Clinical Evidence of Ventilator-Induced Lung Injury Vortex in Patients with SARS-CoV-2
Article Information
Néstor Pistillo1,3*, Pablo Castelluccio1, Ricardo Ciano1, Eduardo Palermo1,2, Sergio Lage1,3, Edgar Amundarain1,3, Osvaldo H Fariña1,2
1Intensive Care Unit, Hospital de Alta Complejidad en Red El Cruce-Néstor C. Kirchner, Buenos Aires, Argentina
2Universidad de Buenos Aires
3Universidad Nacional Arturo Jaureche
*Corresponding Author: Néstor Pistillo, Intensive Care Unit, Hospital de Alta Complejidad en Red El Cruce-Néstor C. Kirchner, Buenos Aires, Argentina
Received: 08 September 2021; Accepted: 15 September 2021; Published: 22 September 2021
Citation:
Néstor Pistillo, Pablo Castelluccio, Ricardo Ciano, Eduardo Palermo, Sergio Lage, Edgar Amundarain, Osvaldo H Fariña. Clinical Evidence of Ventilator-Induced Lung Injury Vortex in Patients with SARS-CoV-2. Anesthesia and Critical Care 3 (2021): 29-43.
View / Download Pdf Share at FacebookAbstract
Background
The concept of ventilator-induced lung injury vortex (VILI vortex) has recently been proposed as a progressive lung injury mechanism in which the alveolar stress/strain increases as the ventilable lung “shrinks”.
Research Question
The objective of this study is to evaluate the incidence of the VILI vortex in patients with SARS-CoV-2.
Study design and methods
Patients were ventilated with low tidal volume (Vt: 6 ±1 ml/kg/PBW) and limited plateau pressure (PPlat <30 cmH2O). FiO2 and respiratory frequency were adjusted to obtain SaO2: 92 to 96% and pH >7.20, respectively. PEEP was scored based on best compliance of respiratory system. Data were registered for 14 days. VILI vortex was defined as a progressive and sustained increase in driving pressure (ΔP) in the context of constant or decreasing tidal volume.
Results
Sixty-five patients were admitted and fifteen of them (23%) progressed to VILI vortex. This phenomenon started subtly but developed fast (ΔP: day 0: 13.1 ±3.6 cmH2O, day 7: 15.6 ±3.6 vs. ΔP day 14: 24.1 ±7.8 cmH2O, p 0.0007). Initially, mechanical impairment was not accompanied by worsening in oxygenation [PaO2/FiO2 day 0: 150 ±73 vs. day 14: 160 ±75), p= NS], thus the PaO2/FiO2 ratio was not sensitive enough for its early detection. Finally, almost all patients with VILI vortex died of refractory hypoxemia (14/15). Negative factors such as delay in invasive mechanical ventilation [14 (11-15) vs. 7.5 (5-11) days, p= 0.0005], fluid overload [965 (501-1280) vs. 471 (206-.730), ml/day, p= 0.002], presence of bacteremia (60 vs. 20%, p 0.002), persistent fever (46.6 vs. 20%, p: 0.03) and need for noradrenaline >0.1 γ/kg/min (80 vs. 34%, p= 0.001) prevailed in VILI vortex group.
Interpretation
VILI vortex
Keywords
<p>Acute respiratory distress syndrome, Ventilator-induced lung injury, Pneumonia, SARS-CoV-2, Refractory hypoxemia, Mechanical ventilation</p>
Acute respiratory distress syndrome articles Acute respiratory distress syndrome Research articles Acute respiratory distress syndrome review articles Acute respiratory distress syndrome PubMed articles Acute respiratory distress syndrome PubMed Central articles Acute respiratory distress syndrome 2023 articles Acute respiratory distress syndrome 2024 articles Acute respiratory distress syndrome Scopus articles Acute respiratory distress syndrome impact factor journals Acute respiratory distress syndrome Scopus journals Acute respiratory distress syndrome PubMed journals Acute respiratory distress syndrome medical journals Acute respiratory distress syndrome free journals Acute respiratory distress syndrome best journals Acute respiratory distress syndrome top journals Acute respiratory distress syndrome free medical journals Acute respiratory distress syndrome famous journals Acute respiratory distress syndrome Google Scholar indexed journals Ventilator-induced lung injury articles Ventilator-induced lung injury Research articles Ventilator-induced lung injury review articles Ventilator-induced lung injury PubMed articles Ventilator-induced lung injury PubMed Central articles Ventilator-induced lung injury 2023 articles Ventilator-induced lung injury 2024 articles Ventilator-induced lung injury Scopus articles Ventilator-induced lung injury impact factor journals Ventilator-induced lung injury Scopus journals Ventilator-induced lung injury PubMed journals Ventilator-induced lung injury medical journals Ventilator-induced lung injury free journals Ventilator-induced lung injury best journals Ventilator-induced lung injury top journals Ventilator-induced lung injury free medical journals Ventilator-induced lung injury famous journals Ventilator-induced lung injury Google Scholar indexed journals Pneumonia articles Pneumonia Research articles Pneumonia review articles Pneumonia PubMed articles Pneumonia PubMed Central articles Pneumonia 2023 articles Pneumonia 2024 articles Pneumonia Scopus articles Pneumonia impact factor journals Pneumonia Scopus journals Pneumonia PubMed journals Pneumonia medical journals Pneumonia free journals Pneumonia best journals Pneumonia top journals Pneumonia free medical journals Pneumonia famous journals Pneumonia Google Scholar indexed journals SARS-CoV-2 articles SARS-CoV-2 Research articles SARS-CoV-2 review articles SARS-CoV-2 PubMed articles SARS-CoV-2 PubMed Central articles SARS-CoV-2 2023 articles SARS-CoV-2 2024 articles SARS-CoV-2 Scopus articles SARS-CoV-2 impact factor journals SARS-CoV-2 Scopus journals SARS-CoV-2 PubMed journals SARS-CoV-2 medical journals SARS-CoV-2 free journals SARS-CoV-2 best journals SARS-CoV-2 top journals SARS-CoV-2 free medical journals SARS-CoV-2 famous journals SARS-CoV-2 Google Scholar indexed journals Refractory hypoxemia articles Refractory hypoxemia Research articles Refractory hypoxemia review articles Refractory hypoxemia PubMed articles Refractory hypoxemia PubMed Central articles Refractory hypoxemia 2023 articles Refractory hypoxemia 2024 articles Refractory hypoxemia Scopus articles Refractory hypoxemia impact factor journals Refractory hypoxemia Scopus journals Refractory hypoxemia PubMed journals Refractory hypoxemia medical journals Refractory hypoxemia free journals Refractory hypoxemia best journals Refractory hypoxemia top journals Refractory hypoxemia free medical journals Refractory hypoxemia famous journals Refractory hypoxemia Google Scholar indexed journals Mechanical ventilation articles Mechanical ventilation Research articles Mechanical ventilation review articles Mechanical ventilation PubMed articles Mechanical ventilation PubMed Central articles Mechanical ventilation 2023 articles Mechanical ventilation 2024 articles Mechanical ventilation Scopus articles Mechanical ventilation impact factor journals Mechanical ventilation Scopus journals Mechanical ventilation PubMed journals Mechanical ventilation medical journals Mechanical ventilation free journals Mechanical ventilation best journals Mechanical ventilation top journals Mechanical ventilation free medical journals Mechanical ventilation famous journals Mechanical ventilation Google Scholar indexed journals Positive end expiratory pressure articles Positive end expiratory pressure Research articles Positive end expiratory pressure review articles Positive end expiratory pressure PubMed articles Positive end expiratory pressure PubMed Central articles Positive end expiratory pressure 2023 articles Positive end expiratory pressure 2024 articles Positive end expiratory pressure Scopus articles Positive end expiratory pressure impact factor journals Positive end expiratory pressure Scopus journals Positive end expiratory pressure PubMed journals Positive end expiratory pressure medical journals Positive end expiratory pressure free journals Positive end expiratory pressure best journals Positive end expiratory pressure top journals Positive end expiratory pressure free medical journals Positive end expiratory pressure famous journals Positive end expiratory pressure Google Scholar indexed journals Inspiratory flow rate articles Inspiratory flow rate Research articles Inspiratory flow rate review articles Inspiratory flow rate PubMed articles Inspiratory flow rate PubMed Central articles Inspiratory flow rate 2023 articles Inspiratory flow rate 2024 articles Inspiratory flow rate Scopus articles Inspiratory flow rate impact factor journals Inspiratory flow rate Scopus journals Inspiratory flow rate PubMed journals Inspiratory flow rate medical journals Inspiratory flow rate free journals Inspiratory flow rate best journals Inspiratory flow rate top journals Inspiratory flow rate free medical journals Inspiratory flow rate famous journals Inspiratory flow rate Google Scholar indexed journals Extra corporeal membrane oxygenation articles Extra corporeal membrane oxygenation Research articles Extra corporeal membrane oxygenation review articles Extra corporeal membrane oxygenation PubMed articles Extra corporeal membrane oxygenation PubMed Central articles Extra corporeal membrane oxygenation 2023 articles Extra corporeal membrane oxygenation 2024 articles Extra corporeal membrane oxygenation Scopus articles Extra corporeal membrane oxygenation impact factor journals Extra corporeal membrane oxygenation Scopus journals Extra corporeal membrane oxygenation PubMed journals Extra corporeal membrane oxygenation medical journals Extra corporeal membrane oxygenation free journals Extra corporeal membrane oxygenation best journals Extra corporeal membrane oxygenation top journals Extra corporeal membrane oxygenation free medical journals Extra corporeal membrane oxygenation famous journals Extra corporeal membrane oxygenation Google Scholar indexed journals Compliance of the respiratory system articles Compliance of the respiratory system Research articles Compliance of the respiratory system review articles Compliance of the respiratory system PubMed articles Compliance of the respiratory system PubMed Central articles Compliance of the respiratory system 2023 articles Compliance of the respiratory system 2024 articles Compliance of the respiratory system Scopus articles Compliance of the respiratory system impact factor journals Compliance of the respiratory system Scopus journals Compliance of the respiratory system PubMed journals Compliance of the respiratory system medical journals Compliance of the respiratory system free journals Compliance of the respiratory system best journals Compliance of the respiratory system top journals Compliance of the respiratory system free medical journals Compliance of the respiratory system famous journals Compliance of the respiratory system Google Scholar indexed journals
Article Details
Topics central to the study
VILI vortex
Abbreviations
ARDS: Acute respiratory distress syndrome
VILI: Ventilator-induced lung injury
Vt: Tidal volume
ΔP: Driving pressure
VI: Inspiratory flow rate
RR: Respiratory rate
PEEP: Positive end expiratory pressure
MP: Mechanical power
FRC: Functional residual capacity
ICU: Intensive care unit
VCV: Volume-controlled ventilation
PPlat: plateau pressure
PPeak: Peak pressure
ECMO: Extra corporeal membrane oxygenation
CRS: Compliance of the respiratory system
SMP: Specific mechanical power
APACHE II: Acute physiology and chronic health disease classification system II
SOFA: Sepsis related organ failure Assessment
ΔΔP: VILI vortex intensity
LDH: Lactate dehydrogenase
P-SILI: Self-inflicted lung injury
Introduction
Mechanical ventilation is an essential tool for the treatment of patients with acute respiratory distress syndrome (ARDS). However, as with other strategies, it is not free of complications. Inadequate ventilation may have a negative impact on pulmonary and systemic hemodynamics, and it could both cause structural damage to pulmonary parenchyma and activate inflammation [1]. This process is known as ventilator-induced lung injury (VILI) and can promote the development of multiple organ failure and eventually death [2-4]. VILI results from the interaction between the mechanical load applied to the ventilable lung and its capacity to tolerate it. Factors such as tidal volume (Vt), driving pressure (ΔP), inspiratory flow rate (VI), respiratory rate (RR), excessive inspiratory effort, high levels of FiO2 and, in some cases, positive end-expiratory pressure (PEEP), have been involved in damage mechanism [2-9]. In that sense, the concept of mechanical power (MP) tries to encompass most of these factors within a measurable unit [10,11]. Furthermore, the decrease in ventilable lung volume (baby lung concept) [12], the heterogeneous lung compromise in ARDS [13], and the presence of cofactors that have a negative impact on the lung (fluid overload, presence of sepsis or shock) could increase its susceptibility to damage [14]. Since the mechanical conditions of the lung change dynamically with the progression of the disease, the ventilatory strategy needs constant adjustments to maintain a balance between the load and the size of the ventilable lung (concept of ergonomic ventilation). In fact, a protective ventilatory strategy of low tidal volume (Vt: 6 ml/kg/PBW) and limited plateau pressure (PPlat <30 cmH2O) may cause damage if the functional residual capacity (FRC) decreases significantly, thus making a lower number of alveoli (including capillaries) withstand a higher mechanical load per unit. The concept of VILI vortex has recently been proposed as a progressive lung injury mechanism in which the alveolar stress/strain increases as the ventilable lung “shrinks”. This positive feedback inexorably leads to the acceleration of lung damage, with potentially irreversible results [15]. Little is known about the clinical aspects of this condition. Understanding its behavior could contribute to changing its potential devastating impact. The objective of this study is to evaluate the incidence of the VILI vortex in patients with SARS-CoV-2, to establish a connection between this phenomenon and mortality, identifying the factors that influence its development.
Materials and Methods
Inclusion criteriaThis single-center prospective observational study evaluated consecutively admitted adult patients with ARDS severe acute respiratory distress syndrome due to COVID-19 (SARS-CoV-2) and needed volume-controlled ventilation (VCV) for at least 7 days. These eligibility criteria were decided to allow a minimum monitoring of the mechanical variables. Those who did not meet such requirement were excluded.
ARDS was defined according to the Berlin criteria16 while the diagnosis of COVID-19 pneumonia was established based on CT images showing such condition and on real-time polymerase chain reaction (RT-PCR) test for coronavirus disease-2019.
Exclusion criteria
Patients with do-not-resuscitate orders and pregnant women. Cardiac arrest before ICU admission. Extra corporeal membrane oxygenation (ECMO) requirement within the first 24 h of ICU admission and chronic obstructive pulmonary disease with gold class 3 or 4, or home oxygen therapy17.
ProcedurePatients were administered sedatives and analgesics (propofol and fentanyl) to obtain a score of -5 on the RASS scale. The protective ventilatory strategy included: low tidal volume ventilation (6 ml/kg/PBW), plateau pressure limitation (PPlat: <30 cmH2O) and prone positioning for severe hypoxemia. FiO2 and PaCO2 were adjusted to achieve SaO2 between 92 and 96% and pH >7.20, respectively. PEEP was titrated according to the best compliance of the respiratory system (CRS), without exceeding the PPlat limit set. Neuromuscular blockers were administered to patients suffering from severe ARDS (PaO2/FiO2 <100) and to the ones with hypoxemia associated with asynchronies despite the administration of sedoanalgesia, PPlat >32 cmH2O or that needed prone positioning to improve oxygenation.
Mechanical variables and PaO2/FiO2 were registered daily for 14 days or until initiating assisted ventilation. These data were obtained in passive mechanical conditions.
Ventilator-induced lung injury vortex was defined as a progressive increase in driving pressure (ΔP) as Vt remained constant or even decreased.
Refractory hypoxemia was defined as PaO2/FiO2 <100 despite the optimization of mechanical ventilation and prone positioning.
Respiratory data assessment
Peak airway pressure, plateau pressure, and total PEEP were measured. The value of driving pressure (ΔP: PPlat - PEEP total), static compliance of the respiratory system (CRS= Vt/ΔP) and specific mechanical power (SMP: mechanical power/CRS) were calculated. Mechanical power was computed as: Peak airway pressure x Vt - (ΔP x Vt/2) x respiratory rate x 0.098.
Clinical complications assessmentThe following variables and complications were also observed during the period of analysis: incidence of: 1) pneumonia associated with mechanical ventilation14, 2) need for noradrenaline over 0·1 γ/kg/min for more than 24 h, 3) positive blood cultures, 4) accumulated fluid balance, dialysis treatment, 5) clinical and/or echocardiographic evidence of heart failure, 6) lactate ≥2 mmol/L in at least two consecutive samples, 7) presence of persistent fever (≥38º at least once a day for three consecutive days), and 8) the highest value of ferritin, D-dimer, C-reactive protein, troponin I and LDH obtained during the first 14 days of invasive mechanical ventilation.
Statistical analysis
Dichotomic data are reported as frequencies and percentages while continuous data are presented as mean (standard deviation) or median (interquartile range) as appropriate, depending on the normality of distribution. Kolmogorov–Smirnov test was used to test normality of distribution for continuous variables. Comparisons between the two groups (VILI vortex vs. No VILI vortex) and intragroup (day: 7 vs. day: 14) were analyzed by either an independent t-test or the Mann–Whitney test. Differences in categorical variables were assessed using the χ2 or Fisher's exact test.
The mortality predictive model following refractory hypoxemia as well as mortality for any reason were analyzed on day 90 by means of ROC curves. Survival difference between both groups was analyzed through longrank test. Cox's proportional hazards regression test was used to study the relationship between VILI vortex intensity and accumulative risk of death from refractory hypoxia. The difference of driving pressure (ΔΔP) between days 7 and 14 was regarded as VILI vortex intensity expression. That was the period in which we more frequently observed VILI vortex development. Data analysis was conducted with MedCalc 11.4.3.0 statistical software (Mariakerke, Belgium) and a p-value of <0.05 was considered statistically significant.
Ethical Approval
This study was conducted in the ICU dedicated to COVID-19 patients of an intensive care unit at a university hospital in Argentina. Approval was obtained from local institutional review board (resolution number: 68/2019) and informed consent followed the ethical committee’s recommendations.
Study RegistrationThe present analysis was registered at www.clinicatrials.gov (NCT04174313).
Results
General characteristics of the populationSixty-five patients with ARDS secondary to COVID-19 pneumonia were studied. Median age: 60 years (IQR, 55-65 years), female/male 25/40, APACHE II: 18 points (IQR, 15-22 points), SOFA: 6 points (IQR, 3.5-8 points) and Murray’s score: 2.5 points (IQR, 2.5-3 points). Forty per cent of the patients needed dialysis and 6.1% had cardiovascular complications. Pneumonia associated with mechanical ventilation was a common event in patients ventilated for more than 7 days (40%). The average days of hospitalization were prolonged 33 days (IQR, 24 to 49 days) and the mortality rate was high (50.7%).
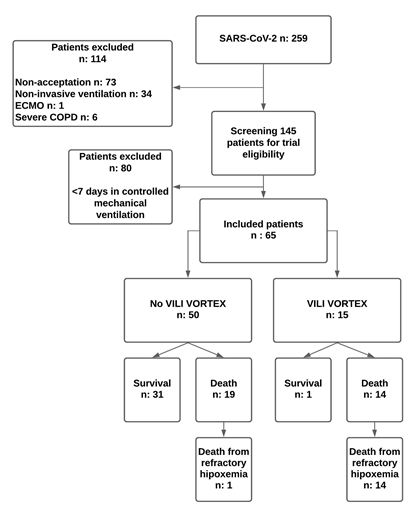
Figure 1: Shows the distribution of the population with SARS-CoV-2 studied.
VILI vortex clinical characteristics
Fifteen patients with ARDS due to COVID-19 (23%) clinically progressed to VILI vortex. Table 1 compares the clinical differences between both groups whereas Table 2 shows respiratory data. The VILI vortex generally started at the beginning of the second week. The increase in ΔP occurred in the context of stable Vt and PEEP, while the average PPlat remained within safe ranges, below 30 cm H2O. The torpid increase in ΔP was not initially accompanied by the PaO2/FiO2 ratio (table 3 and figure 2).
|
Parameters |
NO VILI VORTEX (n: 50) |
VILI VORTEX (n: 15) |
P= |
|
Age, years: |
60 (54-66) |
59 (55-60) |
NS |
|
Female/male: |
18/32 |
06-Sep |
NS |
|
APACHE II: |
18 ±16.5 |
22 ±5.9 |
0.02 |
|
SOFA: |
5 (3-7) |
7 (5-9) |
NS |
|
Days of hospitalization in ICU: |
33 (24-52) |
25 (23-44) |
NS |
|
Hypertension: |
25 (50) |
9 (60) |
NS |
|
Diabetes mellitus: |
11 (22) |
4 (26.6) |
NS |
|
Obesity: |
11 (22) |
5 (33.3) |
NS |
|
Ischemic heart disease: |
6 (12) |
2 (13.3) |
NS |
|
COPD: |
7 (14) |
3 (20) |
NS |
|
Cancer/immunosuppression: |
6 (12) |
4 26.6) |
NS |
|
Intubation delay, days: |
7.5 (5-11) |
14 (11-15) |
0.0005 |
|
Refractory hypoxemia: |
1 (2) |
14 (93.3) |
<0.0001 |
|
Mortality: |
19 (38) |
14 (93.3) |
0.0001 |
Table 1: General characteristics
Data are presented as absolute frequency (% of the included patients), as median and interquartile range or media ± standard deviation. Acute physiology and chronic health evaluation (APACHE II), sepsis-related organ failure assessment (SOFA).
|
Parameters |
NO VILI VORTEX (n: 50) |
VILI VORTEX (n:15) |
P= |
|
PaO2/FiO2, mmHg: |
150 ±73 |
174 ±40 |
NS |
|
Vt, ml/kg/PBW: |
6.3 ±0.7 |
6.5 ±05 |
NS |
|
pH: |
7.29 ±0.1 |
7.28 ±0.1 |
NS |
|
PCO2, mmHg: |
49 ±10 |
44 ±18 |
NS |
|
FiO2: |
60 (50-85) |
60 (50-70) |
NS |
|
Vt, ml: |
0.39 ±0.1 |
0.39 ±0.1 |
NS |
|
RR, bpm: |
24 (22-26) |
25 (20-28) |
NS |
|
PPlat, cmH2O: |
21.9 ±3.9 |
25.1 ±2.9 |
0.001 |
|
PPeak, cmH2O: |
27.9 ±5.1 |
31 ±3.5 |
0.008 |
|
PEEP, cmH2O: |
10 (8-12) |
10 (8-12) |
NS |
|
ΔP, cmH2O: |
12.3 ±2.7 |
13.1 ±3.6 |
NS |
|
CRS, ml/cmH2O: |
34 (27.4-39.4) |
27 (21-40) |
NS |
|
MV, L/min: |
9.3 ±3 |
9.4 ±2 |
NS |
|
Prone position: |
14 (28) |
4 (26.6) |
NS |
|
SMP, J/min/ml/cmH2O: |
0.36 ±1.8 |
0.41 ±1.5 |
NS |
Table 2: Comparison of mechanical parameters at the start of controlled mechanical ventilation
Data are presented as absolute frequency (% of the included patients), as median and interquartile range or media ± standard deviation. Volume tidal (Vt), respiratory rate (RR), plateau pressure (PPlat), peak pressure (Ppeak), driving pressure (ΔP), static compliance of the respiratory system (CRS), specific mechanical power (SMP).
|
Parameters |
Day 7 |
Day 14 |
P= |
|
PaO2/FiO2, mmHg: |
206 ±67 |
160 ±75 |
NS |
|
Vt, ml/kg/PBW: |
6.1 ±0.7 |
5.7 ±0.8 |
NS |
|
pH: |
7.3 ±0.1 |
7.3 ±0.1 |
NS |
|
PCO2: |
56.8 ±17 |
60 ±18 |
NS |
|
FiO2: |
50 (40-60) |
60 (40-70) |
NS |
|
Vt, ml: |
0.36 ±0.05 |
0.33 ±0.06 |
NS |
|
RR, bpm: |
26 (24-28) |
29 (25-32) |
0.01 |
|
PPlat, cmH2O: |
24.9 ±4.6 |
29.8 ±5 |
0.009 |
|
PPeak, cmH2O: |
30.6 ±6 |
37.7 ±5 |
0.001 |
|
PEEP, cmH2O: |
8 (8-10) |
8 (6-10) |
NS |
|
ΔP, cmH2O: |
15.3 ±3.6 |
24.1 ±7.8 |
0.0007 |
|
CRS, ml/cmH2O: |
22.3 (19-30) |
14 (12-19) |
NS |
|
MV, L/min: |
9.6 ±1.8 |
10.4 ±2.7 |
NS |
|
MP, J/min: |
11 ±2.6 |
12.3 ±2.8 |
NS |
|
SMP, J/min/ml/cmH2O: |
0.48 ±0.2 |
0.85 ±0.3 |
0.0003 |
Table 3: Comparison of mechanical parameters evolution in the VILI vortex group
Data are presented as median and interquartile range or media and standard deviation. Volume tidal (Vt), respiratory rate (RR), plateau pressure (PPlat), peak pressure (Ppeak), driving pressure (ΔP), static compliance of the respiratory system (CRS), mechanical power (MP), specific mechanical power (SMP).
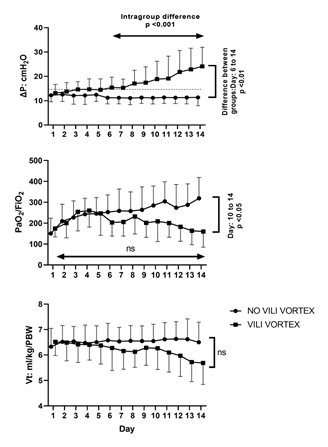
Figure 2: Ventilatory and gasometric parameters evolution during the first 14 days of mechanical ventilation
Whereas driving pressure increased from day 7 in the group that developed VILI vortex, the PaO2/FiO2 ratio remained stable and did not initially accompany the worsening of mechanical variables.
Analysis death related variables
The cause of death in the VILI vortex group was refractory hypoxemia in all cases whereas in the No VILI vortex group death for respiratory insufficiency was exceptional (figure 3). In this sense, VILI vortex (ΔΔP) predicted with good sensitivity and specificity the risk of death from both refractory hypoxemia (sensibility: 86.7% and specificity: 96%, AUC: 0.97 and from any cause (sensibility: 72.7% and specificity: 81.2%, AUC: 0.78). For each cmH2O of ΔΔP increase, the relative risk of death from refractory hypoxemia increased 1·3 times (95% IC: 1.2 to 1.4, p= 0.001).
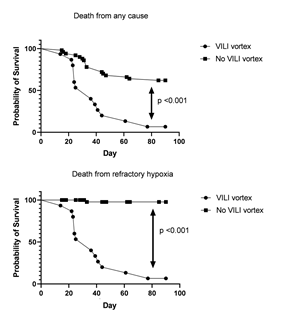
Figure 3: Comparison of survival curve between groups
Complications
Patients with VILI vortex had positive blood cultures; moderate to severe shock, persistent fever, and the observed fluid balance was considerably more positive, table 4.
|
Parameters |
NO VILI VORTEX (n: 50) |
VILI VORTEX (n:15) |
P= |
|
Intranosocomial pneumonia: |
19 (38) |
7 (46.6) |
NS |
|
Noradrenaline > 0.1 γ/kg/min: |
17 (34) |
12 (80) |
0.001 |
|
Renal replacement therapy: |
18 (36) |
7 (46.6) |
NS |
|
Persistent fever: |
10 (20) |
7 (46.6) |
0.03 |
|
Blood cultures: |
10 (20) |
9 (60) |
0.002 |
|
Lactate level > 2 mmol/L: |
16 (32) |
7 (46.6) |
NS |
|
Fluid balance, ml/day: |
471 (206-730) |
965 (501-1280) |
0.002 |
|
Ferritin level, ng/ml: |
2489 ±283 |
2780 ±3337 |
NS |
|
LDH, U/L: |
564 ±210 |
674 ±396 |
NS |
|
Fibrinogen level, mg/dl: |
492 ±101 |
577 ±96 |
NS |
|
CRP, mg/l: |
157 ±101 |
302 ±465 |
NS |
|
Troponin I, pg/ml, media: |
79.2 ±194 |
73 ±178 |
NS |
|
D-dimer, ng/ml FEU: |
3092 ±4554 |
3996 ±4156 |
NS |
Table 4: Complications observed within the first 14 days
Data are presented as absolute frequency (% of the included patients), as median and interquartile range or media and standard deviation.
Discussion
We studied a homogenous population of patients with SARS-CoV-2. The most significant findings of this study were the following: 1) VILI vortex is a relatively frequent phenomenon associated with high mortality due to refractory hypoxemia. 2) Although it usually starts with “safe” values of ΔP, this phenomenon progresses to a catastrophic condition that makes it difficult to maintain a protective ventilatory strategy. 3) Mechanical changes were not initially accompanied by worsening oxygenation, thus the PaO2/FiO2 ratio is not sensitive enough for its early detection. 4) The intensity of VILI vortex (ΔΔP) was positively associated with the risk of death from refractory hypoxemia. 5) Negative factors such as delay in invasive mechanical ventilation, fluid overload, presence of bacteremia, persistent fever and use of noradrenaline (>0·1 γ/kg/min for at least 24 hours) contributed to its development. Although the ventilatory load applied to the lung (SMP) was initially similar for both groups, a small group evolved with driving pressure elevation (VILI vortex) and later death from refractory hypoxemia (14/15). On the other hand, those patients with DP steady values (non-VILI vortex group) had lower mortality which was not related to severe respiratory failure. Individual factors from the patient such as genetic and racial factors or comorbilities, factors related to the pathogenic agent (strain virulence, inoculated virus amount, etc) and hospital-related complications (overinfections, shock, citokynes storms) can make patients evolve in different ways despite the similarity of the treatment they were receiving. As compliance of the respiratory system is associated with the ventilable lung capacity (baby lung) and driving pressure represents the relationship between tidal volume and CRS (ΔP: Vt/CRS), the progressive and sustained increase in DP while Vt remained constant or even decreased may reflect the unfavorable changes that occur in the ventilable lung. These changes may be compatible with VILI vortex, especially during the early stage of ARDS, when the fibrotic component is less present. A 4 cmH2O increase in ΔP in a period of 48 to 72 hours may indicate the start of VILI vortex, although mechanical and gasometric conditions may seem safe, since, as mentioned above, mechnical damage precedes the unfavorable changes in oxygenation. Furthermore, VILI vortex developed within a safe context of Vt and PPlat, thus lung protective measures should not be delayed in case of its early suspicion. Clinical evidence shows that there is a linear relationship between DP above 10 cmH2O and mortality18. Mechanisms related to lung heterogeneity and deformation speed (variable associated with flow) may increase tension at a regional level, even with safe values of PPlat and ΔP [19-21]. Unequal deformation between neighboring alveoli with different elastance may even double local tension [19,20], whereas flow has a reverse impact on the alveoli’s ability to adapt to change (strain rate), among other things [21]. This is an important concept, considering that ΔP is a static variable that is not influenced by flow. Driving pressure accumulative stress above 15 cm H2O and its association with relative risk of death had already been demonstrated [22]. We supported this observation by proving that VILI vortex intensity (ΔΔP), the relative increase of ΔP in a definite period, was also associated with the risk of death from refractory hypoxemia. From a clinical perspective, VILI vortex can lead to an extreme situation since to avoid the rise of driving pressure, it is mandatory to decrease Vt and on the other hand, to increase FiO2, respiratory frequency and inspiratory flow so that it can prevent hypoxia and hypercapnia from worsening. The result is a mechanical power that remains dangerously high in relation to the conditions of the ventilable lung (specific mechanical power). This is the moment when salvage strategies such as CO2 removal devices and ECMO could take place, while prone positioning should be used not only to improve oxygenation, but also to better homogenize intrapulmonary tensions and to promote passive recruitment. Moreover, a better prevention of nosocomial infections, shock control and a more conservative positive fluid balance could have probably mitigated the risk of VILI vortex in patients that remained stable during the first week. Finally, delay in invasive mechanical ventilation probably represents a longer inspiratory effort; Gattinoni and colleagues suggested the development of a self-inflicted lung injury (P-SILI) as one of the lung injury mechanisms involved in ARDS progression [23-25]. Regardless of the nature of inspiratory pressure, either positive, associated with mechanical ventilation, or negative, because of inspiratory effort, transpulmonary pressure (stress) and the alveolar deformation that it causes (strain) are the factors that determine the risk of VILI [26-27].
Limitations
We did not measure the volume of the ventilable lung (FRC): we indirectly defined VILI vortex based on the increase in ΔP, as an indirect expression of ventilable lung shrinkage. The serialized measuring of FRC with CT is not possible in patients in critical condition, while the use of electrical impedance tomography and the measuring of FRC by gas dilution methodology are rather imprecise and have not yet been validated for this specific purpose. Another limitation is that harmful stress and subsequent lung injury are caused by transpulmonary driving pressure (the pressure in the alveoli), but we only have measurements of static airway driving pressure. Airway driving pressure does correlate with transpulmonary driving pressure, but it represents only a surrogate, which might be affected by numerous factors (e.g., resistive pressures, chest wall compliance, and spontaneous breathing). Our data are valid on the early stage of ARDS. In the fibro proliferative phase, the accumulation of inflammatory edema and cellular debris in the alveolar interstitium together with fibrosis formation may contribute to controlling alveolar deformation in the face of tension growth (dissociation between tension and deformation due to an increase in functional specific elastance). Finally, although decubitus changes are likely to alter the value of ΔP, we did not find significant differences in these patients once they were stabilized.
Interpretation
The risk of VILI must be evaluated in a global context, where ventilatory mechanics plays an important but not exclusive role, and therefore should be approached in a comprehensive manner. From a mechanical perspective, in those patients admitted with preserved CRS, the increasing trend of ΔP can better reflect the unfavorable changes occurring in the lung, whereas its initial absolute value, PaO2/FiO2, PPlat and mechanical power is not sensitive enough to anticipate catastrophic VILI vortex.
Acknowledgements
We wish to thank all the nurses and physicians who contributed to conduct the present investigation during COVID-19 pandemic.
Competing interests
The authors all declared that they have no potential conflicts of interest. None of the authors have received financial support.
Funding
The present investigation was conducted by institutional funding.
Contributions
NP is the guarantor author of the study.
NP and OHF conceived the study protocol and participated in the design and coordination of the study; PC, RC, EP, SL and EA collected study data; NP, PC, RC and OHF participated in data interpretation; and drafted the present manuscript; NP, PC, RC, EP, SL, EA and OHF revised the manuscript. All authors read and approved the final version of the manuscript.
References
- Beitler JR, Malhotra A, Thompson BT. Ventilator-induced Lung Injury. Clin Chest Med 37 (2016): 633-646.
- Brower RG, Matthay MA, Morris A, et al. Acute Respiratory Distress Syndrome Network. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med 342 (2000): 1301-1308.
- Hotchkiss JR, Blanch L, Murias G, et al. Effects of decreased respiratory frequency on ventilator-induced lung injury. Am J Respir Crit Care Med 161 (2000): 463-468.
- Amato MBP, Meade MO, Slutsky AS, et al. Driving pressure and survival in the acute respiratory distress syndrome. N Engl J Med 372 (2015): 747-755.
- Kumar A, Pontoppidan H, Falke KJ, et al. Pulmonary barotrauma during mechanical ventilation. Crit Care Med 1 (1973): 181-186.
- Müller-Redetzky HC, Felten M, Hellwig K, et al. Increasing the inspiratory time and I:E ratio during mechanical ventilation aggravates ventilator-induced lung injury in mice. Crit Care 19 (2015): 23.
- Brochard L. Ventilation-induced lung injury exists in spontaneously breathing patients with acute respiratory failure: Yes. Intensive Care Med. 43 (2017): 250-252.
- Rouby JJ. Lung overinflation: The hidden face of alveolar recruitment. Anesthesiology 99 (2003): 2-4.
- Graaff AE, Dongelmans DA, Binnekade JM et al. Clinicians’ response to hyperoxia in ventilat-ed patients in a Dutch ICU depends on the level of FiO2. Intensive Care Med 37 (2011): 46-51.
- Gattinoni L, Tonetti T, Cressoni M, et al. Ventilator-related causes of lung injury: the mechanical power. Intensive Care Med. 42 (2016): 1567-1575.
- Cressoni M, Gotti M, Chiurazzi C, et al. Mechanical power and development of ventilator-induced lung injury. Anesthesiology124 (2016): 1100-1108.
- Gattinoni L, Pesenti A. The concept of “baby lung”. Intensive Care Med 31 (2005): 776-784.
- Gattinoni L, Tonetti T, Quintel M. Regional physiology of ARDS. Crit Care 21 (2017): 3-12.
- Vasques F, Duscio E, Cipulli F, et al. Determinants and Prevention of Ventilator-Induced Lung Injury. Crit Care Clin 34 (2018): 343-356.
- Marini JJ and Gattinoni L. Time course of evolving ventilator-induced lung injury: The "Shrinking Baby Lung". Critical Care Medicine 48 (2020): 1203-1209.
- Ranieri VM, Rubenfeld GD, Thompson BT, et al. acute respiratory distress syndrome: the Berlin Definition. JAMA 307 (2012): 2526-2533.
- Vestbo J, Hurd SS, Agustí AG, et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am J Respir Crit Care Med 187 (2013): 347-365.
- Papazian L, Aubron C, Brochard L, et al. Formal guidelines: management of acute respiratory distress syndrome. Ann Intensive Care 9 (2019).
- Cressoni M, Cadringher P, Chiurazzi C, et al. Lung Inhomogeneity in Patients with Acute Respiratory Distress Syndrome. Am J Respir Crit Care Med 189 (2014):149-158.
- Mead J, Takishima T, Leith D. Stress distribution in lungs: a model of pulmonary elasticity. J Appl Physiol 28 (1970): 596-608.
- Protti A, Maraffi T, Milesi M, et al. Role of strain rate in the pathogenesis of ventilator-induced lung edema. Crit Care Med 44 (2016): 838-845.
- Urner M, Jüni P, Hansen B, et al. Time-varying intensity of mechanical ventilation and mortality in patients with acute respiratory failure: a registry-based, prospective cohort study. Lancet Respir Med 8 (2020): 905-913.
- Marini JJ, Rocco PRM, Gattinoni L. Static and dynamic contributors to ventilator-induced lung injury in clinical practice. pressure, energy, and power. Am J Respir Crit Care Med 201 (2020): 767-774.
- Vieillard-Baron A, Matthay M, Teboul JL, et al. Experts’ opinion on management of hemodynamics in ARDS patients: focus on the effects of mechanical ventilation. Intensive Care Med 42 (2016): 739-749.
- Marini JJ, Hotchkiss JR, Broccard AF. Bench-to-bedside review: microvascular and airspace linkage in ventilator-induced lung injury. Crit Care 7 (2003): 435-444.
- Chiumello D, Carlesso E, Brioni M, Cressoni M. Airway driving pressure and lung stress in ARDS patients. Crit Care 20 (2016):
- Grieco DL, Chen L, Brochard L. Transpulmonary pressure: importance and limits. Ann Transl Med 5 (2017):
