A Very Aggressive Case of Adult Omental Rhabdomyosarcoma: Case report and Literature Review
Article Information
Samer H. Dbouk*, Hussein Mcheimeche, Bassam F Matar, Maureen Chbat, Mohamad Rakka
Department of General Surgery, Al Zahraa University Hospital, Jnah, Beirut, Lebanon
*Corresponding Author: Samer H. Dbouk, Department of General Surgery, Al Zahraa University Hospital, Jnah, Beirut, Lebanon
Received: 10 November 2020; Accepted: 20 November 2020; Published: 27 November 2020
Citation: Samer H. Dbouk, Hussein Mcheimeche, Bassam F Matar, Maureen Chbat, Mohamad Rakka. A Very Aggressive Case of Adult Omental Rhabdomyosarcoma: Case report and Literature Review. Journal of Surgery and Research 3 (2020): 428-434.
View / Download Pdf Share at FacebookAbstract
The greater omentum is a common location for metastatic tumors. On the other hand, the primary tumors of the omentum are rare. Intra-abdominal rhabdomyosarcoma (RMS) is extremely rare in adults. Few cases have been reported in the literature and the most of them were in the pediatric population. Omental rhabdomyosarcoma (RMS) in an adult, is an even rarer case that deserve the presentation. We are reporting a case of intra-abdominal rhabdomyosarcoma in a 54-year-old lady who presented with constipation and increasing abdominal girth. Investigations revealed disseminated omental rhabdomyosarcoma. We decided to start chemotherapy, but unfortunately she passed away on the tenth day of treatment.
Keywords
Omental Rhabdomyosarcoma
Rhabdomyosarcoma articles
Omental Rhabdomyosarcoma articles Omental Rhabdomyosarcoma Research articles Omental Rhabdomyosarcoma review articles Omental Rhabdomyosarcoma PubMed articles Omental Rhabdomyosarcoma PubMed Central articles Omental Rhabdomyosarcoma 2023 articles Omental Rhabdomyosarcoma 2024 articles Omental Rhabdomyosarcoma Scopus articles Omental Rhabdomyosarcoma impact factor journals Omental Rhabdomyosarcoma Scopus journals Omental Rhabdomyosarcoma PubMed journals Omental Rhabdomyosarcoma medical journals Omental Rhabdomyosarcoma free journals Omental Rhabdomyosarcoma best journals Omental Rhabdomyosarcoma top journals Omental Rhabdomyosarcoma free medical journals Omental Rhabdomyosarcoma famous journals Omental Rhabdomyosarcoma Google Scholar indexed journals metastatic tumors articles metastatic tumors Research articles metastatic tumors review articles metastatic tumors PubMed articles metastatic tumors PubMed Central articles metastatic tumors 2023 articles metastatic tumors 2024 articles metastatic tumors Scopus articles metastatic tumors impact factor journals metastatic tumors Scopus journals metastatic tumors PubMed journals metastatic tumors medical journals metastatic tumors free journals metastatic tumors best journals metastatic tumors top journals metastatic tumors free medical journals metastatic tumors famous journals metastatic tumors Google Scholar indexed journals Intra-abdominal rhabdomyosarcoma articles Intra-abdominal rhabdomyosarcoma Research articles Intra-abdominal rhabdomyosarcoma review articles Intra-abdominal rhabdomyosarcoma PubMed articles Intra-abdominal rhabdomyosarcoma PubMed Central articles Intra-abdominal rhabdomyosarcoma 2023 articles Intra-abdominal rhabdomyosarcoma 2024 articles Intra-abdominal rhabdomyosarcoma Scopus articles Intra-abdominal rhabdomyosarcoma impact factor journals Intra-abdominal rhabdomyosarcoma Scopus journals Intra-abdominal rhabdomyosarcoma PubMed journals Intra-abdominal rhabdomyosarcoma medical journals Intra-abdominal rhabdomyosarcoma free journals Intra-abdominal rhabdomyosarcoma best journals Intra-abdominal rhabdomyosarcoma top journals Intra-abdominal rhabdomyosarcoma free medical journals Intra-abdominal rhabdomyosarcoma famous journals Intra-abdominal rhabdomyosarcoma Google Scholar indexed journals laparoscopic cholecystectomy articles laparoscopic cholecystectomy Research articles laparoscopic cholecystectomy review articles laparoscopic cholecystectomy PubMed articles laparoscopic cholecystectomy PubMed Central articles laparoscopic cholecystectomy 2023 articles laparoscopic cholecystectomy 2024 articles laparoscopic cholecystectomy Scopus articles laparoscopic cholecystectomy impact factor journals laparoscopic cholecystectomy Scopus journals laparoscopic cholecystectomy PubMed journals laparoscopic cholecystectomy medical journals laparoscopic cholecystectomy free journals laparoscopic cholecystectomy best journals laparoscopic cholecystectomy top journals laparoscopic cholecystectomy free medical journals laparoscopic cholecystectomy famous journals laparoscopic cholecystectomy Google Scholar indexed journals postprandial vomiting articles postprandial vomiting Research articles postprandial vomiting review articles postprandial vomiting PubMed articles postprandial vomiting PubMed Central articles postprandial vomiting 2023 articles postprandial vomiting 2024 articles postprandial vomiting Scopus articles postprandial vomiting impact factor journals postprandial vomiting Scopus journals postprandial vomiting PubMed journals postprandial vomiting medical journals postprandial vomiting free journals postprandial vomiting best journals postprandial vomiting top journals postprandial vomiting free medical journals postprandial vomiting famous journals postprandial vomiting Google Scholar indexed journals cholecystitis articles cholecystitis Research articles cholecystitis review articles cholecystitis PubMed articles cholecystitis PubMed Central articles cholecystitis 2023 articles cholecystitis 2024 articles cholecystitis Scopus articles cholecystitis impact factor journals cholecystitis Scopus journals cholecystitis PubMed journals cholecystitis medical journals cholecystitis free journals cholecystitis best journals cholecystitis top journals cholecystitis free medical journals cholecystitis famous journals cholecystitis Google Scholar indexed journals leukocytosis articles leukocytosis Research articles leukocytosis review articles leukocytosis PubMed articles leukocytosis PubMed Central articles leukocytosis 2023 articles leukocytosis 2024 articles leukocytosis Scopus articles leukocytosis impact factor journals leukocytosis Scopus journals leukocytosis PubMed journals leukocytosis medical journals leukocytosis free journals leukocytosis best journals leukocytosis top journals leukocytosis free medical journals leukocytosis famous journals leukocytosis Google Scholar indexed journals bowel loops articles bowel loops Research articles bowel loops review articles bowel loops PubMed articles bowel loops PubMed Central articles bowel loops 2023 articles bowel loops 2024 articles bowel loops Scopus articles bowel loops impact factor journals bowel loops Scopus journals bowel loops PubMed journals bowel loops medical journals bowel loops free journals bowel loops best journals bowel loops top journals bowel loops free medical journals bowel loops famous journals bowel loops Google Scholar indexed journals transverse colon articles transverse colon Research articles transverse colon review articles transverse colon PubMed articles transverse colon PubMed Central articles transverse colon 2023 articles transverse colon 2024 articles transverse colon Scopus articles transverse colon impact factor journals transverse colon Scopus journals transverse colon PubMed journals transverse colon medical journals transverse colon free journals transverse colon best journals transverse colon top journals transverse colon free medical journals transverse colon famous journals transverse colon Google Scholar indexed journals microscopic residual tumor articles microscopic residual tumor Research articles microscopic residual tumor review articles microscopic residual tumor PubMed articles microscopic residual tumor PubMed Central articles microscopic residual tumor 2023 articles microscopic residual tumor 2024 articles microscopic residual tumor Scopus articles microscopic residual tumor impact factor journals microscopic residual tumor Scopus journals microscopic residual tumor PubMed journals microscopic residual tumor medical journals microscopic residual tumor free journals microscopic residual tumor best journals microscopic residual tumor top journals microscopic residual tumor free medical journals microscopic residual tumor famous journals microscopic residual tumor Google Scholar indexed journals
Article Details
Introduction
Rhabdomyosarcoma (RMS) is a malignant soft tissue tumor. It constitutes more than 50% of soft tissue sarcomas (STS) in children [1]. Whereas, it is extremely rare in adults and it accounts for less than 1% of all malignancies [2]. It can originate in any site of the body, but the most common locations are the head, neck and genitourinary tract [3]. In any age, intra- abdominal RMS and especially the omental involvement, is very rare, and only few cases have been reported in the literature [4]. Here, we are reporting a very rare case of omental rhabdomyosarcoma in adult.
Case presentation
A 54-year-old lady, smoker, with no known food and drug allergies, nor any past medical or surgical history, presented to the emergency department for acute onset of right upper quadrant abdominal pain associated with nausea and postprandial vomiting. Abdominal examination was only remarkable for positive Murphy sign. Laboratory studies revealed mild leukocytosis (WBC 12 x 103/µL) with neutrophil shift (PMN 85%). Abdominal ultrasonography was done and showed acute calculus cholecystitis for which she was scheduled to undergo laparoscopic cholecystectomy.
Under general anesthesia, the abdomen was insufflated and 2 trocars were inserted. Exploration revealed mild amount of clear peritoneal fluids in addition to adhesions between the omentum, liver and the anterior abdominal wall. The gallbladder was found to be severely inflamed. Peritoneal fluids were taken for bacterial culture and cytology examination. Due to the technical difficulty related to the adhesions, the operation was converted to open and cholecystectomy was then done. The post-operative course was uneventful and the patient was discharged home on the second postoperative day in good condition. The pathological examination of the gallbladder revealed chronic cholecystitis without malignancy. Unfortunately, the cytological examination of the peritoneal fluids demonstrated clusters of dysplastic epithelial cells consistent with carcinoma. The patient was then contacted and advised to come back for further workup but she was reticent because of the corona outbreak.
She presented after one month for evaluation of constipation and increasing abdominal girth. On physical examination: Her abdomen was distended with mild diffuse tenderness and dullness on percussion. Laboratory tests were normal except for mild leukocytosis (WBC 13 x 103/µL). A CT scan of the abdomen and pelvis revealed a moderate amount of ascites with a large intra-abdominal mass located at the level of left mid-lower abdomen, measuring 12 x 9 cms, causing displacement of the adjacent bowel loops (Figure 1). Tumor markers including C.E.A, CA19-9, AFP, CA 125 were within normal range.
Faced with this presentation of ascites and intra-abdominal masses and the inability to identify the origin of the tumor, we decided to perform a diagnostic laparoscopy. Exploration revealed a large amount of clear peritoneal fluids with diffuse omental thickening and adhesions, in addition to a large, hard and nodular mass located within the greater omentum. It was adherent to several small bowel loops and the transverse colon. The liver, spleen and ovaries appeared normal. As the mass was unrespectable, biopsies were taken.
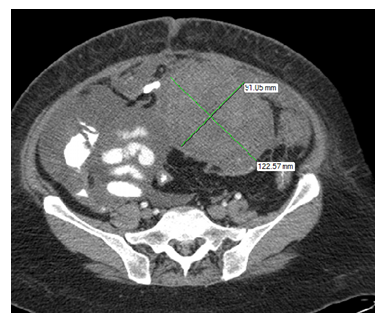
Figure 1: Showing a large, well defined intra-abdominal mass, measuring 12 x 9 cms
The histopathological examination revealed fibro-adipose tissues with multiple foci of infiltrating poorly differentiated neoplasm, consists of spindle mesenchymal cells with multiple better differentiated rhabdoid and muscle fiber cells, consistent with rhabdomyosarcoma (Figure 2).
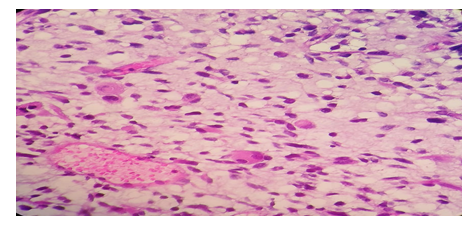
Figure 2: H and E; 40x40 X showing mixture of spindle cells, small round to ovoid cells and prominent rhabdoid cells having an eosinophilic cytoplasm and eccentric nuclei
Immunohistochemistry revealed strong staining for Desmin, Myogenin, CD34 and MYOD-1, therefore confirming the diagnosis of RMS (Figures 3, 4 and 5).
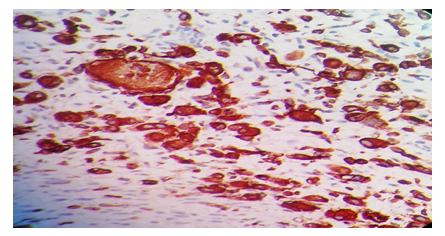
Figure 3: Immunohistochemistry, 40 × 40 X shows tumor cells showing desmin positivity
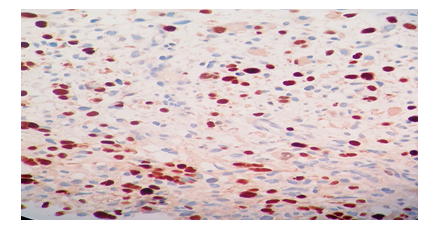
Figure 4: Immunohistochemistry, 40 × 40 X shows tumor cells showing Myogenin positivity
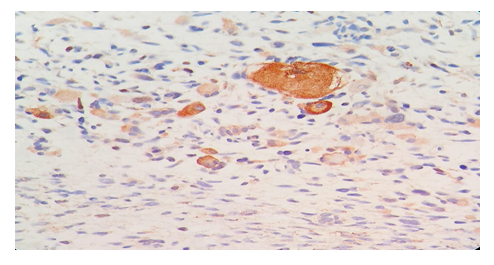
Figure 5: Immunohistochemistry, 40 × 40 X shows tumor cells showing MYOD-1 positivity
The patient was then started on VAC-IE regimen (vincristine, doxorubicin, cyclophosphamide alternating with ifosfamide and etoposide). Unfortunately, her condition began to deteriorate rapidly after the tenth day of chemotherapy until she was admitted to the intensive care unit and passed away.
Discussion
RMS is the most common soft tissue sarcoma in children. Omental RMS is an extremely rare malignancy. Only few cases have been reported in the literature and almost all of them were in the pediatric population [4]. Its pathogenesis is still poorly understood [7]. Some evidence suggests that RMS can arise from aberrant development of non-myogenic cells [8]. And mutations in macrophage inhibitory factor (MIF) and p53 are responsible for tumor progression [7]. The most common symptoms of intra-abdominal RMS –including omental RMS- are abdominal discomfort, abdominal mass and distention [9].
The imaging of choice for intra- abdominal RMS is Magnetic Resonance Imaging (MRI), which can determine the tumor origin and the relationship between the tumor and the adjacent organs. RMS is usually hyperintense on T2 and hypointense on T1 with heterogenous enhancement [6]. Computed Tomography CT scan, in the other hand, is important for staging and for assessing spread to lungs, as lung is the most common site of metastasis [8]. We note that recently PET-CT and PET -MRI are gaining a huge role in the management of RMS, from assessing response after neoadjuvant treatment to detecting residual tumor to local recurrence, metastatic spread and even have a role in the diagnosis of RMS. They give an insight on the tumor biology [10].
The world health organization (WHO) classifies this disease into 4 categories which differ by their histomorphology, most frequent localization, prevalence, IHC, genetics: Embryonal, alveolar, pleomorphic, and spindle cell/sclerosing RMS [11]. Pleomorphic subtype RMS is the most common subtype in adults [12]. Concerning the histological staining in helping to diagnose RMS, the myogenin and MyoD1, contrary to desmin, are highly sensitive and specific myogenic nuclear transcription factor for RMS and present early in the skeletal muscle differentiation [13].
Due to its rarity, the optimal treatment of adult RMS is uncertain [14]. Currently, the management of adult RMS is in line with the multidisciplinary treatment protocol which is proposed by the Intergroup Rhabdomyosarcoma Study (IRSG) group for treating children with RMS and includes complete surgical resection of the tumor with free margins, radiation and chemotherapy. Surgery and radiotherapy are used for the treatment of primary tumor site; however, chemotherapy is used to prevent tumor spread [15].
IRSG recommends that chemotherapy should be given to all patients with RMS and it improves survival. The recommended combination of chemotherapy includes vincristine, actinomycin-d, etoposide or ifosfamide and cyclophosphamide. Irnotecan, a topoisomerase I inhibitors is also recommended. Concerning radiotherapy, all patients must receive radiotherapy to ablate the microscopic residual tumor and to obtain long term control [16].
Chemo-radiotherapy can also be used as neo-adjuvant therapy that results in down staging of the disease. Thus, it allows complete excision of the tumor [17]. The prognosis is related to the presence of metastasis at presentation and the response to chemotherapy [18]. Adult RMS is very aggressive tumor and has a low 5-year survival rate (27%) compared to that observed in pediatric population [19].
Since RMS in children is a different entity from that of adults in terms of sensitivity to chemo-and radiotherapy, some studies showed that the prognosis in adults can be similar to that observed in children, if they are treated aggressively by using the pediatric protocol [18].
Conclusion
Adult omental RMS is an extremely rare and highly malignant neoplasm with a poor prognosis, even after surgical and chemotherapy interventions. Early diagnosis, complete resection, and appropriate radiotherapy and chemotherapy are the keys to managing this disease. Further studies are needed to develop the optimal treatment protocol for improving the prognosis of patients with this rare but deadly cancer.
References
- Clavel J, Steliarova-Foucher E, Berger C, et al. Hodgkin’s disease incidence and survival in European children and adolescents (1978–1997): report from the Automated Cancer Information System project. European Journal of Cancer 42 (2006): 2037-2049.
- Weiss SW, Goldblum J, Weiss SW, et al. Enzinger and Weiss’s Soft Tissue Tumors. 4th ed., St. Louis: CV Mosby (2001): p.785-835
- WU M. Spunt SL. SoO tissue sarcomas of childhood. Cancer Treat Rev 30 (2004): 269-280.
- Leung RS, Calder A, Roebuck D. Embryonal rhabdomyosarcoma of the omentum: two cases occurring in children. Pediatric Radiology 39 (2009): 865-868.
- Lanjekar A, Sathawane R, Gaikwad R, et al. Embryonal Rhabdomyosarcoma: A Rare Case Report. International Journal of Research and Review 6 (2019): 185-188.
- Bajaj G, Tirumani H, Whisman M, et al. Comprehensive Review of Abdominopelvic Mesenchymal Tumors With Radiologic Pathologic Correlation And Update On Current Treatment Guidelines–Part 2. InSeminars in Ultrasound, CT and MRI (2020).
- Aljabban I, Grant CN. Embryonal rhabdomyosarcoma of the urachal ligament presenting as small bowel obstruction: Case report and review of the literature. Journal of Pediatric Surgery Case Reports 53 (2020): 101357.
- Alkhormi AM, Alqifari A, Aljarbou OZ, et al. Primary duodenal embryonal rhabdomyosarcoma in adults: a case report. AME Case Reports 3 (2019).
- E Evans KJ, Miller Q, Kline AL. Solid omental tumors. New York: WebMD LLC (2011): c1994-c2013.
- Gennaro N, Marrari A, Renne SL, et al. Multimodality imaging of adult rhabdomyosarcoma: the added value of hybrid imaging. The British Journal of Radiology 93 (2020): 20200250.
- Christopher DM. World Health Organization classification of tumours. Pathology & Genetic: Tumours of Soft Tissue and Bone (2000).
- Furlong MA, Mentzel T, Fanburg-Smith JC. Pleomorphic rhabdomyosarcoma in adults: a clinicopathologic study of 38 cases with emphasis on morphologic variants and recent skeletal muscle-specific markers. Modern Pathology 14 (2001): 595-603.
- Carroll SJ, Nodit L. Spindle cell rhabdomyosarcoma: a brief diagnostic review and differential diagnosis. Archives of Pathology and Laboratory Medicine 137 (2013): 1155-1158.
- Little DJ, Ballo MT, Zagars GK, et al. Adult rhabdomyosarcoma: outcome following multimodality treatment. Cancer: Interdisciplinary International Journal of the American Cancer Society 95 (2002): 377-388.
- Crist WM, Anderson JR, Meza JL, et al. Intergroup rhabdomyosarcoma study-IV: results for patients with nonmetastatic disease. Journal of Clinical Oncology 19 (2001): 3091-3102.
- Yadav SK, Sinha DK, Ahmed A, et al. Primary intra-abdominal rhabdomyosarcoma in an adult: an unusual presentation and review of literature. Indian Journal of Surgical Oncology 6 (2015): 119-122.
- Agarwala S. Pediatric rhabdomyosarcomas and nonrhabdomyosarcoma soft tissue sarcoma. Journal of Indian Association of Pediatric Surgeons 11 (2006): 15.
- Arora A, Jaiswal R, Anand N, et al. Primary embryonal rhabdomyosarcoma of the liver. Case Reports (2016) 2016.
- Sparreboom B, Litton B, Yaxley J. A rare case of adult rhabdomyosarcoma. Polish Journal of Radiology 82 (2017): 395.
