Comparison of Crystalloid Preloading versus Ephedrine Prophylaxis to Prevent Spinal Anesthesia Induced Hypotension during Cesarean Delivery: A Randomized Clinical Trial
Article Information
Yonatan Mehari Andemeskel* Daniel Habtom Brhane, Jemal Abulkereem MohammedAli, Teklit Mebrahtu Habtetsion
Orotta College of Medicine and Health Sciences, Asmara, Eritrea
*Corresponding Author: Yonatan Mehari Andemeskel, Orotta College of Medicine and Health Sciences, Asmara, Eritrea.
Received: 20 February 2024; Accepted: 11 March 2024; Published: 18 March2024
Citation: Yonatan Mehari Andemeskel, Daniel Habtom Brhane, Jemal Abulkereem MohammedAli, Teklit Mebrahtu Habtetsion. Comparison of Crystalloid Preloading versus Ephedrine Prophylaxis to Prevent Spinal Anesthesia Induced Hypotension during Cesarean Delivery: A Randomized Clinical Trial. Anesthesia and Critical care 6 (2024): 17-25.
View / Download Pdf Share at FacebookAbstract
Hypotension in patients who take spinal anesthesia is a potentially serious issue. Expansion of the intravascular volume can be achieved by preloading with crystalloid fluids, which is a common practice in elective cases. Ephedrine, as a prophylaxis, has been recently practiced to prevent such occurrence. This study aimed to compare the efficacy between preloading with crystalloid fluid and administering ephedrine as a prophylaxis in the prevention of spinal anesthesia-induced hypotension during elective cesarean delivery. A total of 60 parturient mothers (30.98+4.69 years of age) appointed for elective cesarean delivery were randomly allocated to each group. The crystalloid group (n=30) received the usual management which is a crystalloid fluid 15 minutes prior to spinal anesthesia and the second group, ephedrine group (n=30) were given prophylactic ephedrine soon after receiving spinal anesthesia. Vital signs including blood pressure and pulse rate were recorded in time and patients were observed for the occurrence of complications related to spinal anesthesia. The primary endpoint was the change in the mentioned vital signs. The secondary endpoint included the occurrence of spinal anesthesia related complications. The overall mean SBP in the ephedrine group (M= 117.20, SD=10.19) was significantly greater (p=0.004) than the crystalloid group (M=106.43, SD=13.96). No significant difference was observed on the overall measure of pulse rate (p=0.238). The incidence of hypotension (46.7% vs 3.3%) as well as nausea and vomiting (46.7% vs 0%) were significantly higher in the crystalloid group (p<0.0001). In conclusion, administration of ephedrine as a prophylaxis is better than crystalloid preloading in the prevention of spinal-induced hypotension and associated complications.
Keywords
<p>Hypotension, Prophylactic Ephedrine, Crystalloid, Spinal Anesthesia</p>
Hypotension articles; Prophylactic Ephedrine articles; Crystalloid articles; Spinal Anesthesia articles
Article Details
1. Introduction
Spinal Anesthesia is a commonly practiced mode of regional anesthesia for patients undergoing caesarean section. It is an accepted and appropriate choice of anesthesia, providing high quality of sensory and motor block with adequate muscle relaxation and avoiding the risk of aspiration and other risks of general anesthesia [1-10]. However, spinal anesthesia is not without a risk. The most common maternal complication after spinal anesthesia during caesarian delivery is hypotension, in which the incidence reaches up to 80% if preventive measures have not been taken [1, 9, 11-19]. Hypotension is generally characterized as a reduction of systolic arterial pressure greater than 20% of the baseline [20]. Profound hypotension could be a potentially serious issue resulting in adverse maternal and fetal outcomes if not managed effectively and urgently [18, 19, 21-24]. The hypotension happens from sympathetic nerve blockade followed by diminished peripheral vasodilation, vascular resistance, and peripheral pooling leading to reflex increase in heart rate [25-29]. Placental blood flow is directly related to maternal blood pressure, thus a reduction in maternal blood pressure can lead to placental hypo-perfusion and fetal asphyxia. [18, 19, 23, 30, 31]. Developing post-spinal maternal hypotension has various clinical manifestations such as nausea, vomiting, and dizziness, which often interferes with the surgery [8, 31, 32]. The high incidence of post-spinal hypotension suggests the need for multimodal protocols for its prevention and management [33]. Various methods have been considered appropriate to prevent this problem [18, 23, 34-36]. The use of crystalloid preloading has been advocated for decades as an effective regimen in the expansion of intravascular fluid volume and thus reducing hypotension [7, 32, 33, 37]. However, it doesn’t offer full protection and recent evidences has questioned its value [4, 14, 21, 29, 34, 38, 39]. Despite all crystalloid preloading regimens, the incidence of post-spinal hypotension remains still high [32].The use of vasopressors is more widely accepted as an effective method for decreasing post-spinal hypotension than crystalloid loading [1, 21, 40, 41]. Of the available vasopressors, ephedrine has been considered for long as the primary drug for the treatment of post-spinal hypotension, especially in obstetric mothers [14, 18, 30, 34]. It can be the vasopressor of choice to control spinal induced hypotension, but administering it as a prophylaxis to prevent spinal-induced hypotension is a newly developing technique [23]. In our country, parturient mothers who undergo elective caesarian delivery under spinal anesthesia are expected to receive crystalloid preloading preoperatively. Although a formally documented literature doesn’t exit, hypotension is a common experience despite receiving crystalloid preloading in which at times challenge to the anesthetic management of such incidents. Therefore, this study was conducted with the purpose of comparing the efficacy of preloading a parturient with crystalloid fluid versus administering ephedrine as a prophylaxis soon after spinal anesthesia is given to prevent the occurrence of hypotension. In the current study, we hypothesized that administration of ephedrine prophylaxis could have a better outcome in the prevention of spinal anesthesia induced hypotension.
2. Methods
Study design & Setting:
This was a single center, prospective, double blinded, randomized comparative clinical trial conducted from December 2021 to February 2022. The study was conducted in Orotta National Referral Maternity Hospital, located in Asmara, the capital of Eritrea. The hospital is the only national referral maternity hospital providing healthcare at a tertiary level to all patients with gynecology, maternal and obstetric cases from all zones of the country. After subject assessment, 63 parturient mothers appointed for elective cesarean delivery were allocated to each parallel group (crystalloid or ephedrine) in a 1:1 ratio. The study protocol was approved by the ethical committee of Orotta College of Medicine and Health Sciences and Ministry of Health at the Department of Research and Human Resource Development with an approval date on 21/01/2022 (Reff No: 01/0/22). The clinical trial was registered in the Pan African Clinical Trial Registry (Clinical Trial Registration Number: PACTR202302481261045, Date registered: 03/02/2023). The study was performed in accordance with the principles of the Declaration of Helsinki, and after explaining the objective of the study, written informed consent was obtained from each participant prior to enrollment. All methods were performed in accordance with relevant guidelines and regulations.
Participant selection and Randomization:
The study comprised sixty parturient mothers who were scheduled for elective cesarean delivery under spinal anesthesia within the study period. Additional inclusion criteria included, patients with physical status of ASA I and II, those with full term of a single pregnancy, and mothers with the age range of 18 to 40 years. Patients who didn’t consent to participate in the study, those who were not fit for spinal anesthesia, those with a history of allergic reactions to local anesthetics, those with preceding or pregnancy-induced hypertension, patients with coagulopathy of any cause, patients with severe cardiac, respiratory, hepatic or renal disease and pre-eclamptic and eclamptic patients and those who developed emergency conditions during the intraoperative period were excluded from the study. Sample size was calculated using a sample size formula for the difference of two independent samples given by [42]: n ≥ (Zα/2+Zβ)2 *2*σ2 / d2, where Zα/2 is the critical value set at α = 0.05 given by 1.96, Zβ is the critical value of the Normal distribution at β (0.84), σ2 is the population variance (324), and d is the difference needed to be detected (15); was used to get 23 individuals in each group. The study being undergone for the first time, due care was made to avoid unexpected attrition and hence 20% potential non-response was suggested to recommend at least 29 patients in each group. Enrollment and random allocation of the patients to either group were conducted by one independent health care personnel who was not involved in the study in any respect. Patients were randomly assigned following simple randomization procedures. A computer generated list of random numbers was used and patients were randomly assigned to either of the two groups using sealed envelope method. Study participants, perioperative care providers and the data analyst were blinded to the allocated groups. Only the anesthetist attending the surgery knew the allocated groups to enable drug administration, monitoring and management.
Data collection tools:
The key elements of socio-demographic and clinical variables of the patients were obtained using a socio-demographic and clinical form. The variables were age, height, weight, gravid, parity, previous C/S, reason for C/S, and level of block. A checklist was adopted from a similar study conducted in Egypt [22]. The tool was used to score the change in the vital signs. The recorded and assessed vital signs were blood pressure and pulse rate. Content validity of the socio-demographic form and the checklist was insured by expert opinions from the anesthesia department. Pretest was done among 10 patients for the purpose of assessing the comprehension and applicability of the checklist in ascertaining the acquisition of the desired data.
Interventions:
Patients initially were encountered during the preoperative assessment period. After obtaining consent, a thorough pre-anesthetic evaluation was done by taking a detailed history and conducting a physical examination. Routine preoperative laboratory investigations were conducted for the evaluation of patient physical status. No premedication was given. Patients were kept fasting for the night of 8 hours period of time. During the preoperative period patient enrollment and randomization was conducted, and patients were allocated in either of the two groups. In the preparation room, an intravenous 18G cannula was inserted. Upon arrival to the operating room, patients were attached to vital sign monitoring instruments and continuous monitoring was carried out with noninvasive blood pressure evaluation and pulse oximetry. Baseline vital signs were recorded before the start of the procedure. The crystalloid group received the usual preoperative management which is a one liter of preloading normal saline 15 minutes before spinal injection. During the intraoperative period, the intravenous line was secured with a lactated Ringer’s solution for all patients in both groups. During spinal injection, patients were made in sitting position, spinal puncture was performed under aseptic precautions with a G25 Fr at the L3/4 or L4/5 interspace, and 2 ml of 5% hyperbaric bupivacaine was administered. Patients were made in a supine position immediately after spinal injection. They were followed for block progression and the level of the block was recorded. The ephedrine group were started on the trial management which is ephedrine prophylaxis; initially 5mg was given 1 minute after the spinal injection. Another 5mg was given again 1 minute later. Then, the patients received 1mg of ephedrine every 1 minute until the 15th minute. The dose of ephedrine was made based on a previously conducted study [22]. Patients in both groups received 10 IU oxytocin (5 IU intravenously and 5 IU in 500 ml of the running ringer lactated solution) after the expulsion of the fetus from the uterus.
Outcome measurements:
The primary endpoint of this study was the measurement of the vital signs namely systolic blood pressure and pulse rate. They were recorded immediately in the first minute after spinal injection and then every three minutes in the prepared checklist until the surgery was finished. The secondary endpoint was recording the occurrence of complications which are hypotension, which is defined as a 20% reduction of blood pressure from the baseline, as well as nausea and vomiting. Hypotension was treated immediately with 5 mg bolus IV ephedrine. Patients were also vigilantly followed during the postoperative period for the usual anesthetic follow-up or for the occurrence of any additional conditions related to the intervention conducted. After completing with each patient, the completeness and consistency of each checklist was checked throughout the procedure and after the data from all the subjects was collected data was analyzed. Data Analysis: Data was entered in the SPSS (Version 26) software for analysis. Preliminary cleaning and exploratory investigations for normality assessments were done before conducting the main analysis. Demographic data was described using mean (SD) or frequency (percentage) as appropriate. Mean (SD) values of the recordings at various time periods were computed. The primary endpoint was the vital signs including systolic blood pressure and pulse rate after the administration of spinal anesthesia. Comparison of those vital signs across the two groups were conducted using the independent samples t-test, after assessing the equality of variance through Levene’s Test. Secondary endpoints, incidence of hypotension as well as nausea and vomiting, were assessed using chi-square test. The effect of age, height, weight and parity were adjusted using ANCOVA. Statistical significance was considered significant at p<0.05.
3. Results
Of the 65 parturient mothers who were assessed for eligibility, 63 of them who fulfilled the inclusion criteria were randomly allocated into the two study groups, and 60 patients completed they study (fig. 1). Two patients didn’t satisfy the inclusion/exclusion criteria; 3 patients were excluded due to persistent hypotension that did not respond to the rescue doses of ephedrine. As indicated in Table 1, demographic variables of the patients showed no significant difference between the two groups except for parity. The most common (83.3%) cause for the cesarean section in both groups was a previous history of cesarean delivery. The rest of the demographic and clinical characteristics of the patients are depicted in Table 1.
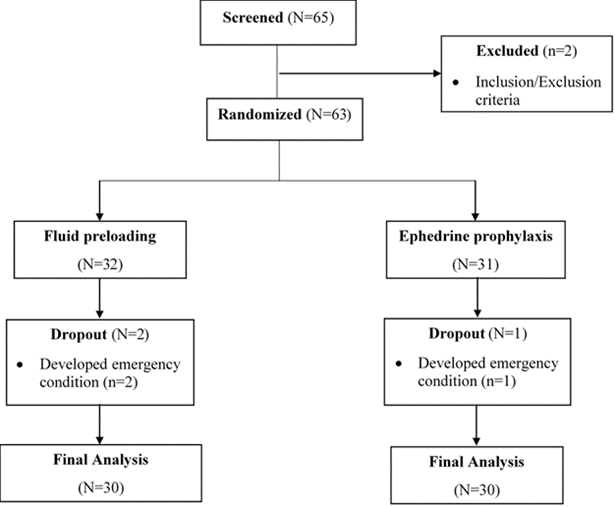
Table 1: Demographic and clinical characteristics of the study participants
|
Variables |
Preloading Group |
Ephedrine Group |
p-value |
|
(n=30) |
(n=30) |
||
|
Age, M + SD |
32.13 + 4.981 |
29.83 + 4.145 |
0.057 |
|
Height, M + SD |
162 + 6.953 |
163.87 + 6.447 |
0.285 |
|
Weight, M + SD |
69.93 + 6.554) |
67.23 + 5.594) |
0.091 |
|
Reason for C/S, N (%) |
0.603 |
||
|
Previous C/S |
21(70) |
25 (83.3) |
|
|
CPD |
2 (6.7) |
1 (3.3) |
|
|
Malpresentation |
3 (10) |
1 (3.3) |
|
|
Others |
4 (13.3) |
3 (10) |
|
|
Previous Hx of C/S, N (%) |
0.360f |
||
|
Yes |
21 (70) |
25 (83.3) |
|
|
No |
9 (30) |
5 (16.7) |
|
|
Parity, N (%) |
0.039f |
||
|
Zero |
8 (26.7) |
3 (10) |
|
|
One |
3 (10) |
13 (43.3) |
|
|
Two |
12 (40) |
7 (23.3) |
|
|
Three |
5 (16.7) |
4 (13.3) |
|
|
> Four |
2 (6.7) |
3 (10) |
|
|
Level of spinal anesthesia block, N (%) |
0.401f |
||
|
T5 |
0 (0) |
3 (10) |
|
|
T6 |
23 (76.7) |
20 (66.7) |
|
|
T7 |
6 (20) |
6 (20) |
|
|
T8 |
1 (3.3) |
1 (3.3) |
|
Effect of crystalloid preloading versus ephedrine prophylaxis on the vital signs: Blood pressure of every patient in both groups was recorded every three minutes and the mean value for every record as well as the overall perioperative mean systolic blood pressure was computed. As it can be observed in table 2, the baseline systolic blood pressure of the two study groups was comparable with no significant difference at both bivariate and multivariate levels. After the administration of spinal anesthesia, monitoring blood pressure was continued and significant differences were observed in the measures of systolic blood pressure and it was higher in the ephedrine group in the 7th (p<0.05), 10th (p<0.05), 13th (p<0.05), 16th (p<0.001), 19th (p<0.0001), 22nd (p<0.0001), 25th (p<0.001) minutes. Analysis at multivariable level showed significant difference at 16th (p=0.002), 19th (p<0.0001), 22nd (p=0.001) minutes. Meanwhile, the overall perioperative mean systolic blood pressure was also highly significant (p=0.004) at multivariable level (Table 2). Regarding the score of pulse rate, the baseline was similarly comparable with non-significant difference (p>0.05) at all times (Table 3).
Table 2: Comparison of systolic blood pressure between the two study groups at bivariate and multivariate levels
|
Bivariate Analysis |
Multivariable Analysis τ |
|||||
|
Preload M (SD) |
Ephedrine M (SD) |
MD (95% CI) |
p-value |
Effect size |
Power |
|
|
Baseline |
132.50 (13.23) |
130.2 (12.84) |
2.30 (-4.44, 9.04) |
0.441 |
0.011 |
0.119 |
|
1 min |
117.13 (21.88) |
111.47 (17.46) |
5.57 (-4.57, 15.90) |
0.275 |
0.022 |
0.191 |
|
4 mins |
104.27 (21.39) |
110.57 (20.02) |
-6.30 (-17.01, 4.41) |
0.088 |
0.053 |
0.4 |
|
7 mins |
105.4 (17.90) |
115.77 (15.68) |
-10.37 (-19.06, -1.67)* |
0.052 |
0.068 |
0.496 |
|
10 mins |
108.07 (19.24) |
119.00 (14.09) |
10.93 (-19.65, -2.27)* |
0.057 |
0.065 |
0.497 |
|
13 mins |
107.63 (18.20) |
116.93 (15.08) |
-9.30 (-17.94, -0.66)* |
0.137 |
0.04 |
0.317 |
|
16 mins |
103.17 (16.58) |
117.33 (15.38) |
-14.17 (-22.43, -5.90)** |
0.002 |
0.166 |
0.897 |
|
19 mins |
100.60 (13.96) |
117.7 (12.37) |
-17.10 (-24.79, -9.41)*** |
<0.0001 |
0.255 |
0.96 |
|
22 mins |
102.28 (12.04) |
120.73 (13.65) |
-18.47 (-27.58, -934)*** |
0.001 |
0.318 |
0.928 |
|
25 mins |
100.25 (13.80) |
117.3 (15.38) |
-17.05 (-28.89, -5.21)** |
0.017 |
0.39 |
0.72 |
|
28 mins |
105.00 (12.19) |
117.6 (11.76) |
-12.60 (-30.07, 4.87) |
0.219 |
0.346 |
0.204 |
|
Overall |
106.43 (13.96) |
117.20 (10.19) |
-10.77 (-17.08, -4.45)** |
0.004 |
0.14 |
0.83 |
τMultivariable analysis was conducted after controlling the effect of age, parity, height and weight.
*p<0.05 **p<0.001 ***p<0.0001
Table 3: Comparison of pulse rate between the two study groups using independent samples t-test
|
Pulse rate |
Preload group M (SD) |
Ephedrine group M(SD) |
MD (95% CI) |
|
Baseline |
96.63 (13.61) |
98.97 (13.96) |
-2.333 (-9.460, 4.793) |
|
1st min |
92.77 (20.28) |
99.5 (18.75) |
-6.733 (-16.826, 3.360) |
|
4th mins |
93.2 (22.66) |
98.23 (22.51) |
-5.033 (-16.704, 6.637) |
|
7th mins |
93.23 (15.42) |
93.8 (20.23) |
-0.567 (-9.862, 8.729) |
|
10th mins |
93.1 (13.11) |
90.1 (19.91) |
3 (-5.740, 11.740) |
|
13th mins |
94.63 (15.00) |
95.17 (17.69) |
-0.533 (-9.007, 7.941) |
|
16th mins |
93.33 (15.27) |
99.57 (16.07) |
-6.233 (-14.335, 1.868) |
|
19th mins |
90.4 (14.00) |
94.74 (14.32) |
-4.339 (-12.570, 3.892) |
|
22nd mins |
91.21 (15.69) |
91.07 (12.67) |
0.144 (-10.024, 10.312) |
|
25th mins |
89.38 (17.05) |
91.7 (14.89) |
-2.325 (-18.284, 13.634) |
|
28th mins |
92 (19.76) |
87.2 (13.07) |
4.8 (-19.631, 29.231) |
|
Overall |
91.47 (11.30) |
95.30 (13.50) |
-3.83 (-10.27, 2.60) |
Incidence of spinal anesthesia related complications:
This incidence of complications was also computed (Table 4). The occurrence of hypotension was highly significant (p<0.0001) in the crystalloid group with 3.3% of the patients in the ephedrine group experiencing hypotension requiring rescue doses of ephedrine.
None of the patients experienced any adverse events related to the interventions given.
Table 4: Incidence of complications among the study groups
|
Complications |
Preloading n (%) |
Ephedrine n (%) |
chi square value |
p-value |
|
|
Hypotension |
18.26 |
<0.0001 |
|||
|
Yes |
14 (46.7) |
0 |
|||
|
No |
16 (53.3) |
30 (100) |
|||
|
Nausea & Vomiting |
15.02 |
<0.0001 |
|||
|
Yes |
16 (46.7) |
1 (3.3) |
|||
|
No |
16 (53.3) |
29 (96.7) |
|||
4. Discussion
The study was conducted with the goal of comparing the efficacy of ephedrine infusion versus crystalloid preloading in reducing the incidence of hypotension in patients undergoing spinal anesthesia for cesarean section. Spinal Anesthesia is a popular and commonly practiced mode of regional anesthesia for such a procedure, providing rapid onset and a dense neuraxial block [5, 22, 33, 43]. However, spinal anesthesia is often associated with significant maternal hypotension, a commonly occurring physiologic derangement occurring due to sympathetic blockade followed by peripheral vasodilation with potentially significant consequences on maternal comfort, organ perfusion, and fetal well-being [4, 5, 8, 43-47]. Development of hypotension is also associated with various unpleasant maternal manifestations such as nausea, vomiting, and dizziness, which often interfere with surgery [38, 48, 49].
Prevention of post-spinal hypotension in cesarean delivery has been frequently investigated for many years. There has been a considerable dispute over the use of different techniques for the prevention of post-spinal maternal hypotension. Despite the different available preventive and management techniques, it still remains with a higher incidence and challenging anesthetists [33, 45]. Hypotension becomes exaggerated by the deficiency of intravascular volume added to the sympathetic block by spinal anesthesia [22]. Techniques currently in use for the prevention of hypotension include crystalloid preloading and administration of vasopressors [6]. The traditional approach and the most commonly recommended method for the prevention of hypotension is optimizing venous return with crystalloid preloading with the aim of filling the dilated vessels, expanding the intravascular compartment and preventing the occurrence of hypotension due to such sympathetic blockade [14, 22, 50-52]. The mother is usually given one up to two liters of fluid, 15-20 minutes before the spinal block [3, 51]. However, strategies in which even higher crystalloid volumes are directed to increase venous return are not very effective in the treatment of arterial hypotension [4, 14, 32, 34, 53]. It has been mentioned that crystalloid solutions are poor plasma volume expanders and their administration before spinal block cannot eliminate or reduce the incidence of spinal induced hypotension [46, 54-56]. The limitation in the efficacy of crystalloid in the prevention of hypotension could be attributed to its rapid distribution to the extravascular space [57]. Hiroshi Uyeadma found in their study found that only 28% of the infused fluid remained in the intravascular space over 30 minutes, indicating that crystalloid fluids have a short intravascular half-life as they get rapidly distributed into the interstitial space suggesting the necessity of a greater increase in blood volume [29]. The use of vasopressors for the prevention of hypotension during cesarean delivery is well established and ephedrine is the traditionally and most widely used vasopressor in current practice [6, 22, 48, 58]. Several studies conducted in different sites, all came to the finding that the use of prophylactic ephedrine in either bolus or infusion form is a more efficient form than fluid preloading without causing significant tachycardia or hypertension [1, 2, 22, 23, 31, 43, 56]. Due to diminished action on adrenergic receptors, administration of ephedrine preserves sufficient utero-placental blood flow while maintaining maternal blood pressure. Moreover, Ashraful Anam reported that, although it was not statistically significant (p > 0.05), a higher heart rate was recorded in the ephedrine group and this could be explained both by the effect of rescue ephedrine and by baroreceptor-mediated reflex increase in heart rate in patients who became hypotensive [1]. Similar to these results, in this current study, the use of ephedrine as a prophylaxis maintained blood pressure better compared to crystalloid preloading thus is associated with better control of maternal hypotension. Though not significant, the fact that pulse rate was clinically higher in the ephedrine group despite higher measures of blood pressure could be explained by the effect of ephedrine in the baroreceptor-mediated reflex increase in heart rate. Furthermore, a bolus dose of 5 mg intravenous ephedrine has been mentioned to decrease the occurrence of the severity of hypotension in preloaded pregnant mother undergoing subarachnoid block for caesarian section [2, 22]. The same procedure was followed in this current study as well. Not all researches were in congruent with these findings and a contrasting finding was reported by a similar research conducted in Pakistan. The research concluded that prevention of hypotension to avoid its deleterious effects on both the mother and fetus can be attempted using simpler and more safer methods like preloading using lactated Ringer’s solution than the use of vasopressors reporting that they carry an inherent risk of anaphylaxis and other life threatening adverse effects [59]. For the mother, arterial hypotension causes very unpleasant signs and symptoms, the most common being nausea, vomiting, and dizziness, which are manifestations of cerebral hypo-perfusion. Prolonged episodes of severe hypotension can have serious consequences such as organ ischemia, loss of consciousness, cardiovascular collapse, and utero-placental hypo-perfusion [22, 31, 60]. Moreover, hypo-perfusion of the gut leads to the release of emetogenic substances like serotonin. In all above-mentioned studies, it is reported that the incidence of complications including nausea and vomiting were low in the group who took ephedrine prophylaxis, which was consistent with the findings of this current study. As the intervention is implemented at the only national referral hospital of the country where parturient mothers from all over the country with a wide variety of demographic and clinical characteristics do their visit, the management can be reliably and effectively delivered in different settings.
5. Conclusion
The study concluded that ephedrine as a prophylaxis is more effective than that of fluid preloading in preventing spinal-induced hypotension and maintaining a better hemodynamic status of parturient mothers undergoing caesarean section. The search for the effective dose of ephedrine for prophylactic administration is clinically important and we suggest future studies to be undertaken to identify it.
Acknowledgements
The authors would like to thank Mr. Eyasu Habte Tesfamariam for his great effort in the data analysis and improvement of the manuscript. The authors would also like to show our gratitude to all patient for their willingness and participation in the research.
Funding
No funding bodies played any role in study design, data collection and analysis, decision to publish, or preparation of this manuscript.
Authors’ contributions
YMA: Study conception and design of the study, analysis and interpretation of data, drafting, revising the manuscript critically for important intellectual content, and submission; DHB: Study conception and design of the study, acquisition of data, analysis and interpretation of data; JAM: Study conception and design of the study, acquisition of data, analysis and interpretation of data; TMH: Study conception and design of the study, acquisition of data, analysis and interpretation of data.
Data availability statement
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors declare that they have no competing interests.
References
- Anam MA, et al. Volume Preload versus Ephedrine Infusion for Prevention of Hypotension Due to Spinal Anesthesia for Cesarean Section. Med Today 31 (2019): 39-41.
- Vercauteren MP, Coppejans HC, Hoffmann VH, et al. Prevention of hypotension by a single 5-mg dose of ephedrine during small-dose spinal anesthesia in prehydrated cesarean delivery patients. Anesth Analg 90 (2000): 324-327.
- Thiangtham, K. and T. Asampinwat, Intravenous ephedrine infusion for prophylaxis of hypotension during spinal anesthesia for cesarean section. Songklanagarind Med J 27 (2010): 291-300.
- Morgan PJ, Halpern SH, and Tarshis J. The effects of an increase of central blood volume before spinal anesthesia for cesarean delivery: a qualitative systematic review. Anesth Analg 92 (2001): 997-1005.
- Mercier FJ, Augè M, Hoffmann C, et al. Maternal hypotension during spinal anesthesia for caesarean delivery. Minerva Anestesiol 79 (2013): 62-73.
- Varathan S, Ekanayake S, and Amarasinghe U. Comparison of prophylactic intramuscular ephedrine with preloading versus preloading alone in prevention of hypotension during elective caesarean section under subarachnoid block. Sri Lankan J Anaesthesiol 17 (2009).
- Cardoso MMSC, Bliacheriene S, Freitas CRC, et al. Preload during spinal anesthesia for cesarean section: comparison between crystalloid and colloid solutions. Rev Bras Anestesiol 54 (2004): 781-787.
- Rehman SJ, Sharif A, and Gulrez A. Prevention of Hypotention after Spinal Anaesthesia in Elective Caesarean Sections. Pakistan Armed Forces Med J 70 (2020).
- Hawkins JL, Arens JF, Bucklin BA, et al. Practice Guidelines for Obstetric Anesthesia: An Updated Report by the American Society of Anesthesiologists Task Force on Obstetric Anesth Anesthesiol 106 (2007): 843-863.
- Campbell J, Sultan P. Regional Anesthesia for Caesarean Section: A Choice of Three Techniques. British J Hospital Med 70 (2009): 605.
- Ng K. Anestesia raquídea versus epidural para la cesárea. Biblioteca Cochrane Plus (2008).
- Simmons S, Dennis AT, Cyna AM, et al. Analgesia espinal y epidural combinadas versus analgesia epidural en el trabajo de parto (Revisión Cochrane traducida). La Biblioteca Cochrane Plus (2008).
- Reidy J and J Douglas. Vasopressors in obstetrics. Anesthesiol Clin 26 (2008): 75-88.
- Mercier FJ. fluid loading for cesarean delivery under spinal anesthesia: have we studied all the options? Anesth Analg 113 (2011): 677-680.
- Shibli K and Russell I. A survey of anaesthetic techniques used for caesarean section in the UK in 1997. International J Obstetric Anesth 9 (2000): 160-167.
- Klöhr S, Roth R, HofmannT, et al. Definitions of hypotension after spinal anaesthesia for caesarean section: literature search and application to parturients. Acta Anaesthesiol Scand 54 (2010): 909-921.
- Hartmann B, Junger A, Klasen J, et al. The incidence and risk factors for hypotension after spinal anesthesia induction: an analysis with automated data collection. Anesth Analg 94 (2002): 1521-1529.
- Turkoz A, Togal T, Gokdeniz R, et al. Effectiveness of intravenous ephedrine infusion during spinal anaesthesia for caesarean section based on maternal hypotension, neonatal acid-base status and lactate levels. Anaesth Intensive care 30 (2002): 316-320.
- Lee A, WDN Kee and Gin T. Prophylactic ephedrine prevents hypotension during spinal anesthesia for Cesarean delivery but does not improve neonatal outcome: a quantitative systematic review. Canad J Anesth 49 (2002): 588-599.
- Neves JFNPD, Monteiro GA, Almeida JR, et al. Utilização da fenilefrina para controle da pressão arterial em cesarianas eletivas: dose terapêutica versus profilática. Rev Bras Anestesiol 60 (2010): 395-398.
- Butwick AM, Columb, Carvalho B. Preventing spinal hypotension during Caesarean delivery: what is the latest? Oxford University Press (2015): 183-186.
- Salama AK, Goma HM, Hamid BMA. fluid preloading versus ephedrine in the management of spinal anesthesia-induced hypotension in parturients undergoing cesarean delivery: a comparative study. Ain-Shams J Anaesthesiol 9 (2016): 72.
- Rehman A, Baig H, Rajput MZ, et al. Comparison of prophylactic ephedrine against prn ephedrine during spinal anesthesia for caesarian sections. Anaesth Pain Intensive Care 15 (2011): 21-24.
- Loughrey J, Yao N, Datta S, et al. Hemodynamic effects of spinal anesthesia and simultaneous intravenous bolus of combined phenylephrine and ephedrine versus ephedrine for cesarean delivery. Int J Obstet Anesth 14 (2005): 43-47.
- Bhagat H, Malhotra K, Sudhir, et al. Evaluation of preloading and vasoconstrictors as a combined prophylaxis for hypotension during subarachnoid anaesthesia. Indian J Anaesth 48 (2004): 299-303.
- Matthews C. A review of nausea and vomiting in the anaesthetic and post anaesthetic environment. J Perioper Pract 27 (2017): 224-228.
- Loubert C. fluid and vasopressor management for Cesarean delivery under spinal anesthesia: continuing professional development. Canad J Anaesth 59 (2012): 604-619.
- Imam SM, Ali CA, and Hussain S. The efficacy of prophylactic combination therapy using ephedrine iv alongwith fluid preload as compared to fluid preloading alone or IV ephedrine alone in prevention of maternal hypotension during spinal anesthesia for caesarean section. Pakistan J Med Health Sci 6 (20102): 573-576.
- Ueyama H, He YL, Tanigami H, et al. Effects of crystalloid and colloid preload on blood volume in the parturient undergoing spinal anesthesia for elective Cesarean section. Anesthesiology 91 (1999): 1571-1571.
- Alkaissi A, Ussbah Q, Al-Bargouthi A. Prophylactic ephedrine versus phenylephrine for maternal hypotension in women undergoing spinal anesthesia for caesarean section-a randomized double blind clinical trial. J Biomed Sci 6 (2017).
- Kee WDN, Khaw KS, Lee BB, et al. A dose-response study of prophylactic intravenous ephedrine for the prevention of hypotension during spinal anesthesia for cesarean delivery. Anaesth Analg 90 (2000): 1390-1395.
- Cyna AM, Middleton P, Chemali M, et al. Techniques for preventing hypotension during spinal anaesthesia for caesarean section. Cochrane Database Syst Rev 8 (2017).
- Hasanin A, Badawy AA, Fouad R, et al. Post-spinal anesthesia hypotension during cesarean delivery, a review article. Egyptian J Anaesth 33 (2017): 189-193.
- Kee WDN. Prevention of maternal hypotension after regional anaesthesia for caesarean section. Curr Opin Anesthesiol 23 (2010): 304-309.
- Kansal A, Mohta, A K Sethi, et al. Randomised trial of intravenous infusion of ephedrine or mephentermine for management of hypotension during spinal anaesthesia for Caesarean section. Anaesth 60 (2005): 28-34.
- Ko J, Kim C-S, Cho H-S, et al. A randomized trial of crystalloid versus colloid solution for prevention of hypotension during spinal or low-dose combined spinal-epidural anesthesia for elective cesarean delivery. Int J Obstet Anaesth 16 (2007): 8-12.
- Mercier FJ. Cesarean delivery fluid management. Curr Opin Anesthesiol 25 (2012): 286-291.
- Weeks S. Reflections on hypotension during Cesarean section under spinal anesthesia: do we need to use colloid? Canad J Anaesth 47 (2000): 607-610.
- Birnbach D, Soens M. Hotly debated topics in obstetric anesthesiology 2008: a theory of relativity. Minerva Anestesiol 74 (2008): 409-424.
- Sharwood-Smith G and Drummond G. Hypotension in obstetric spinal anaesthesia: a lesson from pre-eclampsia. Oxford University Press (2009): 291-294.
- Abdul H, Shaharbano S, Khojeste J. Ephedrine for prevention of hypotension comparison between intravenous, intramuscular and oral administration during spinal anaesthesia for elective caesarean section. Professional Med J 14 (2007): 610-615.
- Rosner B. Fundamentals of biostatistics. Cengage learning (2015).
- Ahmed HO, Hossam M, Adel A. Volume preload versus ephedrine infusion for prevention of hypotension due to spinal anesthesia for cesarean section. Open J Anesthesiol 6 (2016): 37-44.
- Varshney R. and Jain G. Comparison of colloid preload versus coload under low dose spinal anesthesia for cesarean delivery. Anesth Essays Res 7 (2013): 376.
- Oh A-Y, Hwang J-W, Song In-Ae, et al. Influence of the timing of administration of crystalloid on maternal hypotension during spinal anesthesia for cesarean delivery: preload versus coload. BMC Anesthesiol 14 (2014): 1-5.
- Langesæter E and Dyer RA. Maternal haemodynamic changes during spinal anaesthesia for caesarean section. Curr Opin Anesthesiol 24 (2011): 242-248.
- Seyedhejazi M and Madarek E.The effect of small dose bupivacaine-fentanyl in spinal anesthesia on hemodynamic nausea and vomiting in cesarean section. Pakistan J Med Sci 23 (2007): 747-750.
- Loughrey J, Walsh F, Gardiner J. Prophylactic intravenous bolus ephedrine for elective Caesarean section under spinal anaesthesia. European J Anaesthesiol 19 (2002): 63-68.
- Sng B, Tan H, Sia A. Closed-loop double-vasopressor automated system vs manual bolus vasopressor to treat hypotension during spinal anaesthesia for caesarean section: A randomised controlled trial. Anaesth 69 (2014): 37-45.
- Khan MU, Memon AS, Ishaq M, et al. Preload versus coload and vasopressor requirement for the prevention of spinal anesthesia induced hypotension in nonobstetric patients. J Coll Physicians Surg Pak 25 (2015): 851-855.
- Gunusen I, Karaman S, Ertugrul V, et al. Effects of fluid preload (crystalloid or colloid) compared with crystalloid co-load plus ephedrine infusion on hypotension and neonatal outcome during spinal anaesthesia for caesarean delivery. Anaesth Intensive Care 38 (2010): 647-653.
- Dyer R, Farina Z, Joubert IA, et al. Crystalloid preload versus rapid crystalloid administration after induction of spinal anaesthesia (coload) for elective caesarean section. Anaesth Intensive Care 32 (2004): 351-357.
- Tercanli S, Schneider M, Visca E, et al. Influence of volume preloading on uteroplacental and fetal circulation during spinal anaesthesia for caesarean section in uncomplicated singleton pregnancies. Fetal Diagn Ther 17 (2002): 142-146.
- Tawfik M, Hayes SM, Jacoub FY, et al. Comparison between colloid preload and crystalloid co-load in cesarean section under spinal anesthesia: a randomized controlled trial. Int J Obstet Anesth 23 (2014): 317-323.
- Lotfy, ME. Colloid versus crystalloid co-load with spinal anesthesia during emergent cesarean section and their effect on hemodynamic changes. J Am Sci 10 (2014): 158-163.
- Madhusoodana R, Bhovi A. Comparitive Study of Ephedrine Infusion with the Preload of Crystalloids for Prevention of Hypotension during Spinal Anaesthesia for Elective Caesarean Section. Indian J App Res 4 (2014): 2249-2255.
- Lee A, Kee WDN, Gin T. A quantitative, systematic review of randomized controlled trials of ephedrine versus phenylephrine for the management of hypotension during spinal anesthesia for cesarean delivery. Anaesth Analg 94 (2002): 920-926.
- Comparison of intravenous ephedrine with phenylephrine for the maintenance of arterial blood pressure during elective caesarean section under spinal anaesthesia. Faculty Anaesth (2018).
- Shridhar N, Ekbote A, Murthy S. Effectiveness of Preloading in prevention of Spinal Induced Hypotension during LSCS. J Evol Med Dental Sci 3 (2014): 1420-1425.
- Macarthur A and Riley ET. Obstetric anesthesia controversies: vasopressor choice for postspinal hypotension during cesarean delivery. Int Anesthesiol Clin 45 (2007): 115-132.
