Breast Implants and Sarcopenia with Fat Degeneration of the Pectoralis Major Muscle-A Preliminary Clinical Observation
Article Information
Bouraoui Kotti*
Manar II University, Faculty of Medicine of Tunis, Tunisia
*Corresponding Author: Bouraoui Kotti, “The Office when beauty meets science” A1, Residence Yasmin du lac, Avenue de la Bourse, Lac 2, 1057, Tunis, Tunisia
Received: 22 April 2020; Accepted: 30 April 2020; Published: 04 April 2020
Citation:
Bouraoui Kotti. Breast Implants and Sarcopenia with Fat Degeneration of the Pectoralis Major Muscle-A Preliminary Clinical Observation. Journal of Women’s Health and Development 3 (2020): 092-100.
View / Download Pdf Share at FacebookAbstract
Introduction: Breast augmentation with implants is one of the most performed procedures in plastic surgery all over the world. Despite its popularity, this procedure counts many complications that may require a revision, a replacement or a deposit of the implants.
The author describe a new complication inherited to breast implants and through his observation, wonders about an ethiopathogeny relationship that may be nested with some fat degeneration problems and sarcopenia in the human body.
Methods: The author made a clinical observation while replacing implants with capsular contracture noticing a thinning of the pectoralis major muscle and an embrittlement of its fibers with discoloration and yellowness of its corpus.
Histopathology specimen were analyzed, photographed and data was collected and discussed.
Results: Biopsies were performed on the pectoralis major muscle proving the fatty degeneration and corroborant the atrophy and the sarcopenia.
The most common-sense reason was, according to the author, the implant weight and the mechanical pressure on the fibers that led to a cellular stress and a distortion in the regenerative process of the aggressed tissue.
Conclusion: A new complication related to breast implants was described. The author request more research to consider an eventual extrapolation of this theory to explain the ethiopathogeny of some classical fatty degeneration processes and sarcopenia observed in different tissues of the human body.
Keywords
Breast Implants; Sarcopenia; Fat Degeneration
Breast Implants articles Breast Implants Research articles Breast Implants review articles Breast Implants PubMed articles Breast Implants PubMed Central articles Breast Implants 2023 articles Breast Implants 2024 articles Breast Implants Scopus articles Breast Implants impact factor journals Breast Implants Scopus journals Breast Implants PubMed journals Breast Implants medical journals Breast Implants free journals Breast Implants best journals Breast Implants top journals Breast Implants free medical journals Breast Implants famous journals Breast Implants Google Scholar indexed journals Sarcopenia articles Sarcopenia Research articles Sarcopenia review articles Sarcopenia PubMed articles Sarcopenia PubMed Central articles Sarcopenia 2023 articles Sarcopenia 2024 articles Sarcopenia Scopus articles Sarcopenia impact factor journals Sarcopenia Scopus journals Sarcopenia PubMed journals Sarcopenia medical journals Sarcopenia free journals Sarcopenia best journals Sarcopenia top journals Sarcopenia free medical journals Sarcopenia famous journals Sarcopenia Google Scholar indexed journals Fat Degeneration articles Fat Degeneration Research articles Fat Degeneration review articles Fat Degeneration PubMed articles Fat Degeneration PubMed Central articles Fat Degeneration 2023 articles Fat Degeneration 2024 articles Fat Degeneration Scopus articles Fat Degeneration impact factor journals Fat Degeneration Scopus journals Fat Degeneration PubMed journals Fat Degeneration medical journals Fat Degeneration free journals Fat Degeneration best journals Fat Degeneration top journals Fat Degeneration free medical journals Fat Degeneration famous journals Fat Degeneration Google Scholar indexed journals pre-pectoral implants articles pre-pectoral implants Research articles pre-pectoral implants review articles pre-pectoral implants PubMed articles pre-pectoral implants PubMed Central articles pre-pectoral implants 2023 articles pre-pectoral implants 2024 articles pre-pectoral implants Scopus articles pre-pectoral implants impact factor journals pre-pectoral implants Scopus journals pre-pectoral implants PubMed journals pre-pectoral implants medical journals pre-pectoral implants free journals pre-pectoral implants best journals pre-pectoral implants top journals pre-pectoral implants free medical journals pre-pectoral implants famous journals pre-pectoral implants Google Scholar indexed journals posterior valve articles posterior valve Research articles posterior valve review articles posterior valve PubMed articles posterior valve PubMed Central articles posterior valve 2023 articles posterior valve 2024 articles posterior valve Scopus articles posterior valve impact factor journals posterior valve Scopus journals posterior valve PubMed journals posterior valve medical journals posterior valve free journals posterior valve best journals posterior valve top journals posterior valve free medical journals posterior valve famous journals posterior valve Google Scholar indexed journals Histopathological articles Histopathological Research articles Histopathological review articles Histopathological PubMed articles Histopathological PubMed Central articles Histopathological 2023 articles Histopathological 2024 articles Histopathological Scopus articles Histopathological impact factor journals Histopathological Scopus journals Histopathological PubMed journals Histopathological medical journals Histopathological free journals Histopathological best journals Histopathological top journals Histopathological free medical journals Histopathological famous journals Histopathological Google Scholar indexed journals muscle articles muscle Research articles muscle review articles muscle PubMed articles muscle PubMed Central articles muscle 2023 articles muscle 2024 articles muscle Scopus articles muscle impact factor journals muscle Scopus journals muscle PubMed journals muscle medical journals muscle free journals muscle best journals muscle top journals muscle free medical journals muscle famous journals muscle Google Scholar indexed journals atrophy articles atrophy Research articles atrophy review articles atrophy PubMed articles atrophy PubMed Central articles atrophy 2023 articles atrophy 2024 articles atrophy Scopus articles atrophy impact factor journals atrophy Scopus journals atrophy PubMed journals atrophy medical journals atrophy free journals atrophy best journals atrophy top journals atrophy free medical journals atrophy famous journals atrophy Google Scholar indexed journals sarcopenia� articles sarcopenia� Research articles sarcopenia� review articles sarcopenia� PubMed articles sarcopenia� PubMed Central articles sarcopenia� 2023 articles sarcopenia� 2024 articles sarcopenia� Scopus articles sarcopenia� impact factor journals sarcopenia� Scopus journals sarcopenia� PubMed journals sarcopenia� medical journals sarcopenia� free journals sarcopenia� best journals sarcopenia� top journals sarcopenia� free medical journals sarcopenia� famous journals sarcopenia� Google Scholar indexed journals fibers articles fibers Research articles fibers review articles fibers PubMed articles fibers PubMed Central articles fibers 2023 articles fibers 2024 articles fibers Scopus articles fibers impact factor journals fibers Scopus journals fibers PubMed journals fibers medical journals fibers free journals fibers best journals fibers top journals fibers free medical journals fibers famous journals fibers Google Scholar indexed journals
Article Details
Introduction
Breast augmentation with implants is one of the most performed and rewarding procedures in plastic surgery [1]. According to the International Society of Aesthetic and Plastic Surgery, ISAPS, breast augmentation remains in the first position of the top five plastic surgery procedures worldwide with 6.1% augmentation from 2017 to 2018 [2] and according to the American Society of Aesthetic Plastic Surgery, ASAPS, this procedure increased about 15,2% from 2014 to 2018 in the United Stated while keeping the pool position in the top five most practiced procedures during the last five years [3].
Despite its popularity, this procedure counts many complications [4, 5] that may require a revision, a replacement or a deposit of the implants.
We are reporting and describing a new complication due to breast implants through clinical and histological documented cases.
Methods
The author made a clinical observation while replacing pre-pectoral implants with capsular contracture grade III and IV of Baker classification [6].
While changing the pocket to a sub muscular plan, the author noticed, in the majority of cases, a thinning of the muscle and an embrittlement of its fibers and sometimes a discoloration and yellowness of its body. Histopathology specimen were analyzed, photographed and photos and videos were performed during the surgery.
Results
From January 2019 to June 2019, three patients, were operated by the author for a breast revision after capsular contracture with Baker III and two with Baker IV.
None were able to bring their medical reports. There was no possibility to differentiate between an eventual subfascial approach from a retroglandular technique to all the pre-pectoral implants. The revision was performed after 6 to 18 years from the initial breast augmentation surgery. Three were operated initially using a periareolar incision and two an inframammary incision. One case with inframammary incision was related to a submuscular pocket and one case with periareolar incision had also a submuscular pocket. (in summary there is three retro glandular and two retro pectoral pockets).
Journal of Women’s Health and Development Volume 3 No 2 – June 2020 93
All of them were implanted with textured implants from different commercial brands. Two cases with extra capsular rupture and silicone migration operated in this period were not included. We observed a thinning of the pectoralis major muscle for all these five patients with pre and retro pectoral pockets without concern to their Baker grade.
We observed a yellowness of the fibers for two patients operated with retroglandular implants and pre pectoral pockets. Only these two patients had implants more than 500cc in volume for each. The other volumes were between 320 and 485 cc. This clinical observation was obvious with the patient holding the biggest implants sizes (650cc) (video 1)
Biopsies of the muscle were performed always in the same places behind the posterior valve of the implant and at the lateral border of the muscle for the patients with retro glandular pockets and clinical yellowness. In control a biopsy of the muscle was performed also for a patient with a retro-pectoral pocket and clinical thinning of the muscle but without obvious clinical yellowness on its fibers. The Histopathological examination revealed for the cases with clinical discoloration a fat tissue infiltration and a rarefication of the muscular fibers (Figures 1 , 2, 3 and 4).
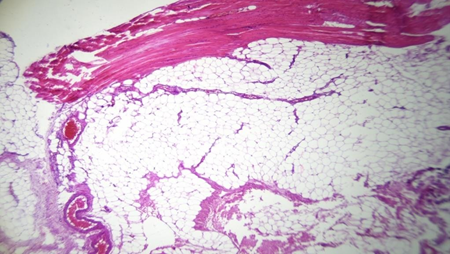
Figure 1: Total infiltration with adipocytes of the pectoralis major muscle
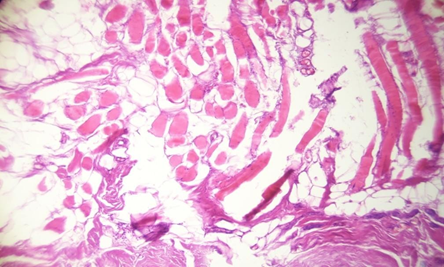
Figure 2: Fatty degeneration and muscular fibers atrophy
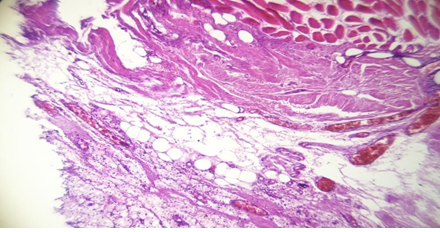
Figure 3: Histologic stigma of chronic inflammation and dystrophic cicatrisation with fat degeneration
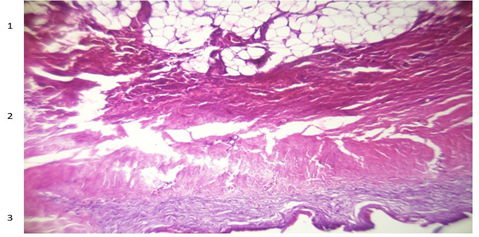
Figure 4: Fat degeneration (1), muscle atrophy (2) and sclerotic tissue (3)
The micrograph of the muscle biopsy harvested on the posterior side of the pectoralis major for the patient with retro- muscular pocket did not show an obvious fat infiltration but a muscle atrophy stigma with a fibro-sclerosis tissue due to the adherent capsule (Figure 5).
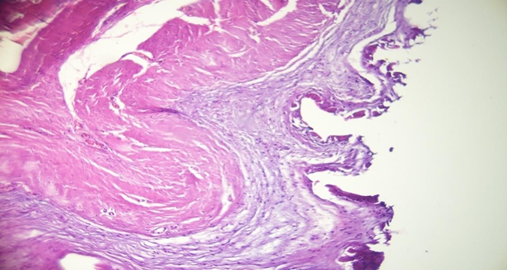
Figure 5: Dystrophic cicatrisation stigma and chronic inflammation of the muscle with sclerotic tissue on the surface.
None of the patients were known with muscular dystrophia, diabetes or other neurological or immunological disease and none reported a significant weight gain during the examination.
Discussion
Preamble
Sarcopenia (Greek ‘sarx’ or flesh + ‘penia’ or loss) is a muscular atrophy first described in 1989 then related, by the same author, in 1997 to the muscular aging process for elder patients [7, 8]. Like osteopenia, it can also be seen in younger patients such as those with inflammatory diseases [9]. Its development may be associated with conditions that are not exclusively seen in older persons, like malnutrition and cachexia [10].
The mechanisms of sarcopenia are not clearly defined. Besides the lack of muscular mobility, sarcopenia is associated with major co-morbidity such as obesity, osteoporosis and type 2 diabetes and insulin resistance for youngers [11, 12] and can be closely nested with muscle fatty degeneration as described for the rotator cuff lesions [13, 14] and the supraspinatus muscle [15]. The fatty degeneration is usually due to the muscle atrophy and not the opposite.
A lot of authors wrote about the fatty degeneration and the muscle denervation on animals and humans [16-18], but we know also that in absence of denervation, after a muscle trauma and when regeneration fails, the fibrotic scar may be infiltrated with adipocytes in addition to fibroblasts [19].
Some authors suggest that some myogenic factors like skeletal muscle satellite cells can spontaneously enter an alternative mesenchymal pathway and generate fat cells [20] while others reject this theory [21] and argue more for other precursors and progenitor cells, than the classical satellite cells, that may interfere in the regenerative repair process under muscle stress [22, 23]. The cellular origin of fatty infiltration remains controversial but its presence surely testifies to a muscular aggression with an aberrant cellular repair in the muscle [24].
Capsular contracture is a multifactorial fibrotic reaction to a foreign body (implant) [25] that known to have lower incidence with the use of textured implants, submuscularplacement [26] with a debate about the inframammary incision as a surgical approach to decrease the capsular contracture rate [27].
In our study, the body inflammatory response with the capsular synthesis happened in different situations with different type of surface texturization, surgical incision and implant placement. In all cases, we observed a clinical thinning of the muscle but not in all cases we observed the yellow discoloration attesting an eventual fat degeneration. We believe that the capsule is not directly involved with our observations.
Roxo et al. found alternatively, a glandular parenchymal reduction with retro-glandular implants and a pectoralis muscle atrophy with retro-muscular implants using a volumetric magnetic resonance imaging [28]. In our study we observed clinically, during the surgery, a muscle atrophy in Baker III and IV grades and a fat infiltration of the muscle for the cases with retro-glandular implants. This clinical observation of muscle flattening is absolutely subjective and its appreciation is related to the surgeon’s experience in the field of breast surgery. Even if we believe that the capsule is not directly involved in this phenomenon, the mechanical pressure inducted by the implant and the capsule on the muscle from one side or the other may lead to a local stress on the muscular cells and induce an inaccurate repair inside the muscle. The weight of the implant on the muscle, especially if it’s nested with a tight initial pocket, significantly contribute, in our opinion, to this distorted muscle reaction with fat degeneration. Indeed, we observed the yellow discoloration and the fatty degeneration only for important implants volumes (more than 500 cc) placed above the muscle.
The direct mechanical pressure of the implant weight on the muscle is, in our thoughts, the responsible of the fat degeneration.
In 2010, the European Working Group on Sarcopenia in Older People (EWGSOP) published a sarcopenia definition [29] that aimed to foster advances in identifying and caring for people with sarcopenia. In 2018, the Working Group (EWGSOP2) updated the original definition [30] focusing on low muscle strength with low muscle quantity and quality to confirm the diagnosis.
In our preliminary study we were not able to measure the pectoralis major strength before and after the surgery but this procedure can be performed in the future for that purpose [31].
As for chicks [32], or denervated muscles [33], the MRI, can also be used for fat degeneration study of the pectoralis major muscle after breast implants and by extrapolation of the gluteus maximus muscle after gluteoplasty with implants.
All our patients are less than 50 YO, none is known with a metabolic, auto-immune or nutritional disease or disorder. We did not find any other risk factor, already described, to explain our ascertainment.
In all the documentation available in literature about muscle fat degeneration and sarcopenia, we did not find the impact of the direct mechanical pressure on the muscular cells due to the weight practiced on the fibers, of the implant in our case, or of the body on the cuff rotator muscles while we sleep on one side or the other and by extrapolation the body weight on the bony tissue of the upper femur that may causes step by step an embrittlement of the bony tissue regeneration process leading to contribute to the osteoporosis in the femoral neck area.
Conclusion
We describe here a clinical observation of a new complication due, probably, to breast implants. Our hypothesis advocates that the weight of the implant and its mechanical pressure on the muscle are an insidious stress that may cause, with time, a fatty degeneration as well as a sarcopenia. We already started to collect more data about muscle reaction, using biopsies and micrographs, during breast implants replacements. More studies need to be conducted to prove that :
- The muscular stress caused by the implant weight is better tolerated with retro-pectoral implants (rather than pre- pectoral), low volumes implants (rather than big volumes) (then, how can we define the limits ?), low and moderate profile implants (rather than extra high profile that may cause, with time, more stress on the environmental tissues with the pushing effect of the implant apex on the gland for retro- glandular placement cases or on the pectoralis major fibers, especially with a lack of a complete muscle costal disinsertion, for retro pectoral placements ).
- By extrapolation, the insidious micro traumatisms and the stress caused by the body weight on the supraspinatus muscle and the rotator cuff muscles as well as the femur during the sleeping hours in lateral positions may cause, after years, an alteration in the regenerative process of the muscle cells or the bony cells and lead to an eventual fatty degeneration and sarcopenia as well as worsen the osteoporosis effect on femoral neck architecture and stability.
Acknowledgements
Thanks to my colleagues cythopathologits for their kind support.
Conflict of Interest
No Disclosure.
References
- Johnson M. Breast implants: history, safety, and imaging. Radiol Technol 84 (2013): 439-515.
- ISAPS international survey on aesthetic/cosmetic procedures performed in Statistical data (2018).
- The American Society of Aesthetic plastic Surgery/Cosmetic (Aesthetic) surgery national data bank statistics (2018).
- Handel N, Cordray T, Gutierrez J, Jensen JA. A long-term study of outcomes, complications, and patient satisfaction with breast implants. Plast Reconstr Surg 117 (2006): 757-767.
- Risks and Complications of Breast Implants. US Food and Drug Administration (2019).
- Spear SL, Baker JL Classification of capsular contracture after prosthetic breast reconstruction. Plast Reconstr Surg 96 (1995): 1119-1123.
- Rosenberg I. Summary comments: epidemiological and methodological problems in determining nutritional status of older persons. Am J Clin Nutr 50 (1989):
- Rosenberg IH. Sarcopenia: origins and clinical relevance. J Nutr 127 (1997): 990-991.
- Schneider SM, Al-Jaouni R, Filippi J, Wiroth JB, Zeanandin G, Arab K, et Sarcopenia is prevalent in patients with Crohn’s disease in clinical remission. Inflamm Bowel Dis 14 (2008): 1562-1568.
- Valter S, Andrea B, Massimiliano M, Marco Clinical definition of sarcopenia. Clin Cases Miner Bone Metab 11 (2014): 177-180.
- Janssen I, Shepard DS, Katzmarzyk PT, Roubenoff R. The health- care costs of sarcopenia in the United States. J Am Geriatr Soc 52 (2004): 80-85.
- Gale CR, Martyn CN, Cooper C, Sayer AA. Grip strength, body composition, and Int J Epidemiol 36 (2007): 228-235.
- Feasson L. Mechanisms of muscular atrophy and fatty degeneration. La Lettre du rhumatologue 396 (2013): 8- 12.
- Melis B, Nemoz C, Walch Muscle fatty infi ltration in rotator cuff tears: descriptive analysis of 1688 cases. Orthop Traumatol Surg Res 95 (2009): 319-324.
- Barry JJ, Lansdown DA, Cheung S, Feeley BT, Ma CB. The relationship between tear severity, fatty infiltration, and muscle atrophy in the supraspinatus. J Shoulder Elbow Surg 22 (2013): 18-25.
- Dulor JP, Cambon B, Vigneron P, Reyne Y, Nouguès J, Casteilla L, et Expression of specific white adipose tissue genes in denervation-induced skeletal muscle fatty degeneration. FEBS Lett 439 (1998): 89-92.
- Gutmann E. The denervated muscle. Prague 1962; Springer Dec (2013).
- Carlson BM. The Biology of Long-Term Denervated Skeletal Muscle. Eur J Transl Myol 24 (2014): 3293.
- Natarajan A, Lemos DR, Rossi Fibro/adipogenic progenitors: A double-edged sword in skeletal muscle regeneration. Cell Cycle 9 (2010).
- Shefer G, Wleklinski-Lee M, Yablonka-Reuveni Z. Skeletal muscle satellite cells can spontaneously enter an alternative mesenchymal pathway. J Cell Sci 117 (2004): 5393-5404.
- Starkey JD, Yamamoto M, Yamamoto S, Goldhamer DJ. Skeletal muscle satellite cells are committed to myogenesis and do not spontaneously adopt nonmyogenic fates. J Histochem Cytochem 59 (2011): 33-46.
- Sirabella D, De Angelis L, Berghella L. Sources for skeletal muscle repair: from satellite cells to reprogramming. J Cachexia Sarcopenia Muscle 4 (2013): 125-136.
- Uezumi A, Fukada S, Yamamoto N, Takeda S, Tsuchida K. Mesenchymal progenitors distinct from satellite cells contribute to ectopic fat cell formation in skeletal muscle. Nat Cell Biol 12 (2010): 143-152.
- Mann CJ, Perdiguero E, Kharraz Y, Aguilar S, Pessina P, Serrano AL, et Aberrant repair and fibrosis development in skeletal muscle. Skelet Muscle 1 (2011): 21.
- Wolfram D, Rainer C, Niederegger H, Piza H, Wick Cellular and molecular composition of fibrous capsules formed around silicone breast implants with special focus on local immune reactions. J Autoimmun 23 (2004): 81-89.
- Headon H, Kasem A, Mokbel Capsular Contracture after Breast Augmentation: An Update for Clinical Practice. rch Plast Surg 42 (2015): 532-543.
- Li S, Chen L, Liu W, Mu D, Luan J. Capsular Contracture Rate After Breast Augmentation with Periareolar Versus Other Two (Inframammary and Transaxillary) Incisions: A Meta-Analysis. Aesth Plast Surg 42 (2018): 32-37.
- Roxo AC, Nahas FX, Salin R, de Castro CC, Aboudib JH, Marques RG. Volumetric Evaluation of the Mammary Gland and Pectoralis Major Muscle following Subglandular and Submuscular Breast Augmentation. Plast Reconstr Surg 137 (2016): 62-69.
- Cruz-Jentoft AJ, Baeyens JP, Bauer JM, Boirie Y, Cederholm T, Landi F, et Sarcopenia: European consensus on definition and diagnosis-Report of the European working group on Sarcopenia in older people. Age Ageing 39 (2010): 412-423.
- Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyère O, Cederholm T, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing 48 (2019): 16-31.
- Beals SP, Golden KA, Basten M, Kelly Strength performance of the pectoralis major muscle after subpectoral breast augmentation surgery. Aesthet Surg J 23 (2003): 92-97.
- Misra LK, Kasturi SR, Kundu SK, Harati Y, Hazlewood CF, Luthra MG, et Evaluation of muscle degeneration in inherited muscular dystrophy by nuclear magnetic resonance techniques. Magn Reson Imaging 1 (1982): 75-79.
- Shabas D, Gerard G, Rossi D. Magnetic resonance imaging examination of denervated muscle. Computerized Radiology 11 (1987): 9-13.
