Computed Tomography Findings of Diffuse Intracranial Calcifications in A Patient with Primary Hypoparathyroidism
Article Information
Ruhaid Khurram*, Rahul Khamar and Srikanth Mandumula
Department of Radiology, Barnet Hospital, Royal Free London NHS Foundation Trust, United Kingdom
*Corresponding Author: Ruhaid Khurram, Department of Radiology, Barnet Hospital, Royal Free London NHS Foundation Trust, United Kingdom
Received: 31 January 2020; Accepted: 07 February 2020; Published: 18 February 2020
Citation: Ruhaid Khurram, Rahul Khamar, Srikanth Mandumula. Computed Tomography Findings of Diffuse Intracranial Calcifications in A Patient with Primary Hypoparathyroidism. Journal of Radiology and Clinical Imaging 3 (2020): 033-037.
View / Download Pdf Share at FacebookAbstract
Primary hypoparathyroidism is a rare endocrine condition characterised by an abnormally low level of parathyroid hormone (PTH) which has significant effects on calcium metabolism, homeostasis and can have a wide range of clinical manifestations. We report a case of a 43-year-old female who was referred to our emergency department following a witnessed generalised tonic-clonic seizure lasting seven minutes. Blood tests and investigations confirmed a diagnosis of primary hypoparathyroidism. Computed tomography (CT) imaging of the brain revealed rare findings of diffuse bilateral intracranial calcifications affecting the cerebellum, brainstem, basal ganglia, internal capsule and numerous other cortical areas.
Keywords
<p>Intracranial; Calcification; Computed tomography; Hypoparathyroidism; Hypocalcaemia</p>
Intracranial articles, Calcification articles, Computed tomography articles, Hypoparathyroidism articles, Hypocalcaemia articles
Intracranial articles Intracranial Research articles Intracranial review articles Intracranial PubMed articles Intracranial PubMed Central articles Intracranial 2023 articles Intracranial 2024 articles Intracranial Scopus articles Intracranial impact factor journals Intracranial Scopus journals Intracranial PubMed journals Intracranial medical journals Intracranial free journals Intracranial best journals Intracranial top journals Intracranial free medical journals Intracranial famous journals Intracranial Google Scholar indexed journals Calcification articles Calcification Research articles Calcification review articles Calcification PubMed articles Calcification PubMed Central articles Calcification 2023 articles Calcification 2024 articles Calcification Scopus articles Calcification impact factor journals Calcification Scopus journals Calcification PubMed journals Calcification medical journals Calcification free journals Calcification best journals Calcification top journals Calcification free medical journals Calcification famous journals Calcification Google Scholar indexed journals Computed tomography articles Computed tomography Research articles Computed tomography review articles Computed tomography PubMed articles Computed tomography PubMed Central articles Computed tomography 2023 articles Computed tomography 2024 articles Computed tomography Scopus articles Computed tomography impact factor journals Computed tomography Scopus journals Computed tomography PubMed journals Computed tomography medical journals Computed tomography free journals Computed tomography best journals Computed tomography top journals Computed tomography free medical journals Computed tomography famous journals Computed tomography Google Scholar indexed journals Hypoparathyroidism articles Hypoparathyroidism Research articles Hypoparathyroidism review articles Hypoparathyroidism PubMed articles Hypoparathyroidism PubMed Central articles Hypoparathyroidism 2023 articles Hypoparathyroidism 2024 articles Hypoparathyroidism Scopus articles Hypoparathyroidism impact factor journals Hypoparathyroidism Scopus journals Hypoparathyroidism PubMed journals Hypoparathyroidism medical journals Hypoparathyroidism free journals Hypoparathyroidism best journals Hypoparathyroidism top journals Hypoparathyroidism free medical journals Hypoparathyroidism famous journals Hypoparathyroidism Google Scholar indexed journals Hypocalcaemia articles Hypocalcaemia Research articles Hypocalcaemia review articles Hypocalcaemia PubMed articles Hypocalcaemia PubMed Central articles Hypocalcaemia 2023 articles Hypocalcaemia 2024 articles Hypocalcaemia Scopus articles Hypocalcaemia impact factor journals Hypocalcaemia Scopus journals Hypocalcaemia PubMed journals Hypocalcaemia medical journals Hypocalcaemia free journals Hypocalcaemia best journals Hypocalcaemia top journals Hypocalcaemia free medical journals Hypocalcaemia famous journals Hypocalcaemia Google Scholar indexed journals parathyroid hormone articles parathyroid hormone Research articles parathyroid hormone review articles parathyroid hormone PubMed articles parathyroid hormone PubMed Central articles parathyroid hormone 2023 articles parathyroid hormone 2024 articles parathyroid hormone Scopus articles parathyroid hormone impact factor journals parathyroid hormone Scopus journals parathyroid hormone PubMed journals parathyroid hormone medical journals parathyroid hormone free journals parathyroid hormone best journals parathyroid hormone top journals parathyroid hormone free medical journals parathyroid hormone famous journals parathyroid hormone Google Scholar indexed journals arterial blood gas articles arterial blood gas Research articles arterial blood gas review articles arterial blood gas PubMed articles arterial blood gas PubMed Central articles arterial blood gas 2023 articles arterial blood gas 2024 articles arterial blood gas Scopus articles arterial blood gas impact factor journals arterial blood gas Scopus journals arterial blood gas PubMed journals arterial blood gas medical journals arterial blood gas free journals arterial blood gas best journals arterial blood gas top journals arterial blood gas free medical journals arterial blood gas famous journals arterial blood gas Google Scholar indexed journals pseudohypoparathyroidism articles pseudohypoparathyroidism Research articles pseudohypoparathyroidism review articles pseudohypoparathyroidism PubMed articles pseudohypoparathyroidism PubMed Central articles pseudohypoparathyroidism 2023 articles pseudohypoparathyroidism 2024 articles pseudohypoparathyroidism Scopus articles pseudohypoparathyroidism impact factor journals pseudohypoparathyroidism Scopus journals pseudohypoparathyroidism PubMed journals pseudohypoparathyroidism medical journals pseudohypoparathyroidism free journals pseudohypoparathyroidism best journals pseudohypoparathyroidism top journals pseudohypoparathyroidism free medical journals pseudohypoparathyroidism famous journals pseudohypoparathyroidism Google Scholar indexed journals pituitary gland articles pituitary gland Research articles pituitary gland review articles pituitary gland PubMed articles pituitary gland PubMed Central articles pituitary gland 2023 articles pituitary gland 2024 articles pituitary gland Scopus articles pituitary gland impact factor journals pituitary gland Scopus journals pituitary gland PubMed journals pituitary gland medical journals pituitary gland free journals pituitary gland best journals pituitary gland top journals pituitary gland free medical journals pituitary gland famous journals pituitary gland Google Scholar indexed journals
Article Details
1. Introduction
In an unenhanced CT scan of the brain, hyperdense areas represent either calcium or acute hemorrhagic extravasation. There are numerous causes of intracranial calcification, many of which are non-pathological. In a non-contrast enhanced CT scan of the brain, areas where physiological calcification is commonly seen include: the pineal gland, habenula, basal ganglia, the choroid plexus and dural coverings including those that constitute the falx cerebri and tentorium cerebelli. In a contrast enhanced scan, vascular structures including the venous sinuses and the pituitary gland also appear hyperdense due to the radiodense property of iodine. The differential diagnoses of pathological intracranial calcifications are vast. Primary hypoparathyroidism is a recognised metabolic causes of intracranial calcification. There have been a few documented cases of this phenomenon in the literature [1-3].
2. Case Description
A 43-year-old female patient was initially referred to our emergency department in April 2018 after experiencing a seven minute generalised tonic-clonic seizure in the community. She was initially brought into our resuscitation department and given several doses of anticonvulsant medication in order to terminate the seizure. On initial assessment after the seizure terminated, she was in the post-ictal phase but had no other focal neurological signs or sequelae. She had no significant medical or surgical history of note except community management for vitamin D deficiency with oral calcium and vitamin D supplementation.
Blood tests including arterial blood gas (ABG), full blood count, renal function tests, liver function tests, creatine kinase and inflammatory markers were taken in the first instance. These were largely unrevealing aside from a raised lactate (8 mmol/L; normal <1.5 mmol/L) on the ABG which was expected secondary to rhabdomyolysis as a consequence of muscle contraction during the seizure. As part of on-going assessment, bone profile tests were requested, in addition to computed tomography (CT) imaging in order to exclude an organic intracranial pathology. Bone profile biochemical tests revealed severe hypocalcaemia (1.34 mmol/L; normal 2.15-2.61 mmol/L) and hyperphosphataemia (1.65 mmol/L; normal 0.74-1.52 mmol/L). These results, in addition to an undetectable parathyroid hormone level (C < 0.4 pmol/L; normal 1.4-6.4 pmol/L) and a normal 25-hydroxyvitamin D level of 67 nmol/L, were in-keeping with a diagnosis of primary hypoparathyroidism. An unenhanced CT examination of the brain by a 64-slice CT scanner was performed. The protocol included a non-contrast CT scan with a 4mm slice thickness. As demonstrated in Figure 1, dense bilateral intracranial calcification was demonstrated involving the cerebellum (A), the brain stem (B), the basal ganglia and internal capsule (C) and numerous other cortical areas (D). There were no other focal cerebral or extra-axial abnormalities noted. She was treated aggressively with intravenous calcium replacement and was subsequently referred to an endocrinologist.
This patient was re-referred to our department for a follow-up CT scan of the brain in January 2019 following a one-week history of new onset muscle stiffness, abnormal gait and mild tremor. On this admission, biochemistry remained consistent with the initial diagnosis revealing hypocalcaemia (1.31 mmol/L, normal 2.15-2.61 mmol/L) and a worsening hyperphosphataemia (2.03 mmol/L, normal 0.74-1.52 mmol/L). As demonstrated in Figure 2 below, there was no significant interval change in the intracranial calcifications as compared to the CT examination from April 2018.
|
Figure SEQ Figure \* ARABIC 1 - Non-contrast computer tomography scan (reported in April 2018) of the brain illustrating the distribution of dense calcification including the cerebellum, brainstem, basal ganglia nuclei, the internal tract and other cortical areas |
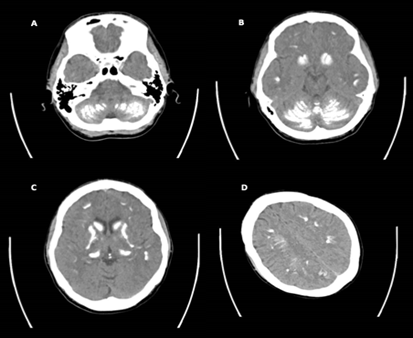
Figure 1: Non-contrast computed tomography scan (reported in April 2018) of the brain illustrating the distribution of dense calcification including the cerebellum, brainstem, basal ganglia nuclei, the internal tract and other cortical areas.
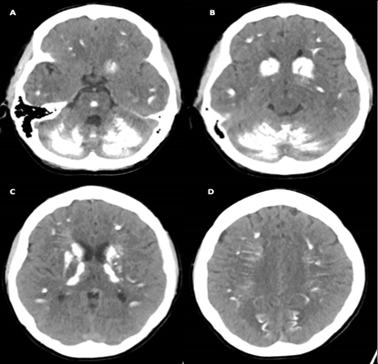
Figure 2: Non-contrast computed tomography scan (reported in January 2019) of the brain illustrating the distribution of dense calcification including the cerebellum, brainstem, basal ganglia nuclei, the internal tract and other cortical areas
3. Discussion
The most commonly observed intracranial physiological calcification is of the pineal gland, with one study observing pineal region calcification in 68% of participants. This was closely followed by the incidence of calcification of the choroid plexus of lateral ventricles (typically of the atrial portions), observed in 58% of participants [4]. The frequency of calcification of both structures showed a steady increase with age in another cross-sectional study [5]. Calcification of the basal ganglia nuclei typically appear as a cluster of irregular punctate calcifications that are bilateral and symmetrical [6]. A study investigating calcification of basal ganglia found that the most common distribution of calcification involved the globus pallidus nuclei, solely and bilaterally. The second most common distribution included both the globus pallidus and caudate nuclei bilaterally [7]. Calcifications of the falx tend to appear as dense flat plaques that are found in the midline between the cerebral hemispheres. Other dural calcifications are typically laminar and are not confined to a particular cortical region. Pathological causes of intracerebral calcification can be classified according to aetiology: congenital, infectious, metabolic, vascular and neoplastic.
The most common metabolic cause is pseudohypoparathyroidism, followed closely by hypoparathyroidism. Whilst the exact mechanism of calcification in both of these conditions isn’t fully understood, it is thought that chronic hyperphosphataemia plays a significant role in promoting ectopic cerebral calcification via elevated levels of calcium-phosphorus product [8]. In such metabolic diseases, the distribution of calcification is often diffuse and bilateral, with the most commonly affected sites including the lentiform and caudate nuclei of the basal ganglia [9]. One notable facet of this case is that despite minimal change in the biochemical corrected calcium level between these two presentations that were nine months apart; there was a notable difference in the severity of the presenting symptoms. When the patient initially presented following a seizure; she was noted to have a serum calcium of 1.34 mmol/L. During her second presentation with symptoms that were less severe including muscle stiffness and gait disturbance, the corrected calcium was found to be 1.31 mmol/L. This suggests that the severity of biochemical hypocalcaemia does not necessarily correlate with the severity of the clinical symptoms of hypocalcaemia. This also highlights the challenges of long-term management of primary hypoparathyroidism. Despite the reported evolution of new symptoms over the nine-month period separating both presentations, there was no significant interval changes reported in the distribution or density of the intracranial calcification noted on the CT scans of the brain.
It can therefore be surmised that an individual patient’s clinical sequelae is independent to the density of the intracranial calcification and more related to the biochemical hypocalcaemia itself. Intravenous calcium replacement is the mainstay of treatment in patients with severe symptomatic hypocalcaemia. Mild hypocalcaemia is often treated with oral calcium supplementation whilst specific secondary causes of hypocalcaemia are managed according to aetiology e.g. supplementation with alfacalcidol in patients with chronic kidney disease. The ultimate goal is to achieve symptom control and to avoid life-threatening complications of hypocalcaemia e.g. seizures, tetany and fatal arrhythmias.
Conflict of Interest
We have no conflicts of interests to declare in writing this case report.
References
- Harada K, Fujikawa T. Intracranial Calcification Due to Hypoparathyroidism. The American Journal of Medicine 131 (2018): e253-e254.
- Mejdoubi M, Zegermann T. Extensive brain calcification in idiopathic hypoparathyroidism. Journal of Neurology, Neurosurgery & Psychiatry 77 (2006): 1328-1328.
- Zisimopoulou V, Siatouni A, Tsoukalos G, et al. Extensive Bilateral Intracranial Calcifications: A Case of Iatrogenic Hypoparathyroidism. Case Reports in Medicine (2013): 1-3.
- Kwak R, Takeuchi F, Ito S, et al. Intracranial physiological calcification on computed tomography (Part 1): Calcification of the pineal region. No To Shinkei 40 (1988): 569-574.
- Bennett J, Maffly R, Steinbach H. The Significance of Bilateral Basal Ganglia Calcification. Radiology 72 (1959): 368-378.
- Brannan T, Burger A, Chaudhary M. Bilateral basal ganglia calcifications visualised on CT scan. Journal of Neurology, Neurosurgery and Psychiatry 43 (1980): 403-406.
- Admassie D, Mekonnen A. Incidence of normal pineal and choroid plexus calcifications on brain CT (computerized tomography) at Tikur Anbessa Teaching Hospital Addis Ababa, Ethiopia. Ethipoian Medical Journal 47 (2009): 55-60.
- Fujita T. Mechanism of intracerebral calcification in hypoparathyroidism. Clin Calcium 14 (2004): 55-57.
- Marques Mendes E, Meireles-Brandão L, Meira C, et al. Primary hypoparathyroidism presenting as basal ganglia calcification secondary to extreme hypocalcemia. Clinics and Practice 8 (2018): 1007.
