Automated Synthesis Method to Produce the PET Tracer [68Ga]Ga-FAPI-46 for Clinical Applications: Development, Optimization and Validation
Article Information
Silvia Migliari*, Maura Scarlattei, Giorgio Baldari, Livia Ruffini
1Nuclear Medicine Division, Azienda Ospedaliero-Universitaria di Parma, via Gramsci 14, 43126 Parma, Italy
*Corresponding author: Silvia Migliari, Nuclear Medicine Division, Azienda Ospedaliero-Universitaria di Parma, via Gramsci 14, 43126 Parma, Italy.
Received: 19 July 2023; Accepted: 24 July 2023; Published: 30 August 2023
Citation: Silvia Migliari, Maura Scarlattei, Giorgio Baldari, Livia Ruffini. Automated Synthesis Method to Produce the PET Tracer [68Ga]Ga-FAPI-46 for Clinical Applications: Development, Optimization and Validation. Journal of Biotechnology and Biomedicine 6 (2023): 336-346.
View / Download Pdf Share at FacebookAbstract
Fibroblast Activation Protein (FAP) is a serine protease selectively expressed in many disorders associated with fibrotic dysregulation. FAP expression in healthy tissues is low, but significantly elevated in sites of tissue remodelling and repair. This specific pattern of expression makes FAP an ideal target for imaging and therapy and then FAP-Specific Small-Molecule Inhibitors (FAPIs) have been developed. The most promising molecule has been found FAPI-46, functionalized with DOTA to obtain a PET probe. Our goal was to develop, optimize and validate a new automated synthesis method to label DOTA-FAPI-46 with Ga-68 and a new quality control system to make the radiopharmaceutical available. The radiopharmaceutical production was optimized scaling down the amount of DOTA-FAPI-46 (50 - 10 μg). The synthesis of [68Ga]Ga-FAPI-46 was done using the Scintomics GRP® module with the already estabilished synthesis template for [68Ga]Ga-DOTATOC/[68Ga]Ga-PSMA. Synthesis efficiency and relevant quality control parameters were evaluated for each produced batch in accordance with the European Pharmacopeia. Best results were yielded with 20 μg DOTA-FAPI-46 and three different batches of validation were obtained with optimal radiochemical yield (67.75%) as well radiochemical purity (99.76%) and molar activity (26.23 GBq/µmol). [68Ga]Ga-FAPI-46 was successfully synthesized and it is available for multi-dose application in clinical settings.
Keywords
[68Ga]68Ga-radiopharmaceuticals; [68Ga]Ga-FAPI-46; Fibroblast activation protein (FAP); PET imaging
[68Ga]68Ga-radiopharmaceuticals articles [68Ga]68Ga-radiopharmaceuticals Research articles [68Ga]68Ga-radiopharmaceuticals review articles [68Ga]68Ga-radiopharmaceuticals PubMed articles [68Ga]68Ga-radiopharmaceuticals PubMed Central articles [68Ga]68Ga-radiopharmaceuticals 2023 articles [68Ga]68Ga-radiopharmaceuticals 2024 articles [68Ga]68Ga-radiopharmaceuticals Scopus articles [68Ga]68Ga-radiopharmaceuticals impact factor journals [68Ga]68Ga-radiopharmaceuticals Scopus journals [68Ga]68Ga-radiopharmaceuticals PubMed journals [68Ga]68Ga-radiopharmaceuticals medical journals [68Ga]68Ga-radiopharmaceuticals free journals [68Ga]68Ga-radiopharmaceuticals best journals [68Ga]68Ga-radiopharmaceuticals top journals [68Ga]68Ga-radiopharmaceuticals free medical journals [68Ga]68Ga-radiopharmaceuticals famous journals [68Ga]68Ga-radiopharmaceuticals Google Scholar indexed journals [68Ga]Ga-FAPI-46 articles [68Ga]Ga-FAPI-46 Research articles [68Ga]Ga-FAPI-46 review articles [68Ga]Ga-FAPI-46 PubMed articles [68Ga]Ga-FAPI-46 PubMed Central articles [68Ga]Ga-FAPI-46 2023 articles [68Ga]Ga-FAPI-46 2024 articles [68Ga]Ga-FAPI-46 Scopus articles [68Ga]Ga-FAPI-46 impact factor journals [68Ga]Ga-FAPI-46 Scopus journals [68Ga]Ga-FAPI-46 PubMed journals [68Ga]Ga-FAPI-46 medical journals [68Ga]Ga-FAPI-46 free journals [68Ga]Ga-FAPI-46 best journals [68Ga]Ga-FAPI-46 top journals [68Ga]Ga-FAPI-46 free medical journals [68Ga]Ga-FAPI-46 famous journals [68Ga]Ga-FAPI-46 Google Scholar indexed journals Fibroblast activation protein (FAP) articles Fibroblast activation protein (FAP) Research articles Fibroblast activation protein (FAP) review articles Fibroblast activation protein (FAP) PubMed articles Fibroblast activation protein (FAP) PubMed Central articles Fibroblast activation protein (FAP) 2023 articles Fibroblast activation protein (FAP) 2024 articles Fibroblast activation protein (FAP) Scopus articles Fibroblast activation protein (FAP) impact factor journals Fibroblast activation protein (FAP) Scopus journals Fibroblast activation protein (FAP) PubMed journals Fibroblast activation protein (FAP) medical journals Fibroblast activation protein (FAP) free journals Fibroblast activation protein (FAP) best journals Fibroblast activation protein (FAP) top journals Fibroblast activation protein (FAP) free medical journals Fibroblast activation protein (FAP) famous journals Fibroblast activation protein (FAP) Google Scholar indexed journals PET imaging articles PET imaging Research articles PET imaging review articles PET imaging PubMed articles PET imaging PubMed Central articles PET imaging 2023 articles PET imaging 2024 articles PET imaging Scopus articles PET imaging impact factor journals PET imaging Scopus journals PET imaging PubMed journals PET imaging medical journals PET imaging free journals PET imaging best journals PET imaging top journals PET imaging free medical journals PET imaging famous journals PET imaging Google Scholar indexed journals radiopharmaceutical practices articles radiopharmaceutical practices Research articles radiopharmaceutical practices review articles radiopharmaceutical practices PubMed articles radiopharmaceutical practices PubMed Central articles radiopharmaceutical practices 2023 articles radiopharmaceutical practices 2024 articles radiopharmaceutical practices Scopus articles radiopharmaceutical practices impact factor journals radiopharmaceutical practices Scopus journals radiopharmaceutical practices PubMed journals radiopharmaceutical practices medical journals radiopharmaceutical practices free journals radiopharmaceutical practices best journals radiopharmaceutical practices top journals radiopharmaceutical practices free medical journals radiopharmaceutical practices famous journals radiopharmaceutical practices Google Scholar indexed journals harmonisation articles harmonisation Research articles harmonisation review articles harmonisation PubMed articles harmonisation PubMed Central articles harmonisation 2023 articles harmonisation 2024 articles harmonisation Scopus articles harmonisation impact factor journals harmonisation Scopus journals harmonisation PubMed journals harmonisation medical journals harmonisation free journals harmonisation best journals harmonisation top journals harmonisation free medical journals harmonisation famous journals harmonisation Google Scholar indexed journals Trifluoroacetic acid articles Trifluoroacetic acid Research articles Trifluoroacetic acid review articles Trifluoroacetic acid PubMed articles Trifluoroacetic acid PubMed Central articles Trifluoroacetic acid 2023 articles Trifluoroacetic acid 2024 articles Trifluoroacetic acid Scopus articles Trifluoroacetic acid impact factor journals Trifluoroacetic acid Scopus journals Trifluoroacetic acid PubMed journals Trifluoroacetic acid medical journals Trifluoroacetic acid free journals Trifluoroacetic acid best journals Trifluoroacetic acid top journals Trifluoroacetic acid free medical journals Trifluoroacetic acid famous journals Trifluoroacetic acid Google Scholar indexed journals radiosynthesis articles radiosynthesis Research articles radiosynthesis review articles radiosynthesis PubMed articles radiosynthesis PubMed Central articles radiosynthesis 2023 articles radiosynthesis 2024 articles radiosynthesis Scopus articles radiosynthesis impact factor journals radiosynthesis Scopus journals radiosynthesis PubMed journals radiosynthesis medical journals radiosynthesis free journals radiosynthesis best journals radiosynthesis top journals radiosynthesis free medical journals radiosynthesis famous journals radiosynthesis Google Scholar indexed journals carboxylic acid articles carboxylic acid Research articles carboxylic acid review articles carboxylic acid PubMed articles carboxylic acid PubMed Central articles carboxylic acid 2023 articles carboxylic acid 2024 articles carboxylic acid Scopus articles carboxylic acid impact factor journals carboxylic acid Scopus journals carboxylic acid PubMed journals carboxylic acid medical journals carboxylic acid free journals carboxylic acid best journals carboxylic acid top journals carboxylic acid free medical journals carboxylic acid famous journals carboxylic acid Google Scholar indexed journals endotoxins articles endotoxins Research articles endotoxins review articles endotoxins PubMed articles endotoxins PubMed Central articles endotoxins 2023 articles endotoxins 2024 articles endotoxins Scopus articles endotoxins impact factor journals endotoxins Scopus journals endotoxins PubMed journals endotoxins medical journals endotoxins free journals endotoxins best journals endotoxins top journals endotoxins free medical journals endotoxins famous journals endotoxins Google Scholar indexed journals
Article Details
Abbreviations:
GMP: Good manufacturing practice; GRP: Good radiopharmaceutical practices; GC: Gas chromatography; HPLC: High pressure liquid chromatography; ICH: international conference of harmonisation; LAL test: Limulus amebocyte lysate test; QC: Quality control; PET: Positron emission tomography; RCY: Radiochemical yield; RCP: Radiochemical purity; TLC: Thin Layer Chromatography; TFA: Trifluoroacetic acid
Background
Fibrosis is a common pathological process which can effect many organs, including the lung, heart, kidney and liver. Fibrosis also influences tumor invasion and metastasis, and occurs in many different pathologies, including atherosclerosis, diabetes, viral infections and autoimmune diseases such as scleroderma, rheumatoid arthritis, Crohn’s disease, ulcerative colitis, and systemic lupus erythematosus. The fibrotic process crucially influences disease progression, effective drug delivery and treatment outcome. Being fibrosis a dynamic process, image-guided tools may provide early detection and staging, allowing treatment initiation at early stages. Thus, there is an urgent clinical need for a sensitive noninvasive tool to diagnose early disease activity, monitor therapeutic response, and advance our understanding of the course of this debilitating disease process. Fibroblast activation protein (FAP) is a type II integral membrane glycoprotein nearly absent in non-diseased adult organs, but greatly upregulated on activated fibroblasts or cancer-associated fibroblasts. This specific pattern of expression makes FAP an ideal target for imaging and therapy. Preclinical and clinical data demonstrate that FAP is a highly specific biomarker of active fibrosis in ILDs [1] and fibrotic remodelling of the heart, and inhibition attenuates fibrosis in mouse models of liver fibrosis [2]. FAP-specific small-molecule inhibitors (FAPIs) with a N-(4-quinolinoyl)glycyl-(2-cyanopyrrolidine) scaffold [9] have been developed and labeled with gallium-68, lutetium-177 or yttrium-90 [3-4]. The most promising molecule has been found FAPI-46, functionalized with 1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic acid (DOTA) (Figure1) [5-6] to obtain a radiolabeled PET probe with high tumor uptake and retention and reduced uptake in normal organs [7].
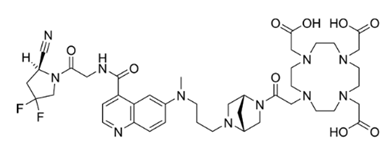
Figure 1: Structure of FAPI-46 (molecular weight: 885.96 g/mol).
Many efforts have been spent by our group in developing and validating radiosynthesis of [68Ga]68Ga-based imaging tools to target different pathways of the fibrogenic process [8-9]. Moreover, we have established a reliable method to optimize the obtained quality of the radioligand scaling down the precursor quantity [10-12]. In this study we have developed a new automated synthesis method to label FAPI-46 with gallium-68 and a new quality control (QC) system to determine its radiochemical purity and the optimal precursor amount, to transfer it in clinical practice for detection and monitoring of the fibrotic process [13].
Results
Labelling and quality controls results of different FAPI-46 loads and of the validated synthesis
The fully automated production of [68Ga]Ga-FAPI-46 was conducted investigating five different precursor amounts of FAPI-46 (10-20-30-40-50 μg) for radiolabelling with gallium-68. Immediately after each synthesis the overall QCs of the final product (Table 1) were performed in order to determine the best precursor amount from which to start to obtain [68Ga]Ga-FAPI-46 and to optimize the entire production process.
|
Peptide (FAPI-46, ug) |
50 (100 μL, 0.056 μmol) |
40 (80 μL, 0.045 μmol) |
30 (60 μL, 0.035 μmol) |
20 (40 μL, 0.022 μmol) |
10 (20 μL, 0.011 μmol) |
|
Radiochemical purity (Radio-UV-HPLC) |
0.9933 |
0.9928 |
0.9915 |
0.998 |
0.9929 |
|
Radiochemical purity (Radio-TLC) |
1 |
1 |
1 |
1 |
1 |
|
pH |
7 |
7 |
7 |
7 |
7 |
|
Radiochemical yield (n.d.c.) |
0.7596 |
0.7351 |
0.72 |
0.6698 |
0.591 |
|
Volume |
10 |
10 |
10 |
10 |
2 - 10 mL |
|
Color |
Colorless |
Colorless |
Colorless |
Colorless |
Colorless |
|
Molar activity (GBq/μmol) |
11.25 |
14.47 |
20.24 |
25.9 |
45.72 |
Table 1: Summary data of [68Ga]Ga-FAPI-46 QCs (10 - 50 μg, n = 3)
The best results were obtained for 20 μg of precursor, therefore once the automated synthesis has been optimized, the production processes have been validated with this amount of precursor, according to regulatory requirements to warrant the robustness of the gallium-68 labelling methods of FAPI-46. Some tested QC parameters were based on the European Pharmacopoeia (11.0/0125) (Table 2).
|
Test |
Batch 1 |
Batch 2 |
Batch 3 |
Acceptance criteria |
|
Radiochemical purity (Radio-UV-HPLC) |
99,84% |
0.9994 |
99,51% |
> 95% |
|
Radiochemical purity (Radio-TLC) |
1 |
1 |
1 |
> 95% |
|
pH |
7 |
7 |
7 |
4 - 8.5 |
|
Radiochemical yield (n.d.c.) |
68,00% |
66,25% |
69,00% |
> 40% |
|
Radioactivity concentration |
57.9 – 96.6 |
56.4 – 95.6 |
58.8 – 96.0 |
> 50 MBq/mL |
|
Radioactivity |
579 - 966 |
564 - 956 |
588 - 960 |
> 150 MBq |
|
Volume |
10 |
10 |
10 |
2 - 10 mL |
|
Color |
Colorless |
Colorless |
Colorless |
Colorless |
|
Molar activity (GBq/μmol) |
26.32 |
25.64 |
26.73 |
1-60 GBq/µmol |
|
Radionuclidic purity |
1 |
1 |
1 |
> 99.9% |
|
Ge-68 breakthrough |
0.000000004 |
4.3E-09 |
4.2E-09 |
< 0.001% |
|
EtOH amount |
0.0373 |
0.0369 |
0.0343 |
< 10% (V/V) (< 2.5 g) |
|
HEPES content |
9.40 μg/mL |
9.40 μg/mL |
9.40 μg/mL |
Less than 200 µg/V of HEPES in test solution |
|
Endotoxins |
< 17.5 IU/mL |
< 17.5 IU/mL |
< 17.5 IU/mL |
< 17.5 IU/mL |
|
Sterility test |
Sterile |
Sterile |
Sterile |
Sterile |
|
Stability over 4h (RCP%) |
0.9647 |
0.965 |
0.9648 |
> 95% |
Table 2: Summary data of three consecutive validation batches of [68Ga]Ga-FAPI-46 (20 μg).
The radiochemical purity (RCP%) was evaluated verifying the presence of free gallium (based on Radio-UV-HPLC) and gallium colloids (based on Radio-TLC). With Radio-UV-HPLC, free gallium-68 was detected at Rt = 1.433 min (Figure 2a), whereas gallium-68 bound to FAPI-46 was detected at Rt = 6.233 min (Figure 2b) with a mean purity of 99.76%.
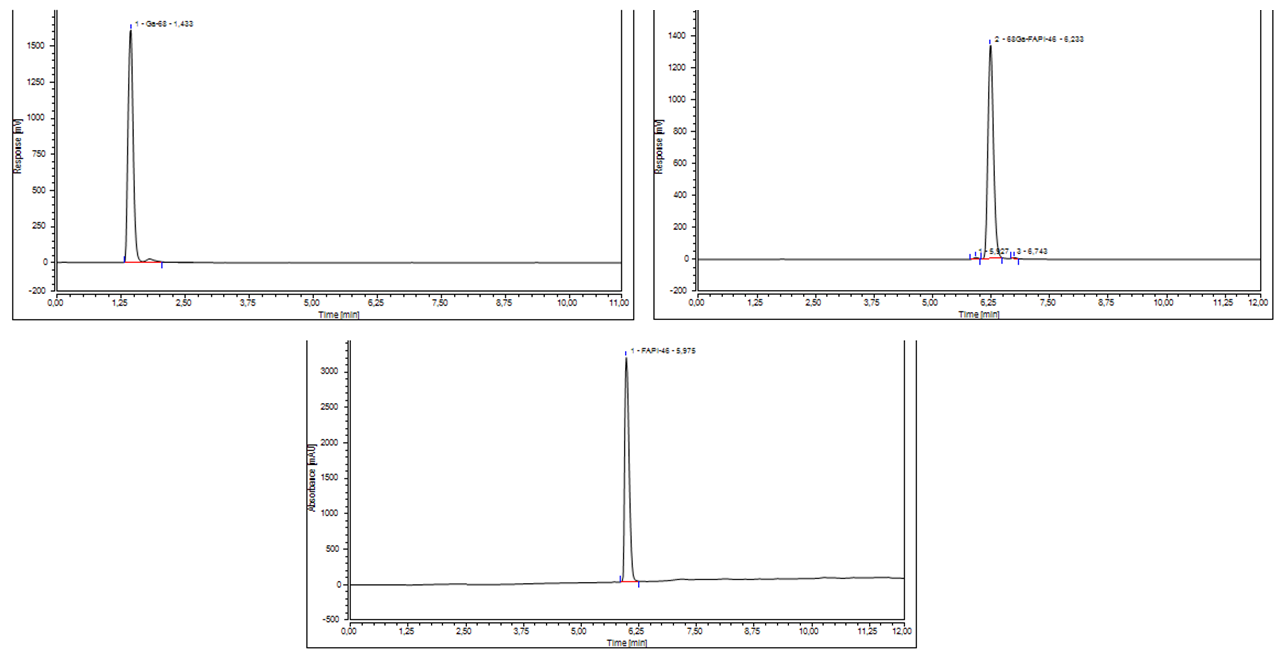
Figure 2: (a) HPLC chromatogram of the radiodetector showing the eluate [68Ga]GaCl3; (b) HPLC chromatogram of the radiodetector showing [68Ga]Ga-FAPI-46.; (c) Radio-UV-HPLC chromatogram of FAPI-46.
The reference solution of FAPI-46 showed a little bit different retention time (Rt = 5.975 min), as seen in Figure 2c, respect to [68Ga]Ga-FAPI-46 (Rt = 6.233 min). The difference of the two retention times is due to the use of different detectors (Radio and UV-VIS), but also to a different charge of the DOTA chelator after incorporation of gallium-68. The different charge results in changed interaction with the column due to a slight change of hydrophobicity of the complete molecule. The absence of 68Ga-chelate in the standard solution, maintains the three free carboxylic acid causing the increased hydrophilicity of the standards and then modifying its Rt.
With Radio-TLC no [68Ga]Ga-colloids could be detected at Rf = 0.2 and the radiopharmaceutical product was detected at Rf = 0.8 (Figure 3).

Figure 3: Radio-TLC chromatogram of [68Ga]Ga-FAPI-46.
In addition the product was tested for endotoxins and their concentration resulted below 17.5 EU/mL for all samples. Sterility was assessed in all samples that resulted sterile. The stability of [68Ga]Ga-FAPI-46 in buffer solution at room temperature was tested up to 4 hours by Radio-UV-HPLC, RadioTLC and pH. As seen in Figure 4 the RCP% of [68Ga]Ga-FAPI-46, determined with Radio-UV-HPLC, stays above 95% under setup conditions. The Radio-UV-HPLC analysis revealed a decrease of RCP% value from 99.80% to 96.47% due to the increase of free Ga-68 (Rt = 1.433 min; reference solution of 68GaCl3 figure 2a) and of products coming from radiolytic decomposition as well as oxidation, identified with the peak at Rt = 5.807 min, next the mean peak of the radiopharmaceutical. All data are provided in Supplemental figure 1.
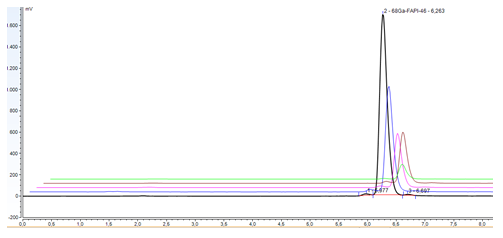
Figure 4: Stability of [68Ga]Ga-FAPI-46 (black dash T0, blue dash T1 h, pink dash 2 h, brown dash 3 h, green dash 4h)
The stability of [68Ga]Ga-FAPI-46 was also confirmed with Radio-TLC test, that didn't detected gallium-68 colloids in the final product (Figure 5). The pH value stays 7 over time.
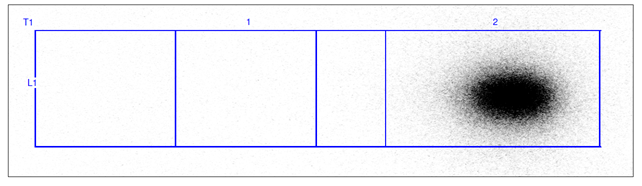
Figure 5: Radio-TLC chromatogram of [68Ga]Ga-FAPI-46 over 4 h.
Discussion
Activated fibroblast have been identified as key effector cells that mediate the extracellular matrix remodeling in pulmonary fibrosis. Positive feedback from fibroblast and an aberrant extracellular matrix lead to progressive fibrotic scarring, functional decline, and ultimately lung failure. FAP is selectively expressed at sites of tissue remodeling and repair and in tumor stromal fibroblasts, suggesting its use as an excellent diagnostic or therapeutic target for disorders associated with fibrotic dIsregulation. Consequently, several FAP-targeting inhibitory compounds have been developed and attracted attention as promising application for radiomolecular precision imaging. We hereby describe the development and validation of a new automated synthesis method and new QC system to label FAPI-46 with gallium-68, which has been shown to be particularly suitable for detecting fibrosis and sensitively monitoring disease activity. The development process of a new radiopharmaceutical involves the setup of the radiosynthesis as well as the quality assessment methods of the final product through the evaluation of release specifications as RCP%, As or Am, RCY%, radionuclidic purity, chemical purity, pH, sterility and stability. One of the parameters of paramount importance is the specific activity or molar activity (As or Am) in the final product, i.e. the ratio of the labeling isotope (in Bq) divided by the amount of the peptide (g) or the unit mole of the compound (mol). For the synthesis setup optimization, molar activity is required to be high due to several biological factors, such as the affinity of the peptide for its receptor and the number of available receptors, limiting the mass dose of peptide that can be administered. Indeed, a suboptimal Am could result in excessive amount of peptide leading to the saturation of the receptor or eventually to the induction of pharmacological side effects. Moreover the use of the same small volume of gallium-68 eluate with higher concentration allows, for a reduced amount of ligand, a faster radiolabeling reaction with quantitative incorporation and high tracer specific activity [14-15]. On the other hand, Am should not be too high because the amount of the peptide should be sufficiently high to allow reasonable radiochemical yield and to guarantee a reliable biodistribution for image quantification, indeed higher amount of the ligand decreases specific radioactivity [16]. To this aim a scale down of five different amounts of FAPI-46 (50-40-30-20-10 μg) was evaluated to produce [68Ga]Ga-FAPI-46, obtaining the optimal results with high Am for precursor amount of 20 μg, as shown in table 1. Current nomenclature rules defined RCY as the ratio of the activity of a specified radionuclide of a specified element after its radiochemical separation or labeling [17-18].
We noticed a decrease RCY% corresponding to a decrease ligand amount (higher RCY% for 50 and lower for 10 µg), (Table 1). This phenomenon could be caused by the possible presence of peptide and radiometal ion contaminants. The chemical purity of the GMP-grade precursor DOTA-FAPI-46 was assessed through RADIO-UV-HPLC analysis that doesn't showed any impurity (figure 2 c), moreover we prepared a stock solution of precursor dissolved in metal free water and then dilution series of five concentrations of the precursor were prepared from the mother solution. The radiometal ion contaminants can adversely affect the labelling yields and RCP% as well as indirectly molar activity, by necessitating higher amounts of peptide precursor to achieve high radionuclide incorporation [16]. However we rigorously pre-purificated the elute Ga-68, as starting material, using SCX cartridge in order to remove unwanted metallic impurities and reduce the volume, as it is especially suitable for radiolabelling of small peptides such as FAPI-46 [15-16]. Therefore, the most likely explanation about the decreased RCY% related to the decreased amount of peptide is that 10 ug load of peptide precursor could be too low to completely complex 68Ga3+ ions, resulting in a less efficient trapping with no quantitative incorporation of the radionuclide [19]. Table 1 shows that the precursor amount of 20 μg allows good RCY%. A turning point in the optimization and definition of a radiopharmaceutical process is the selection of the ideal lowest precursor amount guaranteeing high RCY%, As as well as RCP%.
RCP% should discover all chemicals forms containing the radionuclide and determine their percentage of the total radioactivity of the product [18]. The highest RCP% obtained with 20 μg of DOTA-FAPI-46 together with a good RCY% and high Am led us to validate the entire radiopharmaceutical production process with this precursor amount to get [68Ga]Ga-FAPI-46. In every chromatogram of [68Ga]Ga-FAPI-46, regardless of the amount of precursor, we noticed two peaks near the main peak of the radiopharmaceutical (Figure 2 b). These radiochemical impurities could arise from the synthesis method or radiolysis, indeed radio-peptides could be randomly damaged by radicals that are mainly formed by the radiolysis of H2O and chromatograms of radiolysed radiolabeled products show up as smears because of this event [20]. For releasing a product, all signal above minimum peak height, including the hole smear, should be taken into account for determination of final RCP% [17], but according to Ph. Eur 2.2.46., when a specific impurity contain < 1% of the total peak area on the chromatogram, no quantification is needed (European Pharmacopeia 9.0 2017). Additionally the nature of these impurities was also confirmed through the stability assessment of [68Ga]Ga-FAPI-46, tested in aqueous solution at room temperature up to 4 hours, that showed a decrease of RCP% due to the increase of free Ga-68 and of products coming from radiolytic decomposition as well as oxidation (Figure 4; Supplemental figure 1). However the RCP% stayed above 95% under setup conditions, confirming the radiopharmaceutical quality of [68Ga]Ga-FAPI-46 over time, according to QC parameters based on the European Pharmacopoeia (11.0/0125) (Table 2), allowing patient safety and enabling the daily production and transferability of our production method in other radiopharmaceutical laboratories.
Conclusions
[68Ga]Ga-FAPI-46 was successfully synthesized in a fully-automated way on GRP Scintomics module. All the tested quality parameters for radiochemical purity, pH, endotoxins and sterility were in accordance with European Pharmacopoeia. In addition, the product solution was stable for at least 4 hours after production, as shown by Radio-UV-HPLC. Thus, [68Ga]Ga-FAPI-46 can be integrated in the clinical workflow for routine clinical application.
Material and Methods
Reagents and solvents
All chemicals used for the radiolabelling reaction (saline, ethanol, HEPES buffer solution and water) were of the highest available purity grade and commercially obtained as a single disposable kit (reagents and cassettes for synthesis of [68Ga]68Ga-peptides using cationic purification ABX, Advanced Biochemical Compounds, Radeberg, Germany). The GMP peptides DOTA-FAPI-46 was purchased as lyophilized powder from SOFIE (SOFIE, Dulles, United States). An aqueous mother solutions of DOTA-FAPI-46 (0.5 μg/μL) was prepared in ultrapure water (Sigma Aldrich), kept at -20 °C and the different solutions of precursor used for labeling belong to the same mother solution. Gallium-68 (t1/2 = 68 min, β+ = 89%, and EC = 11%) was obtained from a pharmaceutical grade 68Ge/68Ga generator (1850 MBq, GalliaPharm® Eckert & Ziegler, Berlin, Germany) by elution with 0.1M HCl (Rotem GmbH, Germany). The amount of detected metal impurities as provided by the manufacturer was less than the defined limit in the European Pharmacopeia monograph [21]. The reagents trifluoroacetic acid (TFA), water and acetonitrile used for Radio-UV-HPLC, as well as ammonium acetate and methanol were metal free and purchased from Sigma Aldrich (Saint Louis, Missouri, USA). The aseptic production was conducted in a GMP grade A hot cell (NMC Ga-68, Tema Sinergie). Both 68Ge/68Ga generator and automated synthesis module (Scinotmics GRP® module, Germany) were placed in the hot cell.
Preparation for labelling of DOTA-FAPI-46 with gallium-68
First the commercially available fully automated synthesis platform Scintomics GRP® module was equipped with a disposable single-use cassette (SC-01, ABX). DOTA-FAPI-46 was prepared in 10-20-30-40-50 μg aliquots from the stock solution (0.5 μg/μL). All synthesis reagents were contained in the reagent set. From the reagent set, a syringe containing 1.5 mL of 5M NaCl was attached into the cassette as well as phosphate buffer saline (PBS), ethanol and ethanol/water 1/1 vials in each designated valve. Cation exchange (SCX) cartridge and C18 column (Sep Pak C18 RP) were inserted into appropriate holders into the cassette and module. The reaction mixture containing 3.2 mL 1.5 M 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) buffer solution and an aliquot of DOTA-FAPI-46 was loaded into the reaction vial.
Labelling of DOTA-FAPI-46 with [68Ga]68GaCl3
The synthesis was performed in a fully-automated fashion and each reaction parameter as reaction time, temperature and radioactivity, was monitored in real time. Gallium-68 was obtained eluting a GMP certified 68Ge/68Ga generator (GalliaPharma®, Eckert and Ziegler) with 0.1M HCl, collected and pre-purified on a SCX cartridge which is then eluted with 5M NaCl. The eluate is added into the reaction vial, previously loaded with DOTA-FAPI-46 (10-20-30-40-50 μg in 1.5 M 2-[4-(2-hydroxyethyl)-1-piperazinyl]-ethanesulfonic acid (HEPES buffer solution) at pH = 4-4.5. The mixture was incubated at 95 °C for 10 min. After the completion of labelling reaction the crude product was cooled down and trapped onto Sep Pak C18 RP cartridge, washed with water for injection Ph. Eur., and eluted with 2 mL of Ethanol/Water 1/1. The final product was diluted with phosphate buffered saline (PBS) and sterilized through a 0.2 μm filter (millex GV) into a sterile 25 mL capped glass vial and diluted with PBS for the final formulation (Figure 6). The entire radiopharmaceutical production takes 35 minutes.
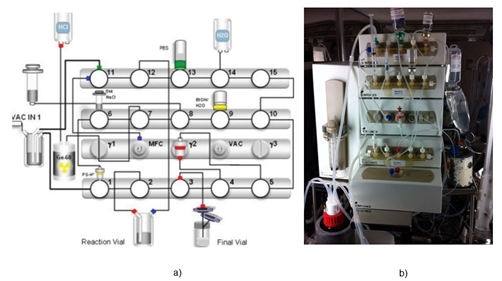
Figure 6: a) Software interface module diagram of the automated Scintomics radiosynthesis system used to prepare [68Ga]Ga-FAPI-46. b) Original Scintomics radiosynthesis module equipped with cassette for the labelling of [68Ga]Ga-FAPI-46.
Quality control and process validation
After synthesis, the radiopharmaceutical product was evaluated for quality control determining the following parameters: total product activity, gallium-68 ion identity via half-life time and gamma spectroscopy, chemical and radiochemical purity by Radio-UV-HPLC and Radio-TLC, pH, radionuclide purity for 68Ge-breakthrough and sterility/endotoxin assay (sterility test and LAL test). Radiochemical purity (RCP) is one of the most important quality criteria to release the final product for the clinical use, as described in the European Pharmacopeia [17; 22-24]. To this aim, a fully validated separation method has to be available according to ICH guidlines [25], enabling optimal separation between different (radio) chemical forms (radioactive impurities) other than the original intact radiopharmaceutical 26-28]. The stability of [68Ga]Ga-FAPI-46 at room temperature was monitored by Radio-UV-HPLC and Radio-TLC for 4 hours. To validate the entire process of radiopharmaceutical production and quality control, three batches of [68Ga]Ga-FAPI-46 were produced in three different days under the same conditions set for typical routine preparations. Every batch was fully characterized from the analytical point of view, with the aim to verify that the product meet the acceptance criteria for all the established quality parameters.
Appearance
The visual inspection of in-house prepared radiopharmaceuticals is necessary before injection into the patient, as a measure of process performance and validation. Presence of particulate in the sample suggests possible failure during radiopharmaceutical synthesis, including purification, sterilizing filtration, and failed environmental control during the setting-up of reagents [25].
Instant Thin Layer Chromatography
Radio-TLC test was used to determine the percentage of [68Ga]Ga-FAPI-46 and gallium-68 colloids in the final product.
For the determination of gallium-68 colloids amount, 1 M ammonium acetate/methanol (1:1 vol/vol) was used as mobile phase and ITLC-SG paper strips (Varian ITLC-SG plates) as stationary phase; gallium-68 colloid (Rf = 0.0-0.2), [68Ga]Ga-FAPI-46 (Rf = 0.8-1). TLC-SG paper strips, used a stationary phase, were counted with a scanner (Cyclone® Plus Storage Phosphor system, Perkin Elmer) and the chromatograms were analysed with OptiQuantTM software.
High Pressure Liquid Chromatography
The Radio-UV-HPLC was additionally used to determine the chemical and radiochemical purity of [68Ga]Ga-FAPI-46 in the final product after the purification with C18 cartridge, at the end of the synthesis. Radio-UV-HPLC was performed on a Dionex Ultimate 3000 HPLC system (Thermo Fisher Scientific) equipped with a AcclaimTM 120 C18 column 3µm 120Å (3.0 mm × 150 mm) and a UV and a γ-detector (Berthold Technologies, Milan, Italy). The used solvents were A) 0.1% TFA in water and B) 0.1% TFA in acetonitrile.
The flow rate of the mobile phase was set at 0.6 mL/min, with a total run of 12 min.
The following phase gradient was used in the Radio-UV-HPLC analysis: 0−8 min from 0% B to 50%, 8-10 min 50% and 10-12 min from 50% to 0% B.
The column temperature was kept at 30°C. The samples were also monitored with UV detector at 220 nm in order to detect chemical impurities in the final product. Activity corresponding to gallium-68 ion and [68Ga]Ga-FAPI-46 was measured by Radio-UV-HPLC γ-detector.
The software system Chromeleon 7 was used to assemble the information.
Ge-68 Breakthrough
The Ge-68 breakthrough was measured by gamma spectroscopy of the final product, using a gamma spectrometer equipped with a high-purity germanium (HPGe detector ORTEC GEM 30P4-76). The γ-ray spectrometry tests included the identification of principal γ-photon (499–521 KeV peak) and Ge-68 content (decay of 499–521 KeV peak ≥48 h) using a large volume counter linked to a multichannel analyser system (HPGe detector ORTEC GEM 30P4-76). The spectra acquisition of Ge-68 was performed at least 48h after the completely decay of Ga-68 produced by the generator. Duration of the acquisition was 180 minutes to obtain a high signal-to-noise ratio. The sample volume was at least 1 ml. Spectrum was analyzed using Genie 2000 software.
Radionuclide identification and activity measurements
Radionuclide purity was determined based on the half-lives, type and energy of the emitted radiations. Half-life was measured with a dose calibrator (Capintec 25-R) at four consecutive intervals (5, 10, 15 and 20 minutes). The expected half-life of gallium-68 is 67.6 minutes and is calculated using the following equation:
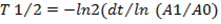
where: dt-time difference,
A1 -ending activity,
A0 -starting activity.
pH evaluation
The pH value of [68Ga]Ga-FAPI-46 was measured using colorimetric pH strips (0-14).
Endotoxin and sterility
Quantitative determination of bacterial endotoxins was performed by the chromogenic method, using Endosafe® nexgenPTS™ (Charles River, Ireland) apparatus. [68Ga]Ga-FAPI-46 samples were previously diluted and then applied in duplicate inside cartridges in parallel with positive control testing. The radiopharmaceutical can be released when the level of endotoxins was less 17.5 IU/mL in accordance with Ph. Eur. (9.0/0125). The sterility of the [68Ga]Ga-FAPI-46 solution was assessed by direct inoculation in a growth broth (Triptic Soy Broth, TSB) which was incubated at 20-25°C, and verified daily over fourteen days [29]. The sample was considered sterile when no microbiological growth was detected.
Residual solvents and HEPES
Potentially present radiolysis products, such as ions and excited molecules could cause undesired and serious side effects [30-31]. Radiolysis may be reduced by utilizing compounds insensitive to radiation or extenuating the process with additives (e.g. radical scavengers). In the clinical context, these radical scavengers should be suitable for human use, such as ascorbic acid or HEPES in the reaction mixture and ethanol in the pre- and post-processing steps. The positive influence of ethanol on radiolabeling yield and radiolysis restrain [32-33] prompted us to use it during the synthesis process in order to obtain a more reliable and repeatable automated method. Ethanol is a class 3 solvent which may remain indeterminate and unmentioned up to 0.5%, but must be declared quantitatively for higher amounts in a pharmaceutical (Ph. Eur. 2019,9.6. 9.7–9.8). In this study we determined the residual ethanol using gas chromatography (GC) and HEPES content by TLC-SG, both according to Ph. Eur. Monograph [21]. The reference solution of HEPES was prepared at a concentration of 200 µg/mL. Two separate spots of reference solution (5 µL) and test solution (a sample of the final product [68Ga]Ga-FAPI-46 were applied on TLC silica gel F254 plate and developed on a path over 2/3 of the plate with use of water : acetonitrile (25 : 75 V/V) solution as a mobile phase. The plate was then exposed to iodine vapor. The spot corresponding to the test solution should not be more intense than the reference solution spot (less than 200 µg/V of HEPES in test solution). HEPES content has been assessed also using the validated HPLC method [34] based on the use of a Waters Xbridge® column C18 (150 mm × 4.6 mm, 3.5 μm), as stationary phase, connected to an UV detector set to a wavelength of 195 nm and a γ-detector (Berthold Technologies, Milan, Italy) and ammonium formate 20 mM pH 9.5, as mobile phase, at an isocratic flow of 0,7 mL/min.
Acknowledgements
Not applicable.
Author contributions
SM, MS, BG and LR have contributed to the organization of the content for this manuscript. SM carried out the labeling procedures and prepared the draft. MS and GB collected data and contributed to data analysis. LR revised the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
All data generated and analysed during this study are included in this published article.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- Yang P, Luo Q, Wang X, et al. Comprehensive Analysis of Fibroblast Activation Protein Expression in Interstitial Lung Diseases. Am J Respir Crit Care Med 207 (2023): 160-172.
- Lee J, Byun J, Shim G, et al.Fibroblast activation protein activated antifibrotic peptide delivery attenuates fibrosis in mouse models of liver fibrosis. Nat Commun 1516 (2022).
- Lindner T, Loktev A, Altmann A, et al. Development of Quinoline-based Theranostic ligands for the targeting of fibroblast activation protein. J Nucl Med 59 (2018): 1415-1422.
- Nader M, Valla DF, Vriamont C, et al. [68Ga]/[90Y]FAPI-46: Automated production and analytical validation of a theranostic pair. Nucl Med Biol 110-111 (2022): 37-44.
- Loktev A, Lindner T, Burger EM, et al. Development of fibroblast activation protein-targeted radiotracers with improved tumor retention. J Nucl Med 60 (2019): 1421-1429.
- Mori Y, Dendl K, Cardinale J, et al. FAPI PET: Fibroblast Activation Protein Inhibitor Use in Oncologic and Nononcologic Disease. Radiology 306 (2023): e220749.
- Meyer C, Dahlbom M, Lindner T, et al. Radiation Dosimetry and Biodistribution of 68Ga-FAPI-46 PET Imaging in Cancer Patients. J Nucl Med 61 (2020): 1171-1177.
- Migliari S, Scarlattei M, Baldari G, et al. Scale down and optimized automated production of [68Ga]68Ga-DOTA-ECL1i PET tracer targeting CCR2 expression. EJNMMI radiopharm. Chem 3 (2023).
- Migliari S, Sammartano A, Scarlattei M, et al. A Rapid and Specific HPLC Method to Determine Chemical and Radiochemical Purity of [68Ga]Ga-DOTA-Pentixafor (PET) Tracer: Development and Validation. Curr Radiopharm 14 (2021): 121-130.
- Sammartano A, Migliari S, Scarlattei M, et al. Performance and long-term consistency of five Galliform 68Ge/68Ga generators used for clinical Ga-68 preparations over a 4 year period. Nucl Med Commun (2022).
- Migliari S, Sammartano A, Boss M, et al. Development and Validation of an Analytical HPLC Method to Assess Chemical and Radiochemical Purity of [68Ga]Ga-NODAGA-Exendin-4 Produced by a Fully Automated Method. Molecules 27 (2022): 543.
- Sartori A, Bianchini F, Migliari S, et al. Synthesis and preclinical evaluation of a novel, selective 111In-labelled aminoproline-RGD-peptide for non-invasive melanoma tumor imaging. MedChemComm 6 (2015): 2175-2183.
- Mecozzi L, Mambrini M, Ruscitti F, et al. In-vivo lung fibrosis staging in a bleomycin-mouse model: a new micro-CT guided densitometric approach. Sci Rep 18735 (2020).
- Decristoforo C, Penuelas I, Patt M, et al. European regulations for the introduction of novel radiopharmaceuticals in the clinical setting. Q J Nucl Med Mol Imaging 61 (2017): 135-144.
- Gillings N, Hjelstuen O, Ballinger J, et al. Guideline on current good radiopharmacy practice (cGRPP) for the small-scale preparation of radiopharmaceuticals. EJNMMI radiopharm. Chem 8 (2021).
- Luurtsema G, Pichler V, Bongarzone S, et al. EANM guideline for harmonisation on molar activity or specific activity of radiopharmaceuticals: impact on safety and imaging quality. EJNMMI Radiopharm Chem 6 (2021): 34.
- de Blois E, de Zanger RMS, Chan HS, et al. Radiochemical and analytical aspects of inter-institutional quality control measurements on radiopharmaceuticals. EJNMMI Radiopharm Chem 4 (2019): 3.
- Coenen HH, Gee AD, Adam M, et al. Consensus nomenclature rules for radiopharmaceutical chemistry - Setting the record straight. Nucl Med Biol 55 (2017): 5-11.
- Spreckelmeyer S, Balzer M, Poetzsch S, et al. Fully-automated production of [68Ga]Ga-FAPI-46 for clinical application. EJNMMI Radiopharm Chem 5 (2020): 31.
- Meisenheimer M, Saenko Y, Eppard E. Gallium-68: Radiolabeling of Radiopharmaceuticals for PET Imaging - A Lot to Consider. Medical Isotopes (2019).
- Gallium Chloride (68Ga) solution for labeling (Monograph 2464) In: European Pharmacopoeia European Directorate for the Quality of Medicines.
- Decristoforo C, Penuelas I, Patt M, et al. European regulations for the introduction of novel radiopharmaceuticals in the clinical setting. Q J Nucl Med Mol Imaging 61 (2017): 135-144.
- Gillings N, Hjelstuen O, Ballinger J, et al. Guideline on current good radiopharmacy practice (cGRPP) for the small-scale preparation of radiopharmaceuticals. EJNMMI radiopharm. Chem 8 (2021).
- Todde S, Peitl, PK, Elsinga P, et al. Guidance on validation and qualification of processes and operations involving radiopharmaceuticals. EJNMMI radiopharm. Chem 8 (2017).
- ICH Q8 (R2): Pharmaceutical development (ICH guideline). European Medicines Agency (EMA) (2017).
- ICH guideline Q2(R1) Validation of analytical procedure: text and methodology. European Medicines Agency.
- Levin S. High Performance Liquid Chromatography (HPLC) in the Pharmaceutical Analysis. Analysis and Pharmaceutical Quality (2010): 1-34.
- Zarghi A, Foroutan S, Shafaati A, et al. A Rapid HPLC Method for the Determination of Losartan in Human Plasma Using a Monolithic Column. Arzneimittelforschung 55 (2011): 569-572.
- EDQM Guidelines: Guide for the elaboration of monographs on RADIOPHARMACEUTICAL PREPARATIONS European Pharmacopoeia Edition (2018).
- Gillings N, Todde S, Behe M, et al. EANM guideline on the validation of analytical methods for radiopharmaceuticals. EJNMMI Radiopharm Chem 5 (2020): 7.
- International Atomic Energy Agency (IAEA)/World Health Organization (WHO) guideline on good manufacturing practices for investigational radiopharmaceutical products, Draft working document (2021).
- Annex 2 International Atomic Energy Agency and World Health Organization (WHO) guideline on good manufacturing practices for radiopharmaceutical products (2020).
- International Atomic Energy Agency (IAEA)/World Health Organization (WHO) guideline on good manufacturing practices for investigational radiopharmaceutical products, Draft working document (2021).
- Migliari S, Scarlattei M, Baldari G, Silva C, Ruffini L. A Specific HPLC Method to Determine Residual HEPES in [68Ga]Ga-Radiopharmaceuticals: Development and Validation. Molecules 27 (2022): 4477.
