Aortic Valve Insufficiency Related to Eosinophilic Granulomatosis with Polyangiitis
Article Information
Raphael Cauchois1*, Sebastien Renard2, Gilles Kaplanski1, Pierre-Andre Jarrot1
1Department of Internal Medicine and Clinical Immunology, CHU Conception, AP-HM, Aix Marseille Univ, C2VN, Marseille, France
2Department of Cardiology, CHU Timone, AP-HM, Aix Marseille Univ, Marseille, France
*Corresponding Author: Raphael Cauchois, Department of Internal Medicine and Clinical Immunology, CHU Conception, AP-HM, Aix Marseille Univ, C2VN, Marseille, France.
Received: 07 August 2022; Accepted: 20 August 2022; Published: 12 September 2022
Citation: Raphael Cauchois, Sebastien Renard, Gilles Kaplanski, Pierre-Andre Jarrot. Aortic Valve Insufficiency Related to Eosinophilic Granulomatosis with Polyangiitis. Archives of Clinical and Medical Case Reports 6 (2022): 624-625.
View / Download Pdf Share at FacebookKeywords
Eosinophilic granulomatosis; Aortic Valve; Surgery; Nasal polyposis
Eosinophilic granulomatosis articles; Aortic Valve articles; Surgery articles; Nasal polyposis articles
Eosinophilic granulomatosis articles Eosinophilic granulomatosis Research articles Eosinophilic granulomatosis review articles Eosinophilic granulomatosis PubMed articles Eosinophilic granulomatosis PubMed Central articles Eosinophilic granulomatosis 2023 articles Eosinophilic granulomatosis 2024 articles Eosinophilic granulomatosis Scopus articles Eosinophilic granulomatosis impact factor journals Eosinophilic granulomatosis Scopus journals Eosinophilic granulomatosis PubMed journals Eosinophilic granulomatosis medical journals Eosinophilic granulomatosis free journals Eosinophilic granulomatosis best journals Eosinophilic granulomatosis top journals Eosinophilic granulomatosis free medical journals Eosinophilic granulomatosis famous journals Eosinophilic granulomatosis Google Scholar indexed journals COVID-19 articles COVID-19 Research articles COVID-19 review articles COVID-19 PubMed articles COVID-19 PubMed Central articles COVID-19 2023 articles COVID-19 2024 articles COVID-19 Scopus articles COVID-19 impact factor journals COVID-19 Scopus journals COVID-19 PubMed journals COVID-19 medical journals COVID-19 free journals COVID-19 best journals COVID-19 top journals COVID-19 free medical journals COVID-19 famous journals COVID-19 Google Scholar indexed journals CNS Inflammation articles CNS Inflammation Research articles CNS Inflammation review articles CNS Inflammation PubMed articles CNS Inflammation PubMed Central articles CNS Inflammation 2023 articles CNS Inflammation 2024 articles CNS Inflammation Scopus articles CNS Inflammation impact factor journals CNS Inflammation Scopus journals CNS Inflammation PubMed journals CNS Inflammation medical journals CNS Inflammation free journals CNS Inflammation best journals CNS Inflammation top journals CNS Inflammation free medical journals CNS Inflammation famous journals CNS Inflammation Google Scholar indexed journals Cardiac Surgery articles Cardiac Surgery Research articles Cardiac Surgery review articles Cardiac Surgery PubMed articles Cardiac Surgery PubMed Central articles Cardiac Surgery 2023 articles Cardiac Surgery 2024 articles Cardiac Surgery Scopus articles Cardiac Surgery impact factor journals Cardiac Surgery Scopus journals Cardiac Surgery PubMed journals Cardiac Surgery medical journals Cardiac Surgery free journals Cardiac Surgery best journals Cardiac Surgery top journals Cardiac Surgery free medical journals Cardiac Surgery famous journals Cardiac Surgery Google Scholar indexed journals Aortic Valve articles Aortic Valve Research articles Aortic Valve review articles Aortic Valve PubMed articles Aortic Valve PubMed Central articles Aortic Valve 2023 articles Aortic Valve 2024 articles Aortic Valve Scopus articles Aortic Valve impact factor journals Aortic Valve Scopus journals Aortic Valve PubMed journals Aortic Valve medical journals Aortic Valve free journals Aortic Valve best journals Aortic Valve top journals Aortic Valve free medical journals Aortic Valve famous journals Aortic Valve Google Scholar indexed journals treatment articles treatment Research articles treatment review articles treatment PubMed articles treatment PubMed Central articles treatment 2023 articles treatment 2024 articles treatment Scopus articles treatment impact factor journals treatment Scopus journals treatment PubMed journals treatment medical journals treatment free journals treatment best journals treatment top journals treatment free medical journals treatment famous journals treatment Google Scholar indexed journals CT articles CT Research articles CT review articles CT PubMed articles CT PubMed Central articles CT 2023 articles CT 2024 articles CT Scopus articles CT impact factor journals CT Scopus journals CT PubMed journals CT medical journals CT free journals CT best journals CT top journals CT free medical journals CT famous journals CT Google Scholar indexed journals Breast Cancer articles Breast Cancer Research articles Breast Cancer review articles Breast Cancer PubMed articles Breast Cancer PubMed Central articles Breast Cancer 2023 articles Breast Cancer 2024 articles Breast Cancer Scopus articles Breast Cancer impact factor journals Breast Cancer Scopus journals Breast Cancer PubMed journals Breast Cancer medical journals Breast Cancer free journals Breast Cancer best journals Breast Cancer top journals Breast Cancer free medical journals Breast Cancer famous journals Breast Cancer Google Scholar indexed journals Nasal polyposis articles Nasal polyposis Research articles Nasal polyposis review articles Nasal polyposis PubMed articles Nasal polyposis PubMed Central articles Nasal polyposis 2023 articles Nasal polyposis 2024 articles Nasal polyposis Scopus articles Nasal polyposis impact factor journals Nasal polyposis Scopus journals Nasal polyposis PubMed journals Nasal polyposis medical journals Nasal polyposis free journals Nasal polyposis best journals Nasal polyposis top journals Nasal polyposis free medical journals Nasal polyposis famous journals Nasal polyposis Google Scholar indexed journals Neurodegeneration articles Neurodegeneration Research articles Neurodegeneration review articles Neurodegeneration PubMed articles Neurodegeneration PubMed Central articles Neurodegeneration 2023 articles Neurodegeneration 2024 articles Neurodegeneration Scopus articles Neurodegeneration impact factor journals Neurodegeneration Scopus journals Neurodegeneration PubMed journals Neurodegeneration medical journals Neurodegeneration free journals Neurodegeneration best journals Neurodegeneration top journals Neurodegeneration free medical journals Neurodegeneration famous journals Neurodegeneration Google Scholar indexed journals
Article Details
Case Report
Eosinophilic granulomatosis with polyangiitis (EGPA) is a rare small-to-medium size vessel anti-neutrophil cytoplasmic antibodies (ANCA)-associated vasculitis, which can display a broad spectrum of manifestations [1]. Although cardiac features are well-known in this disease, valvular insufficiency is rarely described. Herein, we describe a rare case of EGPA-related aortic insufficiency. A 62-year-old caucasian male with a previous history of surgery for nasal polyposis in 2011, was referred to our internal medicine department for a 3-week history of mild asthma, flu-like symptoms, 5 kilograms-weight loss, rhinosinusitis and electroneuromyography-confirmed mononeuritis multiplex affecting the left median and right peroneal nerves. On examination, besides previous evaluation, we were surprised by a marked diastolic murmur, which was maximal in the third left intercostal space, especially at the end of expiration suggesting important aortic insufficiency. Transthoracic echocardiography (Figure 1) showed normal systolic function, but aortic valvular thickening with partial right coronary cusp (RCC) prolapse leading to severe aortic insufficiency with no arguments for endocarditis on transesophageal echocardiography. Cardiac magnetic resonance imaging identified myocarditis with subepicardial enhancement of inferolateral wall associated with small pericardial effusion. Electrocardiogram was normal. The CT-scan revealed maxillary sinusitis, chest was normal. Laboratory findings revealed an elevated white blood cell count of 18.9G/L (N 4.0-11.0) with absolute eosinophil count of 8.80G/L (N 0.05-0.55), inflammatory syndrome (C-reactive protein 107mg/L (N < 5), fibrinogen 4.9g/L (N 1.80-4.0), elevated troponin (0.48ng/mL, N<0.05), BNP level (47pg/mL, N<100) and normal renal/liver functions. Microbiological samples (blood, urine cultures) were negative. Myeloperoxidase (MPO)-ANCA were detected by enzyme linked immunosorbent assay (128UI, N<20UI) yielded a diagnosis of EGPA. Considering the 2011 Five Factor score, this case of EGPA was severe and the patient was treated according to current recommendations [2]. The treatment combined corticosteroids (3 pulse of methylprednisolone followed with gradual taper of prednisone starting from 1mg/kg/day over 6 month) with 6 pulses of cyclophosphamide, relayed with azathioprine for 24 months. Patient achieved subsequent remission, and echocardiographic controls showed incomplete regression of aortic insufficiency after immunosuppressive therapy. Special attention has been paid to the prevention of complications from corticosteroids and immunosuppressants, in particular infectious events. Although the absence of pathological confirmation, the clinical evidence including the recent outbreak of asthma, non-lytic sinusitis, mononeuropathy multiplex, elevated eosinophils count associated with positive anti-MPO antibodies allowed us to retain the diagnosis of EGPA. After ruling out endocarditis by transesophageal echocardiography, aortic insufficiency was likely related to EGPA, since the patient had no previous history of aortic regurgitation on regular check-up before admission. Furthermore, the MRI-confirmed myocarditis and pericardial effusion suggesting a cardiac tropism of his vasculitis. Cardiac involvement, which represents 40% of cases [3], is rather associated with ANCA-negative phenotype and mainly characterized by myocarditis, pericarditis or endomyocardial fibrosis. This is the first cause of the EGPA-patients death, whose suspected mechanisms are vasculitis-related ischemia and eosinophilic infiltration of the myocardium [4,5]. Contrary to granulomatosis with polyangiitis (GPA, formerly called Wegener’s granulomatosis), 5 aortic valvular insufficiency was rarely described in EGPA. Only three additional cases of EGPA-related aortic insufficiency have been reported [6-8]. ANCA detection was negative in all cases, the diagnosis of EGPA was confirmed by pathological analysis of the required valve replacement revealing eosinophilic infiltrates, granuloma and necrosis on thickened leaflets. In addition to valve replacement, two patients achieved remission with corticosteroids, associated with oral cyclophosphamide in one case. The third died of cardiogenic shock before starting immunosuppressive drugs.
Once the diagnosis ANCA-associated vasculitis has been established, it is essential to conduct clinical and paraclinical exploration of the organs that may be affected, in order to adapt the intensity of the immunosuppressive treatment. International guidelines recommend systematic heart check-up by serum N-terminal pro-brain natriuretic peptide and troponin I measurements, chest imaging, electrocardiography and transthoracic echocardiography, increasing emphasis is however being placed on cardiac magnetic resonance, which could identify cardiac involvement in up to 62% of patients [9]. In conclusion, when faced with acute aortic insufficiency associated with systemic signs, infectious endocarditis must first be ruled out. Next, immune causes such as aortitis, non-infectious endocarditis or ANCA-associated vasculitis should be considered. Physicians should be aware of aortic insufficiency as a rare, but life-threatening complication of EGPA, and may perform a systematic cardiac evaluation in this disease. The treatment of EGPA with cardiac involvement is codified and should be initiated without delay.
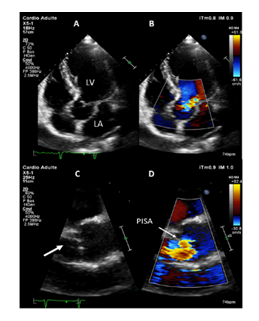
Figure 1: Transthoracic echocardiography. (A-B) Transthoracic apical 5- chamber view (A) showing moderate to severe aortic regurgitation by color Doppler. (C-D) Transthoracic parasternal long axis view. Zoom of the aortic valve showing right coronary cusp prolapse (arrow, C) with moderate to severe aortic regurgitation by color Doppler (D) with obtention of a hemispheric PISA (proximal isovelocity surface area). LA: left atrium; LV: left ventricle.
References
- Kitching AR, et al. ANCA-associated vasculitis. Nat Rev Dis Primers 71 (2020).
- Groh M, Pagnoux C, Baldini C, et al. Eosinophilic granulomatosis with polyangiitis (Churg-Strauss) (EGPA) Consensus Task Force recommendations for evaluation and management. Eur J Intern Med 26 (2015): 545-553.
- Sartorelli S, Chassagnon G, Cohen P, et al. Revisiting characteristics, treatment and outcome of cardiomyopathy in eosinophilic granulomatosis with polyangiitis (formerly Churg-Strauss). Rheumatology (Oxford) 61 (2022): 1175-1184.
- Comarmond C, Pagnoux C, Khellaf M, et al. Eosinophilic granulomatosis with polyangiitis (Churg-Strauss): clinical characteristics and long-term followup of the 383 patients enrolled in the French Vasculitis Study Group cohort. Arthritis Rheum 65 (2013): 270-281.
- Lacoste C, Mansencal N, Ben m'rad M, et al. Valvular involvement in ANCA-associated systemic vasculitis: a case report and literature review. BMC Musculoskelet Disord 12 (2011): 50.
- Doherty L, Kumar P, Bexton R, et al. Aortic regurgitation and Churg-Strauss syndrome. QJM 98(2005): 772-773.
- Song YS, Seol S, Park J, et al. Large left ventricular non-infectious vegetation in patient with eosinophilic granulomatosis with polyangiitis. Cardiovasc J Afr 31 (2020): e1-e4.
- Karthikeyan K, Balla S, Alpert MA. Non-infectious aortic and mitral valve vegetations in a patient with eosinophilic granulomatosis with polyangiitis. BMJ Case Rep 12 (2019).
- Dennert RM, Paassen PV, Schalla S, et al. c. Arthritis Rheum 62 (2010): 627-634.
