A Pilot Lifestyle Behavior Intervention for Overweight or Obese Pregnant Women: Results and Process Evaluation
Article Information
Mei-Wei Chang1*, Alai Tan1, Jonathan Schaffir2, Duane T. Wegener3, Brett Worly2, Katherine Strafford2, Loriana Soma2, Cassandra Sampsell2, Maggie Rosen2
1The Ohio State University College of Nursing, 1577 Neil Avenue, Columbus, OH 43210, USA
2The Ohio State University, Dept. of Obstetrics and Gynecology, 395 West 12th Avenue, 5th floor Columbus, OH 43210
3The Ohio State University Department of Psychology, 1835 Neil Avenue, Columbus, OH 43210, USA
*Corresponding Author: Mei-Wei Chang, Associate Professor, The Ohio State University College of Nursing 1577 Neil Avenue, Columbus, OH 43210, USA.
Received: 03 February 2023; Accepted: 09 February 2023; Published: 27 February 2023
Citation:
Mei-Wei Chang, Alai Tan, Jonathan Schaffir, Duane T. Wegener, Brett Worly, Katherine Strafford, Loriana Soma, Cassandra Sampsell, Maggie Rosen. A Pilot Lifestyle Behavior Intervention for Overweight or Obese Pregnant Women: Results and Process Evaluation. Journal of Pediatrics, Perinatology and Child Health 7 (2023): 10-20.
View / Download Pdf Share at FacebookAbstract
Introduction: This paper presents a pilot lifestyle behavior intervention effect on gestational weight gain and maternal and neonatal outcomes and intervention acceptability.
Materials and Methods: Overweight or obese pregnant participants (N = 70) were randomized to the intervention or usual care group. The 20-week intervention integrated Hope theory and goal-oriented episodic future thinking (GoEFT) to prevent excessive gestational weight gain through stress and emotion management, healthy eating, and physical activity. Intervention participants completed a weekly web intervention module with 2 parts (I and II) and joined individual health coaching sessions (10 sessions). The primary outcome was gestational weight gain (GWG). Secondary outcomes included maternal and neonatal outcomes. Data were collected at 3 time points: baseline (< 17 weeks gestation, T1), 24-27 weeks gestation (T2), and 35-37 weeks gestation (T3). Intervention participants completed a semi-structured interview to evaluate the intervention. We compared GWG at T2 and T3 with T1 for intervention and usual care groups using t-tests and conducted content analysis to identify common themes for intervention acceptability.
Results: There were no significant group differences in GWG at T2 and T3. Maternal and neonatal outcomes were similar between groups. Common themes for intervention acceptability were disked web Part I intervention presented in text, the need for choosing a weekly intervention topic, raising awareness through GoEFT and self-evaluation, increased motivation through GoEFT, and usefulness of pre-written goals and goal progress evaluation.
Conclusions: Results of process evaluation are helpful for researchers to design a lifestyle intervention to prevent excessive gestational wei
Keywords
<p style="text-align:justify">Pregnancy weight gain, goal-oriented episodic future thinking, obesity, gestational diabetes, gestational hypertension, preeclampsia, premature delivery, birth weight</p>
Pregnancy weight gain articles, goal-oriented episodic future thinking articles, obesity articles, gestational diabetes articles, gestational hypertension articles, preeclampsia articles, premature delivery articles, birth weight articles
Article Details
Introduction
Nearly 56% of American women aged 20-39 are overweight or obese.[1] These women are at least two times more likely than normal weight women to experience excessive gestational weight gain, [2,3] exceeding Institute of Medicine (IOM) pregnancy weight gain guidelines. [4] Excessive gestational weight gain is a serious public health problem, because it negatively influences maternal and neonatal outcomes: gestational diabetes, [5] gestational hypertension, [6] cesarean delivery, [7] and fetal macrosomia (birth weight > 4000 gm). [7,8] Excessive gestational weight gain is also a strong predictor of significant postpartum weight retention (retaining ≥ 10 lbs), which is associated with lifelong obesity in mothers [9] and childhood obesity. [10,11] Pregnancy is a teachable moment for weight management and obesity prevention. [12] Therefore, it is imperative to help overweight or obese pregnant women prevent excessive gestational weight gain.
Healthy lifestyle behaviors (healthy eating and physical activity) can prevent excessive gestational weight gain in overweight or obese pregnant women. [13] However, the intervention effect of prior studies has been modest (reducing gestational weight gain by ~3 lbs). [14,15] Integration of Hope theory and goal-oriented episodic future thinking is a promising approach to enhance the effect of such intervention. Yet, prior lifestyle behavior intervention studies have not included Hope theory or goal-oriented episodic future thinking. Hope theory [16] has 3 key concepts: agency thoughts, pathway thoughts, and affect. Agency thoughts refer to motivation to generate and implement plans for achieving goals. Pathway thoughts are the cognitive ability to generate and implement multiple plans for achieving goals. [16] Affect includes perceived stress and emotion.
Goal-oriented episodic future thinking involves picturing goal-relevant future events in the individual’s life. [17] Strong evidence from laboratory studies has shown that episodic future thinking (visualization [picturing] of future events) is associated with emotion regulation, decision-making, and memory. [17,18] Also, episodic future thinking enhances cognitive ability to generate and implement plans for achieving goals by connecting goals and motivation. [19] Moreover, episodic future thinking improves emotion, [18,20-22] which is associated with decreased perceived stress, [23] leading to less likelihood of engaging in emotional eating of unhealthy foods [24] and physical inactivity. [25]
We conducted a pilot lifestyle behavior intervention that incorporated Hope theory and goal-oriented episodic future thinking to help overweight or obese pregnant participants prevent excessive gestational weight gain. The pilot study was designed to evaluate feasibility of recruitment, retention, and intervention acceptability by the intervention participants. This paper presents results of primary and secondary outcomes and intervention acceptability by intervention participants.
Materials and Methods
Participants, Setting, and Procedure: A detailed description of the study procedure, setting, and study criteria has been published. [26] Participants were enrolled between February, 2021 and March, 2022. Briefly, clinicians at our collaborating sites referred pregnant patients to our study. The study research assistants screened the potential participants for eligibility over the phone. Eligible participants completed an electronic consent form and baseline measures. To be eligible to participate, participants were required to have body mass index (BMI) of 25.0-45.0 kg/m2 (calculated using self-reported height and pre-pregnancy weight) and be between 18 and 45 years old. We excluded potential participants with self-reported history of ≥ 3 miscarriages, eating disorder, and diagnosed hypertension and type 1 or 2 diabetes, or contraindications to walking. The study procedure was approved by the Ohio State University Institutional Review Board.
Randomization: Randomization of participants occurred upon completion of baseline measures at or less than 17 weeks gestation. We used a stratified permuted block randomization approach to randomly allocate participants to the intervention (n = 35) or usual care group (n =35). Specifically, participants in each race stratum (White vs. minority) were randomly allocated 1:1 to intervention or usual care using permuted block randomization with varying block sizes of 2 or 4.
Usual Care: All study participants received usual prenatal care from their obstetricians or midwives. The usual prenatal care visit occurs monthly until 28 weeks gestation, every other week between 28 and 36 weeks gestation, and weekly from 36 weeks gestation until delivery. We emailed all study participants a newsletter every other month with general information about pregnancy-related health (e.g., use of over-the-counter medications in pregnancy).
Intervention: We developed a 20-week intervention based on lessons learned from our prior intervention studies [27-29] and working closely with a peer advisory group (N = 5). The newly developed intervention integrated Hope theory and goal-oriented episodic future thinking. In this study, agency thoughts included autonomous motivation and self-efficacy. Pathway thoughts were consideration of future consequences and executive functions. Affect included stress and emotion. Below, we provide examples of Hope concept application. To increase autonomous motivation, participants were asked to evaluate the consistency between a personal value (a happy and healthy family or becoming a better me) and their current behaviors. To boost self-efficacy, our intervention helped participants to identify current and past successful plans for making positive change. To increase consideration of future consequences, participants identified short- and long-term pros of making and cons for not making positive changes. To boost executive functions, we provided practical ways to help participants plan tasks and engage in problem solving. To help reduce stress and promote positive mood, we provided tips for better time management and tips for avoiding or minimizing self-criticism.
The 20-week lifestyle behavior intervention modules had 2 key components: weekly web intervention with 2 parts (using Qualtrics software, a total of 20 modules, 30 min/module) and individual health coaching through Zoom (a total of 10 sessions, 45 min/session). The intervention addressed 3 main topics: stress and emotion management, healthy eating, and physical activity.
Web intervention: Participants used their username and password to access a web intervention module weekly (weeks 1-4) followed by every other week (weeks 6-12) and monthly (weeks 16-20). There were 6 modules addressing stress and emotion management, 10 modules covering healthy eating, and 4 modules addressing promotion of physical activity. Examples modules for improving stress and emotion management were time management and dealing with daily hassles. Healthy eating modules addressed (for example) meal planning and preparing healthy meals at home. Physical activity modules covered indoor and outdoor activities. Each module had 2-6 subtopics. For example, the dealing with daily hassles module included laundry, dishes, and communication (subtopics). The web Part I intervention (written text only) encouraged agency and pathway thoughts and stress and emotion management by asking participants to picture a goal-related event selected by participants from dropdown menus. Part I (25 minutes) also included 32 prewritten goals associated with the intervention subtopics paired with sample plans for achieving the goal. Part II (5 minutes) centered on goal progress evaluation. After participants evaluated their goal, they received a tailored message with focus on affirmation. Participants completed Part I during days 1-4 of the week followed by Part II during days 5-7. They also incorporated goal-oriented episodic future thinking in daily life. Moreover, they implemented self-evaluation of daily life to assess plans for achieving goal.
Health coaching session: There were 10 health coaching sessions (about 45 minutes per session): weekly (weeks 1-4) followed by every other week (weeks 6-12) and monthly (weeks 16-20). The health coaching was designed to reinforce content from the web intervention and helped participants to modify plans as needed. Participants also engaged in goal-oriented episodic future thinking during each health coaching session.
Measurements: Data were collected at 3 time points: baseline (< 17 weeks gestation, T1), 24-27 weeks gestation (T2), and 35-37 weeks gestation (T3). Primary and secondary outcomes were extracted from participants electronic health records. Intervention participants completed a semi-structed interview via Zoom to evaluate the intervention.
Primary Outcome: Gestational Weight Gain: We used the most proximal body weight at enrollment (the same day as randomization) as baseline weight (T1). To compute weekly gestational weight gain, we averaged body weight divided by the number of gestational weeks between the 2 timepoints. To calculate the total gestational weight gain, we subtracted self-reported pre-pregnancy weight from weight measured at T3. The IOM recommends that overweight women (BMI 25.0-29.9 kg/m2) be limited to total gestational weight gain of 15-25 lbs and obese women (BMI ≥ 30.0 kg/m2) to 11-20 lbs. [4] We used this guideline to identify women with gestation weight gain above IOM recommendations.
Secondary Outcomes: Maternal and Neonatal Outcomes: Maternal health outcomes included diagnoses of gestational diabetes, gestational hypertension, and preeclampsia, and incidence of cesarean delivery. The neonatal outcomes were birth weight, gestational age at delivery, Apgar scores, sex, and NICU admission.
Intervention Acceptability by Intervention Participants (Process Evaluation): At completion of the 20-week intervention, intervention participants were invited to evaluate the weekly web intervention and health coaching. The semi-structured interviews were conducted by trained interviewers through Zoom. The interviews were recorded with participants’ permission. Participants were asked to identify barriers and facilitators to intervention adherence and strengths and weaknesses of the intervention. They were also asked to provide suggestions to improve the intervention.
Analysis
Statistical analysis: Descriptive statistics were performed to summarize sample demographics and primary outcome at each timepoint and secondary outcomes. T-tests and Chi-square tests were used to compare sample demographics between groups (intervention vs. usual care). T-tests were also used to test between group differences in the primary outcome. We examined gestational weight gain and weekly gestation weight gain at T2 and T3 from T1. Chi-square tests were used to investigate between group difference in the proportion of participants with total gestation weight gain above IOM recommendations. To investigated between group differences in maternal and neonatal outcomes, we used two-sample T-tests for continuous outcomes (birth weight, 1-min and 5-min Apgar scores) and exact Chi-square tests for categorical outcomes (maternal complications, premature delivery, and sex). All tests were two-sided with a significance level of 0.05. We used SAS version 9.4 (SAS Institute®, Cary, NC) for all the statistical analyses.
Qualitative analysis: The semi-structured interview recordings were transcribed verbatim and reviewed to verify the accuracy of transcription. We developed a code book by reading the first 5 transcriptions. Two research assistants were trained to use deductive processes to identify common themes. [30] They performed coding independently. Discrepancies were discussed with the first author to reach a consensus.
Results
Demographics: Figure 1 presents a CONSORT chart of the pilot lifestyle behavior intervention. The key reasons for not being qualified to participate were BMI outside the range of 25.0 and 45.0 kg/m2 followed by a history of hypertension and ≥ 3 miscarriages. Of participants qualified to participate (n = 111), 41 did not complete baseline measures, leaving 70 participants randomized to the intervention (n = 35) or usual care group (n = 35).
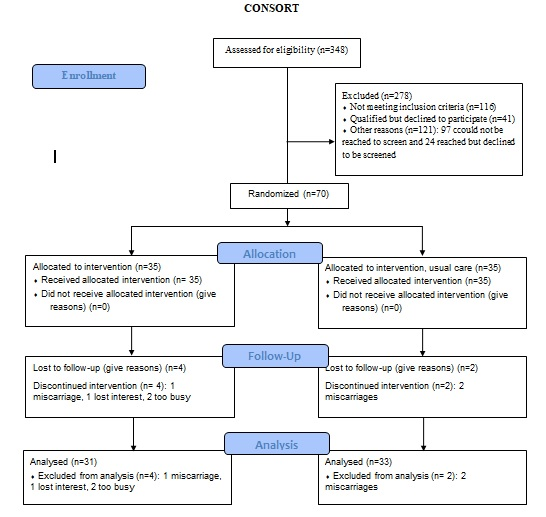
Figure 1: Consort Flowchart.
Table 1 shows demographic characteristics of the study sample (N = 70). There were no significant differences between intervention and usual care groups in these characteristics.
The average age was 32.2 (SD=4.0) years. The average BMI was 31.5 (SD=5.1) kg/m2. Majority of the participants were non-Hispanic White, married, well-educated (at least a bachelor’s degree), and employed full-time. Participants were almost evenly distributed by pre-pregnancy BMI category (overweight vs. obese) and number of pregnancies. Approximately 89% of participants were randomized and enrolled at or prior to 13 weeks gestation.
|
All |
Intervention |
Usual care |
P |
|
|
(N=35) |
(N=35) |
|||
|
Age (years), Mean±SD |
32.2±4.0 |
32.3±4.3 |
32.0±3.8 |
0.702 |
|
Race/Ethnicity, N (%) |
0.111 |
|||
|
Hispanic White |
3 (4.3) |
0 (0) |
3 (8.6) |
|
|
Asian |
3 (4.3) |
1 (2.9) |
2 (5.7) |
|
|
Non-Hispanic Black |
10 (14.3) |
8 (22.9) |
2 (5.7) |
|
|
Non-Hispanic White |
51 (72.9) |
25 (71.4) |
26 (74.3) |
|
|
More than one race |
3 (4.3) |
1 (2.9) |
2 (5.7) |
|
|
Marital Status, N (%) |
1 |
|||
|
Single, never married |
6 (8.6) |
3 (8.6) |
3 (8.6) |
|
|
Married |
63 (90.0) |
31 (88.6) |
32 (91.4) |
|
|
In a domestic partnership |
1 (1.4) |
1 (2.9) |
0 (0.0) |
|
|
Education, N (%) |
||||
|
High school graduate or GED |
1 (1.4) |
1 (2.9) |
0 (0.0) |
0.663 |
|
Some college or technical school, no degree |
2 (2.9) |
1 (2.9) |
1 (2.9) |
|
|
Associate degree |
4 (5.7) |
3 (8.6) |
1 (2.9) |
|
|
Bachelor degree |
27 (38.6) |
10 (28.6) |
17 (48.6) |
|
|
Master degree |
23 (32.9) |
12 (34.3) |
11 (31.4) |
|
|
Professional degree |
11 (15.7) |
7 (20.0) |
4 (11.4) |
|
|
Doctoral degree |
2 (2.9) |
1 (2.9) |
1 (2.9) |
|
|
Employment, N (%) |
0.106 |
|||
|
Employed, full time |
56 (80.0) |
30 (85.7) |
26 (74.3) |
|
|
Employed, part time |
6 (8.6) |
3 (8.6) |
3 (8.6) |
|
|
Unemployed, not currently looking for job |
2 (2.9) |
2 (5.7) |
0 (0.0) |
|
|
Self-employed |
1 (1.4) |
0 (0.0) |
1 (2.9) |
|
|
Homemaker |
3 (4.3) |
0 (0.0) |
3 (8.6) |
|
|
Student |
2 (2.9) |
0 (0.0) |
2 (5.7) |
|
|
Receiving Public Financial Assistance, N (%) |
1 |
|||
|
No |
67 (95.7) |
34 (97.1) |
33 (94.3) |
|
|
Yes |
3 (4.3) |
1 (2.9) |
2 (5.7) |
|
|
Pre-pregnancy BMI category, N (%) |
0.629 |
|||
|
Overweight |
30 (42.9) |
16 (45.7) |
14 (40.0) |
|
|
Obese |
40 (57.1) |
19 (54.3) |
21 (60.0) |
|
|
Pre-pregnancy BMI, Mean±SD |
31.5±5.1 |
31.5±5.1 |
31.0±4.6 |
0.388 |
|
Number of pregnancy, N (%) |
0.443 |
|||
|
1 |
25 (35.7) |
10 (28.6) |
15 (42.9) |
|
|
2 |
19 (27.1) |
11 (31.4) |
8 (22.9) |
|
|
3+ |
26 (37.1) |
14 (40.0) |
12 (34.3) |
|
|
Age of the youngest child (year), Mean±SD |
1.7±2.3 |
1.6±2.4 |
1.7±2.3 |
0.9 |
|
Number of children, Mean±SD |
0.9±1.0 |
0.9±0.7 |
0.8±1.2 |
0.801 |
|
Gestational age at screening (week), Mean±SD |
8.2±1.3 |
8.2±1.3 |
8.3±1.2 |
0.655 |
|
Gestational age at randomization Enrollment (week),* Mean±SD |
11.7±1.36 |
11.7±1.36 |
11.9±1.7 |
0.4 |
*Participants were randomized and enrolled on the same day
Table 1: Sample Demographics (N = 70).
Cohort Retention, Intervention Adherence, and Intervention Contamination: The overall retention rate was 91.4% (64/70). The key reason for dropout was miscarriage. We used Qualtrics software to track web intervention adherence. Every intervention participant (n = 35, 100%) completed the first module (both Parts I and II). The average completed modules was 10.55 (SD=6.42) of the 20; 25% of participants completed at least 16 modules. Regarding health coaching adherence, every intervention participant attended the first session. The average session was 7.9 (SD=3.2) of 10; 71.4 % of participants completed 8-10 sessions. Examining Qualtrics (web intervention) and semi-structured interview data, we did not detect any intervention contamination.
Gestational Weight Gain: Table 2 compares intervention and usual care groups in weekly gestational weight gain at T2 and T3 from T1. There were no significant group differences in gestational weight gain between T1 and T2 (Cohen’s d=0.13), between T2 and T3 (d=0.01), and between T1 and T3 (d=0.12). When examining weekly gestation weight gain, we did not observe significant group differences between T1 and T2 (d=0.11), between T2 and T3 (d=-0.05), and between T1 and T3 (d=0.04).
|
Intervention |
Usual care |
Intervention vs. Usual Care |
|||||
|
N |
Mean (SD) |
N |
Mean (SD) |
Mean Difference (95% CI) |
P |
Cohen’s D |
|
|
Weight, at T1 |
35 |
194.0 (31.6) |
35 |
198.1 (35.3) |
-4.09 (-20.05, 11.88) |
0.61 |
-0.12 |
|
Weight, at T2 |
31 |
202.8 (32.5) |
33 |
206.1 (36.3) |
-3.31 (-20.56, 13.93) |
0.7 |
-0.1 |
|
Weight, at T3 |
31 |
216.8 (32.1) |
33 |
220.1 (36.0) |
-3.25 (-20.33, 13.83) |
0.7 |
-0.1 |
|
GWG, between T1 and T2 |
31 |
10.9 (8.8) |
33 |
9.6 (11.0) |
1.31 (-3.69, 6.31) |
0.6 |
0.13 |
|
Weekly GWG, between T1 and T2 |
31 |
0.9 (0.7) |
33 |
0.8 (0.9) |
0.08 (-0.30, 0.46) |
0.68 |
0.11 |
|
GWG, between T2 and T3 |
31 |
14.0 (7.1) |
33 |
13.9 (12.6) |
0.06 (-5.08, 5.20) |
0.98 |
0.01 |
|
Weekly GWG, between T2 and T3 |
31 |
1.2 (0.6) |
33 |
1.3 (1.1) |
-0.05 (-0.50, 0.41) |
0;84 |
-0.05 |
|
GWG, between T1 and T3 |
31 |
24.9 (12.2) |
33 |
23.5 (10.8) |
1.37 (-4.39, 7.13) |
0.64 |
0.12 |
|
Weekly GWG, between T1 and T3 |
31 |
1.0 (0.5) |
33 |
1.0 (0.5) |
0.02 (-0.22, 0.25) |
0.89 |
0.04 |
The most proximal body weight at enrollment (the same day as randomization) was used as baseline (T1).
Table 2: Comparison of Gestation Weight Gain (GWG) at T2 (24-27 Weeks Gestation) and T3 (35-37 Weeks Gestation) from T1 (Baseline) between Intervention and Usual Care Groups.
Table 3 presents the proportion of participants with a total gestational weight gain below, at, or above IOM recommendations. There were no significant differences in total gestational weight gain between groups. Overall, more than two-third of participants had total gestational weight gain above IOM recommendations. There were no significant group differences in total gestational weight gain above IOM recommendations when stratified by weight status (overweight vs. obese).
|
N (%) |
P |
||
|
Overall (N = 64) |
Intervention |
Control |
0.848 |
|
(n=31) |
(n=33) |
||
|
Below |
3 (9.68) |
2 (6.06) |
|
|
Within |
6 (19.35) |
8 (24.24) |
|
|
Above |
22 (70.97) |
23 (69.70) |
|
|
Overweight (n = 29) |
Intervention |
Control |
1 |
|
(n=15) |
(n=14) |
||
|
Below |
1 (6.67) |
0 (0.00) |
|
|
Within |
4 (26.67) |
4 (28.57) |
|
|
Above |
10 (66.67) |
10 (71.43) |
|
|
Obesity (n =35) |
Intervention |
Control |
0.868 |
|
(n=16) |
(n=19) |
||
|
Below |
2 (12.50) |
2 (10.53) |
|
|
Within |
2 (12.50) |
4 (21.05) |
|
|
Above |
12 (75.00) |
13 (68.42) |
|
Total gestational weight gain was calculated by subtracting self-reported pre-pregnancy weight from weight measured at T3 (35-37 weeks gestation)
Table 3: Proportion of Overweight or Obese Participants Meeting or Not Meeting IOM Gestational Weight Gain Recommendations.
Maternal and Neonatal Outcomes: Table 4 shows maternal and neonatal outcomes. The distribution of maternal and neonatal outcomes were similar between groups. However, we found that premature delivery occurred in 0% intervention but 9.1% in the usual care group.
|
N (%) or Mean±SD |
||
|
Intervention (N=31) |
Usual Care (N=33) |
|
|
Maternal Outcomes |
||
|
Diagnosed with gestation diabetes |
4 (12.9) |
5 (15.2) |
|
Diagnosed with gestation hypertension |
4 (12.9) |
2 (6.1) |
|
Preeclampsia |
2 (6.5) |
2 (6.1) |
|
Cesarean delivery |
9 (29.0) |
9 (27.3) |
|
Ongoing major depression |
4 (12.9) |
7 (21.2) |
|
Ongoing anxiety |
4 (12.9) |
5 (15.2) |
|
Postpartum hemorrhage |
1 (3.2) |
0 (0.0) |
|
Chorioamnionitis |
1 (3.2) |
1 (3.0) |
|
Placenta abstraction |
0 (0.0) |
0 (0.0) |
|
Neonatal Outcomes |
||
|
Birth weight (g) |
3493±327.7 |
3503±483.2 |
|
Birth weight stratum |
||
|
Low birth weight (< 2500g) |
0 (0.0.) |
1 (3.0) |
|
Normal birth weight |
29 (93.5) |
29 (87.9) |
|
Macrosomia (> 4000g) |
2 (6.5) |
3 (9.1) |
|
Apgar score |
||
|
At 1 min |
8.3±1.3 |
8.2±1.0 |
|
At 5 min |
9.0±0.2 |
8.8±0.7 |
|
NICU admission |
0 (0.0) |
0 (0.0) |
|
Premature delivery (< 37 weeks gestation) |
0 (0.0) |
3 (9.1) |
|
Shoulder dystocia |
0 (0.0) |
0 (0.0) |
|
Sex |
||
|
Boy |
15 (48.4) |
18 (54.5) |
|
Girl |
16 (51.6) |
15 (45.5) |
Table 4: Maternal and Neonatal Outcomes (N = 64).
Intervention Acceptability by the Intervention Participants: Of the 31 intervention participants completed the 20-week intervention, 19/31 (61.3%) completed the interview. The main reason for not completing the interview was COVID infection in either the participant or family members. Below, we presented common themes for intervention acceptability by the intervention participants.
Intervention length and frequency: Participants unequivocally commented that the length of the web intervention (30 min) was appropriate. However, they suggested reducing its frequency. This was because the weekly intervention created additional burden on daily life, particularly for participants dealing with COVID-related issues. Although participants suggested shortening the length of the health coaching sessions, they expressed a concern that shorter sessions might not meet their needs.
Disliked web Part I intervention presented in text: Participants consistently reported dissatisfaction with having to read written text in the Part I web intervention. Although they all agreed that the Part I intervention content was very helpful and comprehensive, they felt it was repetitive and boring overtime. As a result, they became lack of interest in continuing the web intervention.
The need for choosing a weekly intervention topic and usefulness of sample plans: Participants consistently reported dissatisfaction with being assigned a main intervention topic for a designated week, because the assigned main topic did not always meet their needs for the week. For example, when they experienced a high level of stress, they desired to learn how to better manage stress and emotion. Yet, they were asked to complete a healthy eating module. However, participants appreciated the health coach providing plans that have worked for peers of the participants with similar goals as input to help them generate their own plans. Participants said these plans were easy and practical to implement into their daily life. They also reported that the health coach helped make them aware of their past successful plans, which empowered and motivated them to generate plans for making positive changes.
Raising awareness through goal-oriented episodic future thinking and self-evaluation: Intervention participants consistently reported that the intervention helped them raise awareness through goal-oriented episodic future thinking (picturing) and self-evaluation. For example, picturing a personal value (a happy and healthy family or becoming a better me) and goal associated short- and long-term pros for making and cons for not making positive changes made them realize inconsistency between their personal value and current behaviors. Daily self-evaluation helped them realize which plans were working or not working for them for achieving goal. Raising awareness motivated participants to generate and implement plans for making continuous positive changes.
Increased motivation through goal-oriented episodic future thinking (picturing): Participants unequivocally reported enjoyment of picturing personal values, goal, pros and cons, and practical plans for achieving their goals. They said it was very easy to incorporate and implement picturing into daily life, because it took less than a minute each time. Most importantly, picturing highly motivated them to generate and implement plans to make continuous positive changes.
Usefulness of pre-written goals and goal progress evaluation: Our intervention participants unanimously said that the list of prewritten goals (web Part I) were comprehensive and helpful, because it would be difficult for them to generate their own. Participants enjoyed evaluating goal progress (web Part II, written text), because it helped them understand their progress in meeting goals. Although they enjoyed receiving tailored feedback with a focus on affirmation, they suggested receiving tailored feedback with a focus on successful plans for achieving their goals.
Request of summaries: Participants consistently requested receipt of a summary showing completed activities for both Parts I and II. They made this request, because they wanted to review their completed activities and track progress of making positive changes overtime.
Discussion
This pilot intervention is one of the first to integrate Hope theory and goal-oriented episodic future thinking to promote stress and emotion management, healthy eating, and physical activity in a real-world setting. Even though the entire project was conducted during the COVID pandemic, we successfully enrolled and retained study participants achieving the study aim. However, the pandemic most likely had a negative effect on the study outcomes, because our intervention was designed without knowing unprecedented pandemic. Our review of all health coaching sessions (for the purpose of monitoring fidelity) has suggested potential improvements to the intervention, which have been included in the discussion below.
Intervention Adherence: Consistent with prior research, [31] our web intervention adherence rate was disappointing and was associated with perception of repetitive and boring written content overtime (web Part I). Another possible factor affecting our web intervention adherence was the use of Qualtrics software, which limited our ability to create an appealing interface. However, our health coaching adherence rate was very high. This was most likely related to participants’ perception of receiving individualized intervention, a recommended strategy for promoting healthy lifestyle behaviors. [32]
Gestational Weight Gain: We did not detect significant differences in excessive gestational weight gain, total gestational weight gain above IOM recommendations, between the intervention and usual care groups. However, the objective of this pilot intervention study was to evaluate feasibility of recruitment, retention, and intervention acceptability by the intervention participants rather than to investigate the intervention effect. We found that 71.0% of intervention and 69.7% of usual care participants experienced excessive gestational weight gain. Our finding is consistent with the findings of a recent meta-analysis of randomized controlled trials aimed to prevent excessive gestational weight gain among overweight or obese participants in which 64.7% of intervention participants and 70.8% of usual care participants gained excessive weight. [13]
Intervention Acceptability by the Intervention Participants
Disliked web Part I intervention presentation presented in text: Because our intervention participants did not enjoy web Part I presented solely in written text, they might have paid little attention to example plans associated with participants’ chosen pre-written goals. This assumption is supported by listening to our health coaching recordings. Our finding suggests that intervention contents presented in written text might not motivate participants to engage in the intervention overtime. Future web interventions might consider minimizing written text and presenting intervention content in other forms. For example, inserting video clips that feature peers of the study population could have great potential to increase intervention adherence.[33]
The need for choosing a weekly intervention topic and usefulness of sample plans: Intervention studies typically assign an intervention topic for a designated week to ensure every participant receives the same intervention with the same sequence. However, this approach was not acceptable by our intervention participants. Nevertheless, participants enjoyed learning about their peers’ successful and practical plans for achieving goal from the health coach. We observed that learning about peers’ successful plans helped intervention participants realized their past success, thus motivated and empowered them to generate and implement plans for achieving their own goals.
Raising awareness through goal-oriented episodic future thinking and self-evaluation: Raising awareness has been identified as one of the key strategies to promote healthy lifestyle behaviors. [34] We identified goal-oriented episodic future thinking as a promising approach to raise awareness. Our findings also supported that self-evaluation provides self-reinforcement for positive behavior change. [34] Listening to the recordings, we learned that participants had challenges in identifying which plans were successful for achieving their goal during the first 2-3 weeks of intervention. Many of them said they just had a good week and were not sure how and why. As the intervention progressed and participants implemented picturing and self-evaluation, they became better able to identify successful and alternate plans for accomplishing goal.
Increased motivation through goal-oriented episodic future thinking (picturing): Lack of motivation has been identified as one of the key barriers to promoting healthy lifestyle behaviors. [35] Effective and practical strategies to increase individuals’ motivation to generate and implement plans remains unknown. We have identified goal-oriented episodic future thinking as a promising approach to increase motivation through facilitating the link between goals and actions and through enhancing the subjective likelihood and/or value of a goal. [19] Listening to the recording, we learned that many women were skeptical about helpfulness of picturing and were hesitated to implement picturing in daily life during the first 2-3 weeks of intervention. This was because they did not think the simple and easy approach could be helpful. Once they implemented this strategy, however, they enjoyed its benefits.
Usefulness of pre-written goals and goal progress evaluation: Goal setting is one of the key intervention ingredients. A previous study has shown that intervention participants had a challenge in setting a goal, [33] which supports our approach: providing prewritten goals for participants to select. Our participants enjoyed evaluating goal progress, because it made them aware and reflected their progress in making positive changes.
Limitations
The pilot study has limitations. The sample size was small, which limits our ability to detect differences between groups. Interpretation of the study findings needs to be cautious. The study was conducted during the COVID pandemic, which might have negatively impacted our study outcomes. Finally, due to the 20-week length of intervention, most intervention participants completed the intervention when they were at or prior to 30 weeks gestation. A longer intervention period might be needed for future studies.
Conclusions
The pilot lifestyle behavior intervention did not prevent excessive gestation weight gain in overweight or obese pregnant participants. However, we successfully identified many intervention strengths (e.g., sample plans, goal-oriented episodic future thinking, self-evaluation, pre-written goals) and some weaknesses (e.g., exclusively textual information). Our finding can be helpful for researchers to design a prevention of excessive gestational weight gain in overweight or obese pregnant participants.
Acknowledgement
This research was funded by the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD). Grant number: R21HD099380
Conflicts of Interest
The authors declare no conflict of interest. The sponsor has no role in the design, execution, interpretation, or writing of the study.
References
- Flegal KM, Carroll MD, Kit BK, Ogden CL. Prevalence of obesity and trends in the distribution of body mass index among US adults, 1999-2010. JAMA. 2012;307(5):491-497.
- Endres LK, Straub H, McKinney C, et al. Postpartum weight retention risk factors and relationship to obesity at 1 year. Obstet Gynecol. 2015;125(1):144-152.
- Kraschnewski JL, Chuang CH, Downs DS, et al. Association of prenatal physical activity and gestational weight gain: results from the first baby study. Womens Health Issues. 2013;23(4):e233-238.
- Institute of Medicine. Weight gain during pregnancy: Reexaming the guidelines. http://nationalacademies.org/hmd/reports/2009/weight-gain-during-pregnancy-reexamining-the-guidelines.aspx. Published 2009. AccessedJuly, 2009.
- Hedderson MM, Gunderson EP, Ferrara A. Gestational weight gain and risk of gestational diabetes mellitus. Obstet Gynecol. 2010;115(3):597-604.
- Ren M, Li H, Cai W, et al. Excessive gestational weight gain in accordance with the IOM criteria and the risk of hypertensive disorders of pregnancy: a meta-analysis. BMC Pregnancy Childbirth. 2018;18(1):281.
- Goldstein RF, Abell SK, Ranasinha S, et al. Association of Gestational Weight Gain With Maternal and Infant Outcomes: A Systematic Review and Meta-analysis. JAMA. 2017;317(21):2207-2225.
- Johansson S, Villamor E, Altman M, Bonamy AK, Granath F, Cnattingius S. Maternal overweight and obesity in early pregnancy and risk of infant mortality: a population based cohort study in Sweden. BMJ. 2014;349:g6572.
- Rooney BL, Schauberger CW. Excess pregnancy weight gain and long-term obesity: one decade later. Obstet Gynecol. 2002;100(2):245-252.
- Lau EY, Liu J, Archer E, McDonald SM, Liu J. Maternal Weight Gain in Pregnancy and Risk of Obesity among Offspring: A Systematic Review. Journal of Obesity. 2014.
- Mamun AA, Mannan M, Doi SA. Gestational weight gain in relation to offspring obesity over the life course: a systematic review and bias-adjusted meta-analysis. Obes Rev. 2014;15(4):338-347.
- Phelan S. Pregnancy: a "teachable moment" for weight control and obesity prevention. Am J Obstet Gynecol. 2010;202(2):135 e131-138.
- Peaceman AM, Clifton RG, Phelan S, et al. Lifestyle Interventions Limit Gestational Weight Gain in Women with Overweight or Obesity: LIFE-Moms Prospective Meta-Analysis. Obesity (Silver Spring). 2018;26(9):1396-1404.
- Muktabhant B, Lawrie TA, Lumbiganon P, Laopaiboon M. Diet or exercise, or both, for preventing excessive weight gain in pregnancy. Cochrane Database Syst Rev. 2015;2015(6):Cd007145.
- Teede HJ, Bailey C, Moran LJ, et al. Association of Antenatal Diet and Physical Activity-Based Interventions With Gestational Weight Gain and Pregnancy Outcomes: A Systematic Review and Meta-analysis. JAMA Intern Med. 2022;182(2):106-114.
- Snyder CR. Hope theory: Rainbows in the mind. , (4), 249–275. . Psychological Inquiry. 2002;13(14):249-275.
- Schacter DL, Benoit RG, De Brigard F, Szpunar KK. Episodic future thinking and episodic counterfactual thinking: intersections between memory and decisions. Neurobiol Learn Mem. 2015;117:14-21.
- Schacter DL, Benoit RG, Szpunar KK. Episodic Future Thinking: Mechanisms and Functions. Curr Opin Behav Sci. 2017;17:41-50.
- Rebetez MM, Barsics C, Rochat L, D'Argembeau A, Van der Linden M. Procrastination, consideration of future consequences, and episodic future thinking. Conscious Cogn. 2016;42:286-292.
- MacLeod AD. Prospection, well-being and memory Memory Studies. 2016;9:266-274.
- Miloyan B, Bulley A, Suddendorf T. Episodic foresight and anxiety: Proximate and ultimate perspectives. Br J Clin Psychol. 2016;55(1):4-22.
- Wu JQ, Szpunar KK, Godovich SA, Schacter DL, Hofmann SG. Episodic future thinking in generalized anxiety disorder. J Anxiety Disord. 2015;36:1-8.
- Bandura A. Social learning theory. Englewood Cliffs, N.J.: Prentice-Hall; 1977.
- Fields SA, Lange K, Ramos A, Thamotharan S, Rassu F. The relationship between stress and delay discounting: a meta-analytic review. Behav Pharmacol. 2014;25(5-6):434-444.
- Chang M, Nitzke S, Guilford E, Adair C, Hazard D. Motivators and barriers to healthful eating and physical activity among low-income overweight and obese mothers. J Am Diet Assoc. 2008;108(6):1023-1028.
- Chang M, Tan A, Schaffir J, et al. Mediation by Executive Functions in the Associations Between Perceived Stress, Prenatal Distress, Emotional Control, and Dietary Intake in Overweight or Obese Pregnant Women. Journal of Pediatrics, Perinatology and Child Health. 2022;6(6).
- Chang MW, Nitzke S, Brown R, Resnicow K. A community based prevention of weight gain intervention (Mothers In Motion) among young low-income overweight and obese mothers: design and rationale. BMC Public Health. 2014;14:280.
- Chang MW, Nitzke S, Buist D, Cain D, Horning S, Eghtedary K. I am pregnant and want to do better but i can't: focus groups with low-income overweight and obese pregnant women. Matern Child Health J. 2015;19(5):1060-1070.
- Chang M, Nitzke S, Brown R. Design and Outcomes of a Mothers In Motion Behavioral Intervention Pilot Study. J Nutr Educ Behav. 2010;42(3 Suppl):S11-21.
- Morse JM, Richards L. Readme first for a user’s guide to qualitative methods. : . Thousand Oaks, California: Sage Publications; 2002.
- Wangberg SC, Bergmo TS, Johnsen JA. Adherence in Internet-based interventions. Patient Prefer Adherence. 2008;2:57-65.
- Raynor HA, Champagne CM. Position of the Academy of Nutrition and Dietetics: Interventions for the Treatment of Overweight and Obesity in Adults. J Acad Nutr Diet. 2016;116(1):129-147.
- Chang MW, Brown R, Nitzke S. Results and lessons learned from a prevention of weight gain program for low-income overweight and obese young mothers: Mothers In Motion. BMC Public Health. 2017;17(1):182.
- Burgess E, Hassmén P, Pumpa KL. Determinants of adherence to lifestyle intervention in adults with obesity: a systematic review. . Clin Obes. 2017;7: 123-135.
- Hardcastle SJ, Hancox J, Hattar A, Maxwell-Smith C, Thøgersen-Ntoumani C, Hagger MS. Motivating the unmotivated: how can health behavior be changed in those unwilling to change? Frontiers in Psychology. 2015;6.
