Chemical composition and uses of Honey: A Review
Article Information
Dessie Ashagrie Tafere*
College of Natural and Computational Science Department of Chemistry, Mekdela Amba University, Ethiopia
*Corresponding Author: Dessie Ashagrie Tafere, College of Natural and Computational Science, Department of Chemistry, Mekdela Amba University, Ethiopia
Received: 28 June 2021; Accepted: 05 July 2021; Published: 13 July 2021
Citation: Dessie Ashagrie Tafere. Chemical composition and uses of Honey: A Review. Journal of Food Science and Nutrition Research 4 (2021): 194-201.
View / Download Pdf Share at FacebookAbstract
Honey is an organic natural substance that is produced from the nectar of flowers by Apis mellifera and is a sweet, flavorful liquid. It is one of the most popular natural sweet substances. From a chemical point of view, it could be defined as a natural food mainly composed of sugars and water together with minor constituent such as minerals, vitamins, amino acids, organic acids, flavonoids and other phenolic compounds and aromatic substances. Its composition is particularly variable, depending on its botanical and geographical origins. Honey is used to feed bees during the winter. For centuries, honey has been used as food and as natural medicine, being prescribed by physicians of many ancient cultures for the treatment of a wide variety of ailments.
Keywords
<p>Honey, chemical composition, uses</p>
Honey articles, chemical composition articles, uses articles
Honey articles Honey Research articles Honey review articles Honey PubMed articles Honey PubMed Central articles Honey 2023 articles Honey 2024 articles Honey Scopus articles Honey impact factor journals Honey Scopus journals Honey PubMed journals Honey medical journals Honey free journals Honey best journals Honey top journals Honey free medical journals Honey famous journals Honey Google Scholar indexed journals chemical composition articles chemical composition Research articles chemical composition review articles chemical composition PubMed articles chemical composition PubMed Central articles chemical composition 2023 articles chemical composition 2024 articles chemical composition Scopus articles chemical composition impact factor journals chemical composition Scopus journals chemical composition PubMed journals chemical composition medical journals chemical composition free journals chemical composition best journals chemical composition top journals chemical composition free medical journals chemical composition famous journals chemical composition Google Scholar indexed journals cereal-based products articles cereal-based products Research articles cereal-based products review articles cereal-based products PubMed articles cereal-based products PubMed Central articles cereal-based products 2023 articles cereal-based products 2024 articles cereal-based products Scopus articles cereal-based products impact factor journals cereal-based products Scopus journals cereal-based products PubMed journals cereal-based products medical journals cereal-based products free journals cereal-based products best journals cereal-based products top journals cereal-based products free medical journals cereal-based products famous journals cereal-based products Google Scholar indexed journals sweetness articles sweetness Research articles sweetness review articles sweetness PubMed articles sweetness PubMed Central articles sweetness 2023 articles sweetness 2024 articles sweetness Scopus articles sweetness impact factor journals sweetness Scopus journals sweetness PubMed journals sweetness medical journals sweetness free journals sweetness best journals sweetness top journals sweetness free medical journals sweetness famous journals sweetness Google Scholar indexed journals grape sugar articles grape sugar Research articles grape sugar review articles grape sugar PubMed articles grape sugar PubMed Central articles grape sugar 2023 articles grape sugar 2024 articles grape sugar Scopus articles grape sugar impact factor journals grape sugar Scopus journals grape sugar PubMed journals grape sugar medical journals grape sugar free journals grape sugar best journals grape sugar top journals grape sugar free medical journals grape sugar famous journals grape sugar Google Scholar indexed journals yeast fermentation articles yeast fermentation Research articles yeast fermentation review articles yeast fermentation PubMed articles yeast fermentation PubMed Central articles yeast fermentation 2023 articles yeast fermentation 2024 articles yeast fermentation Scopus articles yeast fermentation impact factor journals yeast fermentation Scopus journals yeast fermentation PubMed journals yeast fermentation medical journals yeast fermentation free journals yeast fermentation best journals yeast fermentation top journals yeast fermentation free medical journals yeast fermentation famous journals yeast fermentation Google Scholar indexed journals nutritional variety articles nutritional variety Research articles nutritional variety review articles nutritional variety PubMed articles nutritional variety PubMed Central articles nutritional variety 2023 articles nutritional variety 2024 articles nutritional variety Scopus articles nutritional variety impact factor journals nutritional variety Scopus journals nutritional variety PubMed journals nutritional variety medical journals nutritional variety free journals nutritional variety best journals nutritional variety top journals nutritional variety free medical journals nutritional variety famous journals nutritional variety Google Scholar indexed journals melanoidins articles melanoidins Research articles melanoidins review articles melanoidins PubMed articles melanoidins PubMed Central articles melanoidins 2023 articles melanoidins 2024 articles melanoidins Scopus articles melanoidins impact factor journals melanoidins Scopus journals melanoidins PubMed journals melanoidins medical journals melanoidins free journals melanoidins best journals melanoidins top journals melanoidins free medical journals melanoidins famous journals melanoidins Google Scholar indexed journals food spoilage articles food spoilage Research articles food spoilage review articles food spoilage PubMed articles food spoilage PubMed Central articles food spoilage 2023 articles food spoilage 2024 articles food spoilage Scopus articles food spoilage impact factor journals food spoilage Scopus journals food spoilage PubMed journals food spoilage medical journals food spoilage free journals food spoilage best journals food spoilage top journals food spoilage free medical journals food spoilage famous journals food spoilage Google Scholar indexed journals
Article Details
1. Introduction
Honey is a natural substance produced by honeybees (Apis mellifera) from the nectar of flowers which is a sweet, flavorful, viscose liquid [1]. It has been used as a food and medical product since the earliest times. Honey is a complex mixture and presents very great variations in composition and characteristics due to its geographical and botanical origin, its main features depending on the floral origin or the nectar foraged by bees [2]. The composition and quality of honey also depend on several environmental factors during production such as weather and humidity inside the hive, nectar conditions and treatment of honey during extraction and storage. The composition of honey varies with the feeding of the bees. Honey has been reported to contain more than 180 substances and is considered as an important part of traditional medicine. Honey has numerous uses and functional applications worldwide such as in food systems, religious and magical ceremonies as well as in human and veterinary medicine [3]. It is a very important energy food and is used as an ingredient in hundreds of manufactured foods, mainly in cereal-based products, for its sweetness, color, flavor, caramelization, pumpability and viscosity [4].

Figure 1: Honey
2. Chemical composition
The composition of honey basically varies to the floral source, but seasonal, environmental factors and processing conditions are also important.
2.1 Carbohydrates
Sugar in honey is not a single species, but consists of three kinds of sugar. These are the fruit sugar (fructose), which has among the highest (41%), grape sugar (glucose), which has about 34% of ordinary sugar (sucrose) which is between 1 and 2% [5]. The ratio of one type of sugar to other depends of the source, i.e., flower pasture, and to some extent on enzyme invertase, which breaks down regular sugar in grape and fruit. This enzyme is located in the flower from which the bees collect nectar, but it is also present in the bee’s body [6].
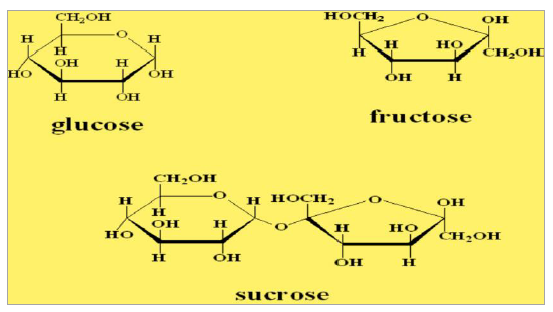
Figure 2: Chemical structure of three kinds of sugar
2.2 Amino acids and proteins
Proteins come in honey from nectar and pollen as a integral parts of plants. Proteins in honey may be in the form of a very complex structure or in the form of simple compounds, i.e., amino acids [7]. The content of amino acids and proteins is relatively small, at the most 0.7 %. Honey contains almost all physiologically important amino acids. The main amino acid is proline is a measure of honey ripeness. The proline content of normal honeys should be more than 200 mg/kg. Values below 180 mg/kg mean that the honey is probably adulterated by sugar addition [8].
2.3 Aroma compounds and phenolics
Honey volatiles are the substances responsible for the honey aroma. Research on honey volatiles started in the early 1960s. Recently, by studying volatiles isolated from honey [9], it was found that most volatile compounds originate probably from the plant, but some of them are added by bees. Until the present time about 600 compounds have been characterized in different honeys. Phenolic acids and polyphenols are plant-derived secondary metabolites. These compounds have been used as chemotaxonomic markers in plant systematics. They have been suggested as possible markers for the determination of botanical origin of honey [10]. Dark colored honeys are reported to contain more phenolic acid derivatives but less flavonoids than light-colored ones [11].
2.4 HMF (HydroxyMethylFurfuraldehyde)
HMF is a six-carbon heterocyclic organic compound containing both aldehyde and alcohol (hydroxymethyl) functional groups [12]. The ring of the structure is centered on furan moieties, whereas the two functional groups, i.e., formyl and hydroxy-methyl groups, are linked at the second and fifth positions, respectively (Figure 3). HMF is a solid, yellow substance that has a low melting point but is highly soluble in water [13].
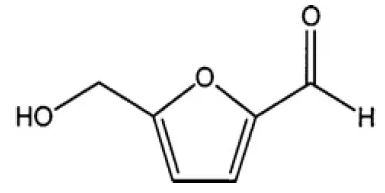
Figure 3: Chemical structure of HMF
HMF is a breakdown product of fructose (one of the main sugars in honey) that is formed slowly and naturally during the storage of honey, and much more quickly when honey is heated. The amount of HMF present in honey is the reference used as a guide to the amount of heating that has taken place: the higher the HMF value, the lower the quality of the honey is considered to be [15]. Some countries set an HMF limit for imported honey (sometimes 40 milligrams per kilogram), and honey with an HMF value higher than this limit will not be accepted [16].
2.5 Minerals and trace elements
Honey contains varying amounts of mineral substances. The main element found in honey is potassium, besides many other elements (as shown in table 1). Potassium, with an average of about one third of the total, is the main mineral element, but there is a wide variety of trace elements Several investigations have shown that the trace element content of honey depends mainly on the botanical origin of honey. Minerals has about 3.68% [17]. Although this part of the honey does not make a large amount, minerals in honey raise the value of honey for human consumption. Honey contains most of the minerals: potassium, chlorine, sulfur, calcium, sodium, phosphorus, magnesium, silicon, iron, manganese and copper [18]. When the observed mean value, dark types of honey are richer in minerals than lighter. Of course, singles can find a darker species that are poorer than some lighter species [19].
|
Minerals |
Unit |
Average amount in 100g honey |
Recommended daily intake |
|
Calcium |
mg |
4-30 |
1000 |
|
Chlorine |
mg |
2-20 |
|
|
Copper |
mg |
0.01-0.1 |
2 |
|
Iron |
mg |
1-3.4 |
18 |
|
Magnesium |
mg |
0.7-13 |
400 |
|
Phosphorous |
mg |
2-60 |
1000 |
|
Potassium |
mg |
10-470 |
- |
|
Sodium |
mg |
0.6-40 |
- |
|
Zinc |
mg |
0.2-0.5 |
15 |
Table 1: Minerals in honey in relation to human requirements
2.6 Acidity and pH
Acids are also components of honey. Before it was believed that bees by stomach inserted bee venom into the cell honeycomb with honey and make it so conserved. Given that one of the main components of bee venom is formic acid, it was thought that the honey has a formic acid [20]. Even some people urged to other not to use honey because of that. Studies have shown that they are completely different acids that are composed in honey, mostly apple and lemon acid. Honey is a buffer, that means that that its pH does not change by the addition of small quantities of acids and bases. The buffer capacity is due to the content of phosphates, carbonates and other mineral salts [21].
2.7 Water
The water content of honey (water-in-honey) is the quality aspect that determines the ability of honey to remain fresh and to avoid spoilage by yeast fermentation. Raw honey can have a water-in-honey content of less than 14% and the lower the water content the higher the perceived value of the honey [22]. It is internationally recognized that good quality honey should be processed at less than 20% water content. Low water content is desirable because honey may begin to ferment and lose its fresh quality if the water-in-honey is greater than 20%. Unpasteurized honey ferments because it contains wild yeast. However, due to honey's high sugar concentration these yeasts are less likely to cause fermentation in honey with low water content [23].
3. Uses of Honey
3.1 For bees
Bees produce honey to act as a food store for the colony for periods when there are no flowers, or the climate is adverse. For example, during the winters of northern, temperate countries, few plants are flowering between October and March, and bee colonies need honey stores to survive throughout this flowering dearth period, and when it may be too cold to leave the nest [24]. In tropical countries, bees need to survive through seasons when there are no flowers, periods of drought, or when bees are not able to forage because of rain or other adverse weather.
3.2 As food for humans
Honey is a useful source of high-carbohydrate food, and usually contains a rich diversity of minor constituents (minerals, proteins, vitamins and others), adding nutritional variety to human diets [25].
3.3 As a medicine or tonic
3.3.1 Antibacterial activity: Honey has proven antimicrobial activity [26]. Honey inhibits a broad spectrum of bacterial species [27]. The alcohol extracts of honey exhibit an inhibitory effect to array of bacterial species including aerobes and anaerobes, Gram positives, and Gram negatives. Honey has powerful antimicrobial effects against pathogenic and non-pathogenic micro-organisms (yeasts and fungi), even against those that developed resistance to many antibiotics. The antimicrobial effects could be bacteriostatic or bactericidal depending on the concentration that is used [28].
3.3.2 The antioxidant property of honey: Honey is being used since long time both in medical and domestic needs, but only recently its antioxidant property has come to limelight [29]. With increasing demand for antioxidant supply in the food, honey is becoming popular as a source of antioxidant. Oxidative stress results from lack of balancing chemical reaction between the production of free radicals and the natural protective effect of our body resulting into cellular damage and disruption of genetic structure the main antioxidants in honey are the phenols, such as quercetin, hesperetin and chyrsin, and the Maillard products called melanoidins. The phenol quercetin directly binds to and strongly inhibits cellular transcription factors’ activities [30]. The inhibition of the transcription factors surpasses the phosphorylation and activation process which avoids cellular effect of the free radicals. It, also, induces apoptosis (programmed cell death) of human osteosarcoma cells and reduces protein expression levels in human fibrosarcoma cells [31].
3.3.3 Wound healing property of honey: The development of wound infection has deleterious effects on patients by causing increased pain, discomfort and inconvenience and can lead to life-threatening illness or even death [32]. Also, it interrupts the healing process, contributing to extended hospital stays, as well as increased treatment costs in terms of antibiotics, dressings and staff time. Wound healing can be affected by endogenous (pathophysiology) and exogenous (micro-organisms) factors [33]. The risk of wound infection increases as local conditions favor bacterial invasion and growth [34]. Honey is one of naturally existing remedies that has been applied in the treatment of wounds [35]. It promotes faster wound healing through its regenerative tissue growth and epithelization effects, with little or no formation of scars. Furthermore, prostaglandins and nitric oxides play a major role in the healing process. Honey has proven safety for use. External application of honey dressings as well as application to mucous layers of the body cavities didn’t show signs of allergic reaction and side effects [36].
3.4 Other uses
Honey is widely used as a source of sugars for making honey wines and beers, and in the manufacture of many secondary products: breakfast cereals, bakery goods, and a multitude of other value-added products.
|
Bacterial Species |
Disease caused |
|
Bacillus anthracis |
Anthrax |
|
Corynebacterium diphtheriae |
Diphtheria |
|
Escherichia coli |
diarrhoea, septicaemia, urinary infections, wound infections |
|
Haemophilus influenzae |
Ear infections, meningitus, respiratory infections, sinusitis |
|
Klebsiella pneumoniae |
Pneumonia |
|
Mycobacterium tuberculosis |
Tuberculosis |
|
Proteus spp. |
Septicaemia, urinary infections |
|
Pseudomonas aeruginosa |
urinary infections, wound infections |
|
Salmonella spp. |
Diarrhea |
|
Salmonella choleraesuis |
Septicaemia |
|
Salmonella typhi |
Typhoid |
|
Salmonella typhimurium |
Wound infections |
|
Serrata marcescens |
septicaemia, wound infections |
|
Shigella spp. |
Dysentery |
|
Staphylococcus aureus |
Abscesses, boils, carbuncles, impetigo, wound infections |
|
Streptococcus faecalis |
Urinary infections |
|
Streptococcus mutans |
Dental carries |
|
Streptococcus pneumoniae |
Ear infections, meningitis, pneumonia, sinusitis |
|
Streptococcus pyogenes |
Ear infections, impetigo, puerperal fever, rheumatic fever, scarlet fever, sore throat, wound infections |
Table 2: List of bacterial species sensitive to honey/honey products and diseases they cause [28].
4. Conclusion
High-quality honey has a variety of positive nutritional and health effects. The quality of honey depends on its floral origin and chemical composition. Honey has been always taken place in diet not only nutritional aspects but also for health properties. The main nutrition- and health-relevant components are the carbohydrates, which make it an excellent energy source, especially for children and sportsmen. Honey is highly special, nutritious, functional and healthy food. Except nutritious value, it inhibits some food spoilage organisms too. As it has high antioxidant activity it is preferred as not only direct consumption but also supportive or preservative in foods. To make use of the miracles of honey, the tricks of honey should be avoided. Quality honey production and consumption should be increased. Adulteration of honey, decreases income of honest producers and it also has negative effects on consumers’ nutrition and health.
References
- White Jr JW. Honey. Advances in food research 24 (1978): 287-374.
- Machado De-Melo AA, Almeida-Muradian LBD, Sancho MT et al. Composition and properties of Apis mellifera honey: A review. Journal of Apicultural Research 57 (2018): 5-37.
- Ajibola A, Chamunorwa JP, Erlwanger KH Nutraceutical values of natural honey and its contribution to human health and wealth. Nutrition & metabolism 9 (2012): 1-12.
- Nigussie K, Subramanian PA, Mebrahtu G Physicochemical analysis of Tigray honey: An attempt to determine major quality markers of honey. Bulletin of the Chemical Society of Ethiopia 26 (2012).
- Cummings JH, Stephen AM. Carbohydrate terminology and classification. European journal of clinical nutrition 61 (2007): S5-S18.
- Di Pasquale G, Salignon M, Le Conte Y et al. Influence of pollen nutrition on honey bee health: do pollen quality and diversity matter? PloS one 8 (2013): e72016.
- Alvarez-Suarez J, Giampieri F, Battino M. Honey as a source of dietary antioxidants: structures, bioavailability and evidence of protective effects against human chronic diseases. Current medicinal chemistry 20 (2013): 621-638.
- Bogdanov S. Honey composition. The honey book (2009): 27-36.
- Bogdanov S, Martin P. Honey authenticity. Mitteilungen aus Lebensmitteluntersuchung und Hygiene 93 (2002): 232-254.
- Bogdanov S, Ruoff K, Oddo LP. Physico-chemical methods for the characterisation of unifloral honeys: a review. Apidologie 35 (2004): S4-S17.
- Gheldof N, Engeseth NJ. Antioxidant capacity of honeys from various floral sources based on the determination of oxygen radical absorbance capacity and inhibition of in vitro lipoprotein oxidation in human serum samples. Journal of agricultural and food chemistry 50 (2002): 3050-3055.
- Lichtenthaler FW. Unsaturated O-and N-heterocycles from carbohydrate feedstocks. Accounts of chemical research 35 (2002): 728-737.
- Shapla UM, Solayman M, Alam N, et al. 5-Hydroxymethylfurfural (HMF) levels in honey and other food products: effects on bees and human health. Chemistry Central Journal 12 (2018): 1-18.
- Tosi EA, Ré E, Lucero H, et al. Effect of honey high-temperature short-time heating on parameters related to quality, crystallisation phenomena and fungal inhibition. LWT-Food Science and Technology 37 (2004): 669-678.
- Da Silva PM, Gauche C, Gonzaga LV, et al. Honey: Chemical composition, stability and authenticity. Food chemistry 196 (2016): 309-323.
- Rodriguez-Otero JL, Paseiro P, Simal J, et al. Mineral content of the honeys produced in Galicia (North-west Spain). Food Chemistry 49 (1994): 169-171.
- Mattoon WR. The southern cypress (No. 272). US Department of Agriculture (1915).
- Aili SR, Touchard A, Escoubas P, et al. Diversity of peptide toxins from stinging ant venoms. Toxicon 92 (2014): 166-178.
- Ensminger ME, Ensminger AH. Foods & Nutrition Encyclopedia, Two Volume Set. CRC press.
- Machado De-Melo AA, Almeida-Muradian LBD, Sancho MT, et al. Composition and properties of Apis mellifera honey: A review. Journal of Apicultural Research 57 (2018): 5-37.
- Molan PC Authenticity of honey. In Food authentication. Springer, Boston, MA (2006): 259-303.
- Hatjina F, Costa C, Büchler R, et al. Population dynamics of European honey bee genotypes under different environmental conditions. Journal of Apicultural Research 53 (2014): 233-247.
- Maughan R. The athlete’s diet: nutritional goals and dietary strategies. Proceedings of the nutrition Society 61 (2002): 87-96.
- Basson NJ, Grobler SR. Antimicrobial activity of two South African honeys produced from indigenous Leucospermum cordifolium and Erica species on selected micro-organisms. BMC Complementary and Alternative Medicine 8 (2008): 1-4.
- Willix DJ, Molan PC, Harfoot CG. A comparison of the sensitivity of wound-infecting species of bacteria to the antibacterial activity of manuka honey and other honey. Journal of applied bacteriology 73 (1992): 388-394.
- Seil JT, Webster TJ. Antimicrobial applications of nanotechnology: methods and literature. International journal of nanomedicine 7 (2012): 2767.
- Ocampo PS, Lázár V, Papp B, et al. Antagonism between bacteriostatic and bactericidal antibiotics is prevalent. Antimicrobial agents and chemotherapy 58 (2014): 4573-4582.
- Nair HK, Rao KV, Aalinkeel R et al. Inhibition of prostate cancer cell colony formation by the flavonoid quercetin correlates with modulation of specific regulatory genes. Clinical and Diagnostic Laboratory Immunology 11 (2004): 63.
- Anantharaju PG, Gowda PC, Vimalambike MG et al. An overview on the role of dietary phenolics for the treatment of cancers. Nutrition journal 15 (2016): 1-16.
- Hagberg C, Georgi R, Krier C. Complications of managing the airway. Best Practice & Research Clinical Anaesthesiology 19 (2005): 641-659.
- Bowler PG. Wound pathophysiology, infection and therapeutic options. Annals of medicine 34 (2002): 419-427.
- Robson MC. Wound infection: a failure of wound healing caused by an imbalance of bacteria. Surgical Clinics of North America 77 (1997): 637-650.
- Pereira RF, Bartolo PJ. Traditional therapies for skin wound healing. Advances in wound care 5 (2016): 208-229.
- Eteraf-Oskouei T, Najafi M. Traditional and modern uses of natural honey in human diseases: a review. Iranian journal of basic medical sciences 16 (2013): 731.
- Krell R. Value-added products from beekeeping (No. 124). Food & Agriculture Org (1996).
