Circulating Biomarkers and MicroRNAs in the Diagnosis, Prognosis and Treatment of Diabetic Cardiomyopathy-A Review
Article Information
Swathy Moorthy1, Teena Koshy2, Vijaya Srinivasan3, Santhi Silambanan4*
1Department of General Medicine, Sri Ramachandra Medical College and Research Institute, Sri Ramachandra Institute of Higher Education and Research (SRIHER), Chennai, Tamil Nadu, India
2Department of Human Genetics, Sri Ramachandra Medical College and Research Institute, Sri Ramachandra Institute of Higher Education and Research (SRIHER), Chennai, Tamil Nadu, India
3Department of Biochemistry, Sri Ramachandra Medical College and Research Institute, Sri Ramachandra Institute of Higher Education and Research (SRIHER), Chennai, Tamil Nadu, India
4Department of Biochemistry, Sri Ramachandra Medical College and Research Institute, Sri Ramachandra Institute of Higher Education and Research (SRIHER) Chennai, Tamil Nadu, India
*Corresponding Author: Santhi Silambanan, Professor, Department of Biochemistry, Sri Ramachandra Medical College and Research Institute, Sri Ramachandra Institute of Higher Education and Research (SRIHER) (DU), Chennai, Tamil Nadu, India
Received: 26 July 2020; Accepted: 03 August 2020; Published: 28 August 2020
Citation: Moorthy S, Koshy T, SrinivasanV, Silambanan S. Circulating Biomarkers and MicroRNAs in the Diagnosis, Prognosis and Treatment of Diabetic Cardiomyopathy-A Review. Cardiology and Cardiovascular Medicine 4 (2020): 481-497.
View / Download Pdf Share at FacebookAbstract
Background: In the year 2015, diabetes mellitus resulted in 5 million deaths which were predicted to increase by 34% in 2030. The prevalence of cardiomyopathy in diabetics is 12% and is almost 22% in people over 64 years of age. Individuals with diabetes mellitus present with cardiomyopathy in the absence of hypertension, coronary artery disease, or valvular heart disease. Diabetic cardiomyopathy is a cardiac condition presenting with heart failure characterized by diastolic dysfunction and cardiac remodeling, in the absence of signs of coronary artery disease and hypertension. Glucotoxicity and lipotoxicity are the initiators of excessive autophagy, apoptosis, altered excitation contraction coupling and remodeling of extracellular matrix with excessive fibrosis in the myocardium. Since the condition is silent during early stage, use of novel biomarkers may be useful to identify patients who are at risk of cardiomyopathy. So far, only a few studies have explored miRNAs profile in patients with diabetic cardiomyopathy with inconsistent results. Biomarkers and microRNAs have been identified to diagnostic as well as prognostic indicators of the various stages of progression of diabetic cardiomyopathy. Cardiac microRNAs target mitochondrial dysfunction and free radical production thus they have a promising role in implementing targeted therapy in patients with diabetic cardiomyopathy. This review was undertaken to understand the roles played by various biomarkers and microRNAs in the diagnosis and prognosis and therapy in patients with diabetic cardiomyopathy.
Methodology: A literature search was done in PubMed, Embase, Cochrane Library, Pubmed Central, UpToDate, with the focus on biomarkers in Diabetic Cardiomyopathy. The articles selected included those on pathogenesis of diabetic cardiomyopathy, various diagnostic modes, the role played by biomarkers and miRNAs in the diagnosis especially
Keywords
Type 2 Diabetes mellitus; Cardiomyopathy; Systolic dysfunction; Diastolic dysfunction; Myocardial fibrosis; Myocardial apoptosis; Autophagy; Heart failure; Cardiac rehabilitation
Type 2 Diabetes mellitus; Cardiomyopathy; Systolic dysfunction; Diastolic dysfunction; Myocardial fibrosis; Myocardial apoptosis; Autophagy; Heart failure; Cardiac rehabilitation
Article Details
1. Background
In 2015, the International Diabetes Federation (IDF) estimated that 442 million people aged between 20 and 79 years had type 2 diabetes mellitus (T2DM) associated with a global economic cost of 673 billion US Dollars [1]. Further, it was predicted to rise to 642 million by the year 2040 [2]. As per the Framingham Heart Study, T2DM is an independent risk factor for the development of heart failure (HF), and that too with worse prognosis. When compared to nondiabetics, the risk of HF was 2.4-fold and 5-fold higher in diabetic men and women, respectively. In the United Kingdom Prospective Diabetes Study (UKPDS), the incidence of HF is 2.3 cases per 1000 person-years in patients with HbA1c of less than 6%, whereas in diabetics with HbA1c of more than 10%, it is estimated to be 11.9 per 1000 person-years [3]. Global studies have projected that the number of diabetic adults of 135 million in 1995 will increase to 300 million by 2025. 80% of deaths among diabetic patients are due to cardiovascular disease (CVD) with the most common cause being coronary artery disease (CAD) [4]. However, there is an increasing recognition of an additional cardiac insult which has not been given attention is ‘Diabetic Cardiomyopathy’ (DCM) [5, 6]. This review was undertaken to understand the mechanism of action of various biomarkers and microRNAs in assessing the diagnosis and prognosis and role as targeted therapy in patients with diabetic cardiomyopathy.
2. Methodology
A literature search was done in Pubmed, Embase, Cochrane Library, Pubmed Central, UpToDate, with the focus on biomarkers in Diabetic Cardiomyopathy. The articles selected included those on pathogenesis of diabetic cardiomyopathy, various diagnostic modes, the role played by biomarkers and miRNAs in the diagnosis especially in the early stage, in the prognosis that is markers representing the various stages of diabetic cardiomyopathy and also in targeted therapy. The articles on cardiac diseases other than diabetic cardiomyopahty were excluded from this research. Since this was a review article ethics clearance was not needed and hence not obtained. This study did not receive financial grants from any government, private, national or international funding agencies.
3. Introduction
DCM is characterized by left ventricular hypertrophy (LVH), cardiomegaly and HF. Diabetic cardiomyopahty is a complex disease and involves metabolic, structural and functional disturbances. This distinct clinical entity was first proposed by Lundbaek in 1954 as diabetic heart disease, independent of hypertension and CAD that commonly co-existed with T2DM. In 1972, Rubler et al., elucidated the same post-mortem findings in patients with T2DM, and heart failure with reduced ejection fraction (HFrEF) [3] and with normal epicardial coronary arteries in the absence of hypertension, CAD, valvular or congenital heart diseases. In 1995, Bertoni et al., confirmed this from a large United States nationwide case control study. This was further supported by another study stating that despite good control of all risk factors, the risk of HF hospitalisation in T2DM, is higher even though there was little or no increase in the risk of mortality, or macrovascular complications of diabetes mellitus [7]. The mechanisms behind structural and functional alterations within the diabetic heart are many. They include insulin resistance (IR), endothelial dysfunction, fibrosis, cardiac lipotoxicity and energy dysregulation. Dysregulated cardiac metabolism plays a central role in diabetic cardiomyopathy [8].
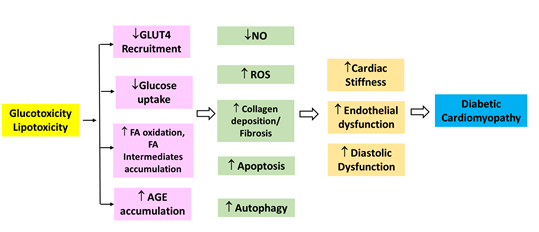
4. Altered metabolic reserve-Glucotoxicity and Lipotoxicity
Excess adiposity and altered fat distribution have been shown to contribute to diabetic cardiomyopathy in T2DM. Glucose-fatty acid cycle or Randle cycle, helps to regulate uptake and utilisation of glucose by the muscle depending on the rate of free fatty acid (FFA) oxidation. In healthy individuals at rest, cardiac metabolism relies predominantly on oxidation of FFA, but during increased cardiac workload, glucose uptake increases by a factor of two, while FFA metabolism does not change. Low cardiac metabolic reserve is the consequence of impaired metabolic flexibility and is associated with increased mortality, in patients with DCM [7]. Diabetic cardiomyopathy is associated with altered levels of peroxisomal proliferator activated receptors (PPAR); as shown by an increase in cardiac PPAR-γ and a decrease in PPAR-α. This results in decreased glucose transport into cardiac myocardium and increased cardiac lipid accumulation. Cardiac glucose uptake is regulated by insulin-mediated recruitment of glucose transporter type four (GLUT4) from the intracellular vesicles to the plasma membrane. High fatty acid concentrations in diabetic hearts may impair insulin signal transduction, thereby decreasing GLUT4 translocation with reduced glucose uptake. Due to altered activities of insulin and counterregulatory hormones there is increased release of FFA from hydrolysis of triglyceride (TGL) in the adipose tissue. There is increased expression of enzymes involved in FFA oxidation, including CD36, carnitine acyltransferase-1 (CAT-1), and acetyl CoA carboxylase (phosphorylated form). In addition, diabetic cardiomyopathy is associated with activation of the enzyme, diacylglycerol acyltransferase (DGAT) which is involved in the synthesis of TGL [9].
In the heart, mitochondria produce maximum ATP from FFA than from glucose. There is altered mitochondrial oxidation of glucose and FFA. Due to this impaired fuel substrate flexibility, diabetic hearts are prone for ischemic injury. Activation of cardiac PPAR-α results in increased expression of genes involved in fatty acid oxidation at the same time they suppress expression of genes involved in glucose utilization. FFA oxidation causes accumulation of citrate in the cytosol, which inhibits glycolysis by inhibiting the activity of phosphofructokinase 1 (the rate-limiting enzyme) in glycolysis. In normal heart, pyruvate, the end product of glycolysis, is transported into mitochondria and further oxidized to acetyl-CoA by pyruvate dehydrogenase (PDH). In diabetic patients PDH activity is impaired, leading to accumulation of pyruvate and other glycolytic intermediates in the heart. Both increased FFA oxidation and PPAR-α activation lead to suppression of PDH, which impairs glucose oxidation. Thus, chronic metabolic derangement results in cardiac lipid accumulation and thus paving the way forward for the onset DCM [9].
PPARα is an essential component in cardiac substrate switching. Decreased PPARα expression in the presence of chronic hyperglycemia will limit the FFA oxidative capacity in the heart. The discordance between the rates of FFA availability and uptake, with that of FFA oxidation results in increased intracellular long chain fatty acyl-CoA concentrations. Since cardiomyocytes are not specialised to store lipid, and cellular lipid overloading underlies the concept of “lipotoxicity” as a potential mechanism for impaired cardiac function. The excess long chain fatty acyl-CoA is then diverted towards non oxidative processes with the production of lipotoxic intermediates such as ceramide and diacyl-glycerol. This leads to impairment of the sarcoplasmic reticulum calcium ATPase 2a (SERCA2a) expression, an essential enzyme in calcium homeostasis, resulting in diastolic dysfunction [7]. Altered glucose and lipid metabolism leading to cardiomyopathy is depicted in Figure 1.
5. Structural and functional changes in cardiac myocardium
Diabetic heart is characterized by myofibrillar hypertrophy with fibrotic strands extending between cardiac myocytes, which tend to cause diffuse myocardial fibrosis [7]. The histopathological studies show hypertrophy of myofibrils, myocardial fibrosis, microangiopathy and acid mucopolysaccharide deposition in the extracellular matrix of myocardium [10]. Alterations in left ventricular geometry lead to LV remodelling, hypertrophy and eventually increased left ventricular mass. LVH in diabetes includes both concentric and eccentric hypertrophy; concentric remodelling is found to have a higher association with cardiovascular mortality than eccentric remodelling. Diastolic dysfunction is apparent as early as the asymptomatic stage. There is significant decrease in ejection fraction and systolic dysfunction and this is found to have a positive correlation with HbA1c levels. The relationship between the metabolic changes within the heart and functional measures such as myocardial strain rates as well as TGL content will help us in better understanding of the management of DCM [7].
Diabetic patients generally exhibit accelerated myocyte death which is seen pathologically as myocyte apoptosis or necrosis. Incomplete FFA oxidation and accumulated toxic fatty acid intermediates lead to generation of reactive oxygen species (ROS). ROS induce lipid peroxidation and cause damage to mitochondrial proteins which subsequently reduce cardiac efficiency. Excessive fatty acids in the heart lead to the generation of ceramide; ceramide can induce apoptosis. This leads to decrease in cardiac function [9]. Suggested mechanisms involved in the pathogenesis of DCM include: increased production of ROS, increased inflammatory cytokines and chemokines, increased glycosylation and phosphorylation of p53 and increased production of angiotensin II. ROS act as signaling molecules in physiological conditions; but upregulation causes pulmonary hypertension and heart failure through the activation of soluble guanylate cyclase (sGC) and NOX1 (NADPH oxidase) [11, 12]. These mechanisms cause myocardial fibrosis and collagen deposition in the diabetic myocardium. Collagen interacts with glucose forming glycated collagen which then undergoes further chemical modification to form advance glycation end-products (AGE), predisposing to myocardial stiffness [3, 6].
The uptake of cardiac FFA exceeds the oxidative capacity of the myocardium. This leads to accumulation of TGL, cardiac steatosis eventually resulting in death of cardiomyocytes, associated with LV systolic dysfunction. This process has been demonstrated in animals and is called lipotoxicity. The change in energy substrate due to the reduction of insulin-mediated glucose uptake, leads to diastolic dysfunction. Generation of lipid intermediates, such as diacylglycerol, may be toxic to microcirculation, by effect on nitric oxide synthase and reduction of myocyte distensibility. Cardiac steatosis has been associated with diastolic dysfunction in diabetic patients [13]. Hyperglycemia, IR and hyperinsulinemia induce formation of AGEs, activation of renin-angiotension-aldosterone system (RAAS), autonomic neuropathy, ER stress, cardiomyocyte death, as well as microvascular dysfunction. These pathophysiological abnormalities promote cardiac stiffness, hypertrophy and fibrosis, resulting in LV diastolic and systolic dysfunction and heart failure [14-16]. The metabolic derangements are more prominent when there is associated metabolic syndrome [17]. Some of the existing glucose-lowering therapies might have independent effects on the myocardium; however the effect which can be beneficial or not depends on the agent [18].
DCM is found to progress in four stages, with each stage representing a more progressive form of the process from heart failure with preserved ejection fraction (HFpEF) to HFrEF. The various stages of progression of DCM is given in Table 1. Patients in stage 1 have no symptoms and have only diastolic dysfunction. Abnormalities in diastolic and systolic function are detected in stages 2 and 3. Patients in stage 4 have symptomatic HF and dilated hearts characterised by fibrosis and disease of large and small coronary arteries [19]. Cardiac dysfunction in diabetes is a continuum, ranging from asymptomatic diastolic dysfunction through subclinical systolic dysfunction and then overt HF with reduced ejection fraction [20].
|
Classification |
Stage 1 |
Stage 2 |
Stage 3 |
Stage 4 |
|
Stage |
Early/middle |
Middle |
Middle/late |
Late |
|
Characteristics |
HF with normal EF, LVH |
HF with normal &¯ EF |
Including HT, microangiopathy |
HFrEF including CAD |
|
Symptoms of HF |
NYHA I |
NYHA II |
NYHA II-III |
NYHA II-IV |
|
Structural features (Imaging, biopsy) |
LVH, LV mass |
LVH, LV mass & thickness Dilatation Fibrosis |
Dilatation Fibrosis Microangiopathy (small vessel disease) |
Dilatation Fibrosis Microangiopathy Macroangiopathy |
|
Functional features |
Diastolic dysfunction |
Diastolic and systolic dysfunction |
- |
- |
|
Cardiac MRI LGE |
Infrequent |
Possible |
Frequent |
Very frequent |
|
Troponins |
Infrequent |
Infrequent |
Positive if concurrent inflammation or ischemia |
Positive in infarction or severe HF |
LGE: Late Gadolinium enhancement; NYHA: New York Heart Association
Table 1: Stages of diabetic cardiomyopathy and/or diabetic heart disease [20].
6. Diagnosis of diabetic cardiomyopathy
DCM has a long latent asymptomatic phase followed by the onset of signs of overt HF. Diagnosis is made by detection of myocardial abnormalities along with the exclusion of other known causes of cardiomyopathies. In the absence of symptoms of overt HF, the American College of Cardiology/American Heart Association (ACC/AHA) recommend imaging for diagnosis of DCM. 2D echocardiography (ECHO) is the widely accepted imaging technique for the assessment of left ventricular function in diabetic patients. Current clinical understanding of DCM, lacks consensus on precise ECHO imaging criteria. The earliest finding in ECHO is LVH and diastolic dysfunction. Diastolic dysfunction not accompanied by any other determinable clinical sign of cardiac disease is categorized as the initial cardiac change in DCM. Prolonged QT in electrocardiogram (ECG) may suggest fibrosis. Cardiac magnetic resonance imaging (CMRI) is another imaging technique which is found to have higher accuracy and precision; thus, has been considered the gold standard in assessing left ventricular mass. CMRI is able to perform myocardial perfusion imaging (MPI) and also can assess myocardial flow reserve (MFR). Further, CMRI has been demonstrated as a useful imaging modality in accurately assessing diastolic function better than ECHO. However, the current use of CMRI is limited because of its high cost, time consumption and the requirement of expertise in handling the procedure [6].
7. Role of biomarkers in cardiomyopathy
Assessment of circulating cardiac biomarkers may complement imaging findings in the diagnosis and prognosis of DCM. The most recognized cardiac biomarker is brain natriuretic peptide (BNP) produced in response to ventricular volume and pressure overload. It gets elevated with the onset of LV dysfunction. But, BNP cannot distinguish between systolic or diastolic HF, which limits its clinical use in the diagnosis of DCM. Hence the utility of biomarkers such as markers of inflammation - hsCRP, IL-6, [7] TNF-alpha and cardiac troponin-T have been studied which can indicate early myocardial changes [22]. G protein coupled receptor kinase 2 (GRK2) is an indicator of early disease of DCM [23]. Heart type fatty acid binding protein (H-FABP) is found to be elevated in the early stages of cardiac injury in diabetic individuals. Insulin-like growth factor binding protein-7 (IGFBP-7), by interaction with IGF-1, regulates insulin receptor activity. IGFBP-7 is found to be an indicator of diastolic dysfunction associated with vascular remodelling, cardiac hypertrophy and fibrosis [22]. Biomarkers such as matrix metallopeptidases-9 (MMP-9) and their inhibitors, transforming growth factor beta (TGF beta) (60) and procollagen type I propeptide levels are elevated in DM with cardiac fibrosis and diastolic dysfunction [3, 24].
Galectin-3 is considered to be an early indicator of fibrosis, inflammation and adverse cardiovascular events in HF patients [19 25]. AGEs are involved in the pathogenesis of apoptosis and fibrosis [26]. Soluble ST2 (sST-2) is said to be involved in cardiac remodeling. Activin-A is found to have a protective role in HF. Chitinase-3-Like protein-1 (CHI3L1 or YKL-40) attenuates fibrosis, apoptosis and induces tissue remodeling [19]. FGF1 is highly expressed in the heart and is induced by all PPAR subtype agonists. FGF2 is expressed in cardiomyocytes and fibroblasts and it causes extracellular matrix deposition, apoptosis and cardiac hypertrophy. FGF21 reduces ROS production and protects the heart against inflammatory or prohypertrophic stimuli. FGF23 is involved in the pathogenesis of LVH via activation of phospholipase Cg and calcineurin–NFAT (nuclear factor in activated T cells) signaling pathways [27]. AGEs may also bind to receptor for AGEs on the cardiac cell membranes, further promoting both pro-fibrotic and pro-inflammatory signaling, and increasing the expression of oxidative stress mediators [28].
GDF-15 is produced by cardiac myocytes, endothelial cells, adipocytes, and macrophages. It is a highly promising biomarker capable of yielding information of several disease pathways, and thus provide the pathophysiological process as found in patients with DCM [29]. The over expression of insulin like growth factor 1 (IGF-1) is reported to inhibit DCM development. Several molecular mechanisms identified AGEs, fibrosis related genes including poly (ADP-Ribose) polymerase 1 (PARP-1), Otsuka Long Evans Tokushima fatty (OLETF) and matrix metalloproteinases 2 (MMP-2), inflammatory cytokines such as interleukin-1beta (IL-1β), IL-6, tumor necrosis factor-alpha (TNF-α) and trans-forming growth factor beta1 (TGF-β1) and altered pathways like mitogen-activated protein kinase (MAPK) signaling and TGF-β signaling [30]. There are studies indicating that pathological signs of DCM are present as early as in the pre-diabetic stage. Worsening glycemic control indicates unfavorable prognosis of DCM and HF in patients with DM.
8. MicroRNAs as biomarkers in diabetic cardiomyopathy
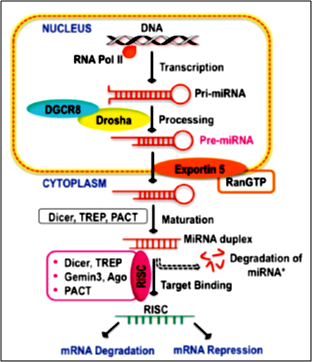
Circulating microRNAs (miRNAs or miRs) have been proposed as new type of biomarkers for cardiovascular diseases, mainly because they are involved in epigenetic mechanisms of cardiomyopathies. MiRs are small non-coding RNA molecules that function as post-transcriptional regulators, decreasing the expression of target genes and regulating multiple cellular pathways [31]. Figure 2 indicates biogenesis of miRs, maturation, and target mRNA-binding. miRNAs’ biogenesis typically starts with RNA polymerase II-mediated transcription from a miRNA gene into primary miRNAs (primiRNAs) and then processed, spliced, and exported as premiRNA from the nucleus by the action of Dosha-DGRB and Exportin 5. In the cytoplasm, premiRNA maturation and target binding is mediated by the action of Dicer, RISC complex, and Argonaute family proteins [32].
The mechanisms by which miRs regulate gene expression and protein translation include mRNA cleavage by binding to the complementary sequence in mRNA or by binding to partially complementary sequence in 3’UTR of target mRNA, leading to repression. Also miRs are shown to upregulate translation process during the arresting phase of cell cycle. Thus it is inferred that miRs show varied expressions in a variety of cardiac diseases suggesting that miRs could play a crucial role in regulating the pathogenesis of these diseases. Hence a miR can modulate various pathological processes by acting on multiple targets, thus producing different and sometimes opposite functions depending upon the duration of the disease [33]. Most of the miRs are found to target genes encoding MAPK, p53, Akt, IRS-1, VEGF, IGF-1R, FGF etc. [34].
Unlike other types of RNA, miRs are highly stable in the extracellular medium. They are packed into lipid vesicles, such as apoptotic bodies, microparticles, and exosomes, and bound with proteins such as AGO2 (argonaute 2) or NPM1 (nucleophosmin 1) or with lipoproteins, to prevent them from degradation by enzymes in the extracellular medium. MiRs are highly resistant to degradation, extremes of pH and temperature, repeated freeze-thaw cycles, or prolonged storage. Extracellular miRs act as a genuine intercellular communication system that regulates gene expression and receptor cell phenotype. MiRs participate in both physiological and adaptive responses such as the onset and development of pathologic conditions, including cardiovascular diseases. Abnormal miR expression levels directly affect expression of the respective target mRNAs and, therefore, miRs are potentially causative elements of disease. The relevance of miR gene expression regulation in cardiovascular development and homeostasis as well as the implications of miRs in various cardiovascular disease are being established [35].
Despite the fact that miRs have been proposed as possible biomarkers, determination of miRs has not been incorporated into clinical practice so far. This is due to various reasons- absence of universal measuring method enabling an easy-to-handle, fast, reliable, and inexpensive determination of miRs. miR expression profiling is a technical challenge: miRs are tiny molecules, each of the miR family members exhibit a high degree of homology, and absolute miR concentrations in body fluids are rather low. Several measurement platforms are currently available to determine miR levels in biological samples. They are Next-Generation Sequencing (NGS), quantitative reverse transcription polymerase chain reaction (qRT-PCR), microarray and Surface Plasmon Resonance (SPR). Each method has its strengths and weaknesses, and selection of the measuring method depends on the specific scientific questions to be addressed. Moreover, to ensure reliable miR measurement, it is necessary to carefully choose the compartment most suitable for measuring the miRs of interest (eg., serum, plasma, blood cells, tissue specimens, or body fluids) and to select an appropriate normalization strategy. Also, miR expression profiles are influenced by genetic heterogeneity and exogenous influences, such as medication, nutrition, or exposure to certain environmental conditions [36]. Studies of circulating miRNAs in chronic heart failure have shown contradicting results, probably due to the variability of patients’ clinical conditions, sex, genetic background and medication; or it could be due to different detection approaches used [37].
miRs are expressed as cardiomyocytes, fibroblasts, endothelial cells, and vascular smooth muscle cells and control virtually all aspects of cardiovascular biology, including cardiac remodeling and fibrosis, apoptosis, inflammation, proliferation, angiogenesis, and metabolism. Abnormal expression miR profiles are associated with disorders such as heart failure, atherosclerosis, and cardiomyopathies of various origins. MiRs show promising results in the pathogenesis as well as molecular therapeutic targets of DCM. MiRs profiling is essential for different pathophysiological processes underlying cardiomyocyte hypertrophy, remodeling, interstitial fibrosis, and heart failure. In this regard, various studies have shown that miR-specific overexpression and repression in vitro and in vivo models including miR-1, miR-126, miR-133a, or miR-208a, play key role in cardiovascular diseases [35].
9. MiRs involved in various disease processes of DCM explained
9.1 MiRs in LVH
Cardiac fibroblasts are secreted into exosomes are enriched with miR-21 as a crucial paracrine signaling mediator of cardiac hypertrophy. Pharmacological inhibition of miR-21, attenuated the development of cardiac hypertrophy in mice infused with angiotensin II (AngII). Few studies have shown that miRs could be transferred between cells [38]. miRs such as miR-9, miR-22, miR-23, miR-27, miR-30, miR-142, miR-190a/b, miR-206, miR-208a/b, miR-221, miR-423-5p and miR-499 are altered in diabetic patients with LVH. MiR-199a/b functions as an important negative regulator of hypertrophy, targeting dual-specificity tyrosine phosphorylation and calcineurin/nuclear factor of activated T-cells (NFAT) signaling, along with miR-23a [39]. MiRs having anti-hypertrophy role includes miR-1, miR-133a, miR-373, miR-378, miR-23b, miR-181a and miR-30c and pro-hypertrophy miRs include miR-208a, miR-195, miR-221, miR-150, miR-199a, miR-214, miR-29a, miR-125b, miR-212 and miR-451 [40]. Elevation in serum and tissue levels of miR-503 indicates a transcript with antiproliferative effects [41]. Several reports support the pro-hypertrophic role of miR-22 [42], miR-1, miR-21, miR-23, miR-133 and miR-208a [43].
9.2 MiRs in cardiac Fibrosis
MiR-133a, constitutively expressed in the heart, protects against myocardial fibrosis without affecting hypertrophy. MiR-133a and miR-30 are involved in myocardial matrix remodelling by regulation of connective tissue growth factor [39]. Myocytes are partially protected from ROS injury by miR-21, via its target programmed cell death protein. However, miR-21 contributes to myocardial fibrosis by stimulating mitogen-activated protein kinase (MAPK) signaling in fibroblasts. Downregulation of miR-29 induces the expression of collagens and enhances the fibrotic response in the myocardium. MiR-133a regulates collagen 1A1 expression, and miR-199 links antiapoptotic Akt signaling with β-adrenergic stimulation. miR-199a-5p is regulated by signal transducer and activator of transcription 3 (STAT), thereby linking cardiomyocyte and endothelial cell function. MiR-9 regulates myocardin expression together with NFATc3, the latter being controlled by miR-23a. Phosphatase and tensin homolog is also derepressed by miR-22, hence protecting rat cardiomyocytes from hypertrophy. MAPK, which is regulated by miR-21 in fibroblasts, is under the control of miR-142. Others report that both miR-221 and miR-27b promote cardiac hypertrophy, whereas miR-206 attenuates cardiac remodeling by inhibiting metalloproteinase inhibitor 3. miR-21, miR-29, miR-1, miR-133, miR-192, miR2001a/b and miR-382 are also involved in fibrosis [39]. miR-29a is involved in collagen IV expression whereas miR-29b is said to block progressive renal fibrosis in diabetic nephropathy. It is not clear whether these microRNAs have a role in diabetic heart [44]. Three cardiac fibroblast-derived circulating miRNAs such as miR-660-3p, miR-665 and miR-1285-3p are upregulated in HF and correlated with HF severity, holding promises as diagnostic biomarker for HF [37]. Another study has shown that miR-21, miR29 and miR-133a are involved in fibrosis [43, 45].
9.3 MiRs in cardiac apoptosis
Several miRs have been associated with apoptosis, including miR195, miR-320, miR-378, miR-34a, miR-30d, miR-24a, miR-483-3p, miR-29, miR-181a, miR-30c [40], miR-195, miR-199a and miR-320 [43] Diabetic milieu is associated with increased levels of miR-1 and miR-206 and correlated with downregulation of the anti-apoptosis factor, IGF-1, Pim-1, and heat shock protein 60 (Hsp-60), thus promoting cardiomyocyte apoptosis [32] Hyperglycemia promotes p53-dependent apoptosis; hence the p53/miR-34 axis may be involved in a hyperglycemia-induced death pathway [46]. High levels of FFAs and the subsequent loss of Mir30c are crucial for activation of autophagy in cardiomyocytes both in cultured H9c2 cells exposed to FFAs and in db/db mice. Autophagy increase in response to high FFAs, lead to cardiac abnormalities in diabetes [47].
9.4 MiRs in Glucose transport and utilization
GLUT4 translocation from the cytoplasmic compartment to the cell membrane was induced by overexpression of miR-223. Thus elevation of miR-223 could be a counter response to maintain normal glucose uptake in diabetic patients [48]. miR-375 negatively regulates glucose-stimulated insulin secretion (GSIS); miR-9 targets the transcription factor, Onecut 2, which inhibits the expression of Granuphilin-a negative regulator of insulin exocytosis [49]. MiR-223 is increased in end-stage ischemic cardiomyopathy, probably due to dysregulation of adaptive mechanism in end-stage patients. MiR-199b is activated by a calcineurin/nuclear factor of activated T cells pathway and miR199b is upregulated in diabetic mice with left ventricular fibrosis and HF [46].
9.5 MiRs and Lipotoxicity
Myocardial steatosis is defined as the accumulation of TGL in the myocardium and precedes the contractile dysfunction and HF as a hallmark of DCM. High serum miR-1 or miR-133a level associated with increased risk of severe myocardial steatosis in patients with T2DM without structural heart disease or inducible ischemia [50].
9.6 MiRs and Heart failure
miRNA-21 level is upregulated in activated fibroblasts of the failing heart. The miR-15 family is found to be significantly increased in cardiac diseases. Expression of six miRNAs (miR-34b, miR-34c, miR-199b, miR-210, miR-223, and miR-650) changed in diabetic patients with HF. MiR-34c and miR-34b are upregulated after acute ischemic damage; these microRNAs are believed to play a fundamental role in the p53 tumor suppressor network [46].
9.7 MiRs and Cardiac remodeling
Genetic deletion of the cardiac-specific miR-208a prevents pathological cardiac remodeling [51]. The miRNAs that are highly related to DCM include miR-9, miR-21, miR-29, miR-30d, miR-34a, miR144, miR150, miR-320 and miR378 [40]. The expression of miR-145 is upregulated and the expression of miR-147 and miR-7 are downregulated in DCM, ultimately inhibiting cardiomyocyte apoptosis. The upregulation of miR-181 inhibited oxidative stress. Upregulation of miR-214 and downregulation of miR-29b attenuate cardiomyocyte fibrosis. Upregulation of miR-155 promotes cardiomyocyte inflammation, which may be an early regulatory mechanism [52]. Mesenchymal stem cells or cardiac progenitor cells genetically modified with miRNAs such as miR-1, miR-21, miR-24, miR-126, miR-155, and miR-221 showed advanced prosurvival properties and regenerative capacity in animal models of cardiovascular disease [53]. Different miRs are either increased or decreased in various stages of DCM as shown in Table 2.
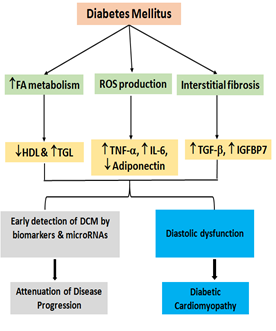
Currently, miR-based therapy is still in its infancy and a number of problems have to be addressed until a broad, reliable, and safe clinical use will be feasible. The development of delivery systems enabling cell-specific uptake and the design of therapeutic molecules without toxic side effects remain a major challenge. Moreover, unwanted off-target effects have to be minimized. After taking these hurdles, miR-based therapy strategies will open up one of the most innovative and promising perspectives in clinical practice [36]. Schematic depiction of the progression of DCM and the role of Biomarkers & miRs to detect early stages of DCM and attenuate disease progression is shown in Figure 3.
|
MicroRNAthophysiological change |
|
|
miR-1 |
Cardiomyocyte differentiation, cardiac arrhythmogenesis |
|
miR-21 |
Proproliferative, anti-apoptotic effect on VSMC, HF-overexpression |
|
miR-19b |
Fibrosis of cardiac myocytes |
|
miR-195 |
Stress-induced hypertrophy |
|
miR-25 |
Cardiac apoptosis |
|
miR-208 |
Cardiac hypertrophy, HF, AMI |
|
miR-132 |
Cardiac hypertrophy & autophagy |
|
miR-133 |
Cardiac hypertrophy & hyperplasia, myocardial matrix remodeling, prolongation of QT interval |
|
miR-206 |
Cardiac apoptosis, cardiac remodeling after a cardiac event |
|
miR-499 |
Cardiac cell differentiation, apoptosis |
|
miR-140 |
Cardiac apoptosis |
|
miR-30e |
Cardiac apoptosis |
|
miR-199a |
Cardiac apoptosis |
|
miR-146a |
Induce inflammation in cardiac myocytes |
|
miR-155 |
Induce inflammation in cardiac myocytes |
|
miR-181 a/b |
Altered Mitochondrial respiration, increased FFA oxidation |
|
miR-347 3b |
Induce inflammation in cardiac myocytes |
|
miR-10a |
Induce inflammation in cardiac myocytes |
|
miR-320 |
Aggravate diabetic cardiomyopathy |
Table 2: Various cardiac specific miRs and their role in pathophysiological process [31, 34, 54].
These miRs are potential serum biomarkers in patients with DCM. Different miRs may impact serials of signaling transductions leading to oxidative stress, apoptosis, sympathetic nervous system activation and RAAS activation, hypertrophic response, fibrosis, oxidative stress and apoptosis, leading to cardiomyopathy in diabetic subjects. These mechanisms are closely correlated and interlinked. Thus they regulate different mechanisms and signaling pathways in promotion or protection of DCM [40].
Developing anti-miRs and miR mimics to treat DCM is an important area of research. Inappropriately elevated miRs which are shown to be causative factor of the disease can be targeted for knockdown or sequestration by the administration of antisense oligonucleotides such as antagomiRs, sponges and erasers. On the other hand, pathological downregulation of miRs can be replenished via the introduction of precursors miRNAs or double-stranded oligonucleotides with their coding sequences, which trigger the maturation of the targeted miRs for their functional implementation. With the growing epidemic of DM and the associated cardiac complications, the value of miR as a new therapeutic target for DCM and HF has invited huge interest among the basic sciences and clinicians. Alterations in miR profile detected in myocardium and the circulation reflects the underlying molecular pathology of DCM. Identification and characterizations of miRs and the pathways they regulate may pave way for development of novel agents to treat or prevent DCM’
10. Exercise in Cardiac Rehabilitation
Individuals with cardiac disease benefit from participation in regular physical exercises and lifestyle modifications. Cardiac rehabilitation (CR) is commonly used to deliver exercises that reduce physical and mental disability and promote an active lifestyle in patients with DCM [56]. Physical activity is highly restricted when the patient in the later stages of DCM, at this stage only palliative care may be of use with limited advantage. Hence early diagnosis and proper preventive measures, including exercise, are crucial for both prevention and better management of DCM. Early identification of high-risk patients is helpful in initiating personalized exercise program; usually prescribed based on risk stratification of the individual. Exercise, in addition to improving cardiac dysfunction, also enhances the outcome of T2DM. Various cardiac rehabilitation measures are found to retard or slow down the progress of diabetic cardiomyopathy [26].
Moderate to high-intensity exercise can increase the survival rate in patients with T2DM. According to the 2019 American Diabetes Association (ADA) guidelines, 150 minutes or more of moderate-to-vigorous intensity aerobic activity per week is recommended to receive the protective effects of exercise. Moderate exercise can reverse these effects by increasing GLUT-4 expression as well as inducing the expression of insulin-sensitive AMP-activated protein kinase (AMPK) and stabilizes energy metabolism dysfunction in DM [22]. American College of Sports Medicine guidelines has give the protocols for Individuals with CVD who can participate in outpatient CR [56]. Frequency, Intensity, Time, and Type (FITT) principle is followed which involves aerobic, resistance and flexibility exercises.
Those participating in maintenance outpatient exercise programs expend approximately 300 kcal per session. Thus, those who attend three times per week expend less than 1,000 kcal per week in exercise sessions. Based on the recommendations of calorie expenditure for cardiovascular disease risk reduction and for weight management, it is important to encourage patients to perform regular exercise in addition to the institutional program. Regular exercise is reported to prevent cardiac apoptosis, fibrosis, oxidative stress, and microvascular diseases, as well as improve cardiac mitochondrial function. Thus, it improves cardiopulmonary fitness and reduces mortality thus slows the development of DCM in T2DM [10].
11. Conclusion
Patients with DM but without overt HF often have abnormalities of diastolic function and subclinical systolic dysfunction. Abnormalities of diastolic function can occur in patients with DM in the absence of HT. Whether these early suggestions of cardiac dysfunction relate to a distinct DCM or to concurrent condition is not clear. The abnormalities of diastolic function or wall thickness reflect the changes of DCM [20]. At present, screening of asymptomatic people with DCM for features of HF is not being recommended. However, when such a strategy is adopted, careful selection of patients would be critical. HF risk may be greater among patients with a longer rather than a shorter duration of DM. Diabetes duration is a difficult parameter, because the delay in recognizing DM could be number years. Also the abnormal LV function appears to be more common in individuals with DM duration of more than 10years [26, 44].
Left ventricular diastolic dysfunction is usually asymptomatic in 50% of apparently healthy diabetic individuals. It is pertinent to understand the time course of development of myocardial apoptosis and fibrosis which are the indicators of the advancing disease. Cardiac myocyte hypertrophy is commonly attributable hyperinsulinemia due to insulin resistance in the setting of increased after load. It is essential to identify diabetic individuals who are likely to develop DCM by measuring biomarkers and miRs. This will benefit the diabetic individuals by early exposure to cardiac rehabilitation which in turn can improve the quality of life. Even though the clinical use of miRNA in CVD risk stratification is not recommended at present, the combination of circulating miRs with biological markers could substantially improve the diagnostic potential of detection of diabetic cardiomyopathy [57].
Ethics Statement
Since this is a review article ethics approval was not required and hence not obtained. The same has been mentioned in the methodology also.
Funding Details
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. All the data and materials required have been mentioned in the manuscript itself and there is no supplementary material which needs to be attached. If published the authors are giving their consent for sharing the information provided in the article with other researchers across the globe.
Acknowledgements
The authors express their gratitude to the institutes for providing the necessary infrastructure in bringing out this article. The authors also wish to acknowledge the authors who contributed to various articles which were the foundation for this article.
Authors Contributions
Dr Santhi Silambanan: conceptualization and design, methodology, analysis, providing intellectual content of critical importance to the work described, revision of article, final version of the article; Dr Swathy Moorthy: conceptualization and design, methodology, analysis, providing intellectual content of critical importance to the work described, original draft version of article, revision of article final version of the article; Dr Vijaya Srinivasan: methodology, analysis, providing intellectual content of critical importance to the work described, original draft version of article, revision of article; Dr Teena Koshy: methodology, providing intellectual content of critical importance to the work described, original draft version of article, revision of article.
Conflicts of Interest
None declared. All the authors express that there was no conflict of interest in writing this review.
References
- Tripathy JP, Thakur JS, Jeet G, et al. Prevalence and risk factors of diabetes in a large community-based study in North India: results from a STEPS survey in Punjab, India. Diabetol Metab Syndr 9 (2017).
- Park S, Moon S, Lee K, et al. Urinary and Blood MicroRNA-126 and -770 are Potential Noninvasive Biomarker Candidates for Diabetic Nephropathy: a Meta-Analysis. Cell Physiol Biochem 46 (2018): 1331-1340.
- Trachanas K, Sideris S, Aggeli C, et al. Diabetic cardiomyopathy: from pathophysiology to treatment. Hellenic J Cardiol 55 (2014): 411-421.
- Baradan R, Hollander JM, Das S. Mitochondrial miRNAs in diabetes: just the tip of the iceberg. Can J Physiol Pharmacol 95 (2017): 1156-1162.
- Mardikar HM, Deshpande NV, Admane P. Recent advances in the management of acute myocardial infarction. J Assoc Physicians India 59 (2011): 31-36.
- Albakri A. Diabetic cardiomyopathy: a review of literature on clinical status and meta-analysis of tissue doppler diagnostic method and the clinical value of intensive glycemic control. Med Clin Arch 2 (2015): 1-12.
- Athithan L, Gulsin GS, McCann GP, et al. Diabetic cardiomyopathy: Pathophysiology, theories and evidence to date. World J Diabetes 10 (2019): 490-510.
- Hathaway QA, Pinti MV, Durr AJ, et al. Regulating microRNA expression: at the heart of diabetes mellitus and the mitochondrion. Am J Physiol Heart Circ Physiol 314 (2018): 293-310.
- Ting-Wei Lee, Kuan-Jen Bai, Ting-I Lee, et al. PPARs modulate cardiac metabolism and mitochondrial function in diabetes. Journal of Biomedical Science 24 (2017): 5.
- Negishi K. Echocardiographic feature of diabetic cardiomyopathy: where are we now? Cardiovasc Diagn Ther 8 (2018): 47-56.
- Elbatreek MH, Pachado MP, Cuadrado A, et al. Reactive Oxygen Comes of Age: Mechanism-Based Therapy of Diabetic End-Organ Damage. Trends Endocrinol Metab 30 (2019): 312-327.
- Ghosh N, Katare R. Molecular mechanism of diabetic cardiomyopathy and modulation of microRNA function by synthetic oligonucleotides. Cardiovasc Diabetol 17 (2018): 43.
- Quinaglia T, Oliveira DC, Matos-Souza JR, et al. Diabetic cardiomyopathy: factual or factoid? Rev Assoc Med Bras 65 (2019): 61-69.
- Jia G, Hill MA, Sowers JR. Diabetic Cardiomyopathy: An Update of Mechanisms Contributing to This Clinical Entity. Circ Res 122 (2018): 624-638.
- Lee WS, Kim J. Diabetic cardiomyopathy: where we are and where we are going. Korean J Intern Med 32 (2017): 404-421.
- Gulsin GS, Athithan L, McCann GP. Diabetic cardiomyopathy: prevalence, determinants and potential treatments. Ther Adv Endocrinol Metab 10 (2019): 2042018819834869.
- Shaver A, Nichols A, Thompson E, et al. Role of Serum Biomarkers in Early Detection of Diabetic Cardiomyopathy in the West Virginian Population. Int J Med Sci 13 (2016): 161-168.
- Kenny HC, Abel ED. Heart Failure in Type 2 Diabetes Mellitus. Circ Res 124 (2019): 121-141.
- Lee MMY, McMurray JJV, Lorenzo-Almorós A, et al. Diabetic cardiomyopathy. Heart 105 (2019): 337-345.
- Gulsin GS, Athithan L, McCann GP. Diabetic cardiomyopathy: prevalence, determinants and potential treatments. Ther Adv Endocrinol Metab 10 (2019): 2042018819834869.
- Filardi T, Ghinassi B, Di Baldassarre A, et al. Cardiomyopathy Associated with Diabetes: The Central Role of the Cardiomyocyte. Int J Mol Sci 20 (2019): 3299.
- Seo DY, Ko JR, Jang JE, et al. Exercise as A Potential Therapeutic Target for Diabetic Cardiomyopathy: Insight into the Underlying Mechanisms. Int J Mol Sci 20 (2019): 6284.
- Lai S, Fu X, Yang S, et al. G protein-coupled receptor kinase-2: A potential biomarker for early diabetic cardiomyopathy. J Diabetes 12 (2020): 247-258.
- Zhang Y, Sun X, Icli B, et al. Emerging Roles for MicroRNAs in Diabetic Microvascular Disease: Novel Targets for Therapy. Endocr Rev 38 (2017): 145-168.
- Flores-Ramírez R, Azpiri-López JR, González-González JG, et al. Global longitudinal strain as a biomarker in diabetic cardiomyopathy. A comparative study with Gal-3 in patients with preserved ejection fraction. Arch Cardiol Mex 87 (2017): 278-285.
- Marwick TH, Ritchie R, Shaw JE, et al. Implications of Underlying Mechanisms for the Recognition and Management of Diabetic Cardiomyopathy. J Am Coll Cardiol 71 (2018): 339-351.
- Palomer X, Pizarro-Delgado J, Vázquez-Carrera M. Emerging Actors in Diabetic Cardiomyopathy: Heartbreaker Biomarkers or Therapeutic Targets? Trends Pharmacol Sci 39 (2018): 452-467.
- Borghetti G, von Lewinski D, Eaton DM, et al. Diabetic Cardiomyopathy: Current and Future Therapies. Beyond Glycemic Control. Front Physiol 9 (2018): 1514.
- Alberto Domi'nguez-Rodri'guez, Pablo Avanzas, Julia Gonza'lez-Gonza' lez, et al. Growth Differentiation Factor 15, a New Prognostic Marker in Diabetic Cardiomyopathy Scientific letters / Rev Esp Cardiol 69 (2016): 75-85.
- Li H, Li X, Guo j, et al. Identification of biomarkers and mechanisms of diabetic cardiomyopathy using microarray data. Cardiology Journal (2018).
- Copier CU, León L, Fernández M, et al. Circulating miR-19b and miR-181b are potential biomarkers for diabetic cardiomyopathy. Sci Rep 7 (2017): 13514.
- Arunachalam Gnanapragasam, Upadhyay Rohit, Ding Hong, et al. MicroRNA Signature and Cardiovascular Dysfunction. Journal of Cardiovascular Pharmacology 65 (2015): 419-429.
- Rawal S, Manning P, Katare R. Cardiovascular microRNAs: as modulators and diagnostic biomarkers of diabetic heart disease. Cardiovasc Diabetol 13 (2014).
- Quiat D, Olson EN. MicroRNAs in cardiovascular disease: from pathogenesis to prevention and treatment. J Clin Invest 123 (2013): 11-18.
- David De Gonzalo-Calvo, Eduardo Iglesias-Gutie´ rrez, Vicenta Llorente-Corte´ s. Epigenetic Biomarkers and Cardiovascular Disease: Circulating MicroRNAs. Rev Esp Cardiol 70 (2017): 696-698.
- Kreth S, Hübner M, Hinske LC. MicroRNAs as Clinical Biomarkers and Therapeutic Tools in Perioperative Medicine. Anesth Analg 126 (2018): 670-681.
- Li H, Fan J, Yin Z, et al. Identification of cardiac-related circulating microRNA profile in human chronic heart failure. Oncotarget 7 (2016): 33-45.
- Rahimi M, Sadat Aghabozorg Afjeh S, Omrani M, et al. The Therapeutic Effects and Role of MiRNAs in Coronary Artery Disease: A Systemic Review. IBBJ 3 (2017): 47-54.
- Heggermont WA, Heymans S. MicroRNAs are involved in end-organ damage during hypertension. Hypertension 60 (2012): 1088-1093.
- Guo R, Nair S. Role of microRNA in diabetic cardiomyopathy: From mechanism to intervention. Biochim Biophys Acta Mol Basis Dis 1863 (2017): 2070-2077.
- Busch A, Eken SM, Maegdefessel L. Prospective and therapeutic screening value of non-coding RNA as biomarkers in cardiovascular disease. Ann Transl Med 4 (2016): 236.
- Colpaert RMW, Calore M. MicroRNAs in Cardiac Diseases. Cells 8 (2019): 737.
- Salinas J, Lin H, Aparico HJ, et al. Whole blood microRNA expression associated with stroke: Results from the Framingham eart Study. PLoS ONE 14 (2019): e0219261.
- Chien HY, Lee TP, Chen CY, et al. Circulating microRNA as a diagnostic marker in populations with type 2 diabetes mellitus and diabetic complications. J Chin Med Assoc 78 (2015): 204-211.
- Wang C, Wan S, Yang T, et al. Increased serum microRNAs are closely associated with the presence of microvascular complications in type 2 diabetes mellitus. Sci Rep 6 (2016): 20032.
- Greco S, Fasanaro P, Castelvecchio S, et al. MicroRNA dysregulation in diabetic ischemic heart failure patients. Diabetes 61 (2012): 1633-1641.
- Chen C, Yang S, Li H, et al. Mir30c Is Involved in Diabetic Cardiomyopathy through Regulation of Cardiac Autophagy via BECN1. Mol Ther Nucleic Acids 7 (2017): 127-139.
- Zhu H, Leung SW. Identification of microRNA biomarkers in type 2 diabetes: a meta-analysis of controlled profiling studies. Diabetologia 58 (2015): 900-911.
- Shantikumar S, Caporali A, Emanueli C. Role of microRNAs in diabetes and its cardiovascular complications. Cardiovasc Res 93 (2012): 583-593.
- Fan B, Luk AOY, Chan JCN, et al. MicroRNA and Diabetic Complications: A Clinical Perspective. Antioxid Redox Signal 29 (2018): 1041-1063.
- Andrew Walayat, Meizi Yang and DaLiao Xiao. Therapeutic Implication of miRNA in Human Disease. In book: Antisense Therapy. Intechopen (2018).
- Huang YM, Li WW, Wu J, et al. The diagnostic value of circulating microRNAs in heart failure. Exp Ther Med 17 (2019): 1985-2003.
- Chistiakov DA, Sobenin IA, Orekhov AN. Strategies to deliver microRNAs as potential therapeutics in the treatment of cardiovascular pathology. Drug Delivery 19 (2012): 392-405.
- Ingrid Fomison-Nurse and Rajesh Katare. Emerging Roles of Micrornas in Diabetic Cardiomyopathy. Nurse and Katare, J Diabetes Metab 5 (2014): 394.
- Shaver A, Nichols A, Thompson E, et al. Role of Serum Biomarkers in Early Detection of Diabetic Cardiomyopathy in the West Virginian Population. Int J Med Sci 13 (2016): 161-168.
- Deborah Riebe, Jonathan K Ehrman, Gary Liguori. ACSM’s Guidelines for Exercise Testing and Prescription 10th Philadelphia: Wolters Kluwer (2018): 332-338.
- Chabior A, Pordzik J, Mirowska-Guzel D, et al. The role of acetylsalicylic acid and circulating microRNAs in primary prevention of cardiovascular events in patients with Diabetes Mellitus Type 2 - A Review. Ann Agric Environ Med 26 (2019): 512-522.