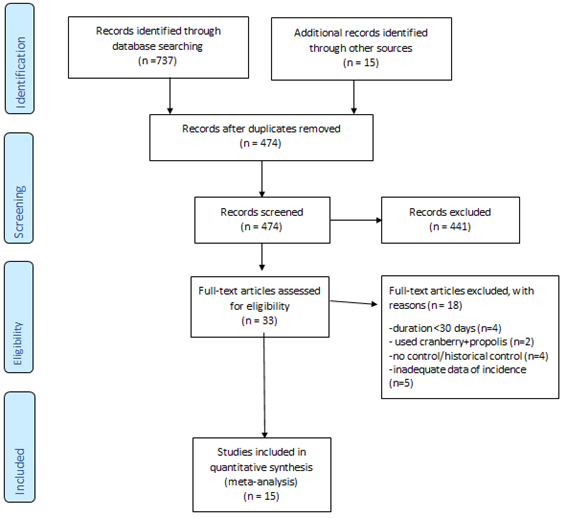
Figure 1: Summary of literature search.
Table 1: Study characteristics of individual studies included in quantitative synthesis of the effect of cranberry extract in reducing r-UTIs.
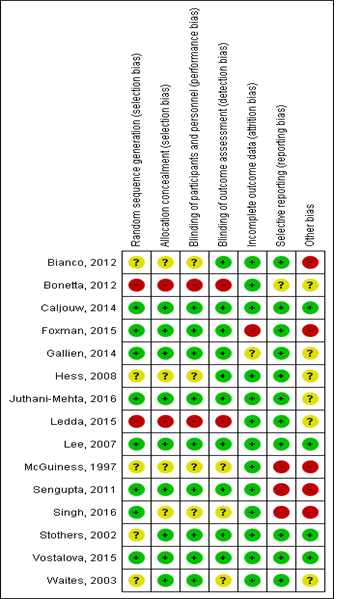
Figure 2: Risk of bias assessment.
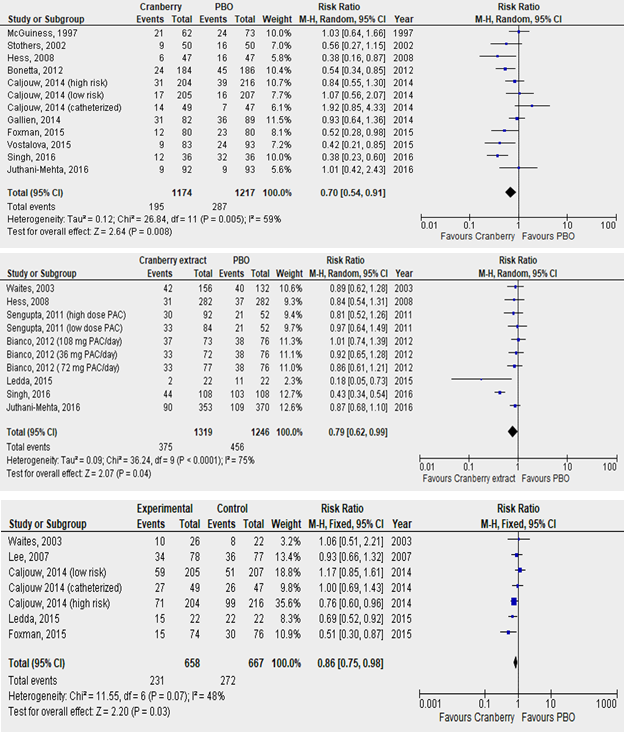
Figure 3: Forest plots depicting pooled relative risk (RR) of recurrent UTI after cranberry extract versus placebo for the three outcomes of culture-confirmed UTI (upper panel), asymptomatic pyuria and bacteriuria (middle panel) and symptomatic UTI (lower panel).
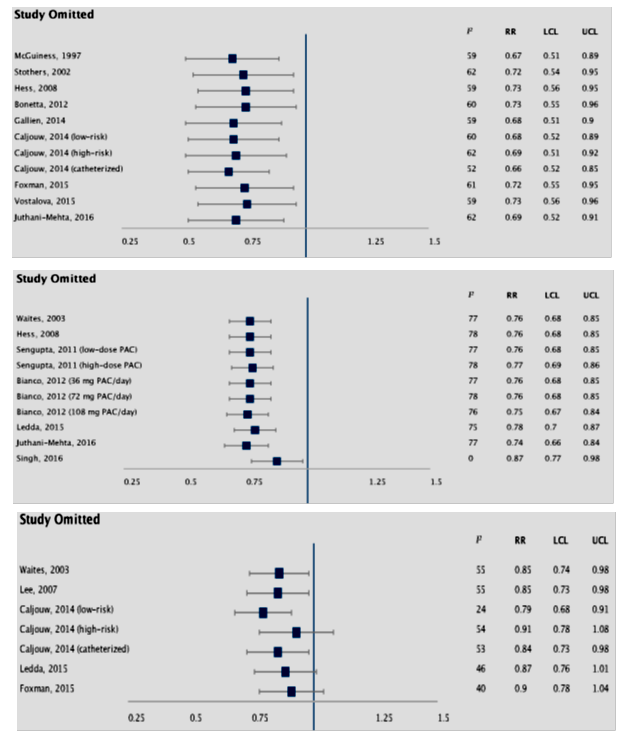
Figure 4: Sensitivity analysis depicting pooled effect sizes and heterogeneity on omitting one study at a time from meta-analysis of the three outcome definitions i.e culture-confirmed UTI (upper panel), asymptomatic pyuria and bacteriuria (middle panel), and symptomatic UTI (lower panel).
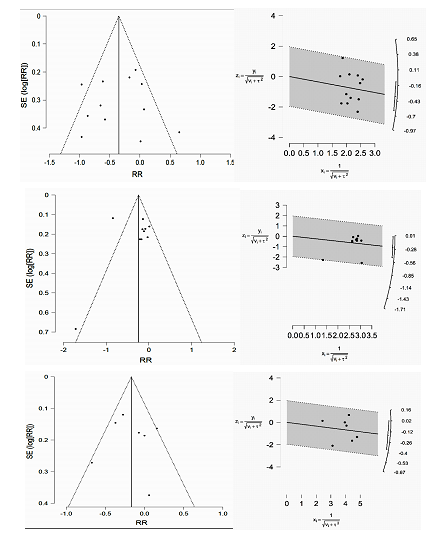
Figure 5: Funnel plots (upper-left, middle-left, and lower-left panels) and radial pots (upper-right, middle-right, and lower-right panels) for culture-confirmed UTI, asymptomatic pyuria/bacteriuria, and symptomatic UTI.
|
Subgroup analysis |
Cohorts, n |
References |
Pooled cumulative UTI incidence |
RR (95% CI) |
Heterogeneity, I2 (%) |
||
|
(Culture-confirmed UTI) |
Cranberry |
PBO |
|||||
|
Overall |
12 |
[21, 23–25, 27–31, 34] |
195/1174 |
287/1217 |
0.70 (0.54, 0.91) |
59 |
|
|
Baseline risk |
|||||||
|
Very high risk (catheterized patients, neurogenic bladder) |
5 |
[27–30, 34] |
84/320 |
106/336 |
0.83 (0.54, 1.28) |
62 |
|
|
High-risk (r-UTI, not catheterized) |
5 |
[21, 23–25, 27] |
85/557 |
156/581 |
0.54 (0.39, 0.74) |
42 |
|
|
Low-risk (no ruti) |
2 |
[27, 31] |
26/297 |
25/300 |
1.05 (0.62, 1.78) |
0 |
|
|
Study duration |
|||||||
|
<6 months |
3 |
[21, 23, 28] |
48/300 |
100/302 |
0.47 (0.35, 0.63) |
0 |
|
|
≥ 6 months |
9 |
[24, 25, 27, 29–31, 34] |
147/874 |
187/915 |
0.83 (0.64, 1.09) |
43 |
|
|
≥ 6 months (excluding catheterized/low-risk) |
4 |
[24, 25, 27, 31] |
58/429 |
88/452 |
0.69 (0.48, 0.98) |
20 |
|
|
Gender |
|||||||
|
Females only |
4 |
[24, 25, 28, 31] |
39/305 |
72/316 |
0.56 (0.39, 0.80) |
0 |
|
|
Females only (excluding catheterized/low-risk) |
3 |
[24, 25, 31] |
27/225 |
49/236 |
0.59 (0.37, 0.94) |
15 |
|
|
Females+males |
6 |
[21, 23, 27, 29, 30, 34] |
156/869 |
215/901 |
0.76 (0.55, 1.06) |
69 |
|
|
Females+males (excluding catheterized/low-risk) |
3 |
[21, 23, 27] |
49/287 |
87/299 |
0.51 (0.28, 0.94) |
72 |
|
|
Dose frequency |
|||||||
|
Once daily |
4 |
[21, 25, 31, 34] |
63/421 |
102/445 |
0.69 (0.44, 1.07) |
54 |
|
|
Once daily excluding catheterized/low-risk |
3 |
[21, 25, 31] |
42/359 |
78/372 |
0.57 (0.38, 0.84) |
17 |
|
|
> Once daily |
8 |
[23, 24, 27–30] |
132/753 |
185/772 |
0.71 (0.50, 1.01) |
66 |
|
|
> Once daily exclude-ing low-risk/catheteri- zed |
4 |
[21, 23, 24, 27] |
76/474 |
132/488 |
0.56 (0.39, 0.81) |
52 |
|
|
Risk of selection bias |
|||||||
|
Low-risk |
8 |
[23, 25, 27–29, 31] |
135/831 |
186/861 |
0.75 (0.53, 1.07) |
66 |
|
|
Low-risk excluding catheterized and low UTI risk |
4 |
[23, 25, 27, 31] |
61/415 |
104/438 |
0.59 (0.36-0.97) |
65 |
|
|
High/unclear risk |
4 |
[21, 24, 30, 34] |
60/343 |
101/356 |
0.62 (0.41-0.95) |
51 |
|
Table 2: Subgroup analysis of the effect of cranberry extract on risk of culture-confirmed UTI.
4. Discussion
We present an updated, outcome specific meta-analysis of cranberry extract for prevention of r-UTI. This is the first meta-analysis exclusive to what is the majorly feasible therapeutic formulation, i.e extract in capsule/tablet form. Key findings include, i) cranberry extract as tablet/capsule is effective in reducing risk of r-UTI with the gold-standard criterion of positive urine cultures in symptomatic patients, with ≈30% reduction in UTI risk, ii) cranberry extract is effective in reducing rates of asymptomatic pyuria/bacteriuria, as well as symptomatic UTI, iii) the benefit of cranberry extract seems restricted to those with history of r-UTI, but ineffective in those at low-risk (i.e. those without recurrent UTI) and in catheterized and/or neuropathic bladder patients. We found that the latter two populations revealed a persistent benefit regardless of outcome definition, and with significantly less heterogeneity. The lack of benefit in catheterized populations is important and not too difficult to explain. The anti-adhesion properties of PACs have largely been studied and demonstrated in gram-negative bacteria, more specifically E. coli. Since catheterized patients are at risk of UTI with a broader class of pathogens, including gram-positive bacteria, it may well be that these anti-adhesive properties fail in these patients. Moreover, the mechanism of catheter-associated UTI is complex, including bio-film formation on catheters, mucosal injury from the catheters, among others. Hence it may represent too over-whelming a risk, or a mechanism not modifiable by PACs. Our findings are all the more robust given that cranberry extract proved to be ineffective in this population regardless of outcome-definition. Furthermore, baseline population risk profile seems to drive much of the heterogeneity in extant literature. Indeed, heterogeneity was significantly attenuated when studies in populations at extremes of baseline risk profile were excluded. A major drawback we encountered in the extant literature is significant publication bias, especially prominent among studies using asymptomatic pyuria/bacteriuria and symptomatic UTI as outcomes. Indeed, previous meta-analyses suffered significant heterogeneity and even conflicting conclusions because of three likely factors, i) pooling studies with different outcome definitions, ii) pooling studies in varied populations with regards to baseline risk profile, and/or c) pooling studies using both juice and cranberry extract. It is worth mentioning that we did not expressly test the last factor since excluded all juice studies. Briefly, there have been 4 meta-analyses in the last decade to examine the effect of cranberries in this context [13-15, 35]. Jepson et al reported a meta-analysis in 2012, predating almost two-thirds of trials included in our analysis [13]. Majority of included trials used cranberry juice, which as noted by the authors themselves at the time, is hardly a feasible long-term therapeutic option. They found no benefit of cranberry “product” in reducing UTI risk (RR 0.86, 0.71-1.04). No separate analysis by formulation was performed. A meta-analysis by Wang et al the same year included 10 studies (10 juice, 4 tablet/capsule), and found significant benefit with cranberry “product” [15]. On subgroup analysis, tablet/capsule group had no benefit (RR 0.79, 0.44-1.44) though cranberry juice seemed to lower UTI risk (RR 0.47, 0.30-0.72). Notably, three of the four tablet/capsule studies were limited to neuropathic bladder patients, a group that our findings suggest does not benefit from cranberry extract. Finally, subgroup analysis by outcome definition revealed cranberry product to lower risk of only symptomatic UTI and not culture-confirmed UTI, highlighting the role of outcome definition in introducing heterogeneity and indeed conflicting findings. Regardless, both these meta-analyses predated majority of trials included in the current meta-analysis.
More recently, Fu et al conducted a meta-analysis of 7 trials exclusive to healthy women [14]. Again, pooling studies using cranberry juice and tablet/capsule, they found no overall benefit. Interestingly, cranberry tablet/capsule subgroup revealed a benefit over juice (RR 0.79, 0.59-1.06 for juice versus 0.48, 0.29-0.79 for tablet/capsule), though based on only two trials using tablet/capsule. Using culture-confirmed UTI as outcome, there was no benefit with cranberry product (RR 0.71, 0.45-1.12), again pooling tablet/capsule and juice trials. There was no separate analysis by formulation for culture-confirmed UTI. In a larger meta-analysis, Luis et al pooled results from a broad set of studies conducted in all age groups including children, pregnant females, and those with neurogenic bladder [35]. Again, trials using both cranberry formulations and all outcome definitions were pooled, as were studies using non-placebo control (antibiotics, other probiotics, etc). Similar to our findings, though there was reduced UTI-risk overall (RR 0.68, 0.57-0.80), there was no benefit in catheterized subjects (RR 0.89, 0.68-1.17). They also found no benefit in young adults (18-35 yrs old), though this subgroup included 3 trials, all of which used cranberry juice, one recruited females with no history of r-UTI, and spanned all three outcome definitions. Finally, their analysis did not address the specific question of formulation, having no sub-group analysis for juice versus tablet/capsule. Hence, so far, the specific question of whether tablet/capsule formulations of cranberry extract can reduce risk of r-UTI when defined using the gold-standard definition, remains unclear despite existing meta-analyses. We demonstrate, for the first time, that cranberry extract in tablet/capsule form is a feasible and effective therapeutic option for those at risk of r-UTI. To our way of thinking, studies using juice as treatment have no more than hypothesis generating and academic value, given the aforementioned barriers to widespread long-term use. In terms of using cranberry extract in tablet/capsule form, there remains a need for large and high-quality RCTs. Moreover, our findings should hopefully help aid patient selection for future investigations, since low-risk patients or catheterized patients could potentially distort findings. Additionally, future studies should use the “gold-standard” definition of UTI, i. e. culture-confirmed UTI in symptomatic patients. Finally, commercially available cranberry extract preparations likely have variable PAC content and bioavailability, making it difficult to make general recommendations based on our results. Hence, future investigations should ideally report PAC bioavailability of the investigated formulation, preferably with measurement of plasma kinetics in at least a subset of included subjects.
Conflicts of Interest
The authors report no conflicts of interest.
References
- Center for Health Statistics N. National Hospital Ambulatory Medical Care Survey: 2017 Emergency Department Summary Tables (2017).
- Simmering JE, Tang F, Cavanaugh JE, Polgreen LA, Polgreen PM. The Increase in Hospitalizations for Urinary Tract Infections and the Associated Costs in the United States, 1998–2011. Open Forum Infect Dis 4 (2017).
- Foxman B. Epidemiology of Urinary Tract Infections: Incidence, Morbidity, and Economic Costs (2002).
- Suskind AM, Saigal CS, Hanley JM, Lai J, Setodji CM, Clemens JQ. Incidence and Management of Uncomplicated Recurrent Urinary Tract Infections in a National Sample of Women in the United States. Urology 90 (2016): 50-55.
- Jennifer Anger, Una Lee, Lenore Ackerman A, Roger Chou, Bilal Chughtai, Quentin Clemens J, et al. Recurrent Uncomplicated Urinary Tract Infections in Women: AUA/CUA/SUFU Guideline. J Urol 202 (2019): 282-289.
- Sobota AE. Inhibition of bacterial adherence by cranberry juice: Potential use for the treatment of urinary tract infections. J Urol 131 (1984): 1013-1016.
- Zafriri D, Ofek I, Adar R, Pocino M, Sharon2 N. Inhibitory Activity of Cranberry Juice on Adherence of Type 1 and Type P Fimbriated Escherichia Coli to Eucaryotic Cells 33 (1989).
- Gupta K, Chou MY, Howell A, Wobbe C, Grady R, Stapleton AE. Cranberry Products Inhibit Adherence of P-Fimbriated Escherichia Coli to Primary Cultured Bladder and Vaginal Epithelial Cells. J Urol 177 (2007): 2357-2360.
- Amy B Howell, Henry Botto, Christophe Combescure, Anne-Béatrice Blanc-Potard, Lluis Gausa, Tetsuro Matsumoto, et al. Dosage effect on uropathogenic Escherichia coli anti-adhesion activity in urine following consumption of cranberry powder standardized for proanthocyanidin content: A multicentric randomized double blind study. BMC Infect Dis 10 (2010).
- Howell AB, Reed JD, Krueger CG, Winterbottom R, Cunningham DG, Leahy M. A-type cranberry proanthocyanidins and uropathogenic bacterial anti-adhesion activity. Phytochemistry 66 (2005): 2281-2291.
- Liska DJ, Kern HJ, Maki KC. Cranberries and Urinary Tract Infections: How Can the Same Evidence Lead to Conflicting Advice?. Adv Nutr 7 (2016): 498-506.
- Jepson R, Mihaljevic L, Craig J. Cranberries for preventing urinary tract infections. In: Cochrane Database of Systematic Reviews. Chichester, UK: John Wiley & Sons, Ltd (2003).
- Jepson RG, Williams G, Craig JC. Cranberries for preventing urinary tract infections. Cochrane Database Syst Rev (2012).
- Fu Z, Liska D, Talan D, Chung M. Cranberry Reduces the Risk of Urinary Tract Infection Recurrence in Otherwise Healthy Women: A Systematic Review and Meta-Analysis. J Nutr 147 (2017): 2282-2288.
- Chih-Hung Wang, Cheng-Chung Fang, Nai-Chuan Chen, Sot Shih-Hung Liu, Ping-Hsun Yu, Tao-Yu Wu, Wei-Ting Chen, et al. Cranberry-Containing Products for Prevention of Urinary Tract Infections in Susceptible Populations A Systematic Review and Meta-analysis of Randomized Controlled Trials. Arch Intern Med 172 (2012): 988-996.
- Merve Eda Eker, Kjersti Aaby, Irena Budic-Leto, Suzana Rimac Brncic, Sedef Nehir E, Sibel Karakaya, et al. A review of factors affecting anthocyanin bioavailability: Possible implications for the inter-individual variability. Foods 9 (2020).
- Ann E Stapleton, James Dziura, Thomas M Hooton, Marsha E Cox, Yuliya Yarova-Yarovaya, Shu Chen, et al. Recurrent urinary tract infection and urinary Escherichia coli in women ingesting cranberry juice daily: A randomized controlled trial. Mayo Clin Proc 87 (2012): 143-150.
- Barbosa-Cesnik C, Brown MB, Buxton M, Zhang L, Debusscher J, Foxman B. Cranberry juice fails to prevent recurrent urinary tract infection: Results from a randomized placebo-controlled trial. Clin Infect Dis 52 (2011): 23-30.
- Higgins J, Savovic J, Page M, Elbers R, Sterne J. Chapter 8: Assessing risk of bias in a randomized trial. In: Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA (editors). Handbook for Systematic Reviews of Interventions version 6.0 (updated July 2019). Cochrane (2019).
- Bianco L, Perrelli E, Towle V, Van Ness PH, Juthani-Mehta M. Pilot randomized controlled dosing study of cranberry capsules for reduction of bacteriuria plus pyuria in female nursing home residents. J Am Geriatr Soc 60 (2012): 1180-1181.
- Bonetta A, Di Pierro F. Enteric-coated, highly standardized cranberry extract reduces risk of UTIs and urinary symptoms during radiotherapy for prostate carcinoma. Cancer Manag Res 4 (2012): 281-286.
- Sengupta K, Alluri K V, Golakoti T, Gottumukkala GV, Raavi J, Kotchrlakota L, et al. A Randomized, Double Blind, Controlled, Dose Dependent Clinical Trial to Evaluate the Efficacy of a Proanthocyanidin Standardized Whole Cran-Berry (Vaccinium Macrocarpon) Powder on Infections of the Urinary Tract 7 (2011).
- Singh I, Gautam LK, Kaur IR. Effect of oral cranberry extract (standardized proanthocyanidin-A) in patients
with recurrent UTI by pathogenic E. coli: a randomized placebo-controlled clinical research study. Int Urol Nephrol 48 (2016): 1379-1386.
- Stothers L. A randomized trial to evaluate effectiveness and cost effectiveness of naturopathic cranberry products as prophylaxis against urinary tract infection in women. Can J Urol 9 (2002): 1558-1562.
- Jitka Vostalova, Ales Vidlar, Vilim Simanek, Adela Galandakova, Pavel Kosina, Jan Vacek, et al. Are High Proanthocyanidins Key to Cranberry Efficacy in the Prevention of Recurrent Urinary Tract Infection?. Phytother Res 29 (2015): 1559-1567.
- Waites KB, Canupp KC, Armstrong S, DeVivo MJ. Effect of cranberry extract on bacteriuria and pyuria in persons with neurogenic bladder secondary to spinal cord injury. J Spinal Cord Med 27 (2004): 35-40.
- Caljouw M, Van den hout W, Putter H, Achterberg W, Cools H, Gussekloo J. Effectiveness of cranberry capsules to prevent urinary tract infections in vulnerable older persons: a double-blind randomized placebo-controlled trial in long-term care facilities. J Am Geriatr Soc 62 (2014): 103-110.
- Foxman B, Cronenwett AEW, Spino C, Berger MB, Morgan DM. Cranberry juice capsules and urinary tract infection after surgery: results of a randomized trial. Am J Obstet Gynecol 213 (2015): 194.e1-194.e8.
- Philippe Gallien, Gérard Amarenco, Nicolas Benoit, Véronique Bonniaud, Cécile Donzé, Jacques Kerdraon, et al. Cranberry versus placebo in the prevention of urinary infections in multiple sclerosis: a multicenter, randomized, placebo-controlled, double-blind trial. Mult Scler 20 (2014): 1252-1259.
- Hess MJ, Hess PE, Sullivan MR, Nee M, Yalla S V. Evaluation of cranberry tablets for the prevention of urinary tract infections in spinal cord injured patients with neurogenic bladder. Spinal Cord 46 (2008): 622-626.
- Manisha Juthani-Mehta, Peter H Van Ness, Luann Bianco, Andrea Rink, Sabina Rubeck, Sandra Ginter, et al. Effect of Cranberry Capsules on Bacteriuria Plus Pyuria Among Older Women in Nursing Homes: A Randomized Clinical Trial. JAMA 316 (2016): 1879-1887.
- Ledda A, Bottari A, Luzzi R, Belcaro G, Hu S, Dugallet M, et al. Cranberry supplementation in the prevention of non-severe lower urinary tract infections: a pilot study. Eur Rev Med Pharmacol Sci 19 (2015): 77-80.
- Lee B B, Haran M J, Hunt L M, Simpson J M, Marial O, Rutkowski S B, et al. Spinal-injured neuropathic bladder antisepsis (SINBA) trial. Spinal Cord 45 (2007): 542-550.
- McGuiness SD, Krone R, Metz LM. A double-Blind, Randomized, Placebo-Controlled Trial of Cranberry Supplements in Multiple Sclerosis. J Neurosci Nurs 34 (2002): 4-7.
- Luís Â, Domingues F, Pereira L. Can Cranberries Contribute to Reduce the Incidence of Urinary Tract Infections? A Systematic Review with Meta-Analysis and Trial Sequential Analysis of Clinical Trials. J Urol 198 (2017): 614-621.
