A Protective Mechanism of Glucogon-like Peptide-1 on AGEs-Induced H9C2 Cardiomyocytes Injury through Inhibiting ROS-Autophagy Pathway
Article Information
Xiang Gu1,2*, Jun Zhang1, Wen-Jing Xia1, Pu-Hua Zhang1, Chang-Chang Yin2, Gao-Peng Guana1,3, Huang Long1
1Department of Cardiology, Affiliated Hospital of Jiujiang University, Jiujiang, P.R China
2Key Laboratory of System Biomedicine of Jiujiang University, Jiujiang, Jiangxi Province, P.R China
3Medicine Graduate School, Nanchang University, Nanchang, P.R China
*Corresponding Author: Xiang Gu, Department of Cardiology, Affiliated Hospital of Jiujiang University, East Xun Yang Road 57, Jiujiang 332000, P.R China.
Received: 26 July 2017; Accepted: 29 July 2017; Published: 02 August 2017
View / Download Pdf Share at FacebookAbstract
Diabetic cardiomyopathy is involved in multiple molecular mechanisms, including advanced glycosylation end products (AGEs), reactive oxygen species (ROS), and autophagy. Glucogon-like peptide-1 (GLP-1) is thought to prevent from the deleterious effects of AGEs through suppressing ROS in diabetes, but the role of GLP-1 in ROSmediated cardiomyocytes injury is unknown. In this study, we tested the hypothesis that Glucogon-like peptide-1 (GLP-1) would attenuate AGEs-induced injury in H9C2 cardiomyocytes by inhibiting ROS-autophagy pathway. Cultured H9C2 cardiomyocytes were treated with one of the following: (1) 1mg/L BSA , (2) 100mg/L AGEs-BSA, (3) 100mg/L AGEs-BSA and 10nmol/L GLP-1, (4) 100mg/L AGEs-BSA and 5nmol/L N-acetyl-L-Cysteine (antioxidant), (5) 100mg/L AGEs-BSA, 10nmol/L GLP-1 and 5μmol/L Rapamycin (autophagy agonist) for 24- hours. We found that AGEs impaired cell viability, increased Intracellular ROS productions, induced apoptosis and triggered autophagy; however, which were reversed remarkably by GLP-1or N-acetyl-L-Cysteine. Furthermore, the effects of GLP-1 of AGEs-induced cell injury were inhibited by Rapamycin except for ROS. These results suggest that GLP-1 may prevent H9C2 cardiomyocytes from AGEs-induced injury partly through inhibiting ROS-autophagy pathway.
Keywords
GLP-1; Ages; ROS; Autophagy; H9C2 Cardiomyocytes; Protective Mechanism
Article Details
1. Introduction
Diabetic cardiomyopathy (DCM) is increasingly recognized as an emerging risk of heart failure and mortality among diabetic patients independent of vascular pathology [1-2]. Advanced glycosylation end products (AGEs) play an indispensable role in the underlying mechanisms of DCM. The increased formation of AGEs secondary to hyperglycemia may alter structural proteins and lead to a series of pathological changes which involve oxidative stress, inflammation, apoptosis, fibrosis, and hypertrophy [3,4]. Recently, autophagy has been regarded as a novel mechanism in diabetic heart [5,6]. Some studies have demonstrated an inhibited autophagic flux in the hearts of type1 diabetic animals, which may be a cardioprotective response against hyperglycemia injury [7-9]. As a result of hyperglycemia, the formations of AGEs have been reported to result in the damages in some tissues by inducing autophagy [10-12].
Glucogon-like peptide-1 (GLP-1) is a kind of gut hormone secreted by the intestinal L cells, which participates in the maintenance of glucose homeostasis, insulin secretion and pancreatic beta-cell function [13,14]. In addition, GLP-1 also has a protective effect on cardiovascular disease [15,16]. Increasing evidences have suggested that the protective role of GLP-1 is concerned with prevention from the deleterious effects of AGEs in diabetes [17], but effects and mechanisms of GLP-1 on DCM are not fully understood. In this study, we investigated the effect of GLP-1 on autophagy in H9C2 cardiomyocytes incubated with AGEs, and evaluated the underlying role of autophagy regulated by GLP-1 in cardioprotection of DCM.
2. Materials and Methods
2.1 Cell Culture and Model of Ages-Induced Cell Injury
The rat cardiomyoblast cell lines H9c2 were offered by the Laboratory of Molecular Biology, the Second Affiliated Hospital of Nanchang University (China). The cell culture procedure was performed according to the previously reported technique [18]. H9C2 cardiomyocytes cultured for 3 days were treated with different doses of AGEs-BSA (Calbiochem, America) to establish a model of AGEs-induced cell injury.
2.2 Group of Experiments and Treatment Protocols
H9C2 cardiomyocytes were respectively treated with 1mg/L BSA (control); 100mg/L AGEs-BSA (AGEs); 100mg/L AGEs-BSA and 10nmol/L GLP-1 (AGEs+GLP-1); 100mg/L AGEs-BSA and 5µmol/L N-acetyl-L-Cysteine (AGEs+NAC); 100mg/L AGEs-BSA, 10nmol/L GLP-1 and 5µmol/L Rapamycin (AGEs+GLP-1+ Rapa). After incubation for 24 h, the effects of GLP-1 (Sigma, America) on AGEs-induced cell injury were appraised.
2.3 Cell Viability Measurements
Cell viability was determined by Cell Counting kit-8 (Beyotime Biotechnology, China). Briefly, following above treatments, the cells were plated onto 96-well plates, and incubated with 10?l CCK- 8 solution for 2 hours at 37°C. Optical density (OD) was measured (450 nm wavelength) using Micoplate reader (Biotek, America).
2.4 Intracellular ROS Measurements
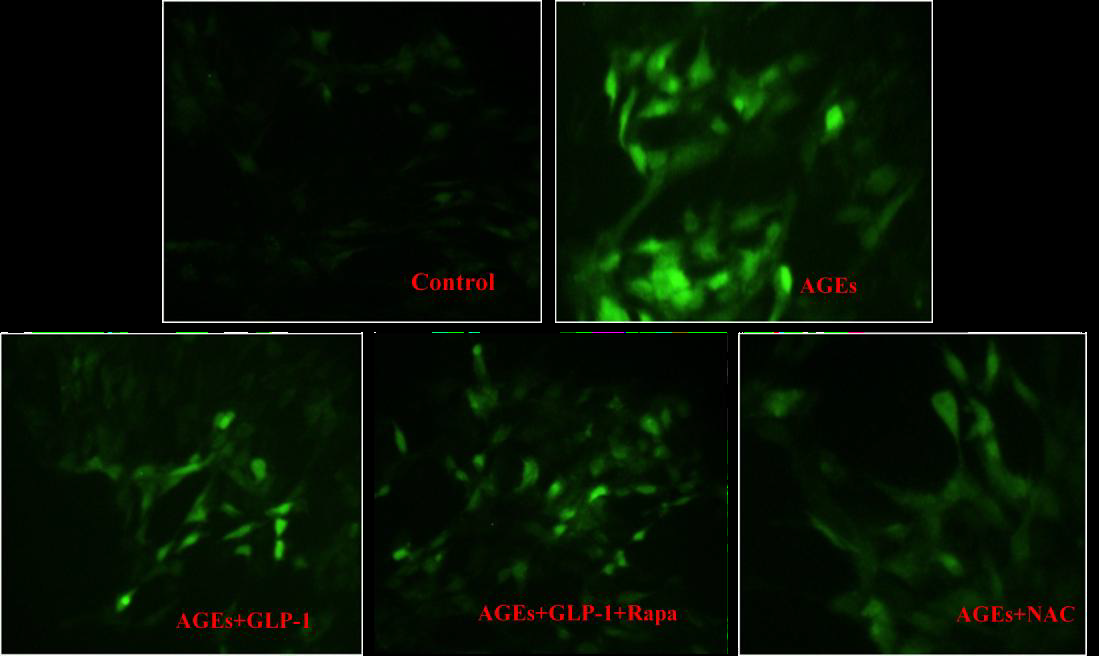
ROS measurement was carried out according to the procedure of ROS assay kit (Beyotime, China). After the treatments for 24 hours, 96-well plates with H9C2 cardiomyocytes were washed two times using PBS. 2, 7-dichlorodi-hydrofluorescein diacetate (DCFH-DA) was added into plates at a dose of 10?mol/L, and incubated in CO2 incubator (Eppendorf, Germany) for 20 minutes. The cells were subsequently washed three times, and then ROS-sensitive dye of cells was assessed under an inverted fluorescence microscope (Olympus, Japan) with an excitation wavelength of 488nm and an emission wavelength of 525nm. Fluorescent readings were acquired through image analysis software (Image J 1. 410).
2.5 Apoptosis Measurements
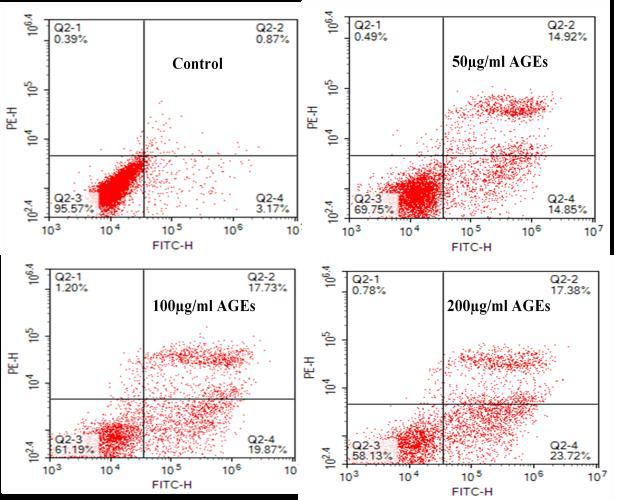
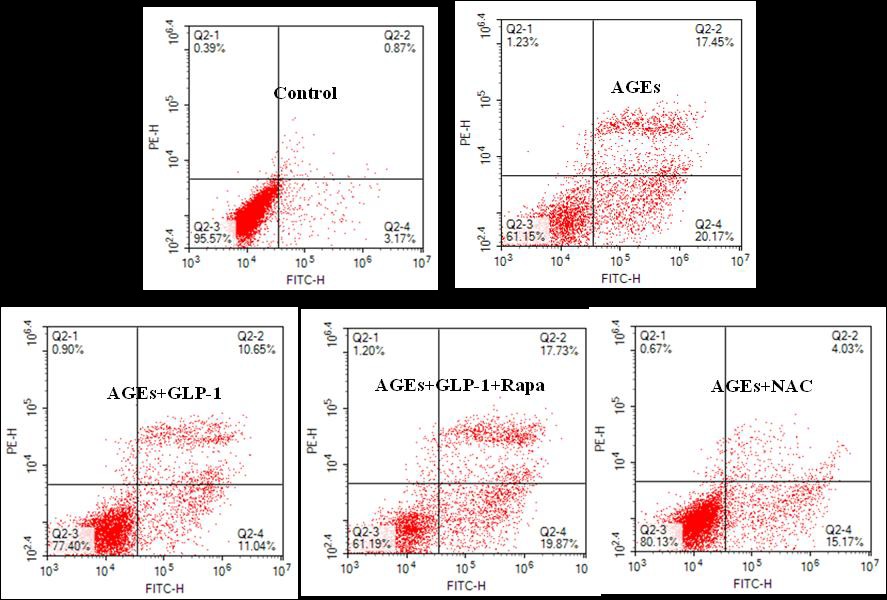
Apoptosis was measured by flow cytometry. In brief, cells from all groups were treated as follow: 0.25% trypsin (Solarbio, China) digestion, cells collection with centrifugation (300g, 4°C), and cell resuspension (1×Binding Buffer). Apoptosis was finally determined by using Annexin V/PI (Zoman, China) double-staining method. The cells were analyzed with a Calibur/Aria flow cytometer (BD, America).
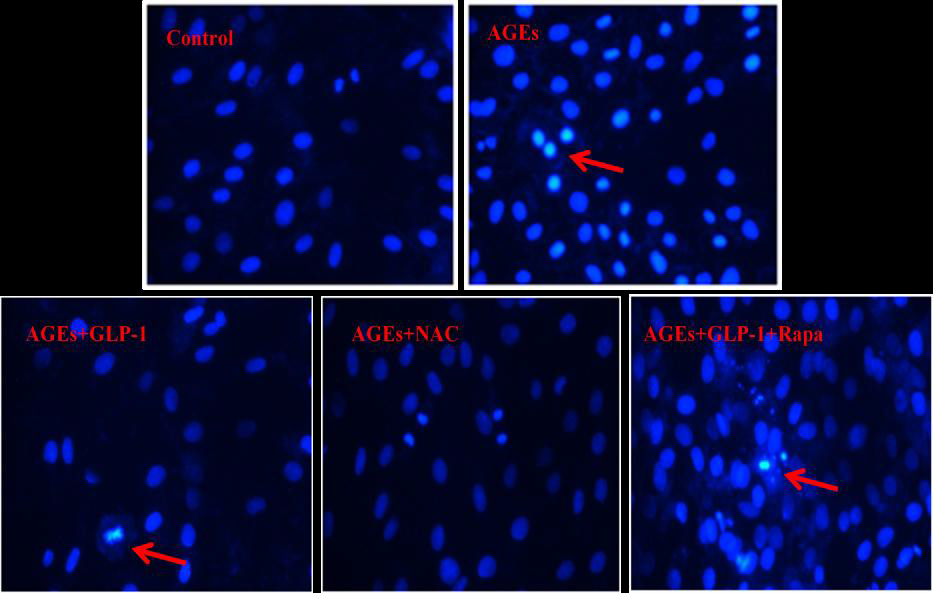
Hoechst 33258 kit (Beyotime, China) was performed to assess the morphology of apoptotic cells. Following the treatment protocols, the culture medium was removed; the cells in wells were washed three times with cold PBS, and fixed with 4% Paraformaldehyde for 5 minutes. After the cells were washed and dried, Hoechst 33258 was added to each well, and the cells were incubated away from light for an additional 5 minutes. The nuclear fluorescence was visualized under a BX50-FLA microscope (Olympus, Japan).
2.6 Autophagosome Formations Measurements
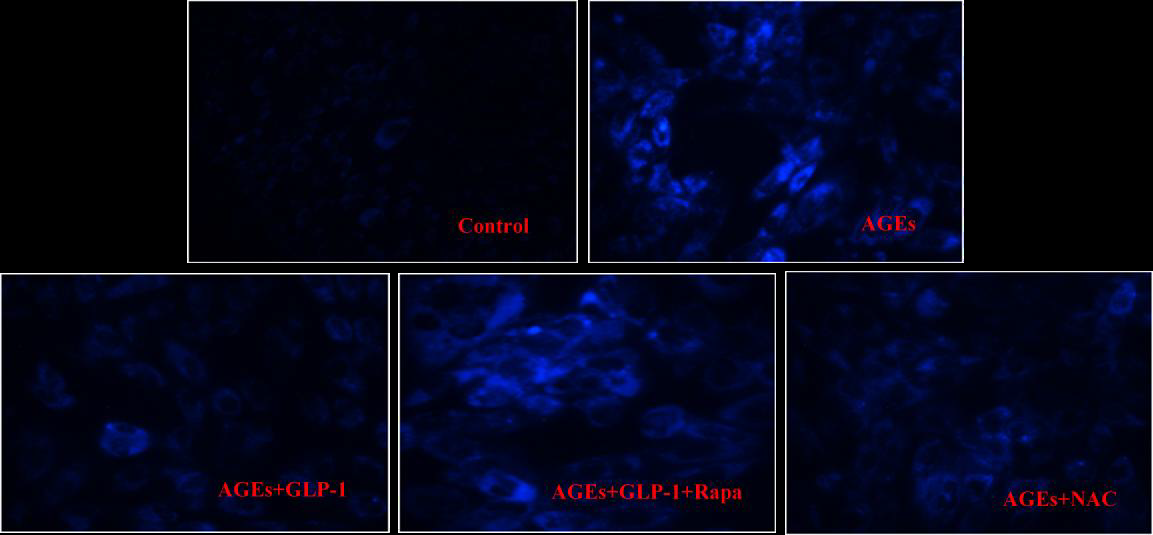
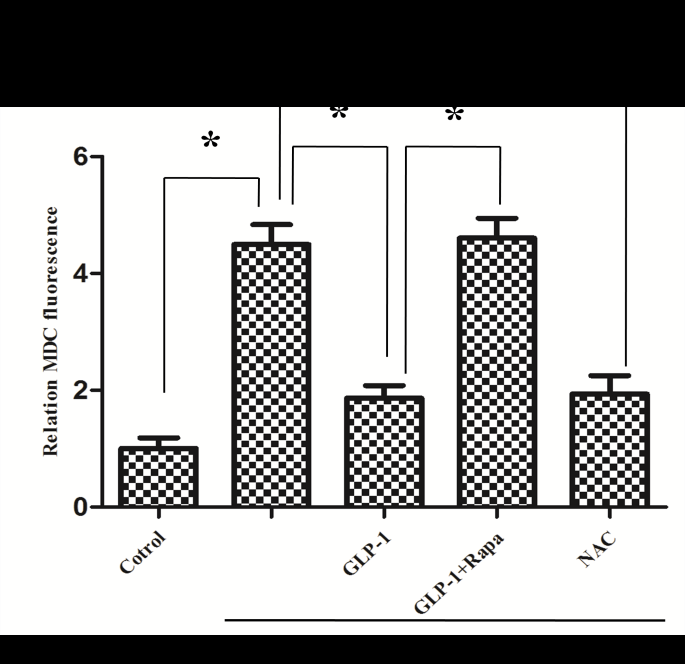
The formations of autolysosomes were detected by Monodansylcadaverine (MDC) staining (Solarbio, China). Cells from all groups in 24-well plates were incubated with 100 ?l MDC solution for 30 min at 37 °C, cells were washed two times with PBS. MDC was observed with a 400-435 nm filter set by fluorescence microscopy (Olympus, Japan).
2.7 Autophagy Associated-Protein Measurements
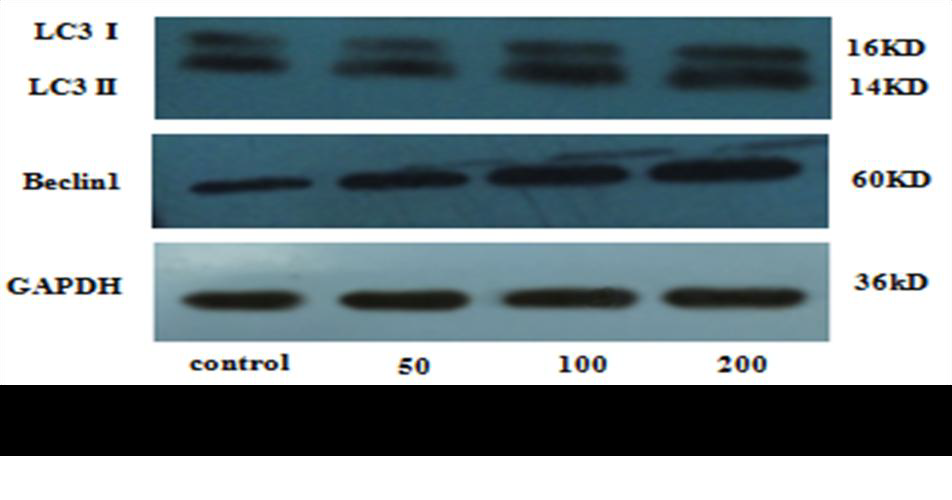
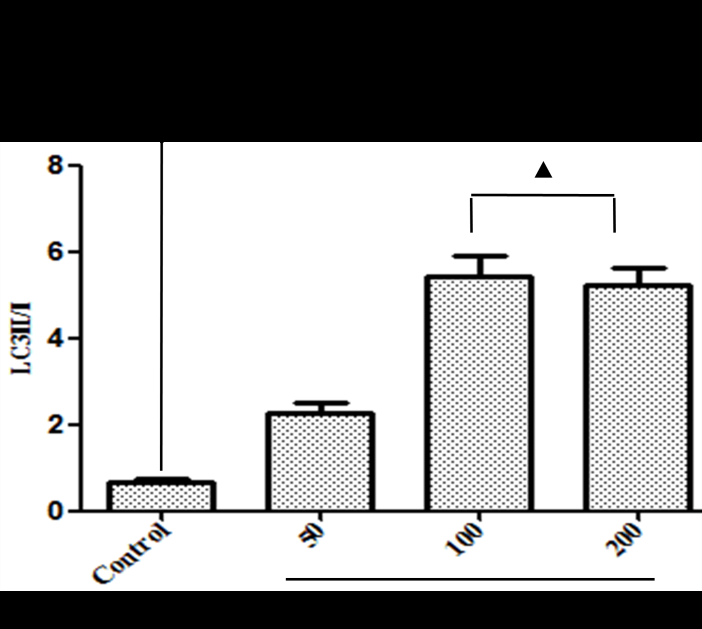
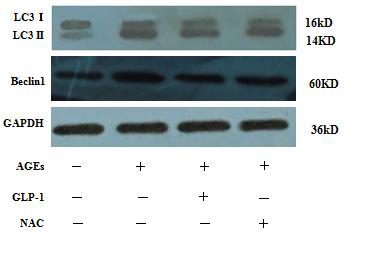
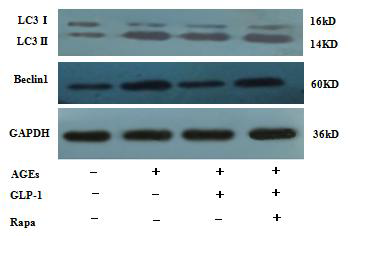
The expressions of autophagy associated-proteins, Beclin-1 and LC3II/I, were analyzed by western blot. Total protein was extracted by cell collection, cell lysis, and protein precipitation. The protein concentration was estimated using a bicinchoninic acid (BCA) assay kit. An equivalent of 30-50?g total protein was resolved on a 5-12% SDS-PAGE and transferred to the polyvinylidene fluoride (PVDF) membrane. Then, the membrane was blocked with 5% non-fat milk in TBST for 2 hours, followed by incubation overnight at 4°C with primary antibodies according to the manufacturer's recommendations. Subsequently, the membranes were washed thrice with TBST for 5 minutes each, incubated with horseradish peroxidase-conjugated secondary antibody in 5% non-fat milk-TBST for 1 hour and washed with TBST four times for 7 minutes each. The intensity of the immunoreactive bands of Beclin-1 and LC3B were quantified by Image J1. 410.
2.8 Autophagic Flux Measurements
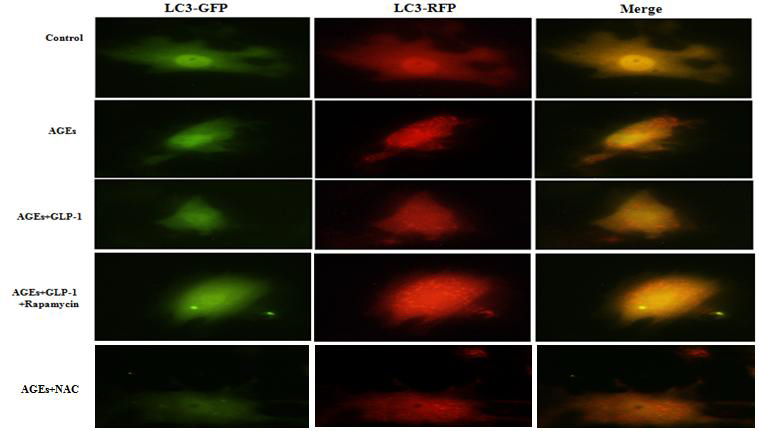
To further confirm the activity of autophagy, autophagic flux was monitored by Ad-mCherry-GFP-LC3 system (Beyotime, China). Briefly, the cells were incubated with mCherry-GFP-LC3 adenoviral vectors for 24 hours at 37°C, and then new medium was replaced in wells, followed by incubation with different reagents for 24 hours at 37°C. Autophagy was observed under a BX50-FLA fluorescence microscope (Olympus, Japan). Autophagic flux was determined by counting the number of GFP, RFP and merged points (dots/cells).
2.9 Statistical Analysis
All experiments were performed at least three times. All data were expressed as the mean ± SD. One-way ANOVA was applied to assess statistical significance between groups. A value of less than 0.05 was considered statistically significant.
3. Results
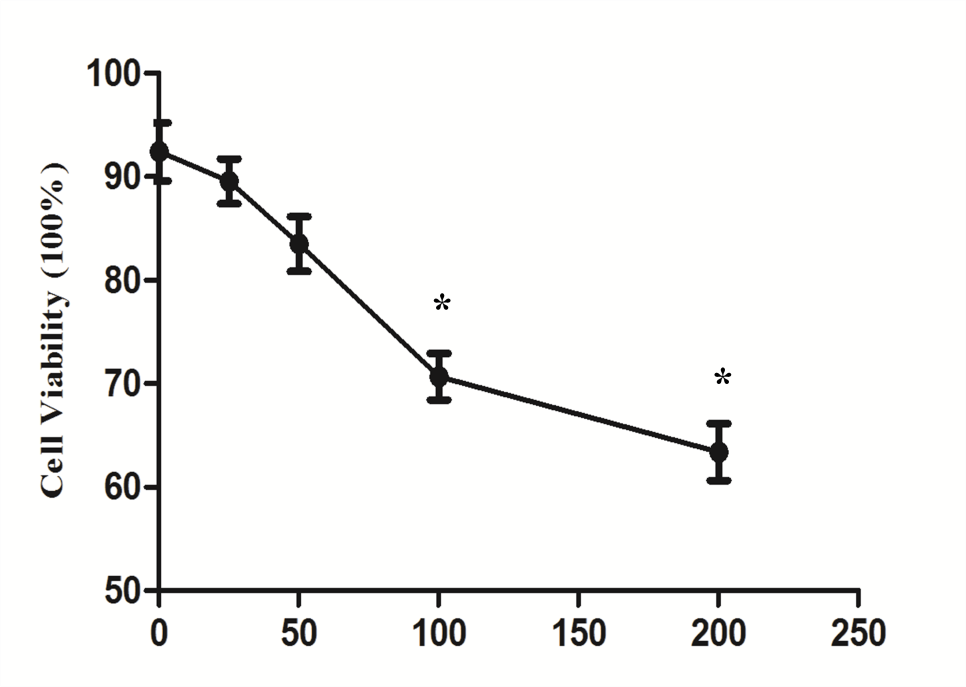
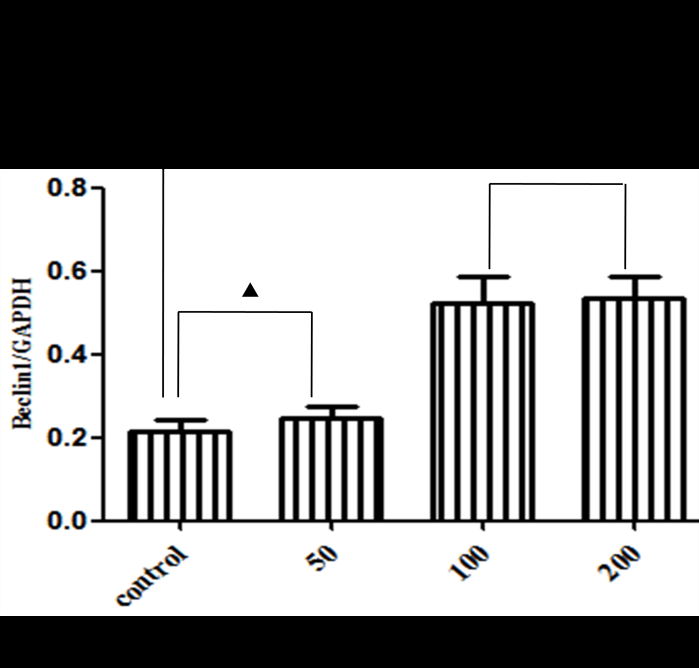
3.1 Ages Induce Cardiomyocytes Injury in A Dose-Dependent Manner
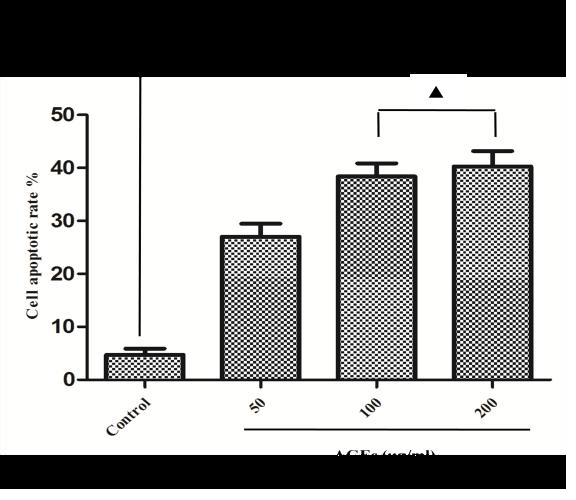
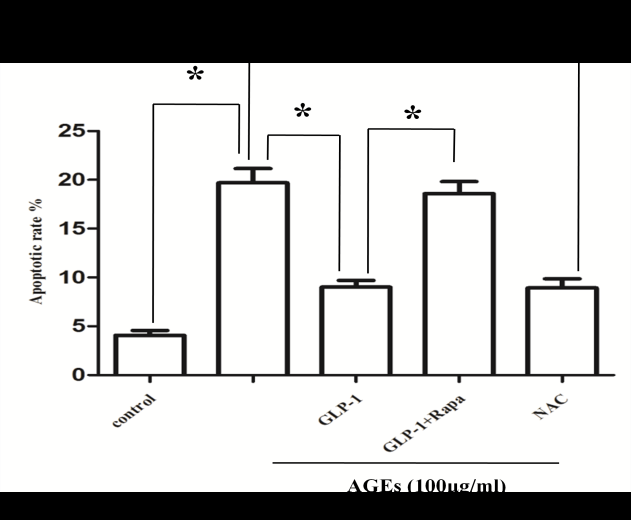
After administration of different doses of AGEs-BSA in H9C2 cardiomyocytes, AGEs resulted in H9C2 cardiomyocytes injury in a dose-dependent manner, which were demonstrated by measurements of cell viability (Figure 1A) and apoptosis rate (Figure 1B and 1C). 100mg/L AGEs was indicated as the dose of following experiment (Figure 1C).
3.2 Ages Induce Cardiomyocytes Injury Through the Increased Formation of ROS
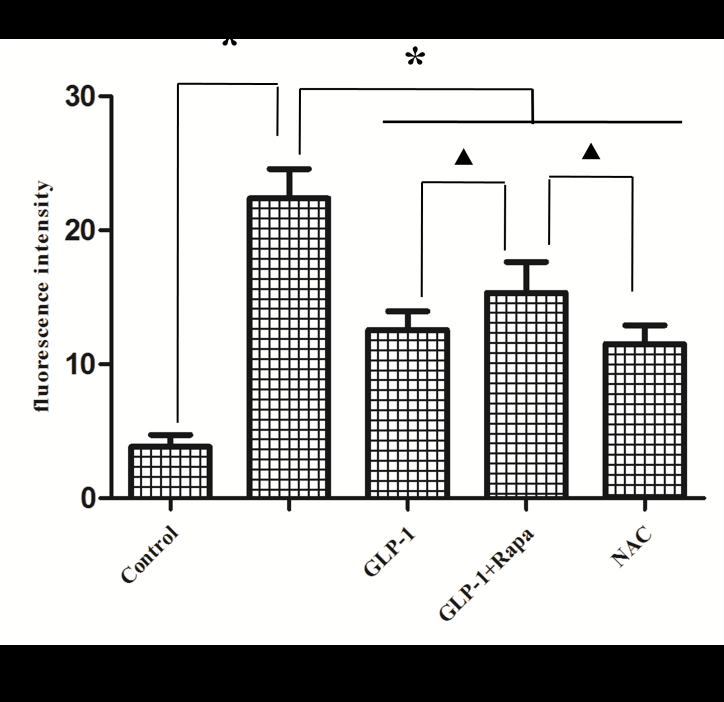
To further determine whether AGEs-induced cell injury is associated with increased ROS, antioxidant (N-acetyl-L-Cysteine, NAC) was applied to investigate the damage mechanism of AGEs. The results showed that NAC reduced Intracellular ROS levels (Figure 3) and apoptotic rate (Figure 2) significantly compared with AGEs. It suggests that AGEs might result in injury to H9C2 cardiomyocytes through the increased formation of ROS.
3.3 GLP-1 Attenuates Cardiomyocytes Injury By Suppressing the Formation of Ages-Induced ROS
To investigate the effect of GLP-1 on AGEs-induced injury in H9C2 cardiomyocytes, the model of AGEs-induced cells injury was treated with a dose of 10nmol/L GLP-1. The results showed that GLP-1 remarkably impaired the adverse effects of AGEs on apoptosis (Figure 2) and ROS levels (Figure 3), which had a similar effect to NAC (Figure 2 and Figure 3). It indicates that GLP-1 protected cardiomyocytes against AGEs-induced injury through interfering with the formation of ROS.
3.4 GLP-1 Inhibits Ages-Induced Autophagy in H9C2 Cardiomyocytes
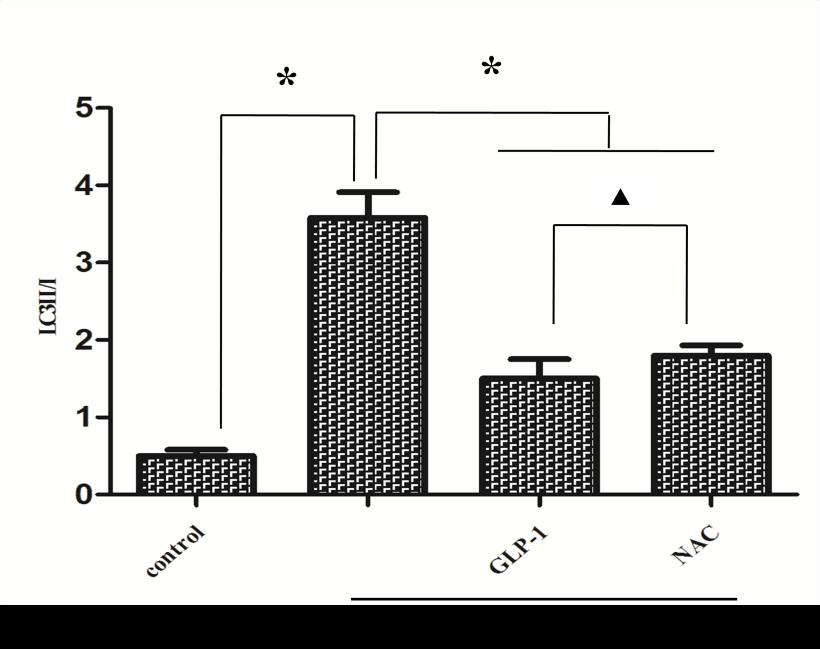
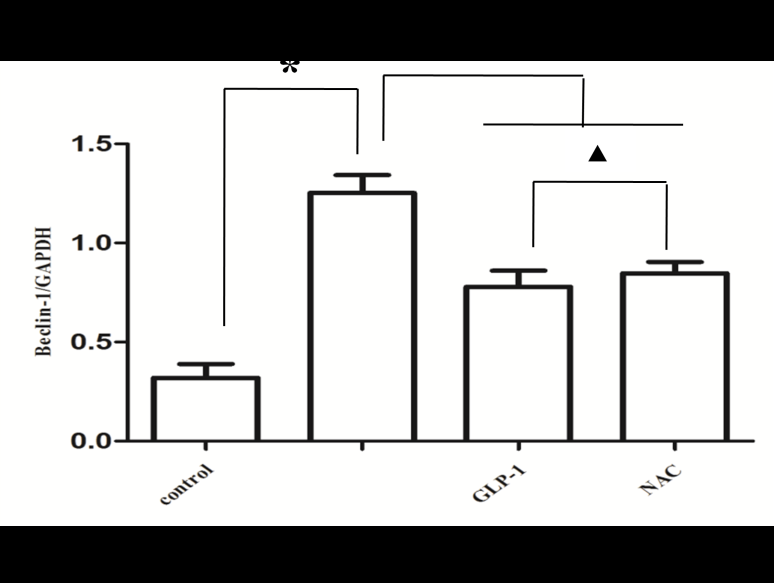
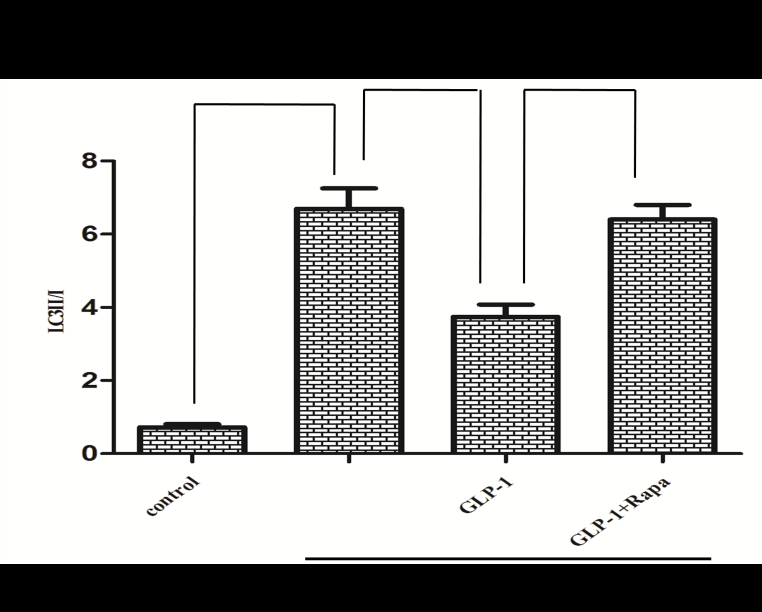
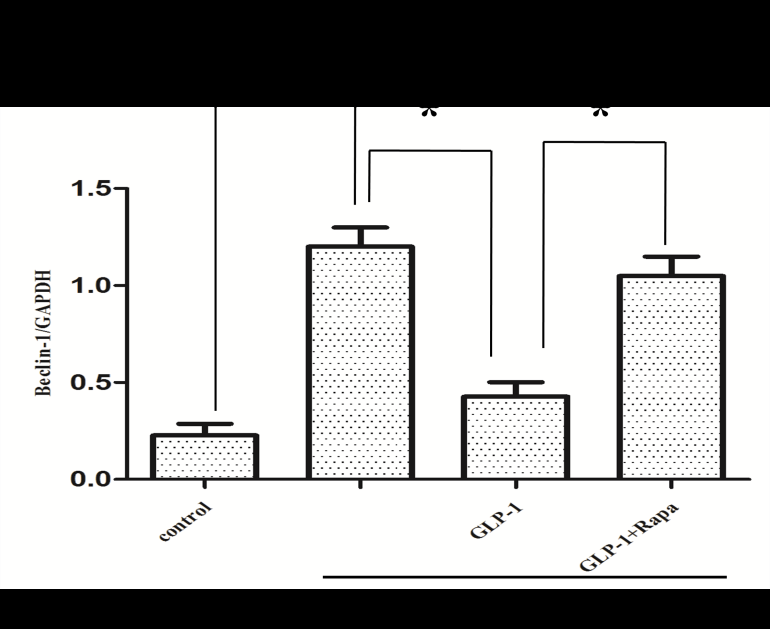
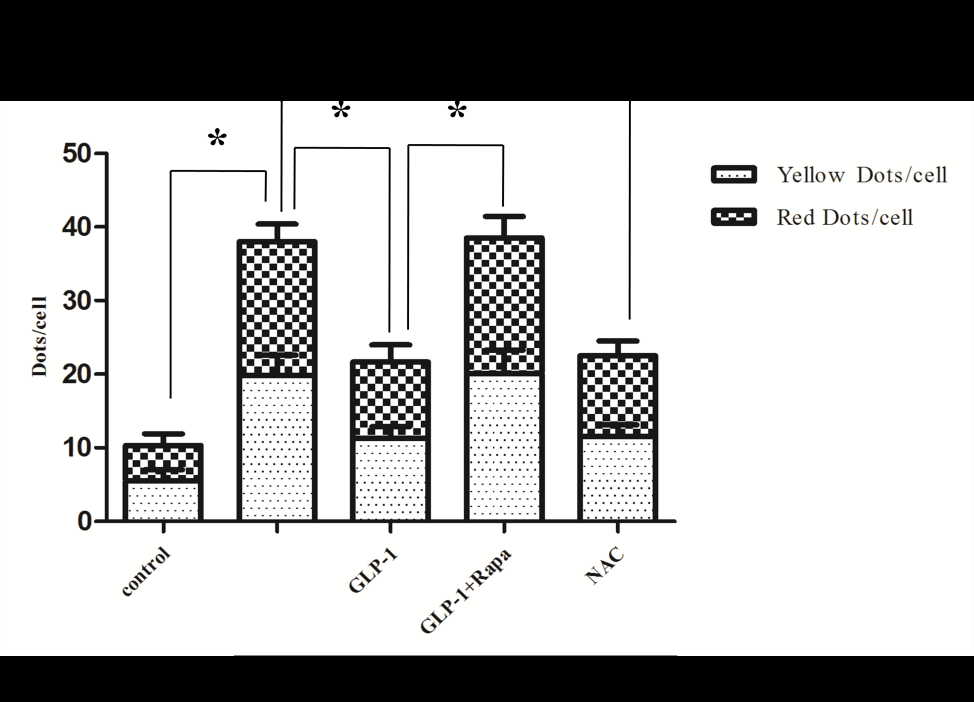
Since there is a tight interaction between ROS and autophagy in related pathological conditions [19,20], we tried to investigate whether GLP-1 had an effect on autophagy through AGEs-induced ROS pathway. The results showed that AGEs induced autophagy significantly at a dose of 100mg/L (Figure 5). After administration of GLP-1, there were lower expressions in the proteins of LC3II/I and Beclin-1 than that in AGEs group, which had no statistical difference compared with administration of NAC (Figure 6). Ad-mCherry-GFP-LC3 also suggested that autophagy was induced at a late stage in cardiomyocytes after incubation with AGEs for 24 hours. As shown in the pictures (Figure 8), there were more red points (representing autolysosomes) and fewer yellow points (representing autophagosomes) in AGEs group. However, after administrations of GLP-1 or NAC, there were fewer fluorescence points whether red or yellow than that in AGEs group (Figure 8). Taken together, our data suggest that GLP-1 might suppress autophagy through ROS-autophagy pathway during AGEs-induced cells injury.
3.5 GLP-1 Prevents Cardiomyocytes from Ages-Induced Injury Partly Through Inhibiting ROS-Autophagy Pathway
To further demonstrate whether GLP-1 participates in the process of protection against AGEs-induced cells injury through inhibiting ROS-autophagy pathway, an autophagy agonist, rapamycin was administrated to the model of AGEs-induced cells injury. The results showed that rapamycin significantly impaired the effects of GLP-1 on autophagy and apoptosis except for ROS (Figure 4, Figure 7, and Figure 8). Combining the results of NAC-inhibiting autophagy, it indicates that GLP-1 might protect cardiomyocytes from AGEs-induced injury partly through inhibiting ROS-autophagy pathway.
Figure 8: GLP-1 treatment inhibits AGEs-induced autophagy in H9C2 cardiomyocytes. (A). Ad-mCherry-GFP-LC3 system was performed to monitor autophagic flux in all groups. As shown in the pictures of merge, yellow points represented autophagosomes, and red points represented autolysosomes. There were fewer fluorescence points whether red or yellow in AGEs group compared with GLP-1 or NAC (*P<0.05).
4. Discussion
Autophagy is a catabolic process in which elderly or damaged organelles and proteins are engulfed by autophagosomes, delivered to and degraded in lysosomes under physiological or pathological conditions such as nutrient deprivation, energy exhaust and oxidative stress. Generally, autophagy is a survival pathway that provides substitutes for metabolism of cells or eliminates unwanted components of cells. However, autophagy is also represented as a mechanism of II programmed cell death while a persistent or intense autophagy is induced [21,22]. Our data demonstrate that autophagy was remarkably triggered in cardiomyocytes after administration of AGEs (100mg/L) for 24 hours; excessive ROS were simultaneously generated in cells, which were both impaired by N-acetyl-L-Cysteine or GLP-1. Our data also suggest that the damaged degree of cells was closely associated with the level of ROS. Recently the diabetic complications induced by AGEs have attracted much more attention, especially in diabetic heart [23]. AGEs-RAGE (receptor for AGEs) axis is regarded as a pivotal mechanism by which AGEs induce the generation of ROS, thereby producing pro-inflammatory mediators, and promoting cardiac fibrosis and hypertrophy via activation of nuclear factor ?-B [4,24]. It indicates that ROS may be an important messenger for AGEs-RAGE axis in the development of DCM.
ROS serve as crucial signaling molecules in the pathogenesis of numerous diseases, including DCM [5,20,25]. Although moderate ROS increase may induce autophagy, thereby triggering a negative feedback mechanism that mitigates oxidative stress and promotes cell survival, excessive ROS could mediate autophagic cell death [26,27,28], which might be involved in impairment of mitochondrial function and concomitant ROS formation [29,30]. Our data suggest that there is a positive correlation between level of ROS and activation of autophagy. Furthermore NAC or GLP-1 administration could significantly lower activity of autophagy and ameliorate cells injury through interfering with ROS generation. It has been reported that GLP-1 counteracts the AGE-induced ROS generation, and prevents AGE-induced necrosis and apoptosis in many cell cultures [31,32,33]. In this study, we first report the effect of GLP-1 on autophagy of cardiomyocytes through AGEs-induced oxidative stress pathway.
To further demonstrate whether autophagy itself also contributes to the cells injury independent of ROS, we administrated rapamycin in H9C2 cardiomyocytes incubated with AGEs and GLP-1 together. The results show that enhanced autophagy with rapamycin markedly promoted apoptosis, and decreased cell viability; but could not significantly increase ROS level in cardiomyocytes. Our findings are consistent with some previous studies in which autophagy had a deleterious effect on cardiomyocytes under overactivated condition [10,34]. Meanwhile, our research also indicates that the effect of GLP-1 on autophagy is involved in ROS-autophagy pathway. Collectively, all above results reveal a potential mechanism that GLP-1 may have protective effect on AGEs-induced injury in H9C2 cardiomyocytes partly through inhibiting ROS-autophagy pathway, but the exact mechanisms of which are fully unknown, and remain to be further explored.
Acknowledgements
This study was supported by the grants from the National Natural Science Foundation of China (No. 81660152).
References
- Dandamudi S, Slusser J, Mahoney DW, Redfield MM, Rodeheffer RJ, et al. The prevalence of diabetic cardiomyopathy: a population-based study in Olmsted County, Minnesota. J Card Fail 20 (2014): 304-309.
- Pham I, Cosson E, Nguyen MT, Banu I, Genevois I, et al. Evidence for a Specific Diabetic Cardiomyopathy: An Observational Retrospective Echocardiographic Study in 656 Asymptomatic Type 2 Diabetic Patients. Int J Endocrinol 2015: 743503.
- Heiko Bugger, E. Dale Abel. Molecular mechanisms of diabetic cardiomyopathy. Diabetologia 57 (2014): 660-671.
- Faria A, Persaud SJ. Cardiac oxidative stress in diabetes: mechanisms and therapeutic potential. Pharmacol Ther 172 (2017): 50-62.
- Kobayashi S, Liang Q. Autophagy and mitophagy in diabetic cardiomyopathy. Biochim Biophys Acta 1852 (2015): 252-261.
- Ouyang C, You J, Xie Z. The interplay between autophagy and apoptosis in the diabetic heart. J Mol Cell Cardiol 71 (2014): 71-80.
- Xu X, Kobayashi S, Chen K, Timm D, Volden P, et al. Diminished autophagy limits cardiac injury in mouse models of type 1 diabetes. J Biol Chem 288 (2013): 18077-18092.
- Xie Z, Lau K, Eby B, Lozano P, He C, et al. Improvement of cardiac functions by chronic metformin treatment is associated with enhanced cardiac autophagy in diabetic OVE26 mice. Diabetes 60 (2011): 1770-1778.
- Zhao Y, Zhang L, Qiao Y, Zhou X, Wu G, et al. Heme oxygenase-1 prevents cardiac dysfunction in streptozotocin-diabetic mice by reducing inflammation, oxidative stress, apoptosis and enhancing autophagy. PLoS One 8 (2013): e75927.
- Hou X, Hu Z, Xu H, Xu J, Zhang S, et al. Advanced glycation end products trigger autophagy in cardiomyocyte Via RAGE/PI3K/AKT/mTOR pathway. Cardiovasc Diabetol 13 (2014): 78.
- Sun K, Wang W, Wang C, Lao G, Liu D, et al. AGEs trigger autophagy in diabetic skin tissues and fibroblasts. Biochem Biophys Res Commun 471 (2016): 355-360.
- Verma N, Manna SK. Advanced Glycation End Products (AGE) Potently Induce Autophagy through Activation of RAF Protein Kinase and Nuclear Factor ?B (NF-?B). J Biol Chem 291 (2016): 1481-1491.
- Garber AJ. Incretin effects on beta-cell function, replication, and mass: the human perspective. Diabetes Care 34 (2011): S258?263.
- Meloni AR, DeYoung MB, Lowe C, Parkes DG. GLP-1 receptor activated insulin secretion from pancreatic ?-cells: mechanism and glucose dependence. Diabetes Obes Metab 15 (2013): 15-27.
- Kocyigit D, Gurses KM, Yalcin MU, Tokgozoglu L. Anti- hyperglycemic agents for the treatment of type 2 diabetes mellitus: Role in cardioprotection during the last decade. Endocr Metab Immune Disord Drug Targets 17 (2017): 19-31.
- Sattar N, Petrie MC, Zinman B, Januzzi JL Jr. Novel Diabetic Drugs and the Cardiovascular Specialist. J Am Coll Cardiol 69 (2017): 2646-2656.
- Puddu A, Mach F, Nencioni A, Viviani GL, Montecucco F. An Emerging Role of Glucagon-Like Peptide-1 in Preventing Advanced-Glycation-End-Product-Mediated Damages in Diabetes. Mediators Inflamm 2013: 591056.
- Cetrullo S, Tantini B, Flamigni F, Pazzini C, Facchini A, et al. Antiapoptotic and antiautophagic effects of eicosapentaenoic acid in cardiac myoblasts explored to palmitic acid. Nutrients 4 (2012): 78-90.
- Li L, Tan J, Miao Y, Lei P, Zhang Q. ROS and Autophagy: Interactions and Molecular Regulatory Mechanisms. Cell Mol Neurobiol 35 (2012): 615-621.
- Yan Y, Finkel T. Autophagy as a regulator of cardiovascular redox homeostasis. Free Radic Biol Med 109 (2016): 108-113.
- Yonekawa T, Thorburn A. Autophagy and Cell Death. Essays Biochem55 (2013): 105-117.
- Lee JH, Lee JH, Jin M, Han SD, Chon GR, Kim IH, et al. Diet control to achieve euglycemia induces significant loss of heart and liver weight via increased autophagy compared with ad libitum diabetic rats. Exp Mol Med 46 (2014): e111.
- Singh VP, Bali A, Singh N, Jaggi AS. Advanced glycation end products and diabetic complications. Korean J Physiol Pharmacol 18 (2014): 1-14.
- Kayama Y, Raaz U, Jagger A, Adam M, Schellinger IN, et al. Diabetic cardiovascular disease induced by oxidative stress. International Journal of Molecular Sciences 16 (2015): 25234?25263.
- Wold LE, Ceylan-Isik AF, Ren J. Oxidative stress and stress signaling: menace of diabetic cardiomyopathy. Acta Pharmacol Sin 26 (2005): 908-917.
- Castino R, Bellio N, Follo C, Murphy D, and Isidoro C. Inhibition of PI3k class III-dependent autophagy prevents apoptosis and necrosis by oxidative stress in dopaminergic neuroblastoma cells. Toxicol Sci 117 (2010): 152-162.
- Castino R, Fiorentino I, Cagnin M, Giovia A, and Isidoro C. Chelation of lysosomal iron protects dopaminergic SH-SY5Y neuroblastoma cells from hydrogen peroxide toxicity by precluding autophagy and Akt dephosphorylation. Toxicol Sci 123 (2011): 523-541.
- Higgins GC, Devenish RJ, Beart PM, and Nagley P. Transitory phases of autophagic death and programmed necrosis during superoxide-induced neuronal cell death. Free Radic Biol Med 53 (2012): 1960-1967.
- Dando I, Fiorini C, Pozza ED, Padroni C, Costanzo C, et al. UCP2 inhibition triggers ROS-dependent nuclear translocation of GAPDH and autophagic cell death in pancreatic adenocarcinoma cells. Biochim Biophys Acta 1833 (2013): 672?679.
- Wang X, Yu S, Wang CY, Wang Y, Liu HX, Cui Y, et al. Advanced glycation end products induce oxidative stress and mitochondrial dysfunction in SH-SY5Y cells. In Vitro Cell Dev Biol Anim 51 (2015): 204-209.
- Puddu A, Storace D, Durante A, Odetti P, Viviani GL. Glucagon-like peptide-1 counteracts the detrimental effects of Advanced Glycation End-Products in the pancreatic beta cell line HIT-T 15. Biochem Biophys Res Commun 398 (2010): 462-466.
- Ishibashi Y, Matsui T, Takeuchi M, Yamagishi S. Glucagon-like peptide-1 (GLP-1) inhibits advanced glycation end product (AGE)-induced up-regulation of VCAM-1 mRNA levels in endothelial cells by suppressing AGE receptor (RAGE) expression. Biochem Biophys Res Commun 391 (2010): 1405-1408.
- Ishibashi Y, Matsui T, Takeuchi M, Yamagishi S. Sitagliptin augments protective effects of GLP-1 against advanced glycation end product receptor axis in endothelial cells. Horm Metab Res 43 (2011): 731-734.
- Xiao R, Teng M, Zhang Q, Shi XH, Huang YS. Myocardial autophagy after severe burn in rats. PLoS One 7 (2012): e39488.